Abstract
An outbreak of severe cutaneous disease associated with an orthopoxvirus in horses in southern Brazil is described. Fourteen Crioulo mares and foals from a husbandry farm developed papules, and vesicles progressing to proliferative and exudative lesions on the muzzle, external nares, and external and internal lips. The vesicles eroded, and the proliferative lesions eventually bled and progressed to moist crusts and scars. The clinical signs lasted approximately 6–12 days, after which the animals progressively recovered. Direct electron microscopy of skin biopsies revealed brick-shaped, 250–300-nm virus particles with orthopoxvirus morphology. Histological examination of the lesions revealed vacuolar degeneration of the cells of the stratum spinosum and the presence of large intracytoplasmic, eosinophilic inclusion bodies. Polymerase chain reaction amplification of poxvirus A-type inclusion body gene confirmed the presence of orthopoxvirus DNA in horse tissues. Inoculation of tissue homogenates into the chorioallantoic membrane of chicken embryonated eggs and intraperitoneally in mice resulted in pock-like lesions with the microscopic appearance of poxvirus-induced histopathology. Taken together, these results demonstrate the association of an orthopoxvirus with the outbreak of cutaneous disease in horses. The origin of the agent causing the outbreak is uncertain because no similar condition has been reported in Brazil.
Historically, different orthopoxviruses of human and animal origin have been implicated in pox-like cutaneous diseases in horses. 2,5–8,11,12,17 In fact, horses seem to serve as alternative hosts for several human and animal pox viruses, developing different clinical syndromes upon infection with each particular virus. 14,15,19 The definitive identification of the virus species associated with the respective disease, however, has been only rarely achieved. As a consequence, the true identity and epidemiology of many Orthopoxvirus infections of horses, sometimes called horsepox, remain uncertain. 19 Even the origin and natural history of Horsepox virus (HSPV), whose entire genome has been sequenced recently, 20 remain unclear.
Horsepox was reported frequently during the 19th and early 20th centuries, and it became progressively rare to the point of being considered extinct. 14,15,19 Classically, HSPV had been associated with different clinical forms, ranging from benign and restricted lesions in the muzzle and mouth (the buccal form, called contagious pustular stomatitis) to a generalized, highly contagious papulonodular form named viral papular dermatitis. 5,12,15,22 A report of viral papular dermatitis in Thoroughbred horses in California described a highly contagious, acute or subacute disease (10–40 days) characterized by papular swellings followed by scab formation over the skin of several parts of the body. 12 A condition clinically and pathologically similar to horsepox was described in a donkey, which developed proliferative, eruptive lesions on the cutaneous surface of the anterior nares, lips, cheeks, gingiva, and eyelids. 6 An exudative skin clinical form reported as “grease” or “greasy heel” also has been described. 1,4,22 Recently, the whole genome of an orthopoxvirus recovered from Mongolian horses suffering from severe pox pustular dermatitis (especially foals and mares), thereby named Horsepox virus, was sequenced and compared to other known orthopoxviruses. 20 Although sequence analysis revealed a close genetic relationship with known Vaccinia virus (VACV)-like viruses, it also identified HSPV as unique, potentially ancestral sequences absent in VACV-like viruses. 20
During the worldwide smallpox eradication program in the 20th century, horses were frequently infected with VACV used in human vaccine, mainly in Europe. 15 Horses naturally or experimentally infected with VACV usually develop a transient and self-limiting disease characterized by pox-like lesions (papulopustular) in the mucous membranes of the mouth and in the skin of the lips and nose. 8,15,19 The eradication of smallpox and the discontinuation of human vaccination in most countries were accompanied by a gradual reduction of the number of horse cases, and the disease eventually became very rare. 1,15,19
