Abstract
In October 2008, a 15-year-old female alpaca (Vicugna pacos) housed at a breeding farm in northern California died after a brief illness characterized by sudden onset of weakness, recumbency, and respiratory distress. Postmortem examination revealed severe hydrothorax and hydropericardium, marked pulmonary edema, and acute superficial myocardial hemorrhage affecting the left ventricle. Bluetongue virus (BTV) was detected in the spleen by quantitative real-time reverse transcription polymerase chain reaction and confirmed by sequence analysis. No antibodies against BTV were detected in the serum using a competitive enzyme-linked immunosorbent assay, confirming acute, fulminant BTV infection.
Keywords
Bluetongue (BT) is an insect-transmitted disease of certain species of wild and domestic ruminants that is caused by infection with Bluetongue virus (BTV; family Reoviridae, genus Orbivirus). 1,7,10,12,15 Whereas BTV is transmitted between ruminants throughout the year in tropical regions, infection is distinctly seasonal in temperate zones, where the vast majority of infections occur during the late summer and autumn months. 3 In the Americas, South American camelids have historically been considered to be resistant to BT, 9 although serological surveys have identified apparently subclinical infection of alpacas. 13 Severe disease has been described in numerous species of wild and domestic ruminants during the ongoing pandemic of BTV infection, 6,14 and cases of lethal BTV (serotype 1) in 2 llamas (Lama glama) and an alpaca (Vicugna pacos) have recently been described in Europe. 4,9
In late October 2008, a 15-year-old female alpaca was submitted to the Davis branch of the California Animal Health and Food Safety Laboratory System (School of Veterinary Medicine, University of California, Davis, CA) for postmortem examination. The alpaca was housed at a breeding farm in Northern California and was noted to be clinically normal on the previous day. She presented with a brief clinical history of acute onset of weakness, recumbency, and respiratory distress and died within 6 hr after onset of clinical signs. No other animals within this herd or elsewhere on the property showed any signs of illness.
At necropsy, large volumes of clear, watery fluid were present within the thoracic (1.5 liter) and pericardial (0.2 liter) cavities. The lungs were diffusely enlarged, heavy, wet, and dark red with abundant pink foam within the lumen of the trachea. Multiple coalescing hemorrhages were present beneath the endocardial surface of the left ventricle (Fig. 1). A single focal hemorrhage (approximately 5 cm in diameter) was present in the pancreas. Representative sections from different organs were fixed by immersion in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4 μm, processed routinely, and stained with hematoxylin and eosin. Histologic examination showed diffuse alveolar edema and congestion of the lung. Multifocally, small adjacent groups of alveoli were expanded by numerous alveolar macrophages with a large amount of clear foamy cytoplasm, interpreted as mild multifocal endogenous lipid pneumonia that was likely unrelated to the cause of death. Acute subendocardial hemorrhage was present in the left ventricle, most prominently adjacent to the papillary muscles. The hemorrhage was characterized by small numbers of erythrocytes with occasional neutrophils that dissected between the connective tissue, Purkinje cells, and cardiomyocytes (Fig. 2).
