Abstract
A 12-year-old chinchilla (Chinchilla lanigera) developed a slow-growing, soft, fluctuating, nonpainful mass on the ventral neck with focally extensive alopecia over a period of approximately 8 months. On postmortem examination, an extensive, multilobulated, cystic, neoplastic mass extended subcutaneously over the ventral and lateral neck with metastatic spread to submandibular lymph nodes, spleen, liver, and lungs. Neoplastic cells were strongly positive for vimentin and pan-cytokeratin but were negative for alpha–smooth muscle actin, S100, and myosin; no intracytoplasmic myofibrils were detected on phosphotungstic acid hematoxylin staining. Histologic and immunohistochemical examination of the mass led to a diagnosis of undifferentiated carcinoma of the salivary gland and contributes to the paucity of knowledge concerning neoplasia in chinchillas.
The chinchilla (Chinchilla lanigera) has been farmed for fur since the late 19th century and more recently has been favored as an experimental model for ototoxicity re-search, 11,12 as well as becoming an increasingly popular pet species. The veterinary literature, however, contains very few reports of acquired diseases in chinchillas. Unlike many other rodents, chinchillas are long-lived (longevity is reported to be up to 20 years), yet reports of neoplasia are rare, leading to speculation that the chinchilla is somewhat resistant to tumor development. 7 Only single reports of tumors exist in the veterinary literature and include neuroblastoma, carcinoma, lipoma, and hemangiosarcoma (unspecified locations) 7 ; a malignant lymphosarcoma with metastatic involvement of the liver, spleen, and kidneys 14 ; and a liver carcinoma. 15 A 5-year retrospective examination of chinchillas that had been presented to the Animal Medical Center in New York revealed only 1 case of neoplasia: a uterine leiomyosarcoma without metastasis in a 1-year-old female. 8 A 35-year retrospective evaluation of chinchilla presentations to the University of Tennessee College of Veterinary Medicine documented only 2 cases of neoplasia: lymphosarcoma and adenocarcinoma of the lung, respectively. 8 A recent case report documented a lumbar osteosarcoma in an 11-year-old male chinchilla with hind-limb paralysis and self-trauma to the distal end of the tail. 16
A 12-year-old female chinchilla was presented for examination at the Royal (Dick) School of Veterinary Studies (The University of Edinburgh, Roslin, Midlothian, UK) for assessment of a slow-growing, right-sided, non-painful submandibular mass, first noticed 5 months earlier. Over the following 2 months, the right submandibular mass increased significantly in size, extending dorsally to the lateral face. The chinchilla lost 5% body weight and developed mild peripheral lymphadenomegally but remained bright and active. Intermittent dysphagia and facial pruritis were also noted. The animal was euthanized 12 weeks after initial presentation because of a sudden deterioration in condition.
Postmortem examination revealed a soft, fluctuant, poorly demarcated, subcutaneous mass (60 mm x 40 mm x 25 mm) on the ventral and lateral neck, covered by sparsely haired skin and small, dark red, superficial crusts. On gross cut section, the mass was mottled pale yellow and contained numerous variably sized, pale yellow, fluid-filled cystic foci. Submandibular lymph nodes were replaced by multiple, coalescing cystic structures containing pale yellow fluid. Multiple, pale yellow, soft to firm, homogeneous masses were present in the liver, lungs, and spleen. The capsular surface of both kidneys was diffusely roughened and multifocally depressed.