A mild verrucous or papillomatous, pox-like disease called Uasin Gishu has been repeatedly reported in some African countries for many years. 10,15 Contrasting with viral papular dermatitis, which is very contagious and presents an acute and relatively short clinical course, 12 Uasin Gishu disease has a prolonged clinical course and seems to be far less contagious. 10 Poxviruses recovered from horses affected by Uasin Gishu disease were antigenically related to Cowpox virus (CPXV) and VACV, yet their identity has not been unequivocally determined in most outbreaks. 7,15
Viral particles morphologically similar to the human Molluscum contagiosum virus (MOCV) have also been demonstrated by electron microscopy (EM) in the skin of horses with poxvirus-like disease. 2,11,17 The equine disease associated with MOCV is characterized by the development and persistence (up to 6 years) of elevated and waxy papules and hairless, hyperkeratotic foci (4–20 mm) on the skin of the face, neck, thorax, limbs, axillary and inguinal areas, and external genitalia. 2,11 Based on a few reports, MOCV in horses seems to be an usually mild, slow, progressive, and mildly contagious disease sharing many macroscopic and microscopic features with Uasin Gishu disease. 11,15 Both diseases are usually mild and thus have irrelevant economic impact for the herds. 10,11,17
An outbreak of severe cutaneous disease associated with orthopoxvirus infection in horses in southern Brazil was investigated in the current report. The disease affected 14 Crioulo horses in a husbandry farm located in the county of Pelotas, state of Rio Grande do Sul, Brazil, in February 2008. Pelotas is located in one of the southernmost areas of the country (subtropical humid climate; average annual temperature: 17.6°C). The affected farm is dedicated to horse husbandry, where mares at reproductive age are brought together from different farms. At the time of the disease, 15 nursing mares and their foals were grazing in the same pasture; 14 animals were affected (9 nursing mares, 4 foals, and 1 adult castrated male). No other animal species were present in the same pasture. The owner initially reported that a mare presented scabby lesions on the muzzle, with the clinical course lasting 8–12 days and without any other clinical sign or changes in behavior. Ten other animals kept in the same pasture progressively started to develop the same clinical signs in a relatively short period of time. Upon examination, the animals did not present systemic signs. Animals during the acute phase (1–5 days after the onset of signs) presented intradermal, painful papules (2–3 mm in diameter) in the muzzle, between and surrounding the nares, and in the internal and external face of the lips. In the muzzle, a serous secretion was usually presented that eventually dried up, originating crusts. The lesions frequently showed a proliferative, verrucous aspect, with occasional exudation and bleeding upon manipulation. In some animals, the proliferative and exudative lesions coalesced and occupied the entire muzzle area between the nares, extending upward (Fig. 1). The eventual detachment of such crusts originated ulcerated areas that progressively became discolored. A similar pattern of lesions (vesicles, eruptions) was observed in the internal face of the lips, yet without the presence of serous secretion. A mare presented small vesicles and papules in the udder; her foal had the described lesions in the muzzle.
During the midterm course (5–8 days after onset), the lesions became more scarce and mild, characterized by proliferative crustosus lesions and a few vesicles. The detachment or disappearance of crusts gave place to uncolored areas. At this time, the mouths did not display the vesiculopapular pattern observed during acute disease. The late disease course (>8 days) was characterized by areas of skin dyspigmentation and hyperkeratosis. The duration of the clinical signs was approximately 6–12 days, after which the animals progressively recovered. According to the owner, after the veterinary assistance and the apparent end of the outbreak, 3 other mares grazing in a neighboring pasture of the same farm developed the disease. The overall duration of the disease in the herd was approximately 90 days. During acute disease, the lesions were treated with a local antiseptic (iodo-polyvinyl-pyrrolidone).
Electron microscopy examination of ultrathin sections of skin biopsies collected from 3 horses during acute disease revealed aggregates of brick-shaped, 250–300-nm diameter particles, with a morphology typical of orthopoxviruses in the cell cytoplasm (Fig. 2). More detailed morphology of orthopoxvirus particles could be observed at higher magnification by EM examination of homogenates of scabs and crusts submitted to negative staining (Fig. 2, inset).
