Abstract
Over the past few decades, there have been increasing numbers of reports of diseases in marine turtles. Furthermore, in recent years, there have been documented instances of apparently new diseases emerging in these species of which the etiology and/or pathogenesis remain unknown. These instances i) raise concern for the survival of marine turtles, and ii) question the health and stability of the benthic marine environments in which turtles live. Knowledge of common disease processes and pathologic changes in lesions, along with a standardized approach to postmortem and sample collection are required to document and understand the host-agent-environment interactions in marine turtle health. This review combines, for the first time, a standardized approach to the postmortem of marine turtles for veterinary clinicians, with a concurrent descriptive review of the gross and microscopic pathologic changes in lesions commonly seen.
Introduction
Veterinarians are playing an increasing role in identifying and determining the contribution of disease processes to population declines in free-ranging marine species. By providing data to wildlife managers on causes of mortality and morbidity, veterinary diagnosticians can play a constructive role as partners in conservation, including establishment of sustainable environmental strategies and reduction of mortality rates of threatened species, where the loss of even a few individuals in some circumstances may have long-term implications. Over the past few decades, there have been significant increases in reports of diseases that cause high mortality in marine animals, including new diseases and new hosts, and that occur predominantly in heavily modified coastal areas undergoing environmental changes (such as catchment management, eutrophication, and increased turbidity) because of urbanization and increased frequencies of toxic algal blooms. 55,142 Globally, these findings are of particular relevance to marine turtles, because the cause of mortality continues to go undiagnosed for many reasons. The significance of these findings are highlighted, with some areas reporting loss of nesting sites 98 and with others reporting loss of numbers of nesting turtles each year. 90 Disease is often suspected as a contributing or primary factor in marine turtle deaths, except for certain harvest or fisheries issues, although the diagnosis of a specific disease(s) has been hampered by a number of factors. These factors may include poor specimen condition, difficulties in accessing these large marine animals along remote coastlines, and a lack of laboratory reagents to diagnose disease with a known degree of confidence. There is also a lack of detailed baseline information on physiology and biology of healthy marine turtles that hampers the recognition of newly emerging diseases.
The aim of the present article is to provide guidance for the diagnostician who is not familiar with marine turtle species, including i) a review of the biology and physiology not routinely encountered in wildlife or domestic species, ii) guidelines for appropriate postmortem, iii) common pathology and causes of mortality, and iv) sampling procedures, both routine and specialized.
Marine turtle species
Current status of marine turtle populations
Marine turtles are an integral part of oceanic ecosystems. 3,18,19,69,70 There are 7 extant species of marine turtle: the green (Chelonia mydas, Linnaeus 1758), the loggerhead (Caretta caretta, Linnaeus 1758), the leatherback (Dermochelys coriacea, Van-delli 1761), the hawksbill (Eretmochelys imbricata, Linnaeus 1766), the flatback (Natator depressus, Garman 1880), the olive ridley (Lepidochelys olivacea, Eschscholtz 1829), and the Kemp's ridley (Lepidochelys kempii, Garman 1880). 56 Six of these species are listed as vulnerable, endangered, or critically endangered on the International Union for Conservation of Nature Red List of Threatened Species. 68 The flatback turtle is listed as “data deficient” because of limited available population information. 68
Marine turtles are protected in most countries. Appropriate permits should be sought before starting any postmortem or other investigation. In addition, there are often legal obligations associated with conducting a postmortem on a marine turtle. As such, the collection of marine turtle carcasses from natural habitats should not be undertaken without recording position coordinates and site information to obtain medical history and provide legal information if investigations by environmental protection agencies and police personnel occur. Multiple photographs of the carcass in situ and surroundings are advised.