Samples of mass, haired skin, lungs, liver, spleen, kidneys, bone marrow, lymph node, adrenal gland, and brain were collected, fixed in 10% buffered formalin, and routinely processed for histologic examination. Sections of tissues 4 μm thick were stained with hematoxylin and eosin, periodic acid–Schiff (PAS), Alcian blue, Masson trichrome, phosphotungstic acid hematoxylin (PTAH), and Congo red stains. Additionally, immunostaining was performed with Envison polymer reagents based on a standard avidinbiotin–peroxidase complex–based method, including reagents for vimentin (clone V9; monoclonal, a dilution 1:100), cytokeratin (pan-CK; clone MNF116, monoclonal, b dilution 1:100), alpha-smooth muscle actin (α-SMA, clone 1A4, monoclonal, c dilution 1:5,000), S100 protein (poly-clonal rabbit anti-S100, polyclonal, b dilution 1:400), myosin (heavy and light chains, monoclonal, c dilution 1:20), major histocompatibility complex class II (MHC class II; clone TAL.1B5, monoclonal, b dilution 1:20), Mac 387 (clone M0747, monoclonal, b dilution 1:400), cluster of differentiation (CD)79 a (HM57, monoclonal, b dilution 1:100), and CD3 (clone A0452, monoclonal, b dilution 1:100) as primary antibodies. For vimentin, α-SMA, S100, myosin, and Mac 387, antigen retrieval involved microwave treatment at 110°C for 15 min. For MHC II, CD79a, and CD3, antigen retrieval involved treatment with sodium citrate (pH 6.0) in a steamer for 3 min. Pan-CK and Mac 387 immunostaining required prior treatment with proteinase K (ready-to-use, b room temperature) for 30 and 5 min, respectively. Ovine colon, ovine lung, and canine tonsil were used as control tissues, and internal controls included chinchilla cutaneous tissue, salivary gland, and skeletal muscle.
Histopathologic examination of the mass revealed a nodular, unencapsulated, neoplastic proliferation of spindloid to polygonal cells surrounded by a moderately abundant fibrous stroma. Cells were arranged in sheets, packets, lobules, and short streams, with a moderately infiltrative growth pattern. This cell proliferation extended from the superficial dermis into the deep dermis, subcutis, and skeletal muscle and was contiguous with salivary glands. Cells had variably distinct cell borders and a moderate amount of eosinophilic, finely granular cytoplasm. They had large, round to oval to reniform, open-faced nuclei with 1–2 medium-sized nucleoli. Anisocytosis, anisokaryosis, and nuclear pleomorphism were marked, and the mitotic index was moderately high (3–5 mitotic figures per high-power field [40x]) with frequent atypical mitoses and multinucleated forms. Frequently, the center of these neoplastic foci contained large pseudocystic spaces (Fig. 1) containing abundant, very pale eosinophilic fluid that stained faintly positive with PAS and Alcian blue. Neoplastic cells surrounding these pseudocysts were frequently flattened and poorly cohesive. Small occlusive neoplastic emboli were noted within lymphatic vessels, and proliferations of identical cells were noted in sections of submandibular lymph node, liver, lungs and spleen, forming short streams, bundles, and small pseudocystic spaces.
Immunohistochemical examination revealed diffuse, strong cytoplasmic labeling of the majority (>95%) of neoplastic cells for vimentin (Fig. 2), and the cytoplasm of approximately 40–60% of cells stained strongly for pan-CK of the neoplastic foci with fewer positively stained cells at the periphery (Fig. 3). Tumor cells were negative for α-SMA, but myofibroblastic stroma was strongly positive (Fig. 4). Immunohistochemistry for S100, myosin, MAC 387, CD3, CD79a, and PTAH stain yielded negative results. Labeling for MHC class II was inconclusive because no staining was observed in any of the section, including surrounding tissues. Normal myoepithelial cells of the adjacent salivary gland were immunoreactive for vimentin, cytokeratin, α-SMA, S100, and myosin, whereas no intracytoplasmic myofibrils were detected on PTAH stain. Skeletal muscle myofibrils stained deeply red with PTAH stain in adjacent tissue. Similar neoplastic foci were present in the spleen, lung, lymph nodes, and liver, occasionally forming pseudocysts, consistent with metastases of the neck mass. Mild, multifocal renal cortical fibrosis with tubular atrophy and mild, multifocal hepatic extra-medullary hematopoiesis were also recorded.
