Abstract
Five cases of postparturient vulvovaginitis and metritis in cattle caused by Clostridium septicum (malignant edema) are described in the current report. The diagnosis was established based on detection of C. septicum by culture and fluorescent antibody test. All animals were Holsteins, and 4 were primiparous (the parity of 1 animal was not reported). All animals developed clinical signs 1–3 days after calving, consisting of swelling of perineal and perivulvar areas, fever, and depression. Perineal, perivulvar, and perivaginal gelatinous and often hemorrhagic edema was consistently observed on gross examination. Longitudinal vulvar, vaginal, cervical, and uterine body tears, covered by fibrinous exudates, were also present. Microscopically, vulvar, vaginal, and uterine mucosae were multifocally necrotic and ulcerated. Large Gram-positive rods, some with subterminal spores, were present within the edematous subcutaneous and submucosal tissues. Clostridium septicum was demonstrated by culture and/or fluorescent antibody test in tissues of most animals. These cases of malignant edema were considered to be produced by C. septicum and predisposed by the trauma occurring during parturition.
Malignant edema and Blackleg are diseases of cattle, sheep, and other species, and they are usually referred to generically as clostridial myositis. Malignant edema is a condition that occurs in cattle, sheep, horses, and other species and is considered to be “exogenous” because microorganisms from the environment gain access to the tissues after skin or mucosal wounds and the development of an anaerobic environment. Blackleg, in contrast, is caused by Clostridium chauvoei alone; it is most often described in cattle, 6,7,11,14 and it is considered to be an “endogenous” disease because latent C. chauvoei spores in the muscle germinate when an anaerobic environment develops in the tissues, usually as a consequence of trauma.
Malignant edema is caused by one or more pathogenic clostridia, including Clostridium septicum, C. chauvoei, Clostridium novyi, Clostridium sordellii, and Clostridium perfringens. Types of trauma that may predispose an animal to develop malignant edema include, but are not restricted to, intramuscular injections, parturition, shearing, castration, and tail docking. 2,6,13