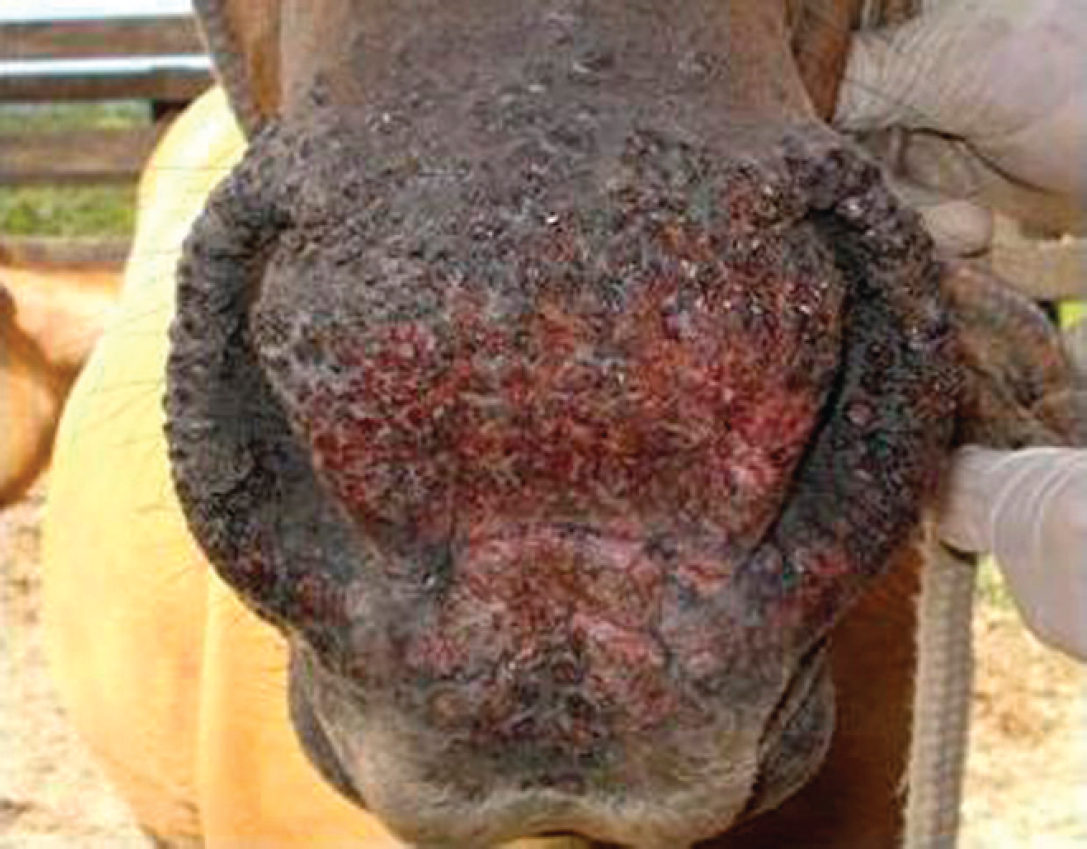
Muzzle of an affected mare during acute disease. Multiple, confluent papules and proliferative lesions in the muzzle, between and surrounding the nares, and extending upward. The external face of the superior lips was also frequently affected.
