Abstract
In the dog, early-stage epitheliotropic T-cell lymphoma (ETCL) can clinically and histologically mimic a large range of inflammatory dermatoses and often progresses rapidly to a more aggressive tumor stage. Early diagnosis of ETCL is essential to proceed with a specific oncologic therapy that is favorable for the prognosis. In the present study, an improved method for the detection of T-cell receptor gamma (TCRγ) rearrangement was developed by designing a new set of consensus primers to amplify the different forms of rearranged canine TCRγ gene sequences by polymerase chain reaction. The amplicons were analyzed by conventional polyacrylamide gel electrophoresis, which requires minimal specific equipment and may be performed in almost every pathology laboratory at low costs. The method proved to be highly specific and sensitive to detect early ETCL in formalin-fixed, paraffin-embedded biopsy specimens, providing an efficient tool for veterinary pathologists to distinguish early neoplastic from reactive cutaneous T-cell infiltrates (tumor-specific marker) or to discriminate T-cell lymphoma from B-cell lymphomas or nonlymphoid neoplasms (T-cell lineage marker). By direct sequencing analysis of amplified TCRγ gene sequences, ETCL was found to rearrange exclusively the joining (J) 4 region, which suggests specific biology for primary cutaneous T-cell lymphomas. Also, a novel (seventh) functional J region in the TCRγ gene, localized approximately 2.3 kb upstream of J5, was identified.
Introduction
Canine mycosis fungoides is a cutaneous epitheliotropic T-cell lymphoma (ETCL) that is relatively rare in dogs. 6 Nevertheless, early-stage ETCL is a frequent differential diagnosis by veterinary dermatopathologists. Early lesions of ETCL can be clinically and histologically confused with a wide range of inflammatory skin diseases, among which are interface dermatoses, such as erythema multiforme and cutaneous lupus erythematosus. 6 The neoplastic cells in ETCL are cluster of differentiation (CD)3+ T memory cells. 1,8 Because these cells are admixed with dendritic cells of Langerhans cell lineage and other types of tissue histiocytes/macrophages, 6 the overall composition of cellular infiltrates in ETCL is highly similar to that of interface dermatoses. As a consequence, the immunohistochemical characterization of skin infiltrates is generally not useful to distinguish between neoplastic and nonneoplastic T-cell infiltrates, and the histologic detection of early-stage ETCL remains a big challenge in dermatopathology. 4,15 At present, only molecular biology approaches based on the analysis of T-cell receptor (TCR) genes can help to distinguish reactive T-cell infiltrates from T-cell lymphomas. 4,9,15
In dogs and other mammals, TCR is a complex gene that consists of α and β, or γ and δ chains. The 4 chains are encoded by 4 distinct loci designated TCRα, TCRβ, TCRγ, and TCRδ. 4,15 The gene loci contain multiple segments coding for the variable (V), diversity (D), joining (J), and constant (C) regions of the TCRs. During T-cell development in the thymus, T lymphocytes somatically rearrange their antigen receptor genes, whereas all the other somatic cells keep the germ line (non-rearranged) forms of the genes. Rearrangement results from splicing the large TCR gene loci, followed by the random joining of a V, D, and J segment (the so-called VDJ recombination) as well as random deletions and insertions of nucleotides between the V and J segments. 4 The countless possible combinations of V, D, and J segments is one of the bases of the large diversity in antigen receptor specificities at the surface of mature T cells. It has been estimated that more than 1014 different antigen specificities can be generated by this mechanism. 5 In addition, there is a high diversity of length and sequence in the fragments that join the recombined V and J segments, each form of which is a highly specific genotypic label for mature T lymphocytes. Each form of TCR gene rearrangement can be considered a specific hallmark for a given T lymphocyte or for a given neoplastic T cell and its lineage, which is the basis of the molecular approaches developed to distinguish monoclonal from polyclonal T-cell populations. Because most T cells from both aβ and γδ lineages rearrange TCRγ (but not necessarily TCRα, TCRβ, or TCRδ) at an early stage of their development, the TCRγ gene is the ideal target for the detection of T-cell clonality in human lymphomas. 2,3,11,12 In the dog, the TCRγ locus is well characterized, consisting of 4 V region segments (V1-V1), 6 J region segments (J1-J1), and 4 C region segments (C1-C1). 15