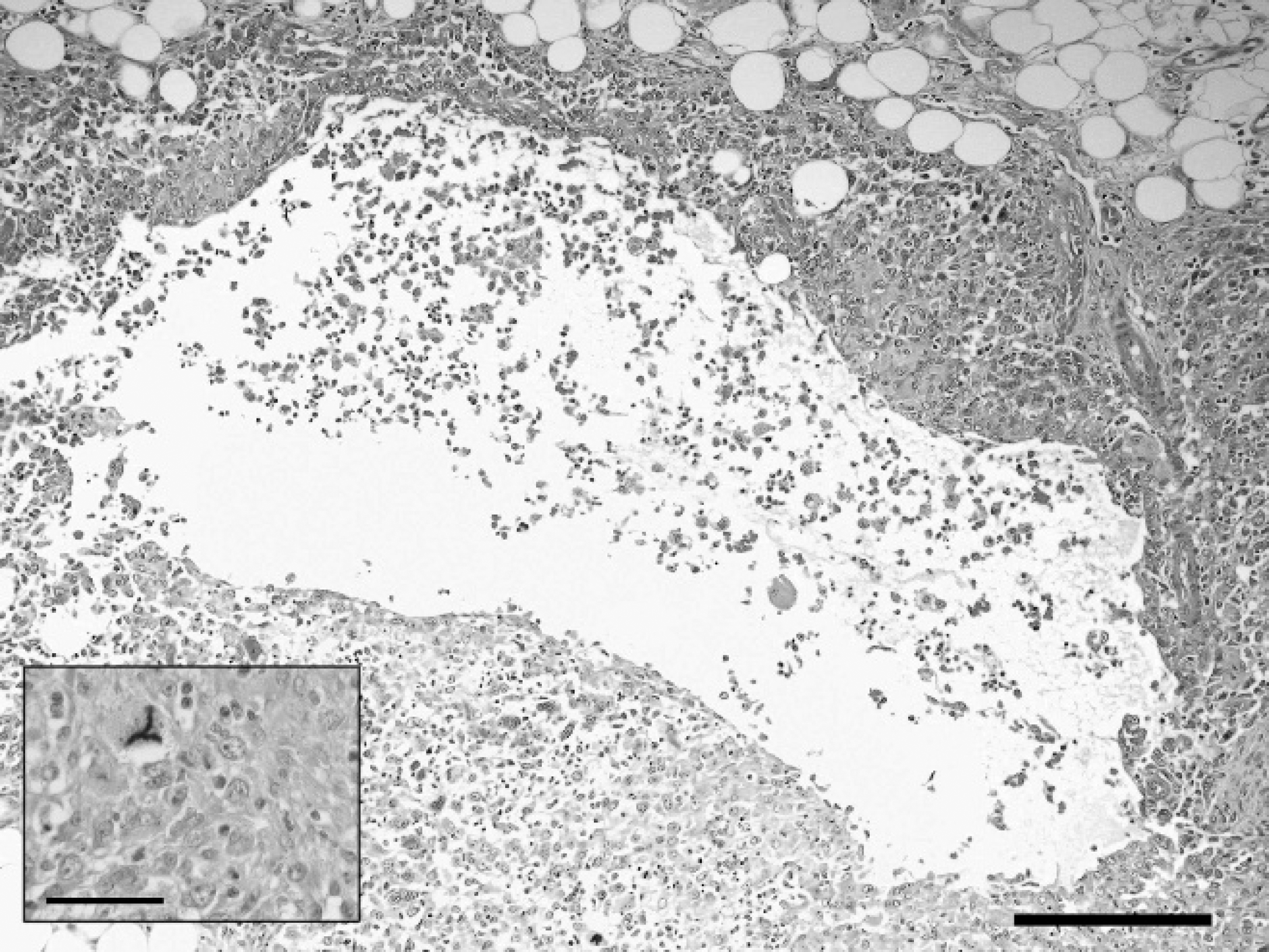
Undifferentiated carcinoma, salivary gland, chinchilla (Chinchilla lanigera). Pseudocysts surrounded by a thick wall of polygonal to spindloid cells. Hematoxylin and eosin. Bar = 200 μm.
