Abstract
A 3-y-old male miniature Dachshund was presented with an ~0.8 cm diameter mass in the right mandibular region. Fourteen months later, the mass was 5 × 4 × 3 cm. Grossly, the mass was encapsulated and was homogeneously gray-white on cut surface. Microscopically, the mass was composed of large, round to polygonal tumor cells that were arranged in solid nests and cords separated by a fibrovascular stroma. Tumor cells had large, round, hypochromatic nuclei containing large prominent nucleoli and abundant eosinophilic cytoplasm containing dark blue granules visible with phosphotungstic acid–hematoxylin stain. Metastasis was observed in the mandibular lymph node. Immunohistochemically, tumor cells were positive for CK AE1/AE3, low-molecular-weight CK (CAM5.2), E-cadherin, mitochondria ATPase beta subunit, and S100, but were negative for vimentin, carcinoembryonic antigen, p63, CK14, CD10, and chromogranin A. Ultrastructurally, tumor cells contained numerous mitochondria. Therefore, the tumor was diagnosed as an oncocytic carcinoma of the mandibular gland.
Primary salivary gland tumors are uncommon in dogs. 5 According to the World Health Organization (WHO) classification of tumors of domestic animals, these malignant epithelial tumors include acinic cell carcinoma, mucoepidermoid carcinoma, cystadenocarcinoma, adenocarcinoma, malignant myoepithelioma (myoepithelial carcinoma), carcinoma or sarcoma in pleomorphic adenoma (malignant mixed tumor), squamous cell carcinoma, and undifferentiated carcinoma. 5 In humans, primary salivary gland tumors are also uncommon, and include basal cell adenocarcinoma, malignant sebaceous tumors, and oncocytic carcinoma as well. 2
Oncocytic carcinoma arising in human salivary glands is an extremely rare tumor composed of malignant oncocytes with atypical nuclear and abundant eosinophilic cytoplasm containing numerous mitochondria, which can be detected with phosphotungstic acid–hematoxylin (PTAH) stains, immunohistochemistry (IHC) using an anti-mitochondria antibody, and ultrastructural analysis. 2 However, to our knowledge, salivary gland oncocytic carcinoma has not been described in dogs.
Herein, we report a case of oncocytic carcinoma that arose in the submandibular gland and metastasized to the mandibular lymph node. A 3-y-old male miniature Dachshund was presented with a mass ~0.8 cm diameter in the right mandibular region. Fourteen months later, the mass measured 5 × 4 × 3 cm. Aside from the mass, no other abnormalities were observed on physical examination, complete blood cell count, or routine serum biochemical profile. Detailed ultrasonographic and radiographic examinations detected no masses in the thoracic or abdominal cavities. The mass was surgically excised and submitted to the Department of Veterinary Pathology, Nippon Veterinary and Life Science University (Tokyo, Japan). No recurrence was noted after 11 mo. Additional radiation and chemotherapy were not performed following surgical excision.
The mass was fixed in 10% neutral-buffered formalin and embedded in paraffin wax. Sections (3 µm) were stained with hematoxylin and eosin, alcian blue at pH 2.5 (AB), mucicarmine, Grimelius, periodic acid–Schiff (PAS), PTAH, and oil red O stains. Serial sections underwent IHC with primary mouse antibodies specific for cytokeratin (CK) AE1/AE3, a low-molecular-weight CK (CAM5.2), b CK14, c CD10, a E-cadherin, a mitochondrial ATPase beta subunit (ATPB), d p63, e α-smooth muscle actin (SMA), a vimentin, a and Ki-67, a and primary rabbit antibodies specific for carcinoembryonic antigen (CEA), a chromogranin A, a glial fibrillary acid protein (GFAP), a and S100. a Antibodies used were validated by a positive reaction with appropriate normal tissue control sections and a negative reaction to replacement with normal mouse or rabbit immunoglobulins. The neoplastic cell proliferation index was assessed by counting both Ki-67–positive and –negative tumor cells in 10 randomly selected fields at 400× magnification and by expressing the count as a percentage (Ki-67–positive cells/total number of cells counted). For ultrastructural examination, pieces of formalin-fixed tumor tissue were refixed in 1% osmium tetroxide, followed by 0.2 M phosphate buffer, and then embedded in epoxy resin. After uranyl acetate and lead citrate staining, ultrathin sections were examined with an electron microscope.