A special form of malignant edema characterized by vulvovaginitis and occasionally metritis developing after parturition has been reported in cattle. 19 However, despite the long history of malignant edema in the veterinary literature, 12,19 to the authors' knowledge, there is no detailed published information on the gross, microscopic, and bacteriologic findings of postparturient malignant edema (vulvovaginitis and metritis) caused by C. septicum in cattle. This report describes 5 cases of postparturient vulvovaginitis and metritis caused by C. septicum in Holstein heifers.
Five cattle submitted to the California Animal Health and Food Safety Laboratory System between 1998 and 2004 for postmortem examination were included in this study. All 5 animals were Holstein, from 4 different herds, and 4 were first-calf heifers; the parity of 1 was not reported. Clinical histories indicated that all animals developed vulvar swelling 1–3 days after calving, followed by fever and depression. Four of the 5 died spontaneously within 24 hr of onset of clinical signs, and 1 animal was submitted live but recumbent and was euthanatized with intravenous administration of an overdose of sodium pentobarbital.
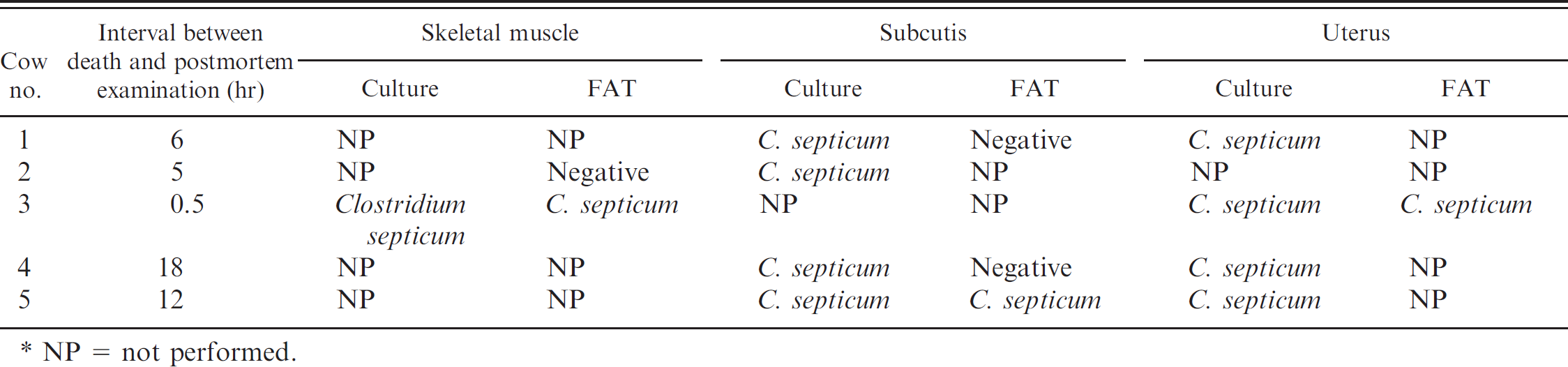
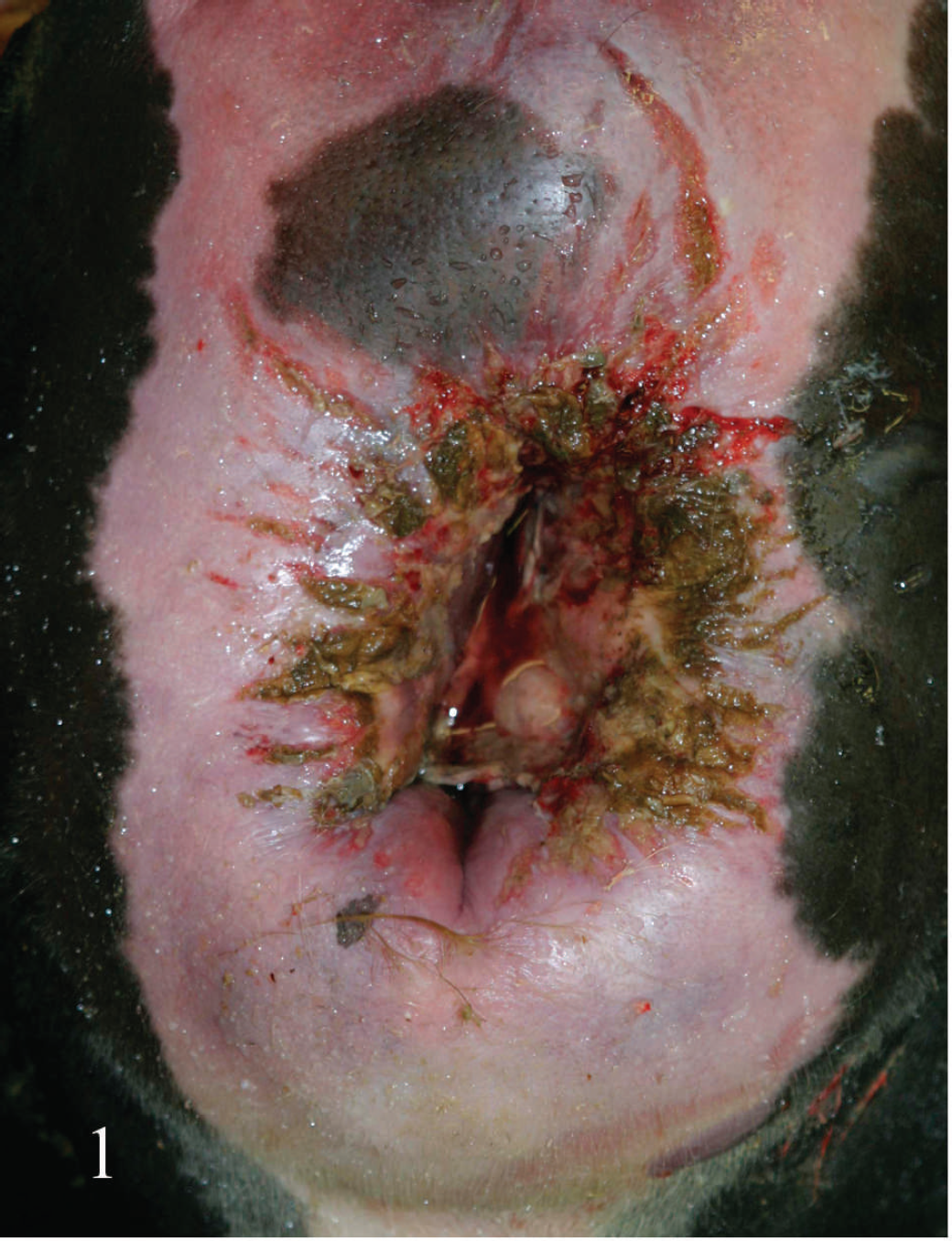
Necropsies were performed on all animals within 18 hr of death (Table 1). All animals were in good nutritional condition and in fresh (cow nos. 1–3) to mildly decomposed (cow nos. 5 and 6) postmortem state, and all had similar gross lesions. The most striking gross changes were marked, diffuse perineal, perivulvar, and perivaginal gelatinous and often hemorrhagic (dark red) edema, extending into the adjacent musculature and subcutaneous tissue (Figs. 1–3). Multiple longitudinal vulvar, vaginal (Fig. 2), cervical, and uterine body tears with variable lengths between 1 and several centimeters, covered with fibrinous exudates, and sometimes adherent necrotic fetal membranes were present. In 1 animal, most skeletal muscles and subcutaneous tissue of the left rear limb were diffusely hemorrhagic and edematous. Multifocal petechiation and occasionally ecchymosis were present on all serous membranes. The lungs were congested and edematous, with a large amount of stable froth present in the trachea and lower airways. No other significant gross abnormalities were observed in any of the animals.