Postmortems are essential if attempting to determine causes of death of individual animals when disease is a suspected factor. When such examinations are performed routinely, this information also contributes significantly to determining the health status of the population, particularly when combined with health assessments of living animals. There are 5 freely available comprehensive instructional guides for the postmortem of marine turtles, some in multiple languages: Wolke and George, 146 Jacobson, 72 Work, 148 Wyneken, 153 and Flint et al. 37 Each of these guides have their strengths in specific areas of turtle postmortem examination, including detailing loggerhead turtle anatomy, 146 providing descriptive sampling techniques, 72 outlining a practical field-guide approach and available in multiple languages, 148 describing detailed anatomy, 153 and including pictorial histologic and gross pathology. 37
Species-specific differences
Despite their overall similarities, some areas of turtle anatomy, physiology, and biology are unique to certain species. For example, in loggerhead and Kemp's ridley turtles, the lungs are more closely attached to the vertebral column than in other species 153 ; leatherback turtles have a soft epidermal layer that covers the skeletal structure that forms the carapace, whereas the other species have a hardened layer 153 ; and the composition of the glands of the gastrointestinal tract vary, depending on herbivorous or carnivorous lifestyles. 88,137 In addition, turtles as a group differ from other groups of animals commonly encountered by diagnosticians and biologists. For example, marine turtles have 2 distal aortas, no diaphragm, 153 a low-pressure cardiovascular system, 140 and the ability to maintain gastric stasis for prolonged periods of time (this accounts for the fact that a turtle may be in rehabilitation for several months and still have “fresh” seagrass in the esophagus). The prosector should be familiar with species differences before commencing a postmortem.
Postmortems
External postmortem
Signalment. As for any species, the external assessment should be careful and systematic; findings should preferably be recorded on a predesigned datasheet (Fig. 1). The signalment should include the species, in addition to individual identification, with a search for any previously applied tags and/or microchips (passive integrated transponder [PIT] tags). Standard tags are usually metal and are applied to the trailing edge of a flipper. PIT tags are approximately 12 × 3 mm in a glass or durable plastic capsule. They are applied in the interdigital space of the proximal tarsals or carpals of the left or right limbs, the cranial aspect of the shoulder, the soft-tissue areas associated with the cranial aspect of the carapace, or in the subcutaneous tissue of the tail. PIT tags are recognized by most microchip scanners. Other identification marks include notching of the scutes and a living tag (this is the transplantation of a patch of carapace to plastron and vice versa, which creates a dorsally unpigmented and ventrally pigmented transplant of living tissue) for which photographs should be taken for identification by an appropriate expert. 