Diagnoses, localizations, and clonality status of 92 analyzed lesions. *
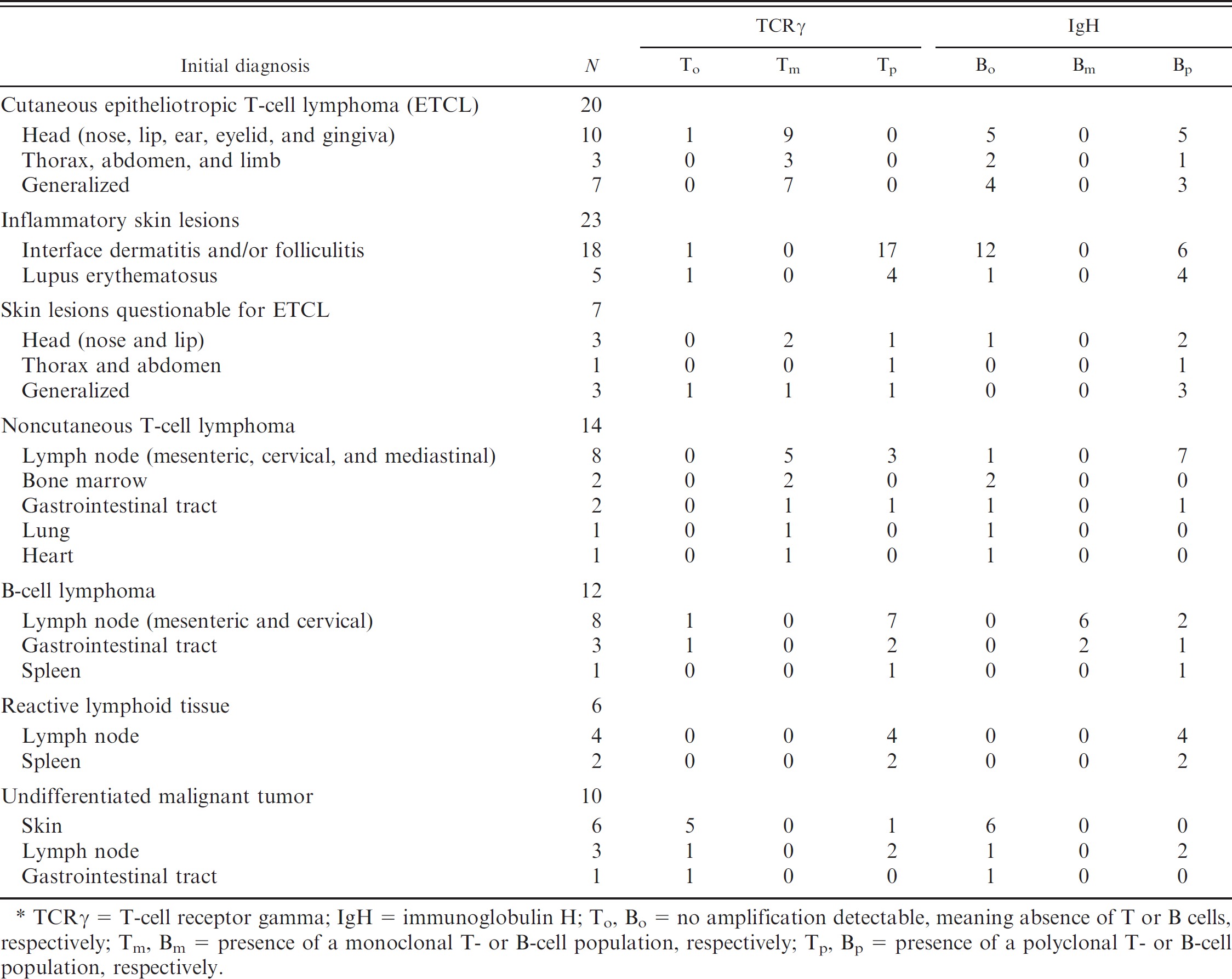
TCRγ = T-cell receptor gamma; IgH = immunoglobulin H; To, Bo = no amplification detectable, meaning absence of T or B cells, respectively; Tm, Bm = presence of a monoclonal T- or B-cell population, respectively; Tp, Bp = presence of a polyclonal T- or B-cell population, respectively.
Clonality assessment of T-cell populations that target the TCRγ gene first used restriction fragment length polymorphism (RFLP) and Southern blotting, 13 which is an efficient but laborious and time-consuming approach. In addition, Southern blotting has 2 major limitations in diagnostic pathology: first, it needs quite large amounts of DNA that cannot be obtained from small biopsy specimens, and, second, it is not suitable for formalin-fixed, paraffin-embedded (FFPE) tissues in which DNA is much too fragmented. An approach much more appropriate for small formalin-fixed biopsy specimens is polymerase chain reaction (PCR). Consensus primers specific for the V and J regions of the TCRγ gene are used to amplify the recombined VJ segments, followed by separation of the PCR products by electrophoresis. Because amplicons are usually shorter than 250 bp, DNA from formalin-fixed tissues can be amplified. Whereas a dominant amplification band reflects the presence of a monoclonal (neoplastic) T-cell population, a smear, in contrast, indicates a polyclonal population. Because of their large size, the germ line (non-rearranged) VJ fragments contained in cells other than T lymphocytes are not amplified and do not interfere with the results.
Primers used to amplify the rearranged T-cell receptor gamma (TCRγ) gene.

In the dog, different sets of consensus primers that recognize conserved parts of the 4 different V regions and the 6 different J regions of TCRγ were reported and tested for the amplification of the rearranged gene from T-cell populations. 4,14,15 The reported approaches seemed to be efficient in distinguishing T-cell neoplasms from inflammatory tissues, but their real usefulness was not evaluated in borderline lesions or early neoplasms. In the present study, new consensus primers were designed for the PCR amplification of the TCRγ gene, and their helpfulness in clinical dermatopathology was tested by analyzing a series of 92 FFPE tissue samples from 20 well-documented cutaneous ETCLs, 7 skin lesions questionable for ETCL (QETCLs), 23 inflammatory skin lesions (ISLs), 6 other reactive tissues, 12 B-cell lymphomas (BCLs), 14 noncutaneous T-cell lymphomas (NCTCLs), and 10 undifferentiated malignant tumors (UMTs).
Materials and methods
Formalin-fixed, paraffin-embedded tissue samples from 92 dogs (47 males and 45 females), aged between 1 and 15 years (mean: 7.6 years), were obtained from the archives of the dermatopathology service of the Institute of Animal Pathology at the University of Berne (Berne, Switzerland). Of the 92 dogs, 55 were mixed breed and 37 pure breed (10 German Shepherd Dogs, 7 Cocker Spaniels, 7 Golden Retrievers, 6 Labrador Retrievers, 3 Huskies, 2 West Highland Terriers, and 2 Airedale Terriers).
The following 50 lesions were selected from electronic archives between 2000 and 2007 by using “mycosis fungoides,” “epitheliotropic,” “interface,” and/or “lupus” as key words: 20 well-documented cutaneous ETCLs, 23 well-documented ISLs, and 7 QETCL skin lesions. Among the 23 ISL cases, 18 cases were reported as interface dermatitis and/or folliculitis (2 of which contained Demodex spp.) and 5 cases as erythematous lupus. In addition, the following 36 lesions were also selected from the electronic archives: 14 well-documented NCTCLs, 12 well-documented BCLs, as well as 10 morphologically and immunohistochemically UMTs. Finally, 6 reactive lymphoid tissue samples (4 lymph node and 2 spleen samples) from necropsies were used as negative controls. The biopsy specimens were reviewed by 2 pathologists by using morphologic and immunohistochemical criteria. The localizations and initial diagnoses are summarized in Table 1.