Summary of anaerobic culture and fluorescent antibody test (FAT) results.*
NP = not performed.
Samples of uterus, vagina, perivulvar skeletal muscle, subcutaneous tissue, skin, liver, kidney, lung, heart, adrenal gland, abomasum, small intestine, and colon were collected and fixed by immersion in 10% neutral buffered formalin. Tissues were processed using standard histologic techniques, embedded in paraffin wax, sectioned at 4 μm, and stained with hematoxylin and eosin. Selected sections were also Gram stained.
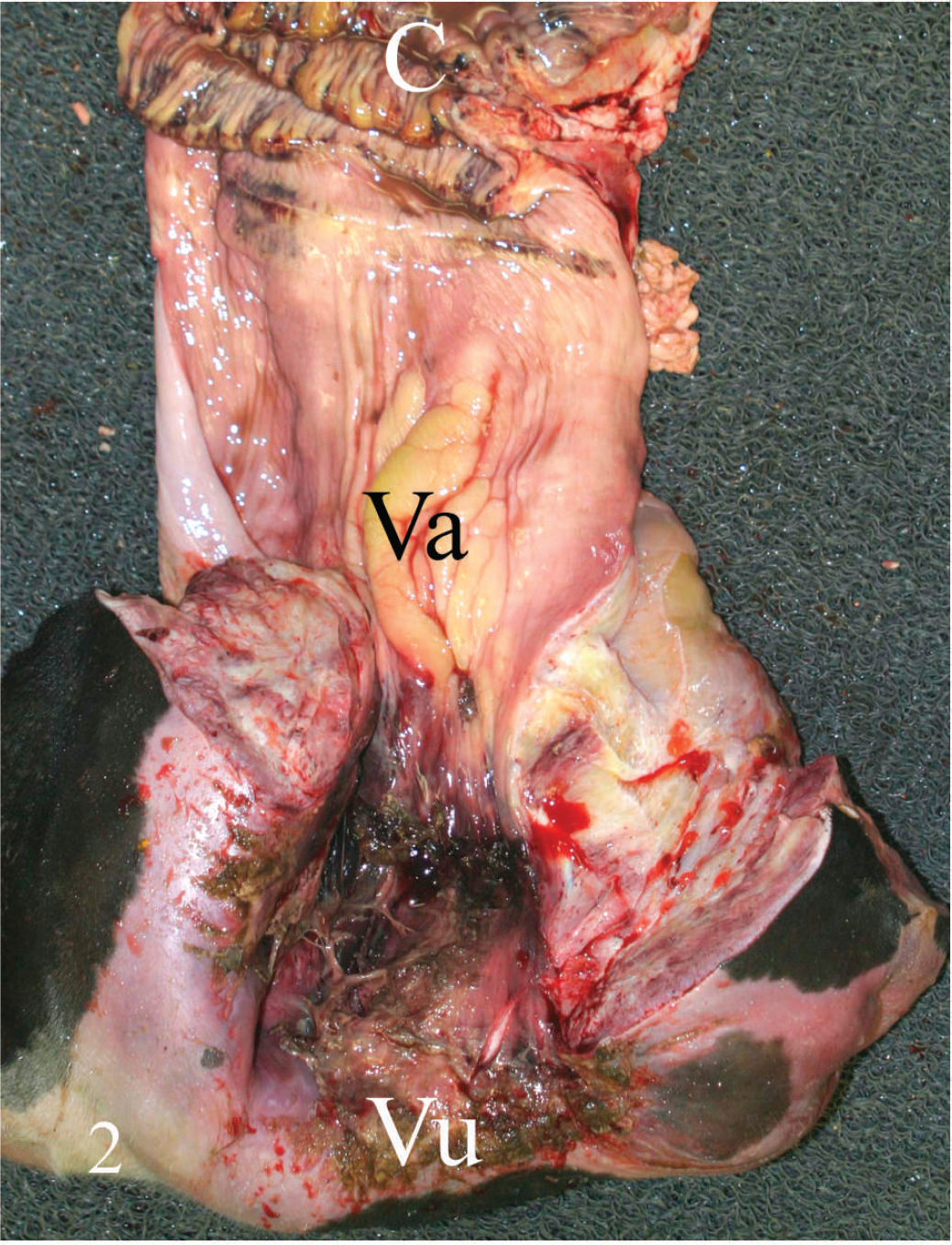
With only a few exceptions described below, similar histopathologic changes were noted in all 5 animals. The vulvar, vaginal, and uterine mucosae were multifocally necrotic and ulcerated. The ulcer beds were markedly edematous and hemorrhagic and infiltrated by large numbers of neutrophils and fewer lymphocytes, macrophages, and plasma cells admixed with cell debris and fibrin. Superficial mucosal capillaries frequently contained thrombi. The submucosa was markedly distended by edema and fibrin (Fig. 3), and neutrophilic, necrotizing vasculitis was frequently observed. Small numbers of neutrophils, lymphocytes, macrophages, plasma cells, and large Gram-positive rods, some of which had subterminal spores, were present within the submucosa. Large numbers of small coccoid bacteria were adhered to the luminal surface of the vaginal mucosa in 1 animal that had been dead only 30 min before necropsy was performed (cow no. 3; Table 1). Exudate composed of fibrin, hemorrhage, necrotic leukocytes and cell debris was adhered to the mucosa. In 1 animal, there were numerous irregularly shaped gas bubbles throughout the uterine wall.