6 It is important to photograph the carcass, with a reference scale included, on which the identification number of the animal and the date are written. Such images can be used in annual stranding and mortality reports, as published, for example, by the Queensland Department of Environment and Resource Management in Australia. 50,52 Standard images include the following: ventral (plastron) and dorsal (carapace) surfaces; the head (dorsal-ventral and rostral-caudal views); forelimbs and hind limbs; any identifying marks (trauma or tags); and any external abnormalities, including wounds, deformities, and missing limbs or eyes. All photographs should allow for the confirmation of species identification by an expert, if required. This may be done by counting the number of costal scales that are located on the carapace, counting the number of pairs of prefrontal scales located immediately rostral to the eyes and assessing the shape of the edge of the carapace. 115 The turtle may need to be washed to remove sand or scraped clean of encrusting organisms so that the features listed above may be clearly visible when photographed. Identification keys to assist in species determination are available through most institutions tasked with monitoring marine turtle populations. 37
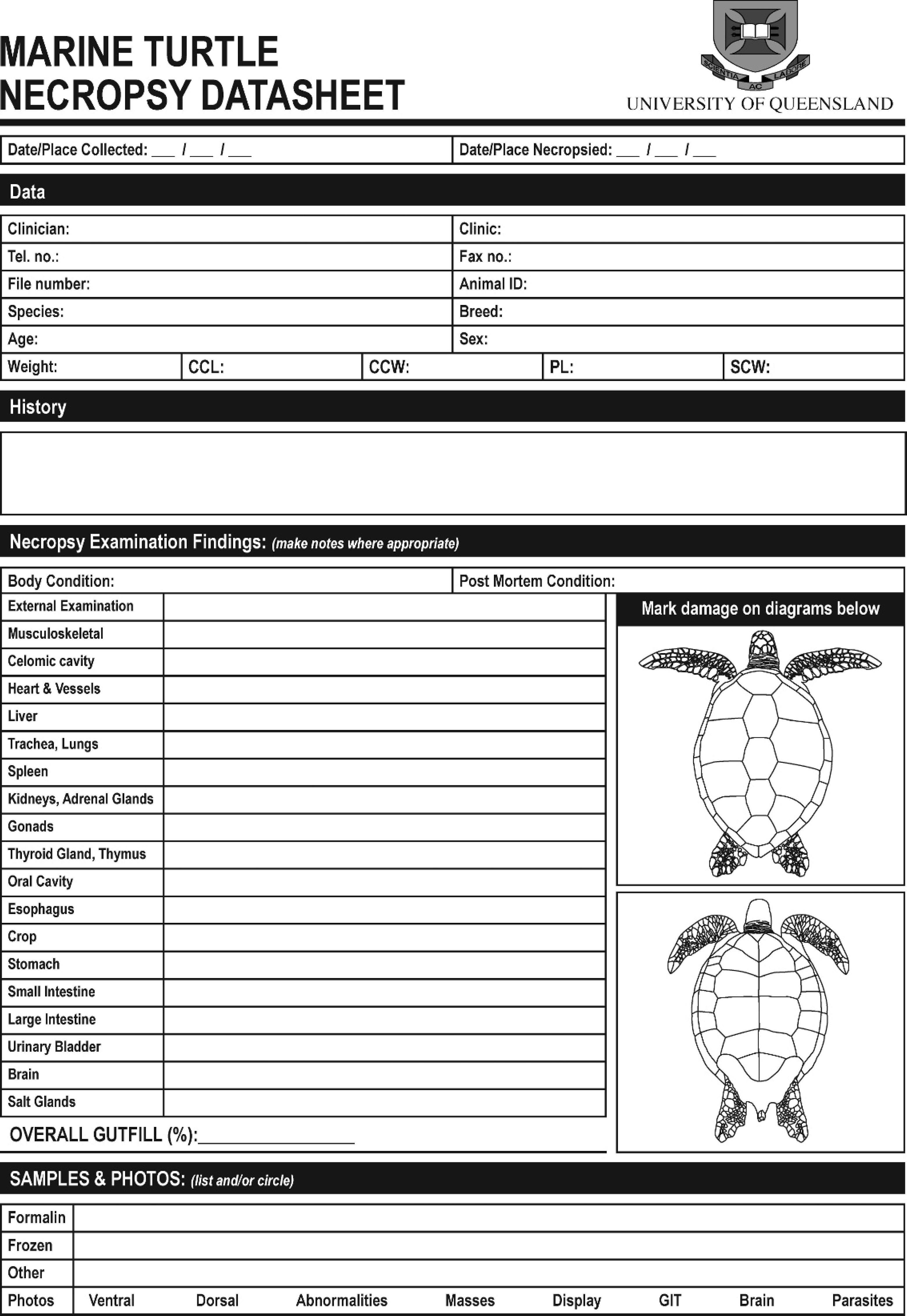
Marine turtle necropsy datasheet providing a reference to record geography, biology, history, morphology, significant gross necropsy findings, the samples collected and method of storage, and photographs taken.
Sex determination. In adult turtles, sex and breeding status may be estimated externally with measurement of tail length (TL), indicating the sex, 87,89 and estimation of fat deposition, indicating breeding condition. However, this method is highly subjective, with much variation between individuals of each sex and species. External determination of sex in immature turtles is only possible if a blood sample can be collected immediately after death and assayed for testosterone. 13 This method is expensive, and the accuracy is also variable. Sex and breeding status in both mature and immature turtles are most accurately determined by internal examination of the gonads.
Age. There are 3 broad age classes: hatchling, juvenile (immature), and adult (mature). Ages are often estimated externally based on midline curved carapace length (CCL) or straight carapace length (SCL; see below), supported by gonad assessment during internal examination; ranges for each age class vary depending on the species and geographic location and may require interpretation by an appropriate expert.
Carcass decomposition. Significant variations in decomposition rate can occur because of the aquatic brine and water temperature in which the carcass is often immersed. The degree of decomposition may be subjectively estimated from corneal opacity, color changes and sloughing of the skin, pooling of blood in dependent areas, degree of bloating, odor, and body temperature. 121,146 The exact time of death, as for any species, is difficult to discern; predictive algorithms in canine and human models usually overestimate the time of death, and none are available for turtles. 121 Carcass decomposition codes for marine turtles in Australian waters 86 have been adapted from standard decomposition codes 120 to offer a guide for the prosector.