Tissue preparation
One paraffin block representative of the lesion of interest was selected for each of the 92 selected cases. From each paraffin block, the following serial sections were performed: 1 initial hematoxylin and eosin (HE)-stained section, 4 consecutive unstained 20-μm-thick sections for DNA extraction, 6 consecutive unstained positive charge–coated sections for immunohistochemistry, and 1 final HE-stained section.
Immunohistochemistry and DNA extraction
All tissues were characterized immunohistochemically with a rabbit anti-CD3 polyclonal antibody a diluted 1:20 and a mouse anti-CD20 (clone B-Ly1) monoclonal anti-body a diluted 1:100 by using the avidin–biotin–peroxidase complex method, 3-amino-9-ethylcarbazol as a chromogen, and hematoxylin for counterstaining. DNA was extracted from 4 consecutive 20-μm-thick tissue sections by using a commercial kit, b according to manufacturer's specifications, and eluted in a final volume of 200 μl.
Primer analysis and design
Primers for amplification of the rearranged canine TCRγ gene were reported and used in different studies. 4,7,14 Recently, the genomic organization of the canine TCRγ gene on chromosome 18 was described, and a new set of primers for the amplification of the rearranged TCRγ gene was proposed. 15 BLAST software (http://blast.ncbi.nlm.nih.gov/Blast.cgi) set on the BLASTn algorithm and low stringency parameters (for “somewhat similar sequences”) was used to identify and localize the reported primer sequences in canine genomic and RNA sequences available in GenBank. The degree of consensus of the reported primers was evaluated by using the “align two sequences” module of the BLAST software. The Oligo 4.0 software c was used to design consensus primers that show improved accordance with the newest genomic and RNA TCRγ sequences available from GenBank.
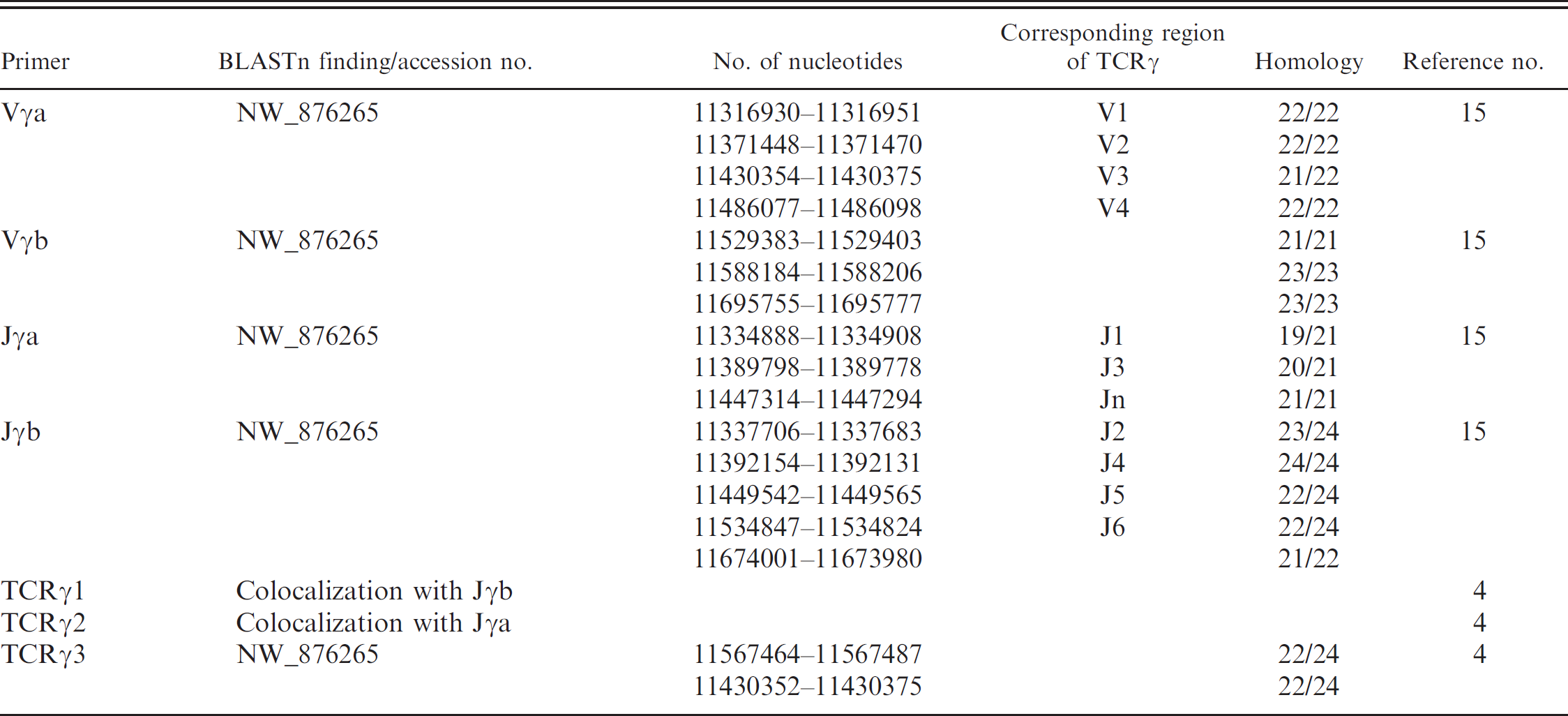
BLAST search of T-cell receptor gamma (TCRγ) primer sequences against genome and “whole genome shotgun” database of the dog.
Polymerase chain reaction
Polymerase chain reaction (volume 30 μl) that contained 2 μl of template DNA, 2 μl of each of the forward and reverse primer at 7.5 pmol/μl, 6 μl of 5 × Q solution, b 3 μl of 10 × PCR buffer, b 1.5 μl of 5 mM of each deoxyribo-nucleotide triphosphate, and 0.5 U of Taq polymerase b were subjected to 35 thermal cycles by using the following profile: 94°C for 30 sec, 55°C for 45 sec, and 73°C for 45 sec. The PCR conditions were the same for TCRγ, immunoglobulin H (IgH), and p16 gene amplifications. Primers designed for the amplification of the rearranged TCRγ gene are described in the Results section and indicated in Table 2. The primers used to amplify the rearranged IgH gene are dIgH-Va, 5′-AGCCTGA GAGCCGAGGACAC-3′ and dIgH-Ja, 5′-TGAGGA GACGGTGACCAGGG-3′. Reactions that lacked template DNA were used as negative controls. As a positive control, a fragment of the highly conserved p16 gene was amplified by using the following specific primers: dcp16-ex2a (5′-CAGGTCATGATGATGGGCAGCG-3′) and dcp16-ex2b (5′-AGCACCACCAGCGTGTCCAGG-3′). All reactions were performed in triplicate.