Within the skeletal muscle, there was multifocal degeneration and occasionally necrosis of myofibers, hemorrhage, vascular congestion, and leukocytic infiltration consisting of neutrophils and fewer plasma cells, lymphocytes, and macrophages (Fig. 4). Necrotizing vasculitis with fibrin thrombi was occasionally observed. Large Gram-positive rods, occasionally with subterminal spores, were present within areas of edema and hemorrhage (Fig. 5).
The subcutaneous tissue was edematous, with small to moderate numbers of scattered neutrophils and mild fibrin leakage. Large Gram-positive rods, some of which contained subterminal spores, were variably present. Acute segmental necrosis of vessel walls and neutrophilic vasculitis were present in some sections. No significant histologic abnormalities were observed in any of the other tissues examined.
Samples of uterus and/or perivulvar subcutaneous tissue and/or perivulvar skeletal muscle (Table 1) were aseptically collected as soon as possible after the necropsy began and inoculated onto 5% sheep blood agar plates for aerobic culture and onto prereduced anaerobically sterilized Brucella plates a for anaerobic culture. All incubations were performed at 37°C for 48 hr. The isolates were identified by conventional biochemical techniques. Briefly, beta hemolysis and absence of lipase and lecithinase activity on blood agar and egg yolk agar plates, respectively, were evaluated. Sensitivity to kanamycin and vancomycin and indole and urease activities were also evaluated. Additionally some of the isolates were also evaluated using a Rapid ANA II strip. b
Impression smears of perivulvar subcutaneous tissue and/or paraffin-embedded sections of this tissue and/or skeletal muscle (Table 1) prepared as soon as possible after the necropsies began were subjected to a direct fluorescent antibody test (FAT) for C. septicum, C. chauvoei, C. sordellii, and C. novyi, as previously described, 4 and counterstained with a 0.2% Evan's blue c solution in phosphate-buffered saline for 20 sec. The conjugates were from Pragma d (cow no. 3) or from VMRD e (cow nos. 1, 2, 4, and 5). Smears prepared with cultures of international standard strains f of these microorganisms were used as controls for the FAT.