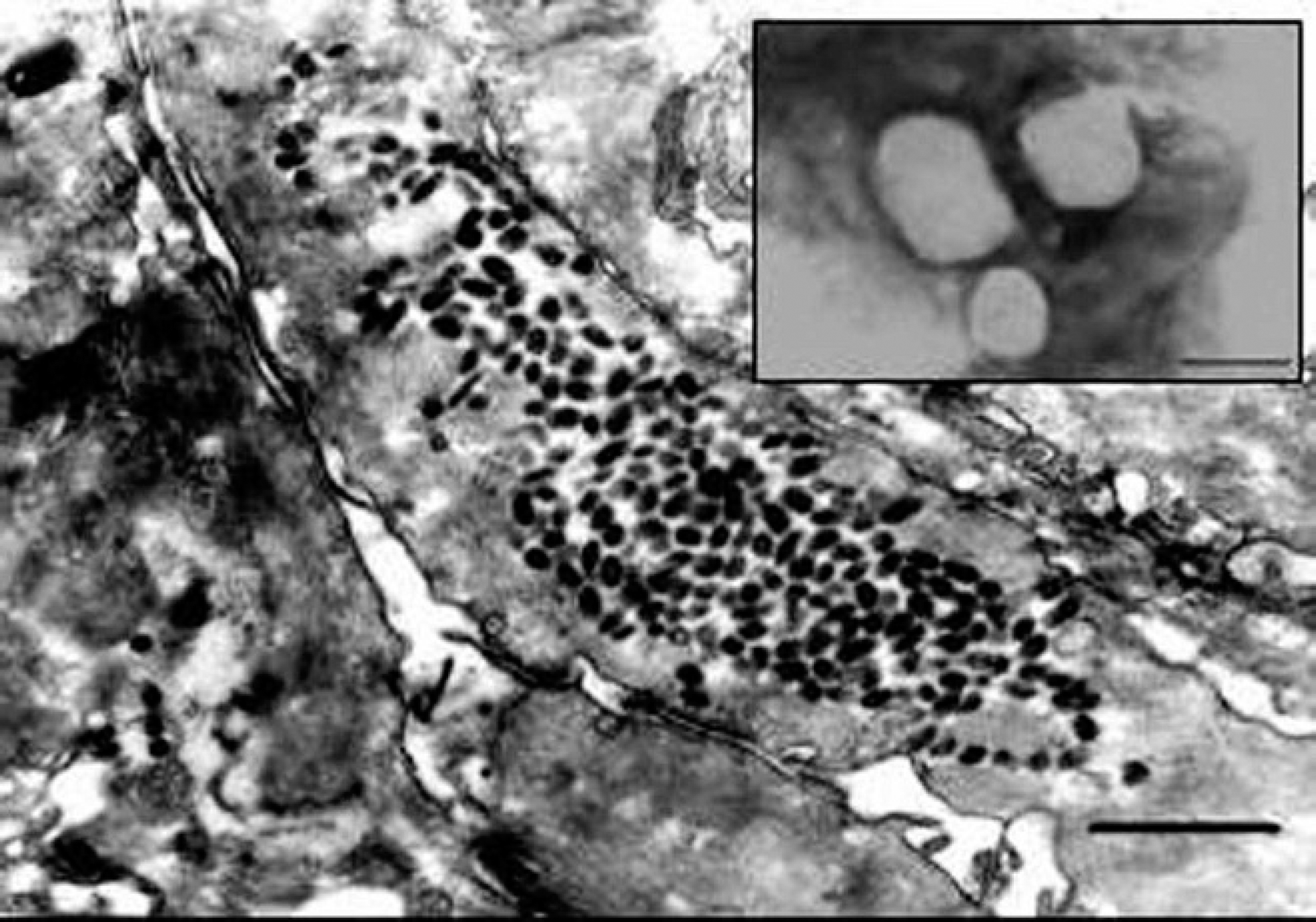
Electron micrograph (EM) of an ultrathin section from an acute lesion in the lip of a mare. A large aggregate of oval or brick-shaped viral particles can be observed in the cell cytoplasm. 27,200x magnification. Bar = 740 nm. Inset: EM examination of homogenates of scabs and crusts upon negative staining: brick-shaped, 250–300-nm viral particles with typical orthopoxvirus morphology. 66,150x magnification. Bar = 300 nm.