Agarose gel electrophoresis
The PCR products were admixed with a loading dye that contained xylene cyanol and were separated in standard 2% agarose gels that contained 2 μg/μl ethidium bromide. TCRγ amplicons were run only for a short time. Under these conditions, the amplified products migrate as a single band, in spite of their variable sizes. The thickness of the band roughly reflects the overall amount of amplified copies, providing the overall amount of T cells (or B cells) present in the examined tissue.
Polyacrylamide gel electrophoresis
Given insufficient separation on 2% agarose gels, TCRγ and IgH PCR products were admixed with a loading dye that contained xylene cyanol and bromophenol blue and then were separated in 20% polyacrylamide gels (29:1 acrylamide:bisacrylamide), 0.75-mm thick, by using a vertical electrophoresis unit. d The gels were run for approximately 4 hr at 150 V and then were stained for 20 min in 0.5 μg/ml ethidium bromide.
Interpretation of PCR results
Only tissues from which the p16 gene was amplifiable were retained. DNA samples with unamplifiable p16 were considered to be of insufficient quality. By analyzing the agarose gel first, the TCRγ and IgH PCR reactions were considered positive when a PCR product was identifiable (whatever its size and the number of products). All reactions for which no TCRγ or IgH amplicon was observed on the agarose gel were considered negative (T0 and B0, respectively). The migration pattern on polyacrylamide gel was then considered for the positive cases. Cases for which the TCRγ or IgH PCR product migrated as a smear were considered polyclonal (Tp and Bp, respectively). Cases for which one or several well-defined bands of adequate size were identifiable (with or without a smear as background) were considered monoclonal (Tm and Bm, respectively).
BLAST search of T-cell receptor gamma (TCRγ) primer sequences against reference and nonreference RNA database of the dog. *
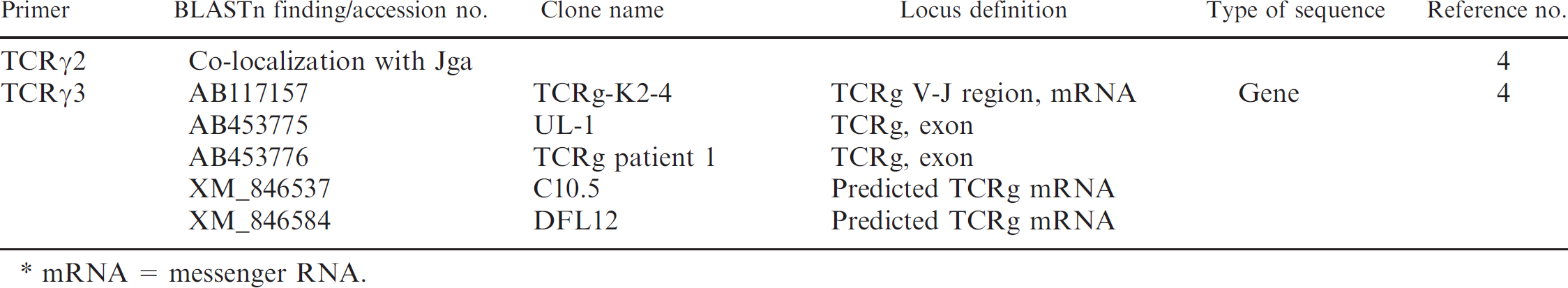
mRNA = messenger RNA.
DNA sequencing
Well-defined single bands identified on the polyacrylamide gels were excised under an ultraviolet lamp by using disposable scalpel blades. DNA was purified from the removed bands by using a DNA purification system e according to the manufacturer's specifications. The purified DNA samples were sent to a DNA sequencing facility f for direct sequencing. The sequencing primers were those used for PCR.
Results
Optimizing consensus primers for the rearranged TCRγ gene
The previously reported TCRγ primer sequences 4,15 were blasted against the “genome” and “whole genome shotgun” GenBank databases from Canis familiaris. Results are summarized in Table 3. The Jγa and Jγb primer sequences 15 were identified in the chromosome 18 contig NW_876265, at positions that corresponded to the J1 and J3 regions and to the J2, J4, J5, and J6 regions, respectively. The TCRγ1 and TCRγ2 primer sequences 4 were found to colocalize (and partially overlap) with Jγb and Jγa primer sequences, respectively. The Vγa primer sequence was also identified in NW_876265, in the V1, V2, V3, and V4 regions. 15 The Vγb primer sequence was found to localize further downstream. The TCRγ3 primer sequence 4 was found to colocalize with the Vγa sequence, but it matched poorly with the V1, V2, V3, and V4 regions from NW_876265. All reported primer sequences 4,15 were also blasted against the “reference RNA” and “nonreference RNA” canine GenBank databases (Table 4). The Vγa and Jγb primer sequences were both identified in 26 messenger RNA (mRNA) sequences from GenBank. In contrast, the Jγa, Vγb, and TCRγ3 primer sequences could be identified only in a few reported mRNA clones. The TCRγ1 and TCRγ2 sequences were confirmed to colocalize (and partially overlap) with Jγb and Jγa primer sequences, respectively.
Given the above observations, the decision to concentrate on the Vγa, Jγa, and Jγb primers was made. 15 By examining the 31 different mRNA clones in which the Vγa, Jγa, and/or Jγb primer sequences may be identified, one or several of the last 3 nucleotides at the 3′-end of the Vγa and Jγb primer sequences were often found to be wrong. Similarly, there was frequently no consensus in the first 3 nucleotides at the 5′-end of the Jγa primer sequence. Therefore, the Vγa and Jγb primer sequences were switched 3 positions toward their 5′-end and the Jγa primer sequence was switched toward its 3′-end, to make them more consensual. The newly designed primers, designated dTCRγ-Va, dTCRγ-Ja, and dTCRγ-Jb, are indicated in Table 2.