Clostridium septicum was isolated and/or demonstrated by FAT in at least one of the tissues in all animals and in both the uterus and subcutis of 4 of 5 animals (Table 1). Staphylococcus spp. was isolated from the uterus in the animal that showed cocci in histologic sections of vaginal mucosa (cow no. 3; Table 1). The location of C. septicum as detected by FAT was consistent with the Gram-positive rods noted in the Gram-stained sections (Fig. 6). No other clostridia were isolated or detected by FAT in any animal.
Vulva of animal no. 3. Observe swelling, tears, and inflammatory exudates on the mucocutaneous junction surface.
Vulva (Vu), vagina (Va), and cervix (C) of cow no. 3. Observe mucosal tears covered by inflammatory exudate in vulva and vagina.
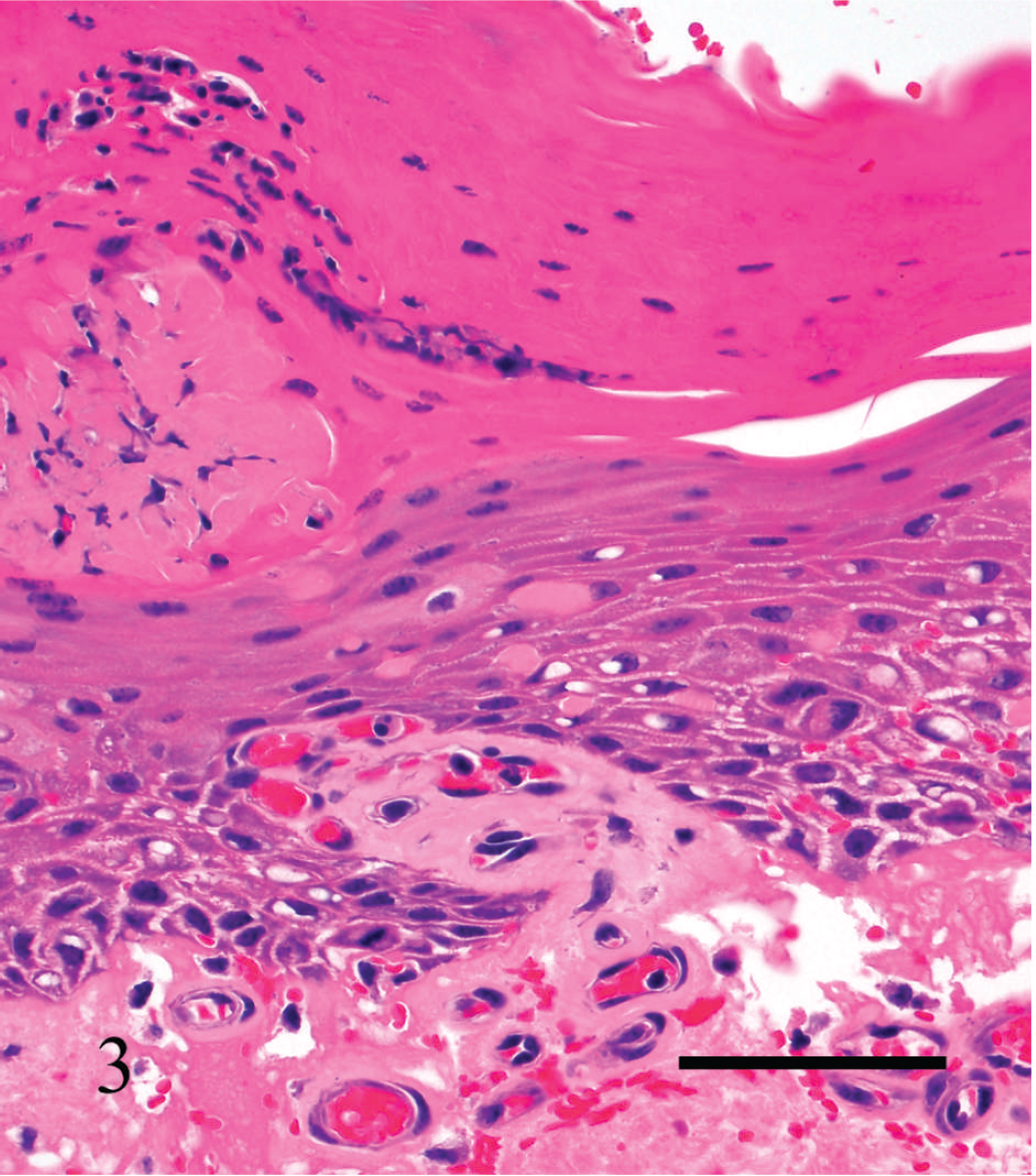
Vaginal mucosa of cow no. 3. Observe degenerative and inflammatory changes in the mucosal epithelium and severe edema and fibrin in the subepithelial tissues. Hematoxylin and eosin. Bar = 100 μm.
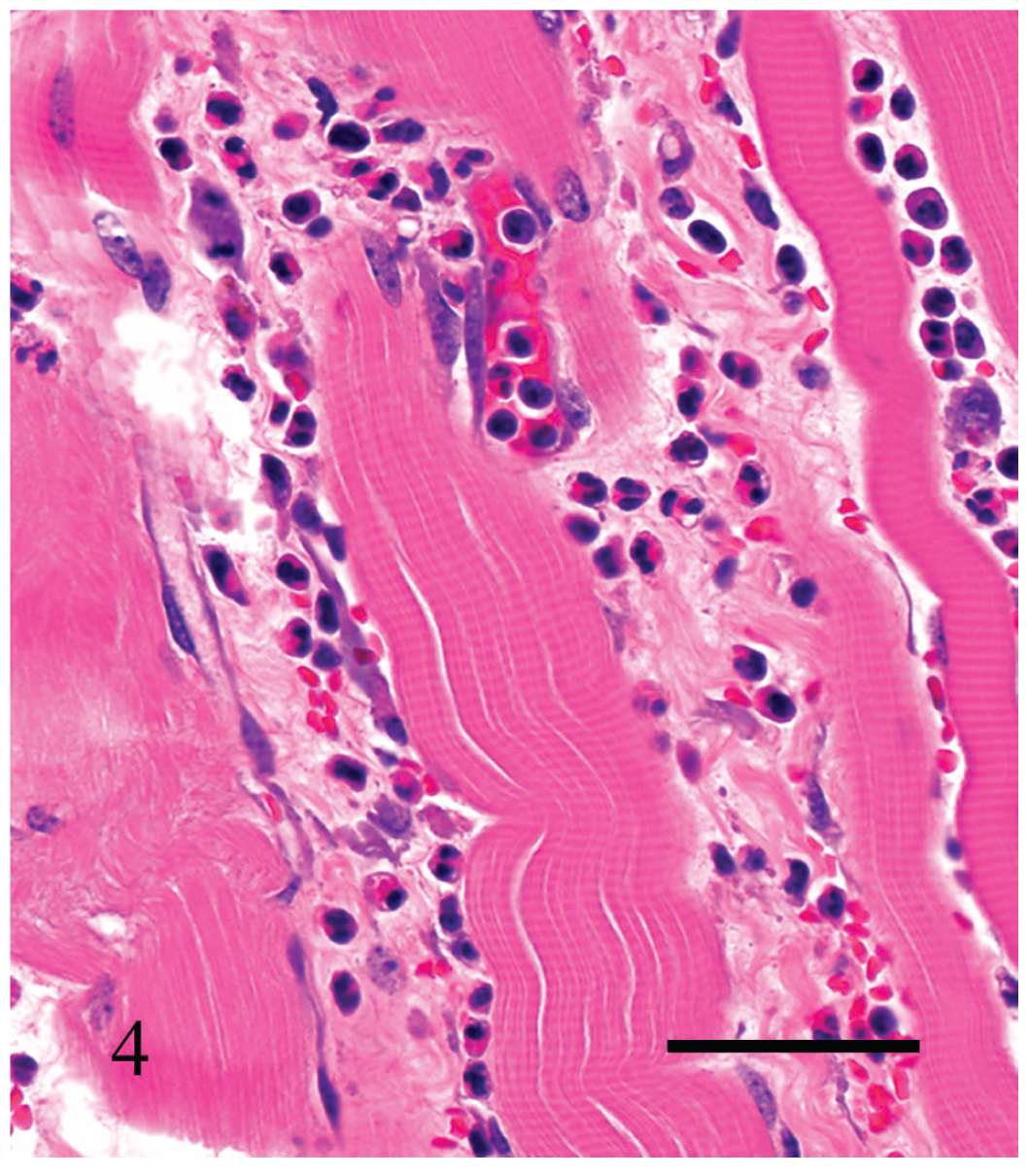
Interstitial proteinaceous edema and mild, mostly neutrophilic inflammatory infiltrate separating striated skeletal muscle fibers in cow no. 3. Hematoxylin and eosin. Bar = 60 μm.
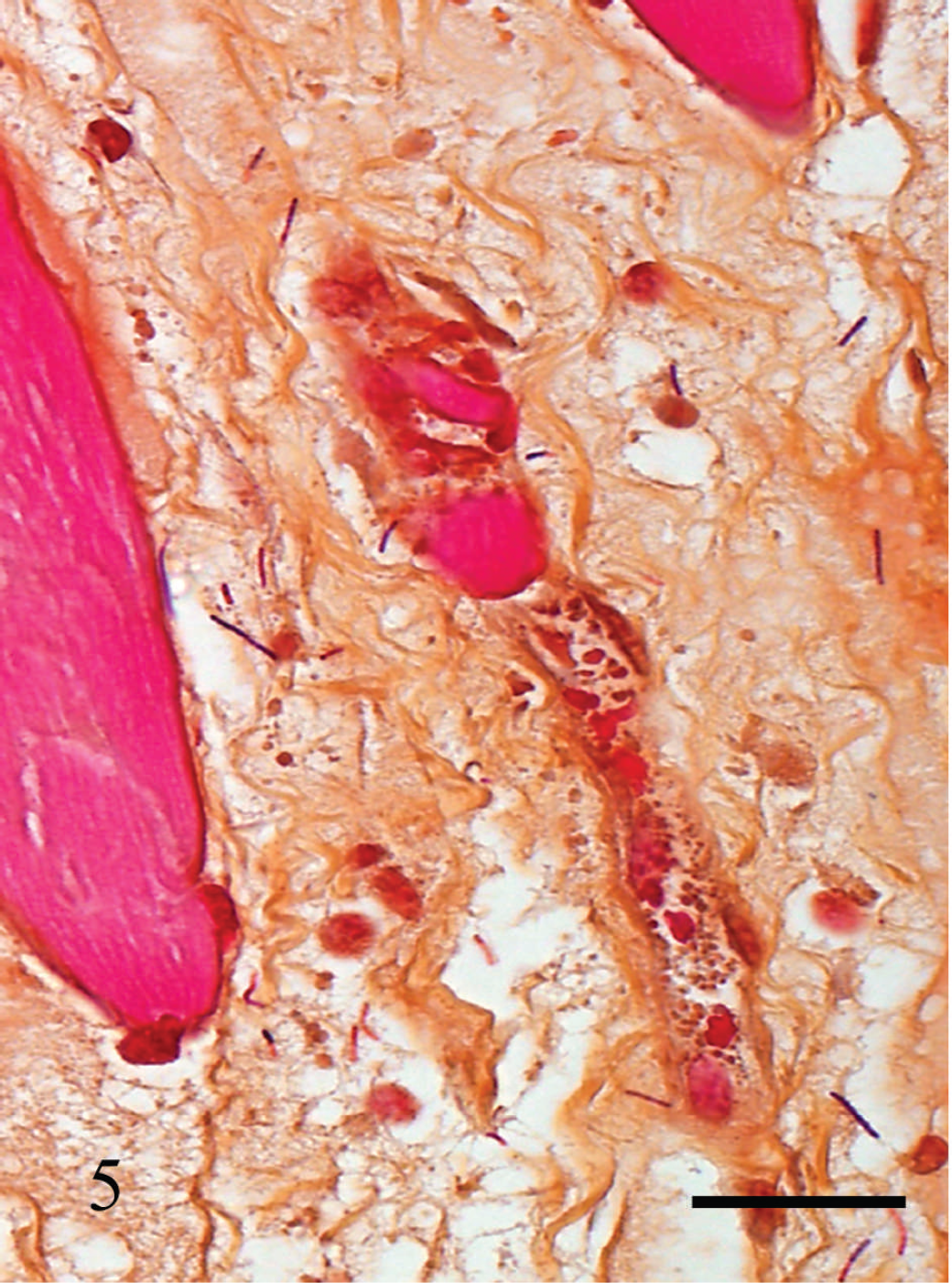
Gram-positive rods present in the interstitial edema of skeletal muscle of cow no. 3. Gram stain. Bar = 100 μm.