Tissues known to regularly coexpress vimentin and cytokeratin in human beings and mammals include normal and neoplastic mammary gland, salivary gland, synovial epithelium, and mesothelial cells. 5 The co-expression of cytokeratin and vimentin, and the contiguity of the tumor with normal salivary gland were considered to be consistent with a diagnosis of malignant neoplasia of the salivary gland with metastases to spleen, liver, lungs, and lymph nodes.
Solid adenocarcinoma, malignant myoepithelioma, and undifferentiated carcinoma were considered the primary differential diagnoses for this tumor. The polygonal to spindloid morphology of the neoplastic cells, unencapsu-lated nature of the tumor, numerous necrotic pseudocystic spaces, and lack of acinar or ductular structures are features commonly seen in malignant myoepitheliomas and undifferentiated carcinomas of rodents and domesticated animals. 1,9 The lack of acinar or ductular structures is the key feature for differentiation from adenocarcinoma in this case.
Although rare, a wide range of salivary gland tumors have been described in other rodents, including mice and rats. 2,3,13 Salivary gland adenomas occur spontaneously in rats and are thought to arise from acinar or ductular epithelial components. Adenocarcinomas of the salivary gland are rare in mice but occur with some frequency in rats, in which they span a spectrum from well differentiated to poorly differentiated and are characterized by areas of solid, acinar, and papillary differentiation. 13 Squamous cell carcinomas are rare but occur spontaneously in transgenic mice with the v-Ha-ras oncogene 2 and polyomavirus-induced tumors are frequently reported in the salivary gland after inoculation of neonatal mice with polyoma-virus. 3
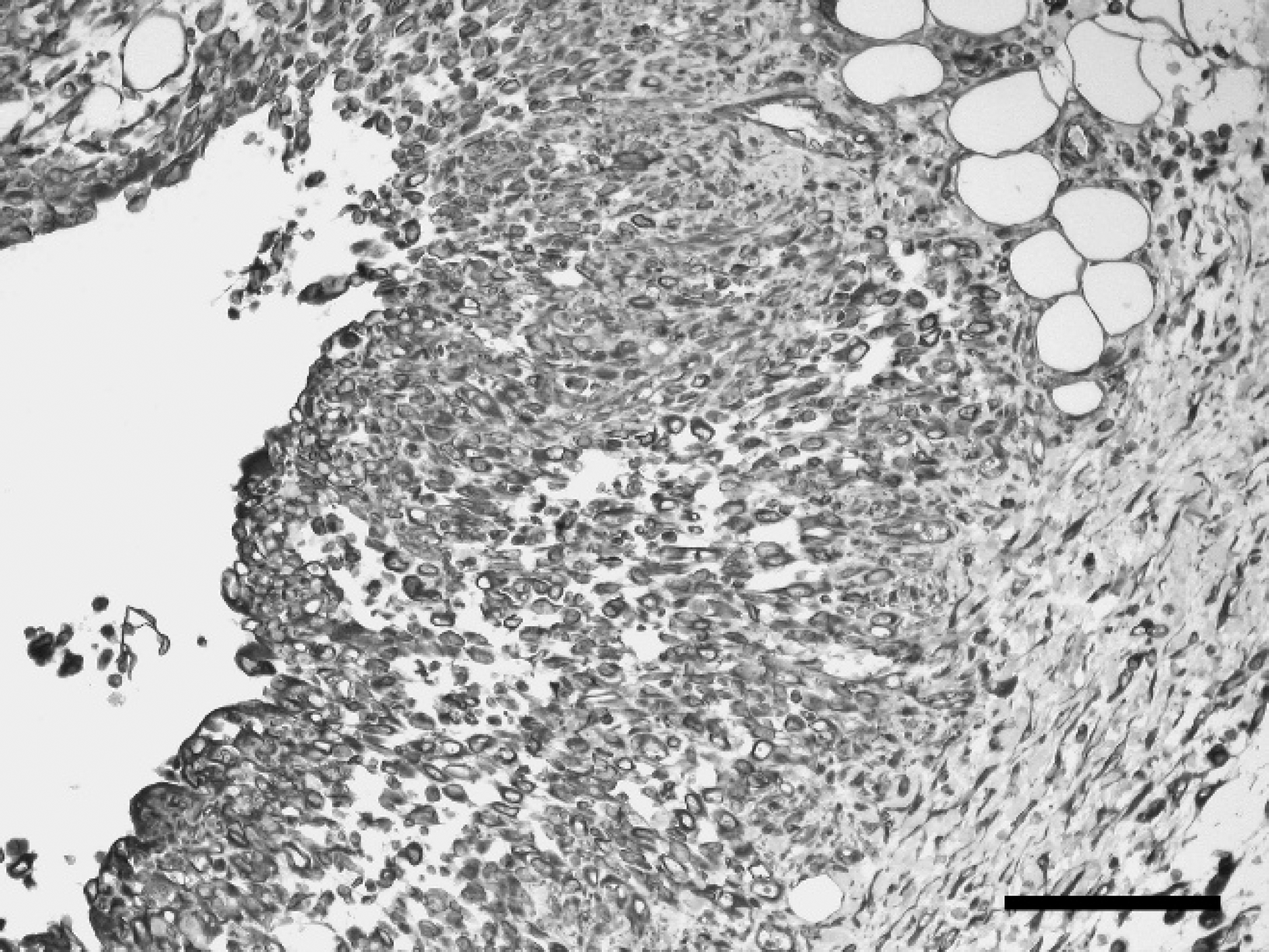
Undifferentiated carcinoma, salivary gland, chinchilla (Chinchilla lanigera). Immunohistochemical staining for vimentin antigen. Diffuse, strong cytoplasmic labeling of the majority (>95%) of neoplastic cells for vimentin. Immunohisto-chemistry. Bar = 100 mm.
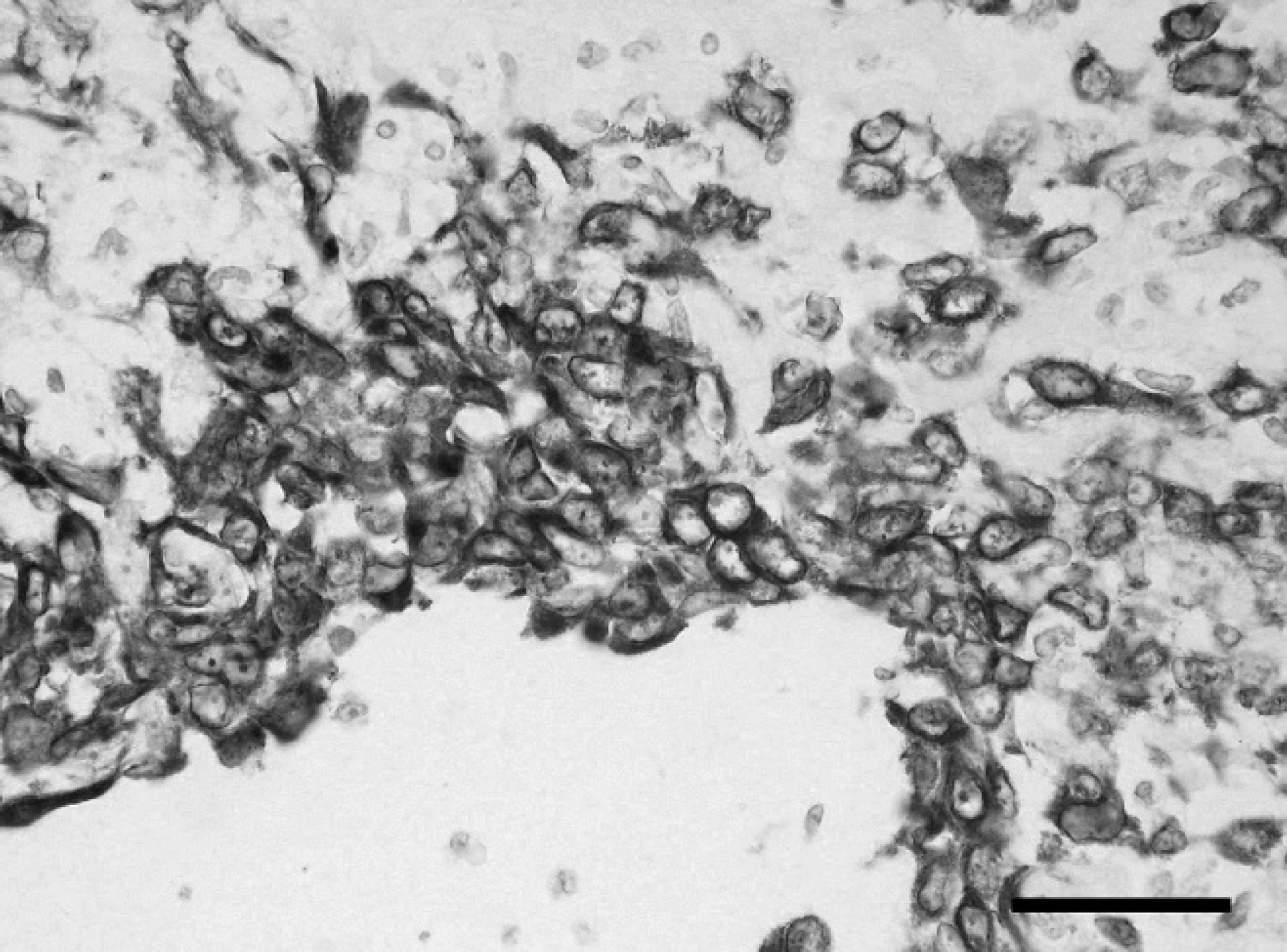
Undifferentiated carcinoma, salivary gland, chinchilla (Chinchilla lanigera). Immunohistochemical staining for cytokeratin. Cytokeratin-positive neoplastic cells were predominantly located toward the center of the neoplastic foci with fewer positively stained cells at the periphery. Immunohistochemistry. Bar = 50 μm.