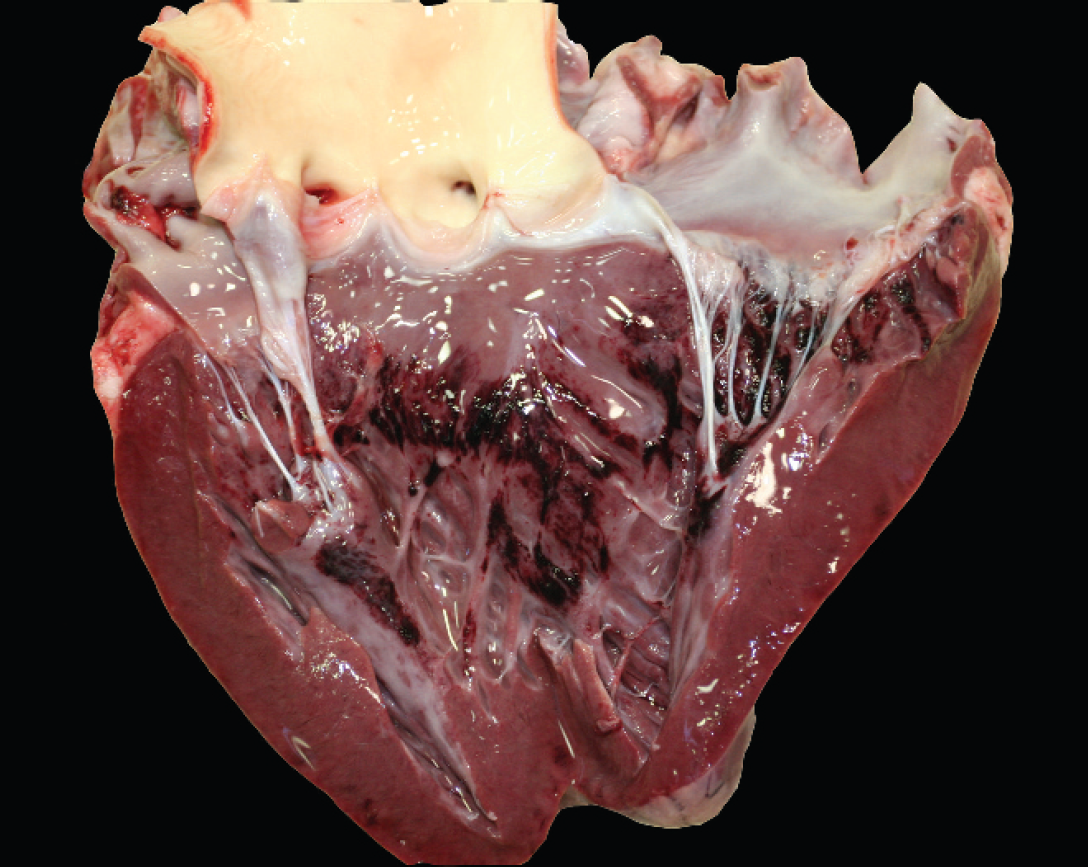
Heart; acute, multifocal to coalescing, subendocardial hemorrhage of the left ventricle.
The presence of hydrothorax, hydropericardium, pulmonary edema, and cardiac hemorrhage were all suggestive but clearly not pathognomonic of BT, and there was no evidence of other lesions that are characteristic of BT in sheep, including erosion and ulceration of the oral cavity; esophagus, facial, and submandibular edema; edema of the dorsal neck musculature around the ligamentum nuchae; and subintimal hemorrhages in the pulmonary artery. 8,10,15 Thus, other potential causes of disease were investigated and excluded. Specifically, oleander toxins were not identified in the stomach or intestinal content from the alpaca. 2 No pathogenic organisms were recovered by aerobic culture of the lung and liver, and there were no abnormal concentrations of heavy metals or selenium in liver samples.
Portions of spleen were evaluated by BTV-specific quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR). A 10% tissue suspension of spleen tissue was prepared by adding denaturation solution a and 2.5-mm zirconia/silica beads in a 2-ml tube and homogenized using a bead beater. b Total RNA was extracted from homogenized spleen tissue using magnetic bead technology c according to the manufacturer's recommendation. Viral RNA amplification was performed essentially as previously described 11 with the primer and probe modifications suggested previously, 5 including the conversion of the molecular beacon probe into a TaqMan probe. A pre–reverse transcription heat-denaturation step for 5 min at 95°C was adapted as previously described. 5 An amplified PCR product of 91 bp (threshold cycle [Ct] value of 24.32) was verified via 4% agarose gel electrophoresis. The PCR product was purified d and then submitted to a commercial sequencing facility. e Sequence data were analyzed using commercial software f and compared with published sequences using a BLAST search (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). Analysis revealed a high similarity (98%, 52/53 nucleotides) to BTV. The presence of BTV nucleic acid in the sample was unequivocally confirmed using a second real-time qRT-PCR assay targeting the NS1 gene. 16