Morphometric measurement. Accurate and complete morphometric measurements are essential to assess body condition and include the following: midline CCL and SCL and width (curved carapace width [CCW] and straight carapace width [SCW]; Fig. 2A), Carr's SCL, plastron length (Fig. 2B), standard depth (SD; Fig. 2C), head width (HW; Fig. 2D), TL (Fig. 2E), body weight (to the nearest 0.5 kg), and the body condition index (BCI) calculated as kg/SCL 3 . 10 Marine turtles may be weighed by using a large cargo net (best used for larger turtles) or leg ropes fixed to the proximal aspect of each flipper, with the animal in dorsal recumbency; the animal is then lifted by using a jib attached to scales (Fig. 3). Lengths and widths should be measured by using a fiberglass tape, rather than a plastic tape, which may stretch and give incorrect readings. Midline CCL is measured from the cranial skin-carapace junction to the caudal margin of the suture of the postcentral scutes along the curvature. SCL is a measure that uses the same landmarks but by using calipers, to produce a measure independent of carapace curvature. The relationship between CCL and SCL changes, depending on the curvature and flexibility of the carapace. Curvature of the carapace varies with age and species. Flexion of the carapace varies with body condition; with poor condition, animals have a highly flexible carapace and plastron. Carapace width (SCW and CCW) is measured the same way at the widest point of the carapace. Carr's SCL is measured from cranial-most aspect of the carapace (over the shoulder) to the caudal most aspect of the carapace by using calipers. Plastron length is measured by using a fiberglass tape at the cranial-most and caudal-most points along the midline of the plastron. SD is also measured by using calipers to determine the maximum depth of the body; the midline apex of the carapace (dorsal) is selected, and the measurement is made at that point at 90° to the corresponding midline of the plastron (ventral). HW is measured by using calipers at the widest part of the quadrate bones located immediately rostral to the auditory canal and caudal to the ocular orbit on the ventrolateral aspect of each side of the skull. HW combined with head shape may be used to confirm species. 153 TL is measured from the caudal aspect of the suture of the postcentral scutes to the distal caudal aspect of the extended tail. This is denoted by a positive measure if the tail extends past the caudal aspect of the suture of the postcentral scutes and by a negative measurement if it does not. Measurements that are standard for a particular region should be used to ensure consistency. For example, Queensland researchers measure the CCL, whereas Hawaiian researchers measure the SCL. Conversion equations are available to compare the different measures, but these often need to be validated with local species. BCI (0–3) quantifies body condition based on a ratio of metric weight and the SCL. A score of less than 1 denotes poor condition, 1.1 is average, and 1.2 and above is “healthy.” 10 This quantitative assessment of BCI by using SCL and weight (kg/SCL 3 ) can be found in a previous study. 10 In some circumstances, the BCI is not necessarily indicative of the health of the animal.

Morphometric measurements of marine turtles.
For example, adult green turtles in northern Australian waters lose significant body condition during periods when they migrate from resident feeding grounds to breeding units and undertake periods lasting many months of energetically demanding reproductive activities. 153

A loggerhead turtle in dorsal recumbency, with leg ropes tied around the proximal aspect of each flipper (arrows), allowing suspension from a jib attached to scales.
Examination of the head, limbs, and skin. The oral cavity and jaw should be closely examined for mandibular or maxillary fractures; hemorrhage; lacerations; ulceration; deformities; tumors; and foreign bodies, including fishing hooks and line, and parasites. Sizes and lengths of any foreign bodies should be recorded, and photographs should be taken. The eyes should also be assessed for evidence of trauma; opacity; and masses, including fibropapillomas (see below); and the degree of retraction (sinking) recorded. Limbs should be assessed for symmetry, trauma, tumors, function, and foreign bodies. The cloaca should be examined for evidence of prolapse, hemorrhage, lacerations, leeches, deformities, tumors, and foreign bodies. Reductions in adipose tissue, fractures and deformities of the tail are common abnormalities and should be noted. 60
Epibionts. Epibionts are all organisms, including ectoparasites, that live on the surfaces of marine turtles, including barnacles, leeches, and algae. Collectively, they comprise the epibiotic load. Normal epibiotic load varies between turtle species (e.g., a normal epibiotic load on a hawksbill turtle would be excessive on a green turtle). Barnacles and leeches should be identified and counted. The distribution, density, and percentage of algal cover on the dorsal surfaces should be estimated, because this may indicate floating behavior; the theory being that certain algae proliferate if the animal is predominantly surface dwelling. If identification of an epibiont is required, then a photograph denoting size and location on the turtle should be taken before collecting a specimen.