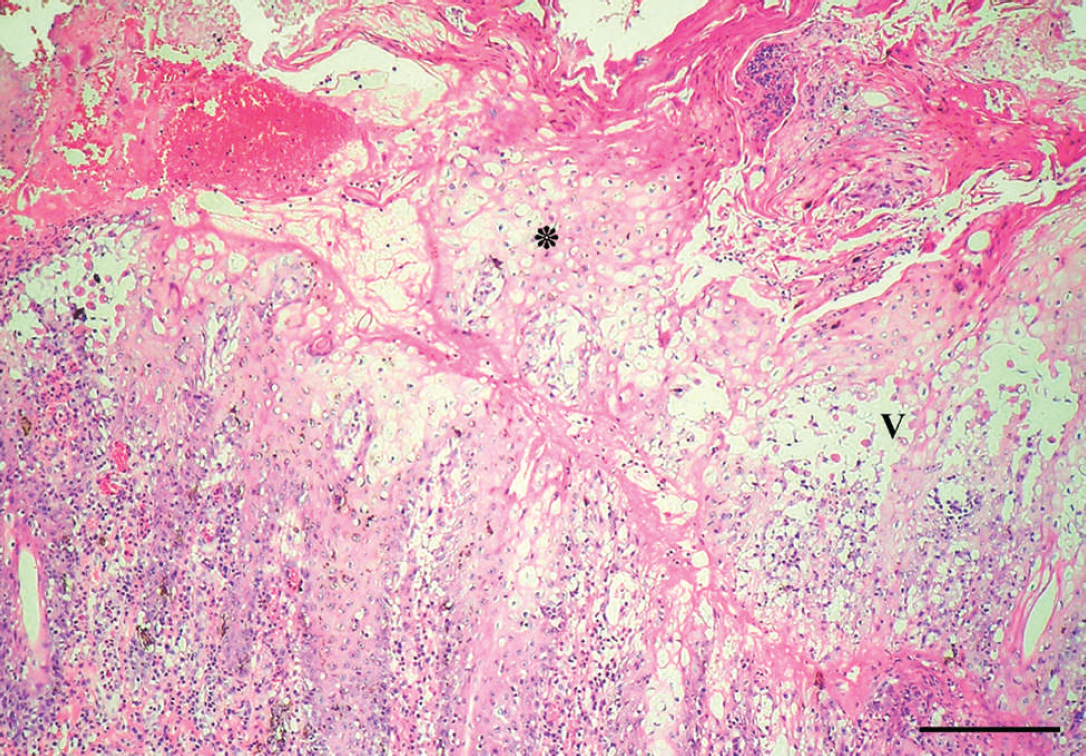
Skin of the muzzle of an affected horse. Marked parakeratosis, ballooning degeneration, and several crusts in the stratum corneum (*). Vesicles and acantholytic cells in the epithelium (V). Moderate dermal inflammatory cell infiltrate of neutrophils and mononuclear cells is seen associated with epithelial damage. Hematoxylin and eosin. Bar = 300 μm.
Histological examination of biopsies obtained from 3 sick horses revealed multiple areas of intense proliferation of the stratum spinosum (acanthosis). Extensive cytoplasmic vacuolation in groups of epithelial cells, parakeratotic hyperkeratosis with necrosis, and keratosis of individual cells were observed in the epithelial layer (Fig. 3). Detachment of the dermal layer and necrosis with lymphocytic and neutrophilic inflammatory infiltrate were observed in some areas. Eosinophilic circular or oval inclusion bodies were observed in the cytoplasm of some vacuolated cells (Fig. 4).
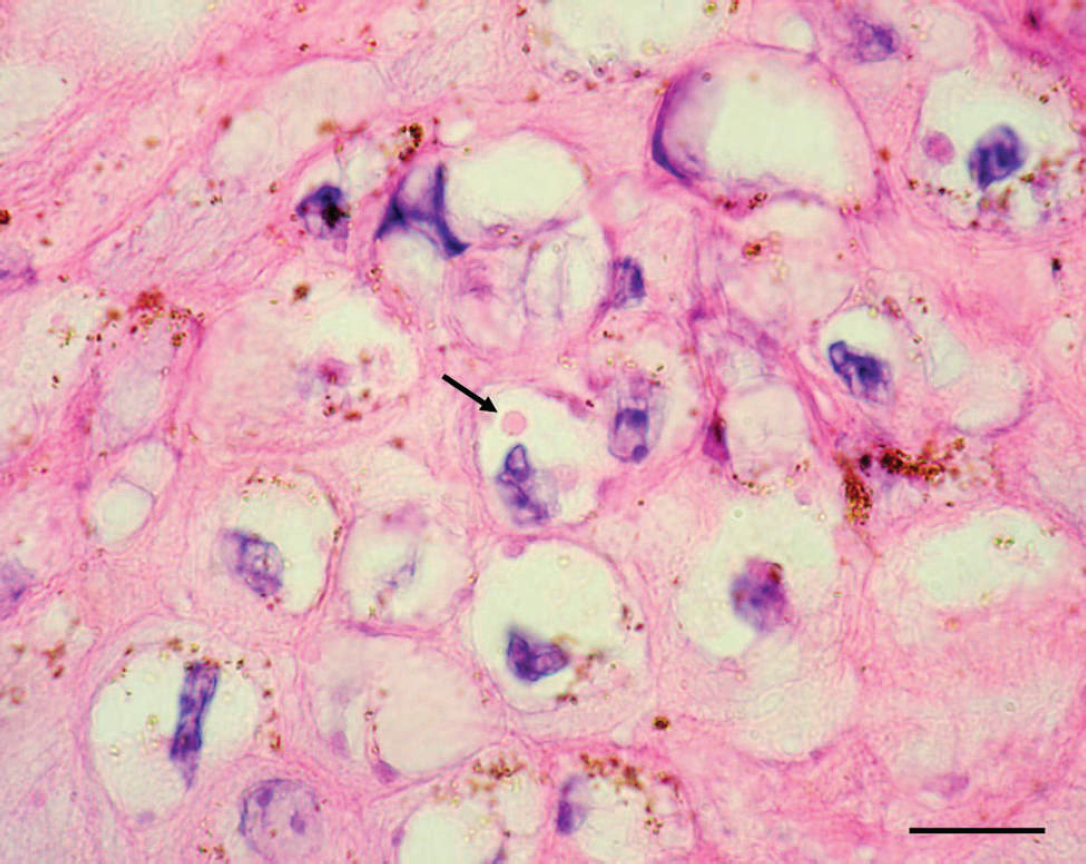
Skin of the muzzle of an affected horse. Eosinophilic, homogeneous, intracytoplasmic inclusion bodies in degenerated keratinocytes (arrows). Hematoxylin and eosin. Bar = 30 μm.