Grossly, the mass was encapsulated and was homogeneously gray-white on cut surface. Microscopically, the mass was composed of large, round to polygonal tumor cells that were arranged in solid nests containing duct-like structures, cords, or scattered individual cells separated by a fibrovascular stroma (Fig. 1A). Large solid nests with central necrosis were often observed. Tumor cells had large, round, hypochromatic nuclei containing large prominent nucleoli, and had abundant eosinophilic cytoplasm, containing dark blue granules on PTAH stain (Fig. 1B). Furthermore, they contained partially diastase-sensitive, PAS-positive granules or lipid revealed by oil red O stain. Tumor cells did not show specific staining with AB, mucicarmine, and Grimelius stains. Frequency of mitotic figures was 0–3 per high power (400×) field. Occasional mandibular glands and lymph nodes were observed at the edge of the mass. A metastatic lesion was observed in the right mandibular lymph node (Fig. 1C).
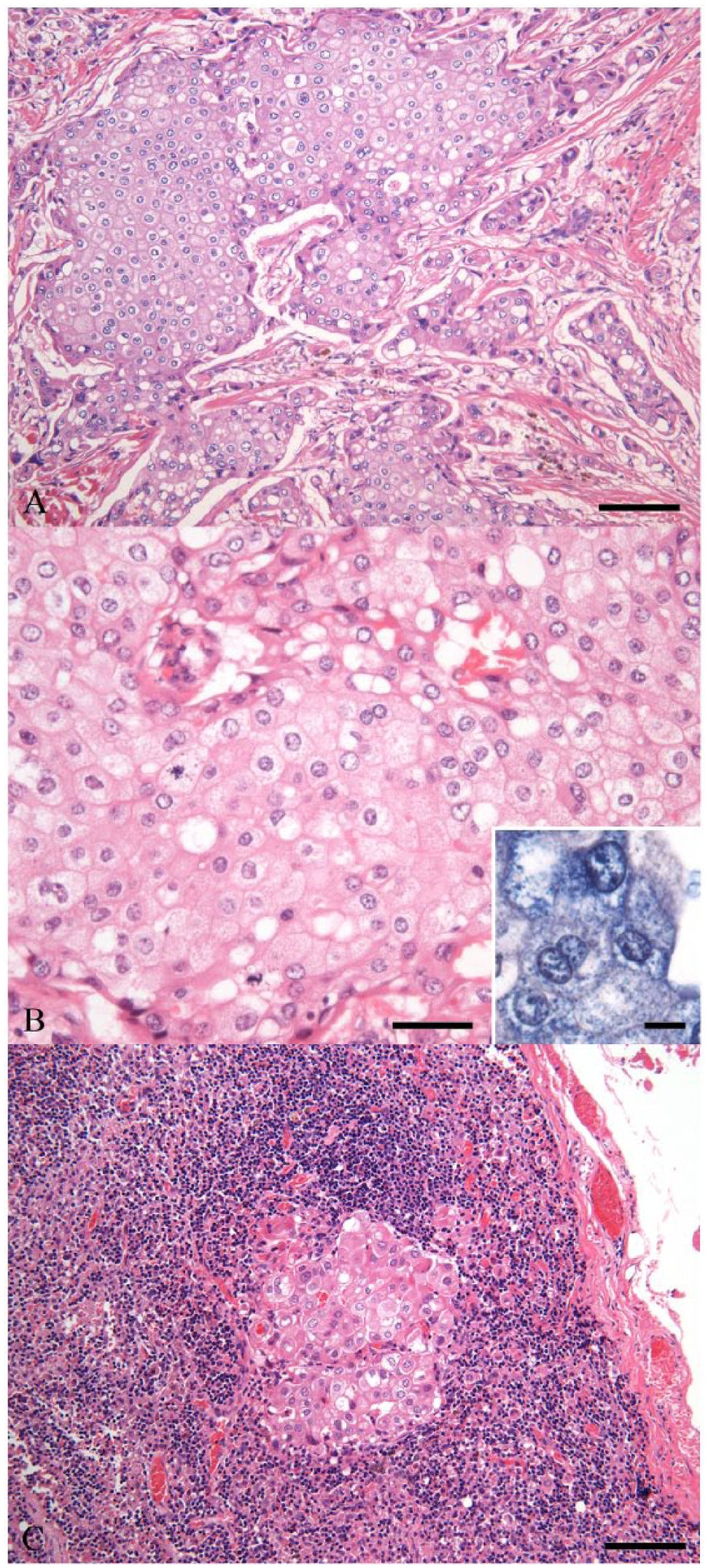
Histologic features of a canine submandibular oncocytic carcinoma. Tumor cells are arranged in solid nests and cords separated by fibrovascular stroma (
Immunohistochemically, most tumor cells were positive for CK AE1/AE3 (Fig. 2A), CAM5.2 (Fig. 2B), E-cadherin, mitochondrial ATPB (Fig. 2C), and S100, but were negative for vimentin, CEA, p63, CK14, SMA, GFAP, CD10, and chromogranin A. The proliferation index of tumor cells was low (7.9%). Immunohistochemical features of metastatic tumor cells in the mandibular lymph node were similar to those of the primary tumor. Ultrastructurally, the cytoplasm of tumor cells contained abundant swollen mitochondria (Fig. 2D). No electron-dense granules or microvilli were observed.