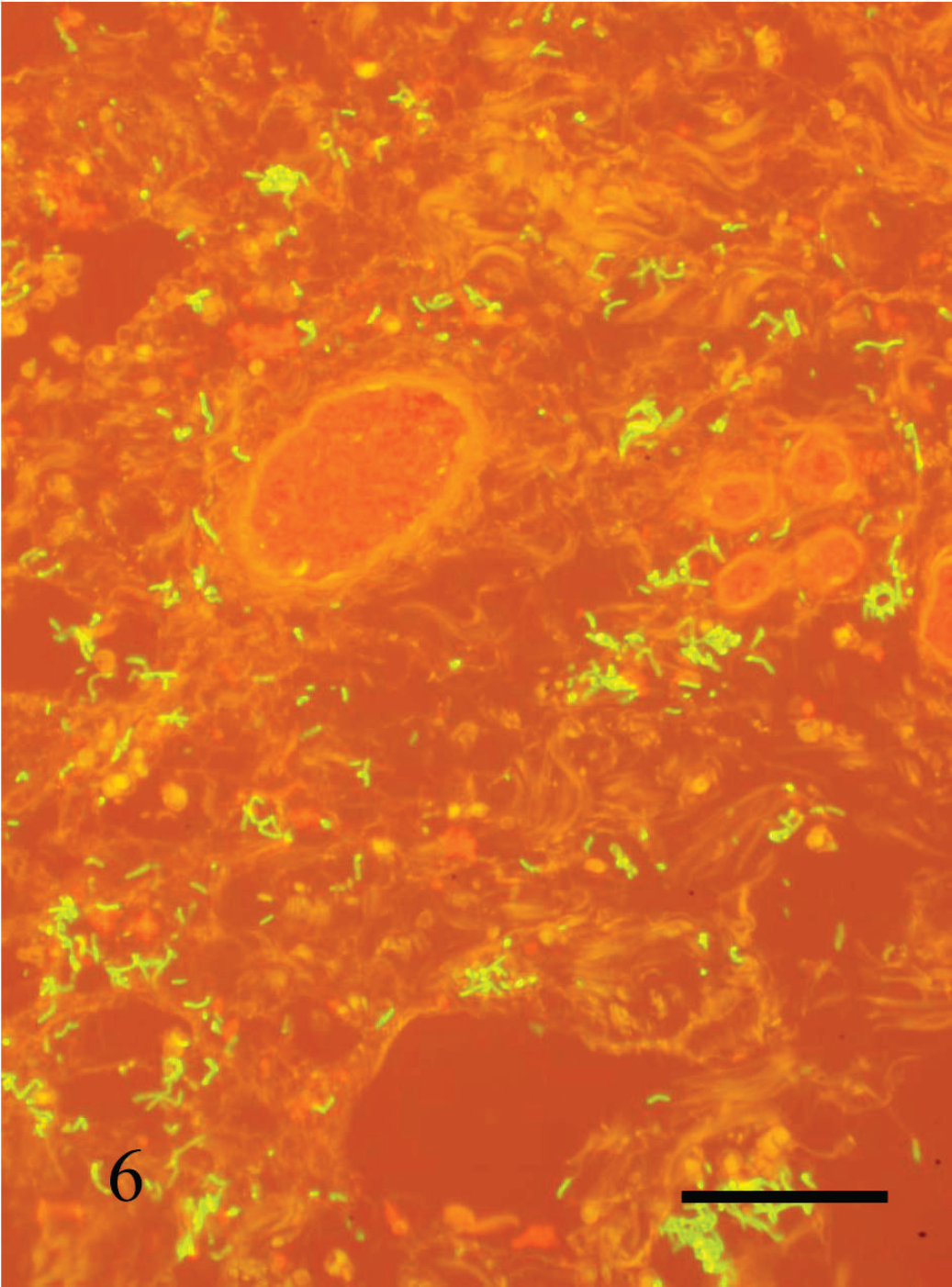
Clostridium septicum in perivulvar subcutaneous edema of cow no. 3. Fluorescent antibody test (C. septicum conjugate and Evan's blue counterstain). Bar = 100 μm.