Identifying a new J region in the canine TCRγ gene
By blasting the dTCRγ-Ja primer sequence against the canine genome, an unexpected location was identified approximately 17 kb downstream of V3 and 2.3 kb upstream of J5, in a 600-bp region, and showed more than 80% homology with the reported J1 and J3 regions. 15 The region was called Jn (for J new). The organization of the canine TCRγ locus is illustrated in Figure 1.
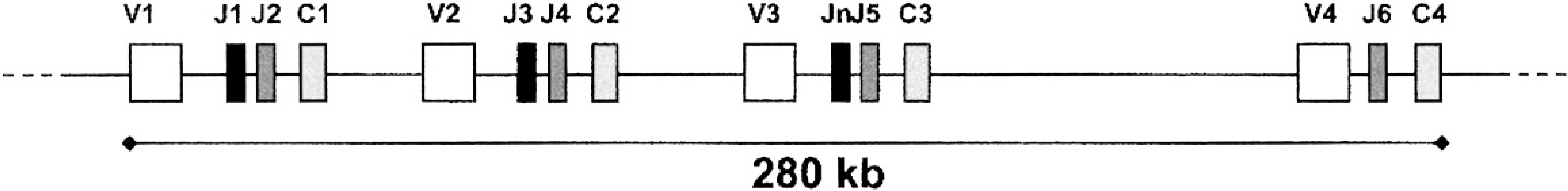
Organization of the canine T-cell receptor gamma (TCRγ) locus. According to the chromosome 18 sequence from GenBank (NW_876265), the gene consists of 4 variable regions (V1-V1), the 6 joining regions described previously (J1-J1), 15 an additional joining region newly identified in the current study (Jn) situated between the V3 and J5 regions, and 4 constant regions (C1-C1).
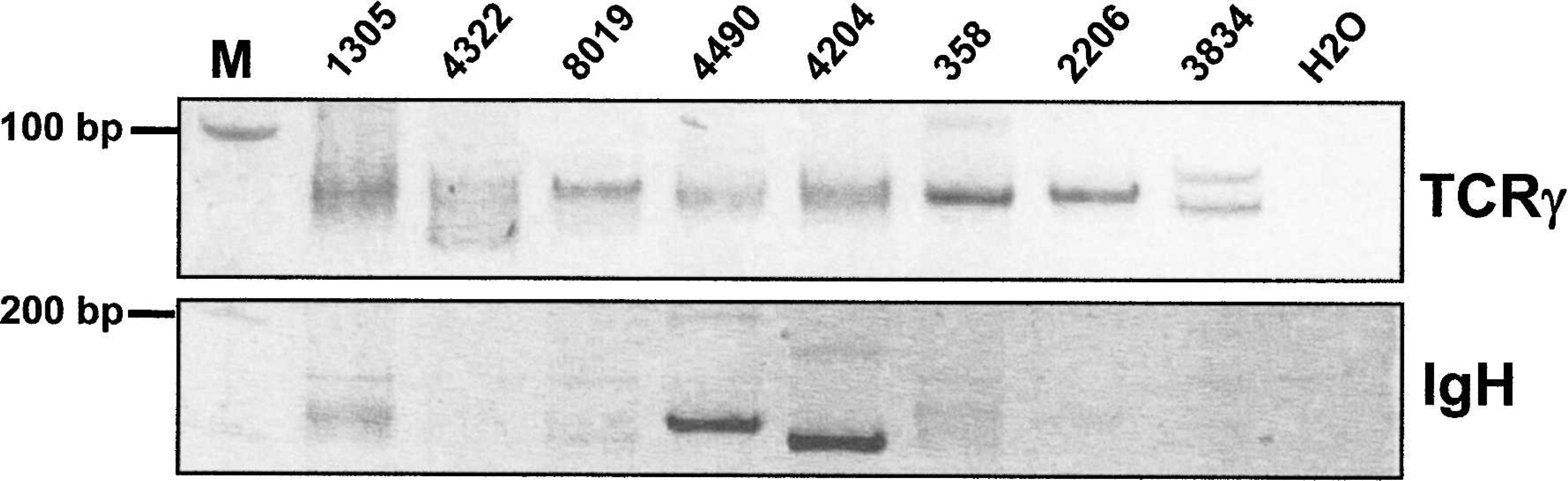
Analysis of the rearranged T-cell receptor gamma (TCRγ) and immunoglobulin H (IgH) genes. DNA extracted from formalin-fixed, paraffin-embedded tissue samples was amplified by polymerase chain reaction by using primers specific for the canine TCRγ locus (dTCRγ-Va and dTCRγ-Jb), as well as primers specific for the canine IgH gene (dIgH-Va and dIgH-Vb), and loaded onto a 20% polyacrylamide gel. The analyses of 2 B-cell lymphomas (nos. 4490 and 4204), 3 epitheliotropic T-cell lymphomas of the skin (nos. 358, 2206, 3834), 2 inflammatory lymphocytic skin infiltrates (nos. 1305, 4322), and 1 skin lesion questionable for epitheliotropic T-cell lymphoma (no. 8019) are illustrated. Well-defined bands reflect monoclonal TCRγ gene rearrangement in case nos. 358, 2206, and 3834, and monoclonal IgH gene rearrangement in case nos. 4490 and 4204. Smears indicative of polyclonal TCRγ and IgH gene rearrangements are observed in case nos. 1305 and 4322. In the TCRγ analysis of case no. 8019, a band can be distinguished over a smear, which indicates the presence of a monoclonal T-cell population. In addition to the monoclonal neoplastic B-cell population of the 2 B-cell lymphomas (nos. 4490 and 4204), the smear in the TCRγ blot allude to the concurrent polyclonal inflammatory T-cell infiltrate. M = 100-bp ladder; H20 = no template DNA control.