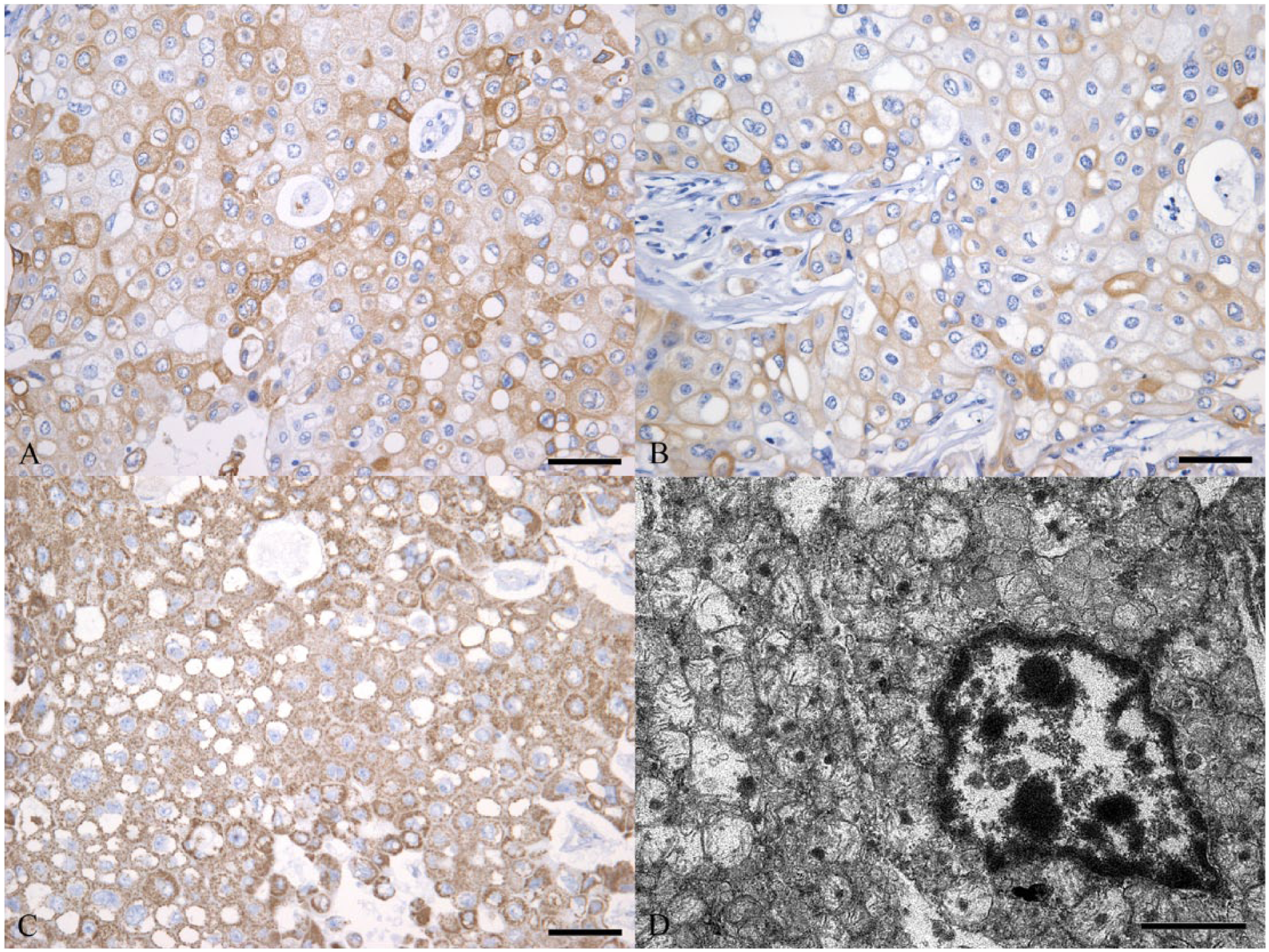
Immunohistochemical and ultrastructural features of a canine submandibular oncocytic carcinoma.