Postmortem removal of the plastron for internal examination. A, cutting through the soft tissue at the plastron-carapace junction, with the turtle in dorsal recumbency.
Internal postmortem
A previous study 153 comprehensively reviewed turtle anatomy to provide a thorough anatomical reference when conducting a postmortem. To optimize access, it is best to place the body in dorsal recumbency, with placement of blocks around the carapace to stabilize it if necessary. The plastron is removed completely by cutting through the soft tissue at the plastron-carapace junction (i.e., the junction of the marginal and inframarginal scutes; Fig. 4A). These incisions are extended through the soft tissue of the ventral aspects of the limbs, neck, and cloaca, with any release of gas from the celomic cavity indicating putrefaction. 146 The plastron is attached at 3 points: 2 “clavicles” (acromion processes of the pectoral apparatus) and the pelvis (lateral pubic processes of the pelvic girdle). The plastron is retracted by using a hook, and all soft tissue is carefully excised as close to it as possible (Fig. 4B) to reveal the pectoral muscles and underlying celomic cavity (Fig. 4C). A circumferential incision is then made through the skin around each forelimb. The caudal aspect of each scapula (coracoid process) is elevated, and the connective tissue of the dorsal surface of the pectoralis major muscle group for each forelimb is excised; in a healthy turtle, the pectoral muscles should be red to pink, firm, and ample. To detach the flipper, it may be rotated medially; complete rotations should remove the flipper by tearing the connective tissues that surround the humeroscapular joint.
Examination of the celomic cavity. Volumes of celomic fluid that create more than small pools (e.g., >200 ml) on either side of the spinal column are considered to be increased; the color and viscosity should be similar to peritoneal fluid in other species. Examination of the celomic surfaces may indicate if penetrating trauma, such as a boat strike, has occurred and should be correlated with external lesions. The estimated depth of fat reserves that line the cavity (internal surfaces of the carapace and plastron) indicates the nutritional status; in a normal turtle in good body condition, it should be firm, tan to gray to green to yellow, and have a thick buttery consistency. Carapace fat should be examined for evidence of serous atrophy (i.e., a watery or gelatinous consistency). As the adipose tissue is replaced by water, black pigmentation can become more apparent. Mesenteric tissues associated with internal organs should also contain ample amounts of adipose tissue and may be excised at this stage to gain access to the internal organs.
Location and removal of the internal organs. Relative and absolute organ sizes vary significantly, because marine turtles may range in size from less than 20 g as hatchlings to up to 500 kg as adults, depending on the species, 115 season, and reproductive cycle. 136 Few studies have examined the relationship between body weight and organ weight. 125 While the internal organs are still in situ, the heart, the singular thyroid gland, and the thymus should be examined, followed by their removal. The thymus is a gray to pink, dense, laterally elongated, multilobed structure located cranially to the thyroid gland, between the subclavian arteries. The heart is removed by cutting transversely through the left and right aorta, pulmonary artery, and brachiocephalic trunk of the right aorta, and the interiors of each chamber and vessel are exposed via longitudinal incisions through the large vessels. The large bilobed liver and gall bladder should also be examined initially in situ, followed by assessment of patency of the common bile duct, and then removal. The normal liver is dark purple to brown, firm and smooth, with sharp borders. The entire gastrointestinal tract is then removed by cutting through the distal esophagus, mesentery, and cloaca. The pancreas is directly adjacent to the (round) spleen alongside the duodenum, just past the level of the common bile duct. After removal of the intestinal tract, the paired distal aortas should be clearly visible along the dorsal wall of the celomic cavity. The tongue and remaining esophagus are freed and removed after disarticulation of the hyoid apparatus along with the trachea; the trachea is excised through the bronchial bifurcation. The paired lungs lie along, and are firmly attached by connective tissue to, the dorsal wall of the celomic cavity, extending approximately two-thirds of its length. To excise them, the connective tissue has to be removed along the lateral border and as close to the carapace as possible to avoid damage. This may be achieved by a combination of sharp and blunt dissection. The kidneys, gonads, and adrenal glands are then removed, the urinary bladder opened and examined, and the ureters traced along the dorsal celomic wall; the adrenal glands are most readily located by palpation adjacent to the cranial poles of the kidneys. 153 The brain is removed with the head either attached or disarticulated by making a transverse cut immediately caudal to the orbits and immediately rostral to the frontoparietal scale (Fig. 5A); removal of the head allows access to the rostral cervical spinal cord. The cut may be extended ventrally to the mandible (Fig. 5B) so the rostral section of the cranium is completely removed, exposing the brain and salt glands (Fig. 5C).