Analyzing the clonality of cutaneous T-cell infiltrates
Fifty lymphocytic infiltrates of the skin were analyzed for TCRγ and IgH gene rearrangement: 20 well-documented cutaneous ETCLs, 23 well-documented ISLs (18 interface dermatitis and/or folliculitis and 5 lupus dermatitis), and 7 QETCLs. A monoclonal rearrangement of the TCRγ gene was observed in 19 of the 20 cutaneous ETCLs (95.0%) (Table 1). The only ETCL case in which there was no identifiable monoclonal rearrangement was presumably a skin infiltrate from a generalized lymphocytic leukemia rather than a primary ETCL. In contrast, there was no evidence of monoclonal rearrangement of the TCRγ and IgH genes in the 23 ISL. A monoclonal rearrangement of TCRγ was detected in 3 of the 7 QETCLs. In 2 of them, the diagnosis of ETCL was confirmed by the clinical follow-up and by novel skin biopsies performed 18 and 25 months later. Two of the 4 dogs with QETCL that lacked monoclonal rearrangement of TCRγ were in good health with no skin lesions 32 and 55 months after biopsy, respectively, which confirmed the absence of ETCL. Polymerase chain reaction analyses and histopathologic features of some neoplastic and inflammatory T-cell infiltrates are illustrated in Figures 2, 3, and 4.
Determining the B- or T-lineage of neoplasms
Thirty-six malignant tumors were analyzed for TCRγ and IgH gene rearrangement: 14 well-documented NCTCLs, 12 well-documented BCLs, and 10 UMTs. A monoclonal rearrangement of the TCRγ gene was identified in 10 of the 14 NCTCLs (71.4%). A monoclonal rearrangement of the IgH gene was present in 8 of the 12 BCLs (66.6%; Table 1). Finally, a monoclonal TCRγ rearrangement was observed in 2 of the 10 UMTs. Both tumors were reclassified as T-cell lymphomas. Examples of B- and T-cell lymphoma analyses are illustrated in Figure 2.
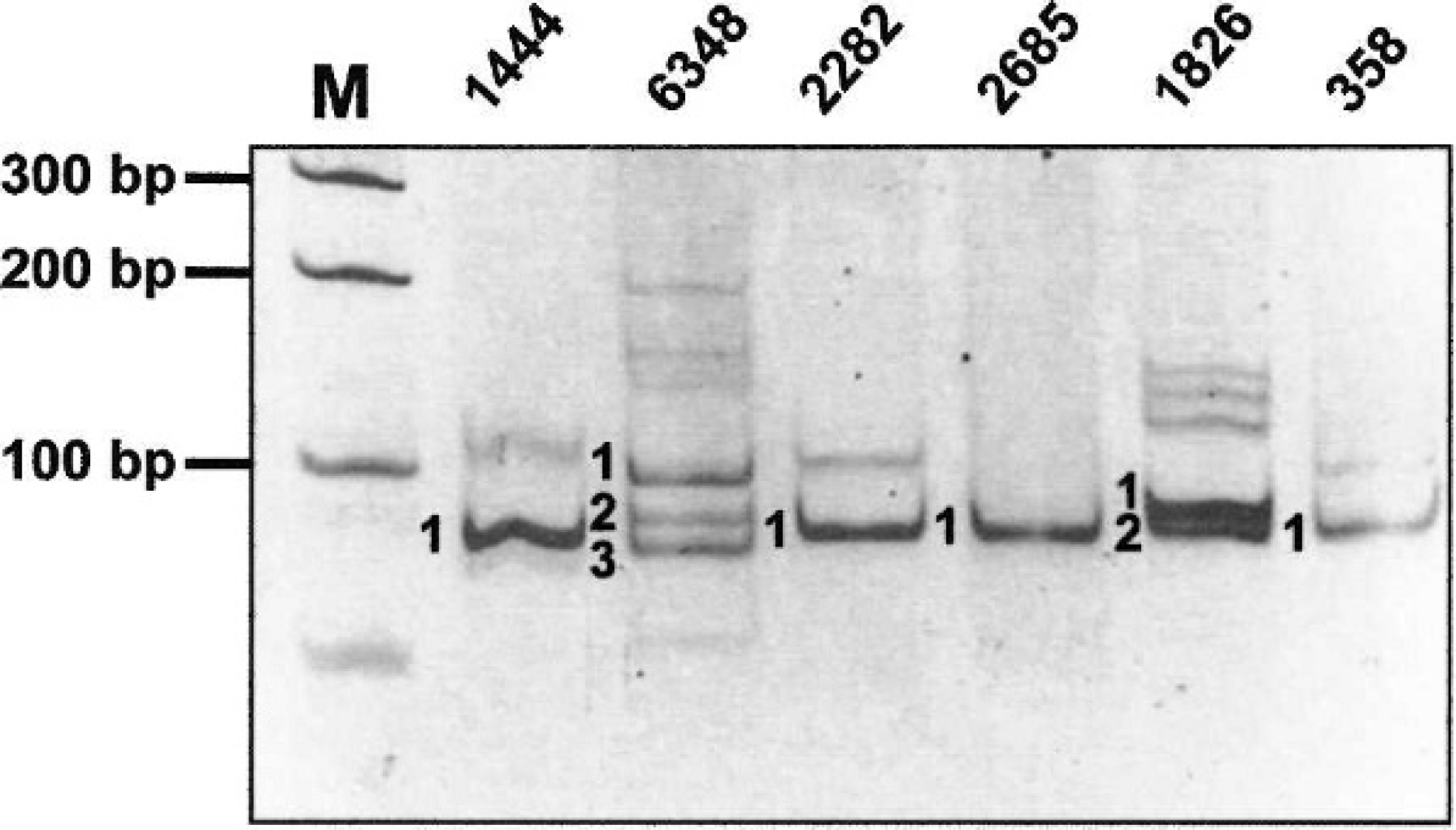
Analysis of the rearranged T-cell receptor gamma (TCRγ) gene in 3 cutaneous epitheliotropic T-cell lymphomas and 3 noncutaneous T-cell lymphomas. DNA extracted from formalin-fixed, paraffin-embedded biopsy specimens was amplified by polymerase chain reaction by using the dTCRγ-Va and dTCRγ-Jb primers, then separated on a 20% polyacrylamide gel. Monoclonal rearrangement of the TCRγ gene is demonstrated by the presence of 1 (nos. 1444, 2282, 2685, and 358), 2 (no. 1826), or 3 (no. 6348) well-defined bands. These bands were excised from the gel, and their DNA was re-amplified and sequenced. Results of DNA sequencing are illustrated in Figure 5. M = 100-bp ladder.