Fragments of a lesion of a sick horse were macerated with sterile sand and minimum essential minimum containing antibiotics and were clarified by centrifugation at 3,000 x g. The supernatant was inoculated into the chorioallantoic membrane (CAM) of 9–12-day-old embryonated chicken eggs. The eggs were incubated at 37°C for 72 hr. At the end of the first passage, multiple white lesions characteristic of pocks and measuring approximately 1–2 mm in diameter were observed in the CAM of the inoculated eggs. The presence of poxvirus-induced lesions was confirmed by histological examination of the CAM. Accentuated vacuolar degeneration of the cytoplasm, eosinophilic intracytoplasmic bodies, and moderate hyperplasia of epithelial cells of the CAM were observed (data not shown).
Inoculation of 6 suckling mice with tissue homogenates was followed by the development of a skin lesion in the abdominal region in 1 animal. The lesion was located adjacent to the inoculation site and had a white, vesicular aspect. Microscopic examination revealed a large vesicle on the epidermis, with accentuated parakeratotic hyperkeratosis, ballooned degeneration, and necrosis of individual cells (data not shown). The vesicle extended to the deepest layer of the dermis, and numerous vacuolated cells were present in the surrounding area. Eosinophilic intracytoplasmic inclusion bodies with circular or oval shape were present in the vacuolated cells (data not shown).