Detailed examination of systems and organs, including common pathologic findings
Integumentary system
Trauma may include evidence of a boat strike (multiple parallel linear lacerations if a propeller has hit the animal [Fig. 6], a single linear laceration from the keel or skeg of an outboard motor, or diffuse concussive fractures from impact), rope or fishing line entanglement (partial or complete linear limb amputations), shark bites, or fractures and deformities. Determining the age or cause of traumatic lesions may be difficult and may be assisted by histologic examination, particularly of the wound margins.
Changes in skin color and condition in reptiles can represent similar pathologic processes as those seen in mammalian species, including septicemia (e.g., red-purple mucous membranes and petechial hemorrhaging of the scutes); burns (e.g., necrotic, sloughing, or black skin); nutritional deficiencies, including calcium and vitamin D3 (e.g., scute lifting or localized sloughing of skin); anemia (e.g., pale skin color); toxicosis (e.g., erythema or discoloration of the skin); and coagulopathies (e.g., subcutaneous hemorrhage). 14,28,102 In cases of extreme emaciation, penetration of the plastron and/or carapace by the pectoral apparatus and/or pelvic girdles is common (Fig. 7). Secondary bacterial infections are frequent in sick and injured (immunosuppressed and/or debilitated) animals and may manifest as infected traumatic lesions, infections of the carapace that cause sloughing of the scutes (this should not be confused with lifting of the scales as part of the normal growth process), abscess formation, or stomatitis. 102,152 Reported bacterial infections in marine turtles included Vibrio, Aeromonas, Salmonella, Pseudomonas, Bacteroides, Fusobacterium, Flavobacterium, Clostridium, and Mycobacterium spp. 21,40,41,51,97,104,126,133,145
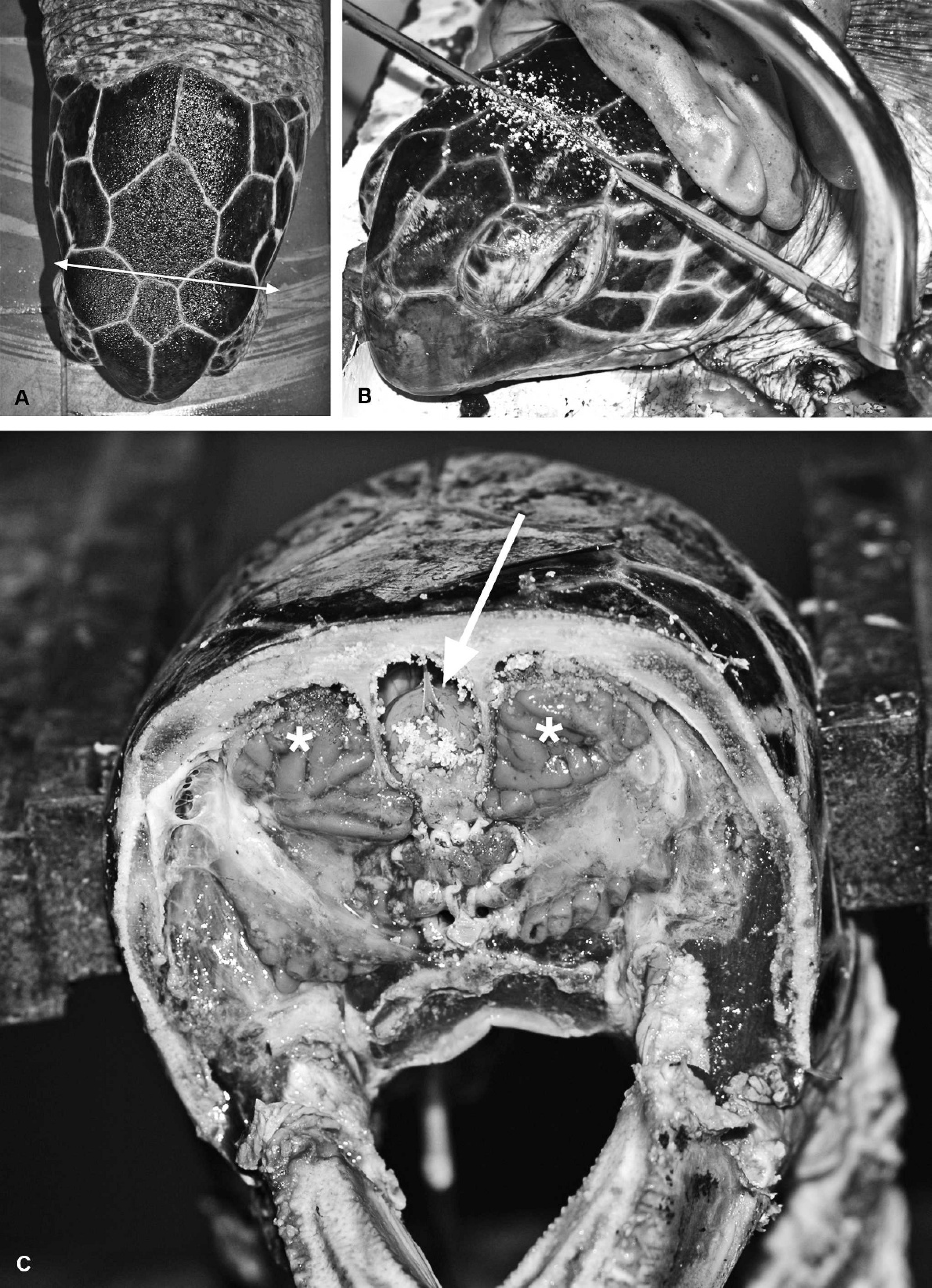
Postmortem removal of the brain.