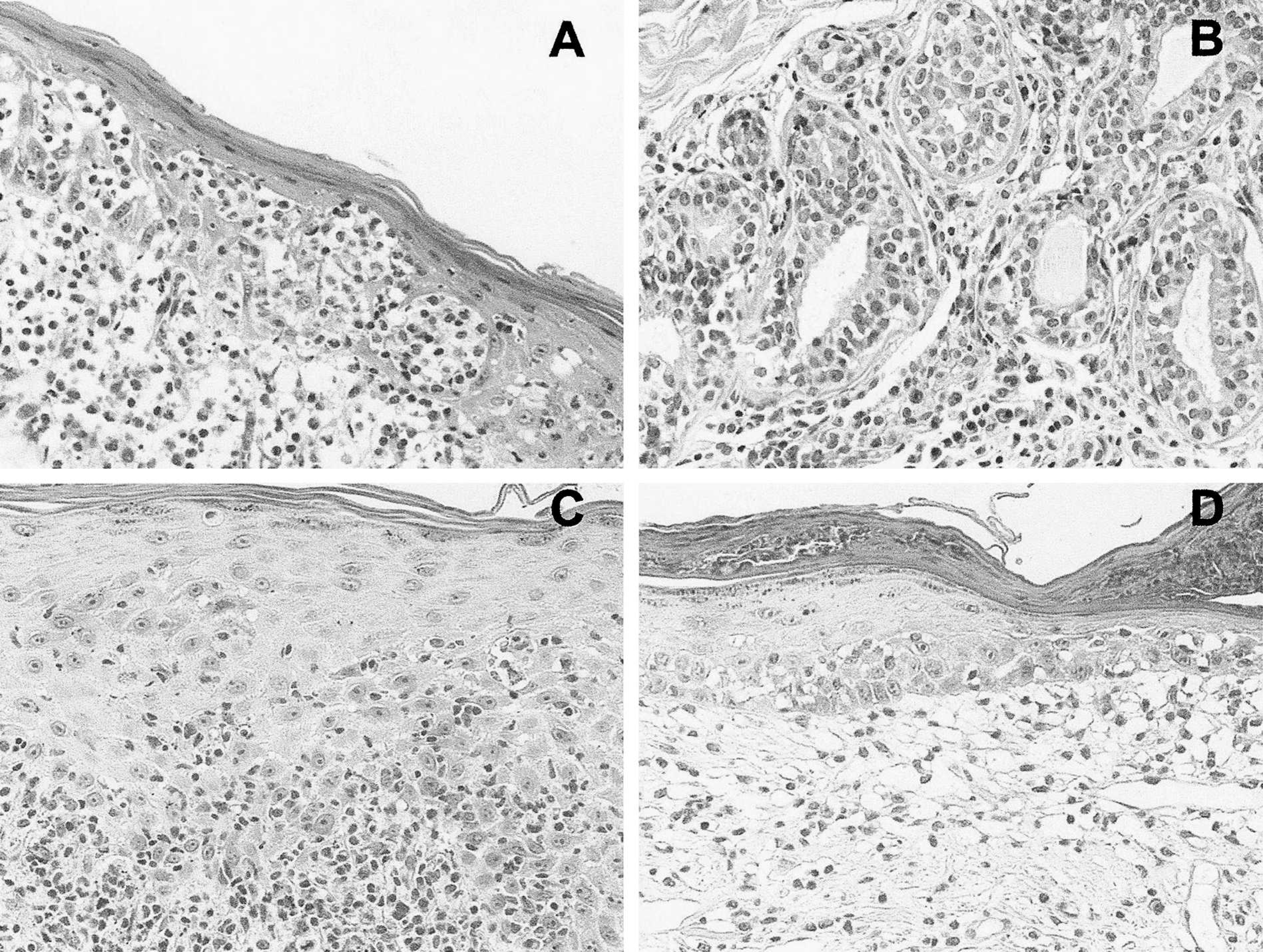
Histopathologic pattern of neoplastic and nonneoplastic T-cell infiltrates of the skin.
Determining the rearrangement profile of TCRγ in ETCL
To determine the rearrangement profile of the TCRγ gene present in ETCL, a total of 20 well-defined bands from 11 cutaneous ETCL and 3 NCTCL cases were excised from the polyacrylamide gels, reamplified by PCR, and subjected to direct DNA sequencing. Results are presented in Figure 5. The fragment length was situated between 57 and 89 bp. Up to 3 different rearrangement profiles were observed in the same tumor, which presumably corresponded to distinct allele patterns. Fifteen of the 16 bands isolated from cutaneous ETCL and 2 bands isolated from a NCTCL corresponded to Vx-J4 rearrangements. One band isolated from a cutaneous ETCL as well as the 2 remaining bands from NCTCL cases corresponded to Vx-J2 rearrangements (Fig. 5).
Discussion
In veterinary pathology, as in human pathology, it is difficult to distinguish between neoplastic and reactive lymphocytic infiltrates in the skin. Early-stage ETCL can mimic, clinically and histologically, a large range of inflammatory dermatoses. Because ETCL can progress rapidly to aggressive tumors, early diagnosis is essential. Monoclonality is a hallmark of neoplasia and probably the only tumor-specific marker available to date. Looking for the clonality status of a cell population, therefore, remains the most reliable test to distinguish neoplasia from hyperplasia or inflammatory infiltrates. This is particularly true for lymphoid cell populations, in which monoclonality may be demonstrated at the genetic level, because during maturation, B-lymphocyte precursors rearrange their immunoglobulin and T lymphocytes rearrange their receptor (TCR) genes. In this context, 2 studies described PCR-based approaches for the amplification of the rearranged TCRγ gene in dogs. 4,15 The methods may help to distinguish between neoplastic and inflammatory T-cell populations. They use consensus primers for the highly conserved V and J regions that should ideally recognize all forms of rearranged TCRγ gene.