An attempt to identify the virus species associated with the outbreak was performed by using a polymerase chain reaction (PCR) that amplifies a segment of the CPXV acidophilic-type inclusion body (ATI) gene. This PCR has been used to identify and differentiate orthopoxviruses based on the size of the amplicons. 13 Skin fragments of 3 affected horses were submitted to DNA extraction and PCR amplification using a pair of primers (ATI-up 5′-AATACAAGGAGGATCT-3′ and ATI-rev 5′-CTTAA-CTTTTTCTTTCTC-3′) under reaction conditions described previously. 13 Polymerase chain reaction using the ATI primers and the DNA extracted from the lesions as template generated an amplicon of approximately 1,650–1,700 bp (data not shown). The obtained amplicon was similar in size to those obtained by amplification of CPXV (1,673 bp) and VACV (1,603 bp) genomes. 13
Although poxvirus-associated diseases have been described occasionally in horses, 2,6,7,11,12,17,21 the definitive identification of the virus species associated with the respective disease has been only rarely achieved. In fact, many clinical reports of pox disease in horses seemed to be associated with viruses of cattle or of human origin poorly characterized. 2,6,8,11,12,17,21 Even the epidemiology and natural history of HSPV, whose entire genome has been sequenced, 20 is rather unclear. The secular trend of poxvirus-associated diseases in horses reinforces this observation: as human and bovine poxvirus infections were being progressively controlled, and some were eventually eradicated by vaccination, the incidence of poxvirus disease in horses also progressively weaned, and the disease eventually became very rare. 15,19
The most remarkable histopathological findings in the current study were somewhat similar to those observed in several equine conditions associated with different ortho-poxviruses. 2,5,6,9,11,15,17,19,21 The gross findings in the outbreak reported here showed remarkable similarity to what has been described for the buccal form of horse-pox, 5,6,15 with some similarity to the condition reproduced in horses by inoculation of VACV. 18 The papuloeruptive, proliferative, and exudative lesions in the muzzle, surrounding the nares and face, and in the lips and cheeks, plus the presence of large, eosinophilic, intracytoplasmic, oval or circular inclusions in vacuolated keratinocytes are considered pathognomonic of the buccal form of horse-pox. 6 Likewise, the acute and severe clinical course and the highly contagious nature of the disease were more like horsepox (papular dermatitis, estomatitis) 6,12 than Uasin Gishu disease or horse MOCV, which are generally mild, long-lasting, mildly contagious, and chronic diseases. 2,10,11,15,17,21 Differential diagnosis also included Dermatophilus congolensis and dermatophytosis (Microsporum spp., Tricophytum spp.), which were discarded based on clinicopathological and epidemiological data.
According to equine clinicians and experienced Brazilian pathologists, such a clinical condition is extremely rare, or even nonexistent, in Brazil. The observations in the present study corroborate the current concept that poxvirus diseases of horses, especially those called horsepox, are currently very rare. 15,19 The origin of the virus causing the outbreak is merely speculative. Because the affected farm often houses mares coming from different origins, it is reasonable to speculate that the infection might have been introduced by an infected mare.
Beginning in the late 1990s, several episodes of exanthema affecting dairy cattle and milkers have been reported in southeastern Brazil. 3,16 In cows, the clinical features resembled those of cowpox, including pustules on the teats and udder that progressed from vesicles to pustules and healed within 3 weeks, leaving a scar. 3 Biological and genetic analyses showed that the orthopoxviruses associated with these episodes are more like VACV than CPXV, and genome sequencing demonstrated a remarkable homology with a VACV vaccine strain (Cantagalo strain) formerly used in the smallpox eradication program in the country. 3 It has been suggested that such a VACV strain may have persisted in nature in a yet unknown indigenous animal host. 3 Although no cases of equine disease have been reported in the outbreaks described in the current study, the observations serve to illustrate the capacity of these viruses to remain silent in nature for decades and eventually re-emerge as human and/or animal pathogens. To the authors' knowledge, similar clinical conditions in cattle or horses have not been reported to veterinary diagnostic services in central and southern Brazil in recent decades. It should not be discarded that the virus associated with the outbreak might have originated from a domestic or indigenous wild animal.
The definitive identification of poxviruses associated with equine diseases has not been an easy task throughout the history of veterinary medicine. Horses appear to be alternate hosts for a number of poxviruses of human and animal origin, which may cause similar, yet differentiable, clinical signs. In addition to viruses that have been shown to cause disease in horses (e.g., VACV, Uasin Gishu disease, MOCV, CPXV, and HSPV), it is reasonable to speculate that other, yet unidentified or unclassified orthopoxviruses might be occasionally associated with poxvirus disease in horses. In this sense, the definitive identification of the poxvirus reported in the present study may either reinforce the involvement of one of these above-mentioned viruses in pox disease in horses or add a new virus to the list of poxviruses causing equine disease. After the acceptance of the manuscript, additional molecular analyses by partial nucleotide sequencing of the HA gene revealed a remarkably high nucleotide sequence homology (95–100%) with Brazilian VACV isolates.