The pathologic and microbiologic findings in these cases confirmed a diagnosis of malignant edema due to infection with C. septicum. Other common causes of vulvovaginitis in cattle include Bovine herpesvirus 1, Chlamydophila spp., Ureaplasma spp., and Porphyromonas levii. These causes were ruled out by histopathology and/or bacteriology.
The most likely route of entry of C. septicum was via the vulvar, vaginal, and uterine mucosal tears that occurred during parturition, either directly from the environment or from fecal contamination. Presumably, the mucosal tearing allowed C. septicum to enter the tissues, and the bruising of the deeper tissues set up the anaerobic conditions that allow the organisms to proliferate. Although C. septicum is a frequent postmortem invader, 16 it is usually accepted that if it predominates in specimens obtained soon after death, as in at least 3 of these cases (cow nos. 1–3; Table 1), it is likely to be the etiologic agent. 16 In these 3 cases, postmortem examination was performed between 0.5 and 6 hr after death, and the carcasses were fresh, which lends support to the statement that the C. septicum isolated and demonstrated by FAT was an antemortem pathogen and not a postmortem invader. Also, C. septicum may overgrow other clostridia when cultured, giving a false impression of the relative predominance of this microorganism in tissues, whereas the performance of FAT for multiple clostridial species gives a more accurate indication of the true clostridial population in the tissues. In all the cases reported in the current study, C. septicum was the only anaerobic bacterium isolated from tissues and the only clostridial species detected by FAT. In addition, the absence of bacilli in blood vessels supports the position that C. septicum played a pathogenic role in the cases presented here and was not simply the result of postmortem invasion and overgrowth. Negative FAT results in 3 of the animals from which C. septicum was isolated could be due to preparation of smears from areas in which this microorganism was not present or to lack of sensitivity of the conjugate used for some strains of this microorganism. The latter has been observed previously by the authors with some commercial conjugates (authors' unpublished observations).
Several findings, including pulmonary edema and mucosal petechiation, were interpreted as nonspecific gross findings consistent with terminal septicemia and/or toxemia. Staphylococcus spp. was isolated from the uterus of 1 animal. This microorganism is considered a normal inhabitant of the environment and the skin of cattle. It is likely that in this case the Staphylococcus spp. was a secondary contaminant that invaded the uterus after delivery, perhaps aided by the necrotizing effects of the C. septicum toxins.
Clostridium septicum is a swarming bacterium, a property that enables it to spread quickly along fascial planes. Disease progression is rapid, with clinical signs evident within 24 hr, 18 and is often fatal within 1–4 days. This microorganism has been implicated in malignant edema of several animal species, including cattle. Clostridium septicum also causes “braxy,” a hemorrhagic or necrotizing abomasitis in lambs and calves. 3,15
Clostridium septicum produces alpha toxin (a hemolysin), 17 which is related to the aerolysin toxin produced by Aeromonas hydrophila 17 and epsilon toxin of C. perfringens types B and D. 1 It is not sequentially, structurally, or functionally related to the alpha toxin produced by C. perfringens. The alpha toxin of C. septicum uses glycosylphosphatidylinositol-anchored proteins on the membrane of target cells as its cellular receptor, undergoes proteolytic activation, and interacts with other activated monomers within the membrane to form a transmembrane pore. 5 This pore-forming activity of alpha toxin is essential for C. septicum-mediated myonecrosis and in vivo toxicity. 9,10 Alpha toxin causes rapid myonecrosis, interstitial hemorrhage, and edema when injected into mice 9 and microvascular collapse. 8 In addition to the lethal alpha toxin, C. septicum also produces delta toxin (hemolysin), beta toxin (DNase, leukocidal), gamma toxin (a hyaluronidase), a neuraminidase and hemagglutinin, a chitinase, a weak lipase, and a sialidase, 17 which, although not lethal, are thought to contribute to the pathogenesis of C. septicum disease by facilitating tissue invasion 9 and myonecrosis 10 and by reducing microvascular blood flow. 8
The FAT procedure worked well on formalin-fixed, paraffin-embedded tissue sections, and this technique was useful to correlate the presence of clostridial microorganisms with histologic lesions. This technique would be valuable in confirming diagnoses of malignant edema in cases in which fresh tissues are not available for microbiologic culture. In summary, the current report describes 5 cases of postpartum malignant edema with vulvovaginitis and metritis by C. septicum in cattle.
Footnotes
a.
Hardy Diagnostics, Santa Maria, CA.
b.
Rapid ANA II strip, Innovative Diagnostic Systems Inc., Atlanta, GA.
c.
Sigma-Aldrich, St. Louis, MO.
d.
Pragma, Kent, England.
e.
VMRD Inc., Pullman, WA.
f.
American Type Culture Collection, Manassas, VA.