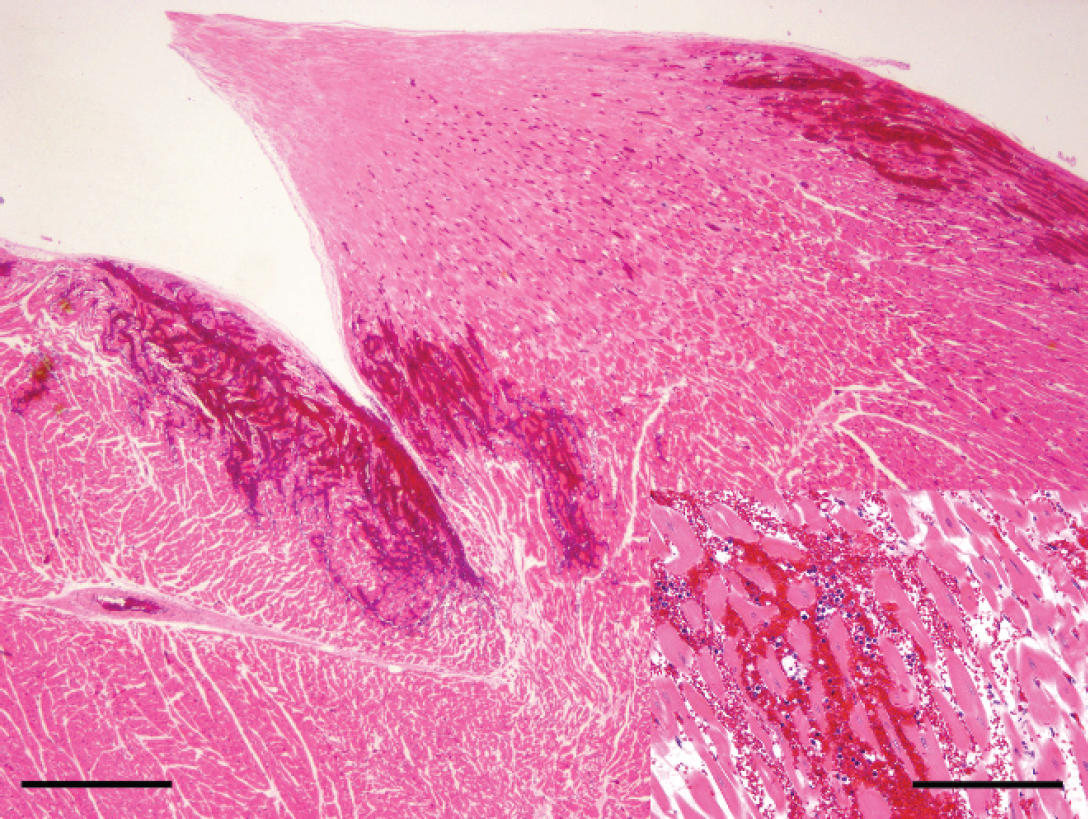
Heart (histologic section of region depicted in Fig. 1); multifocal areas of hemorrhage infiltrate the myocardium at the level of the papillary muscle. Hematoxylin and eosin stain. Bar = 500 μm.
Serum from the alpaca did not contain BTV-specific group antibody, as determined using a competitive enzyme-linked immunosorbent assay (cELISA). The absence of BTV antibodies, coupled with the very strong real-time qRT-PCR value, suggests that this animal died during the acute phase of BTV infection. The animal's contact herd mates were negative for BTV both by real-time qRT-PCR and cELISA; however, other animals on the property tested serologically positive. Although serologic prevalence of BTV has been described in alpacas from Peru, 13 to the authors' knowledge, there is no published information regarding the seroprevalence of BTV infection in the United States, and further epidemiological studies are clearly warranted.
In summary, the sudden onset and severe pulmonary edema, hydrothorax, hydropericardium, and acute myocardial hemorrhage in this alpaca are similar to the described course and lesions of lethal BT in South American camelids affected during the European pandemic. 4,9 The diagnosis of BT in the alpaca is consistent with the enzootic presence of BTV in California, the seasonal nature of the infection, and the simultaneous occurrence of severe BT in sheep in the same general area (unpublished observations). Quantitative real-time RT-PCR followed by sequence analysis confirmed the diagnosis of BTV infection in this animal. To the authors' knowledge, the current case is the first report of lethal BT in a camelid in the Americas.
Footnotes
a.
Ambion®, Applied Biosystems, Foster City, CA.
b.
Mini-Beadbeater, BioSpec Products Inc., Bartlesville, OK.
c.
Ambion® MagMaxTM-96 for microarray assay, Applied Biosystems, Foster City, CA.
d.
Montage® PCR Centrifugal Filter Devices, Millipore Corp., Billerica, MA.
e.
Davis Sequencing, Davis, CA.
f.
Chromas version 2.33, Copyright © 2003–2008 Technelysium Pty Ltd., Tewantin, Queensland, Australia.