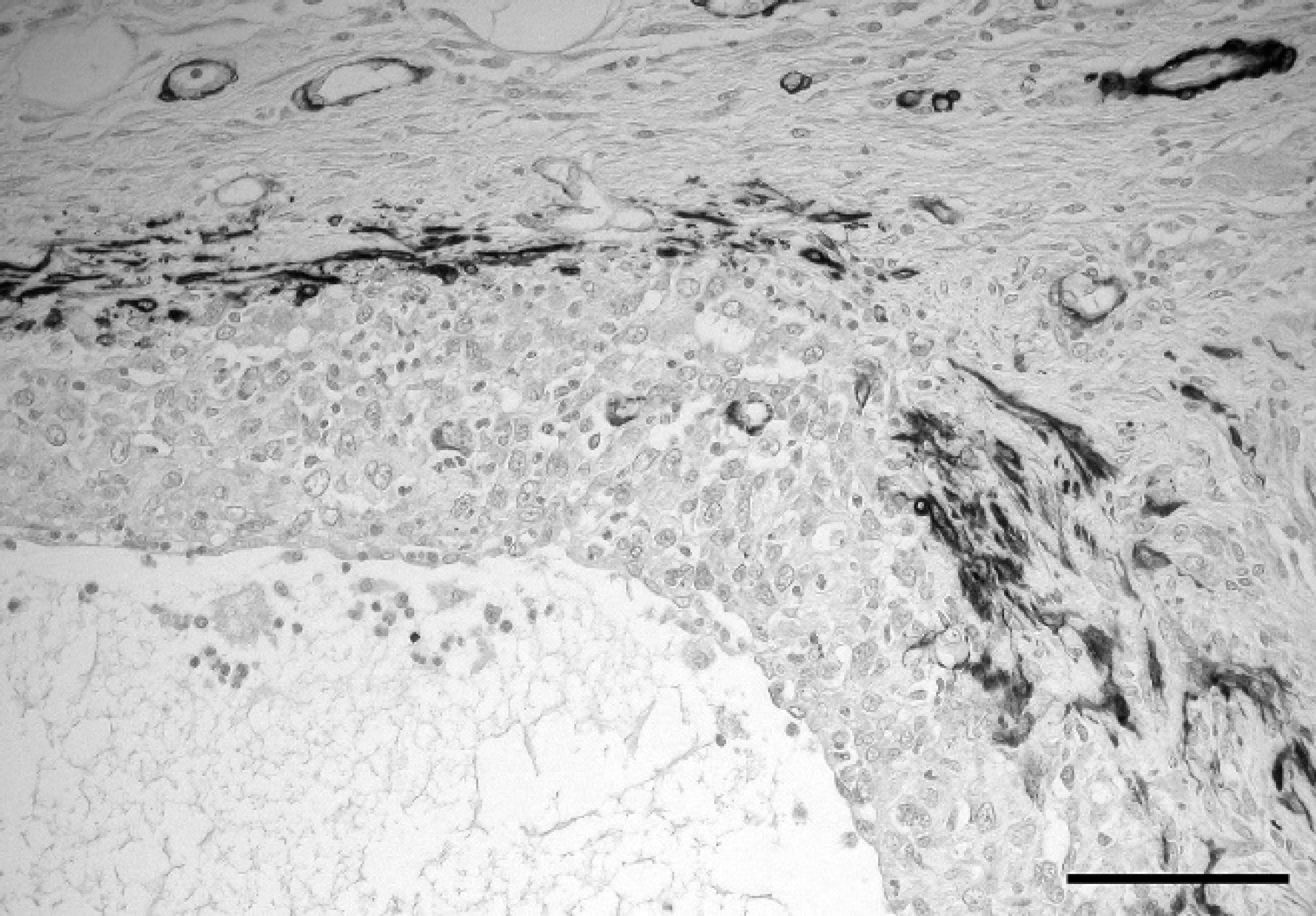
Undifferentiated carcinoma, salivary gland, chinchilla (Chinchilla lanigera). Immunohistochemical staining for alpha-smooth muscle actin (α-SMA). Tumor cells were negative for α-SMA, but myofibroblastic stroma was strongly positive. Immunohistochemistry. Bar = 100 mm.
Salivary gland myoepitheliomas arise infrequently in most strains of mice but are relatively more common in other strains, such as BALB/c and A/J stain mice. 3,17 These tumors can become quite large, with pseudocystic chambers containing serous fluid, and are composed of large, pleomorphic spindle cells with epithelial and mesenchymal features. 1 Myoepitheliomas arise from myoepithelial cells of various exocrine glands and can exhibit a dual epithelial and contractile phenotype, confirmed by immunohisto-chemistry or electron microscopy. In general, normal and neoplastic myoepithelial cells are immunoreactive for S100, contain actin and PTAH-positive filaments, and express the intermediate filaments vimentin and cytokeratin. 3,4,6,10 The negative staining of neoplastic cells for α-SMA, S100, PTAH, or myosin results obtained in this case are sufficient to exclude a diagnosis of malignant myoepithelioma.
It has been speculated that chinchillas have a lower incidence of neoplasia than other small mammals, 7 possibly as a consequence of some undetermined genetic factors, but this remains unproven at the present time. With their increasing popularity as pets, their relatively long lifespan, and the development of clinical veterinary services for geriatric rodents, it is anticipated that the frequency of reports of neoplastic conditions in chinchillas will increase in the future. To the authors' knowledge, the present case represents the first known finding of an undifferentiated carcinoma of the salivary gland in this species.
Acknowledgements. The authors thank the histopathol-ogy departments of the Royal (Dick) School of Veterinary Sciences and Glasgow Veterinary University for excellent technical assistance. JS is funded by the RCVS Trust.
Footnotes
a.
Novocastra Laboratories Ltd., Newcastle upon Tyne, UK.
b.
Dako, Cambridge, UK.
c.
Sigma-Aldrich Chemie GmBH, Munich, Germany.