T-cell receptor gamma (TCRγ) rearrangement profiles analyzed by DNA sequencing. Migration bands from 12 cutaneous epitheliotropic T-cell lymphomas (ETCL) and 2 noncutaneous T-cell lymphomas (NCTCL), including some of the cases illustrated in Figure 3, were excised from the polyacrylamide gels. Subsequently, the corresponding DNA was re-amplified by polymerase chain reaction (PCR) by using the dTCRγ-Va and dTCRγ-Jb primers, and then was purified and subjected to direct DNA sequencing. The sequencing primers were the same as for PCR amplification (sequences in bold). Sizes of the PCR products ranged from 57 to 89 bp. The 4 nucleotides immediately consecutive to the dTCRγ-Jb primer sequence are constant (CAAC, read from right to left). According to the sequence that follows the CAAC segment, 3 distinct patterns (read from right to left) can be distinguished: 1) the ATGGTCGGTCTGGT pattern (double underline) present in the J4 region of chromosome 18 sequence NW_876265, 2) the ATGGTCATTCTGGT pattern (single underline), which also corresponds to the J4 sequence but contains a GG to AT polymorphism, and 3) the GAGGTTGATGAG pattern (shaded), which corresponds to the J2 region.
The primary objective of the present work was to adapt the 2 reported methods to dermatopathology, with the hope of distinguishing early ETCL from nonneoplastic lymphocytic infiltrates in FFPE skin biopsy specimens from the dog. In this perspective, the 2 approaches were first thoroughly analyzed and critically compared. By comparing the primer sequences proposed by the 2 studies to the most recent data from GenBank, it seemed that the described sequences might be suboptimal in terms of sequence consensus. As a consequence, one of the reported primer sets, therefore, was slightly modified to improve the consensual degree (see the section “Optimizing consensus primers for the rearranged TCRγ gene” in the Results section). In addition, the PCR protocol was adapted to enable amplification from DNA isolated from FFPE tissues. The improved and adapted methodology was then tested in 56 neoplastic lesions, 29 nonneoplastic lesions, as well as in 7 skin lesions questionable for T-cell lymphoma, with the aim to evaluate its specificity and sensitivity in daily life conditions. Used as a marker of T-cell lineage, the method demonstrated in the present study showed a sensitivity of 71.4% for noncutaneous lymphomas, reaching 95.0% in epitheliotropic ETCL of the skin. Used as a tumor-specific marker, the method's sensitivity reached 95.0% and its specificity reached 100%. Given its high sensitivity and specificity in the discrimination between neoplastic and reactive cutaneous T-cell infiltrates in paraffin-embedded skin biopsy specimens, the method should be of interest to most dermatopathology laboratories. The described method is based on the electrophoretic separation of very short PCR fragments, the length of which varies between 57 and 89 bp. As a consequence, the method requires highly efficient polyacrylamide gel electrophoresis and cannot be achieved with simple agarose gels. This slightly prolongs the procedure. This requirement, however, should not dissuade pathologists interested in introducing the method in their laboratories, because it remains compatible with general laboratory equipment. Contrary to other approaches, the described method does not require special expensive equipment, such as capillary gel electrophoresis 7 or microchip gel electrophoresis, 10 and the investment, therefore, remains reasonable.
By direct DNA sequencing, it was found that ETCL contained almost exclusively Vx-J4 rearrangements, which corroborates previous observations 15 that T cells from normal skin samples are either V1-J4 or V2-J4. The only case of the series that was initially diagnosed as ETCL but contained a rearrangement profile other than Vx-J4 (case no. 2282, Vx-J2) was actually a generalized T-cell lymphoma with a leukemia component. From the authors' point of view, this case represents a primarily noncutaneous lymphoma with secondary skin involvement rather than true cutaneous ETCL. Taken together, observations of the current study and from other laboratories 15 indicate that ETCL most likely originates from T cells that are normally present in the quiescent or reactive skin. As their normal counterpart, the neoplastic T cells from cutaneous ETCL seem to have a particular biology, quite distinct from that of other T-cell lymphomas. There was one case in the current study that was initially diagnosed as ETCL but had no detectable TCRγ rearrangement. This case was morphologically and immunohistochemically indistinguishable from the other ETCL cases. The absence of detectable monoclonality might be caused by the presence of a rearrangement profile that is not detectable by the authors' PCR (presumably not involving J4), but a more probable explanation could be a deletion that involved all alleles of the rearranged TCRγ locus.
By using the BLAST program to verify the localization of the dTCRγ-Ja primer sequence within the canine genome, an unexpected sequence was found approximately 17 kb downstream of V3 and 2.3 kb upstream of J5, situated in a 600-bp region highly homologous to the J1 and J2 regions. 15 The region corresponds to a Jn, which may probably recombine with V regions during TCRγ gene rearrangement. Given that the sequence of the canine chromosome 18 is not fully ascertained thus far, one can expect, in the near future, the identification of further V or J regions and the discovery of a more complex structure and organization for the TCRγ locus. In summary, it can be concluded that the method developed in the current study for the detection of TCRγ rearrangements by PCR can provide excellent results when distinguishing cutaneous ETCL from nonneoplastic skin lesions and, thus, can be recommended to every dermatopathology laboratory.
Footnotes
a.
Dako Denmark A/S, Glostrup, Denmark.
b.
DNeasy® Blood and Tissue kit, Qiagen Inc., Valencia, CA.
c.
Molecular Biology Insights Inc., Cascade, CO.
d.
Hoefer™ SE 250, Hoefer Inc., San Francisco, CA.
e.
Wizard® PCR Preps, Promega Corp., Madison, WI.
f.
Microsynth AG, Balgach, Switzerland.