Based on morphologic, immunohistochemical, and ultrastructural findings, the tumor was diagnosed as an oncocytic carcinoma of the submandibular gland, according to the WHO classification of human salivary gland tumors. Oncocytes occur from acinic, myoepithelial, and ductal cells of the normal salivary gland as a result of aging; the cytoplasm of oncocytes contains numerous swollen mitochondria.1,4,11 Proliferative disorders of oncocytes are classified as oncocytosis, oncocytoma, and oncocytic carcinoma.2,8 Conversely, oncocytic metaplasia also occurs in salivary gland carcinomas, including acinic cell carcinoma, mucoepidermoid carcinoma, myoepithelial carcinoma, and basal cell adenocarcinoma. 2 Oncocytic carcinoma may originate from intercalated duct cells. 8 However, the pathogenesis of oncocytic tumors remains unclear. Immunohistochemical analysis using various markers is helpful in identifying oncocytic tumor origin. In dogs, CK14, p63, and SMA are particularly useful myoepithelial cell markers, whereas CK AE1/AE3 and CAM5.2 recognizing CK8 are markers of epithelial and luminal cells in the salivary gland. 6 S100 is expressed by both luminal epithelial and myoepithelial cells in canine salivary gland tumors. 9 Approximately 10% of human acinic cell carcinomas are positive for S100.2,10 The tumor arising in the mandibular gland of this dog had highly atypical oncocytic cells that were positive for CK AE1/AE3, CAM5.2, and S100, but negative for vimentin, CK14, p63, and SMA. These findings suggest that this tumor originated from duct and acinic cells rather than myoepithelial cells.
Primary oncocytic tumors arising in the salivary glands, including mandibular and parotid glands, have not yet been reported in dogs, whereas oncocytomas have been reported in the kidney, larynx, and thyroid gland.3,7,12 The presence of excessive numbers of mitochondria—the hallmark morphologic feature of oncocytes—can be confirmed by ultrastructural observation. In the tumor examined herein, we found abundant mitochondria using PTAH stain, IHC, and ultrastructural examination.
Oncocytic carcinomas in humans are high-grade tumors bearing a poor prognosis given the likelihood of local recurrence and regional or distant metastasis.2,13 In this case, tumor recurrence or metastasis was not observed up to 11 mo after surgery, although metastasis to the regional lymph node was observed at the time of surgical resection. Further studies are required to establish the prognosis for oncocytic carcinoma in dogs.
Footnotes
Acknowledgements
We thank Dr. Hidemi Kitagawa for providing the follow-up information and the tumor specimen for this study.
Authors’ contributions
R Nakahira contributed to design of the study and to analysis and interpretation of data. M Michishita and M Bonkobara contributed to conception and design of the study and to acquisition, analysis, and interpretation of data. M Kato, Y Okuno, H Hatakeyama, and H Yoshimura contributed to design of the study and to analysis of data. D Azakami contributed to design of the study and to interpretation of data. K Ochiai contributed to conception and design of the study and to analysis and interpretation of data. K Takahashi contributed to design of the study and to acquisition, analysis, and interpretation of data. All authors drafted the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Dako Denmark, Glostrup, Denmark.
b.
BD, Franklin Lakes, NJ.
c.
BioGenex Laboratories, San Ramon, CA.
d.
Abcam, Cambridge, MA.
e.
NeoMarkers, Fremont, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by Grants-in-Aid for Scientific Research (26292161 and 24248050 to M. Michishita) from the Ministry of Education, Culture, Sports, Sciences and Technology, Japan (MEXT).