Multiple parallel linear grooves in the dorsal carapace of a loggerhead turtle (arrows), consistent with healed propeller lacerations.
Caution should be taken when estimating the clinical significance of various epibiont burdens; as stated above, normal loads can vary significantly between different locations and turtle species. 64,132 For example, healthy loggerhead and hawksbill turtles can have more than 100 different epibiont species, whereas only a few common species would be expected on green turtles. External parasites and other epibionts may be collected by manual extraction and fixed in 95% ethanol 25 for morphologic identification and/or genetic sequencing. 8,105 Epibionts would usually not be considered to be harmful to the host by definition, but they have been suspected of being indicators of poor health and/or contributors to immunosuppression. It has been speculated that high loads of the turtle barnacle Chelonibia testudinaria (Fig. 8A) 93 on animals in poor body condition may indicate an inability to dive to clean by rubbing on abrasive surfaces or by accessing cleaner fish. 57,93 In North Carolina, no correlation between carapace C. testudinaria load and health parameters was found in a study of juvenile logger-head turtles, a species of turtle that has a high epibiont load in normal, healthy individuals. However, results from a more recent study of green turtles in Moreton and Shoalwater Bays, Queensland, Australia, has indicated that small immature animals with high Chelonibia testudinaria counts on the plastron are more likely to be clinically unhealthy than those with low counts. 36,132 The marine leech Ozobranchus margoi was suggested to be associated with fibropapilloma development (see below) 48,58 ; and there is localized inflammation, including fibrous tissue proliferation at sites of burrowing barnacles (Tubicinella cheloniae; Fig. 8B). 101 The barnacle, Platylepis spp., is frequently found on marine turtles, but has not been implicated in any pathology.

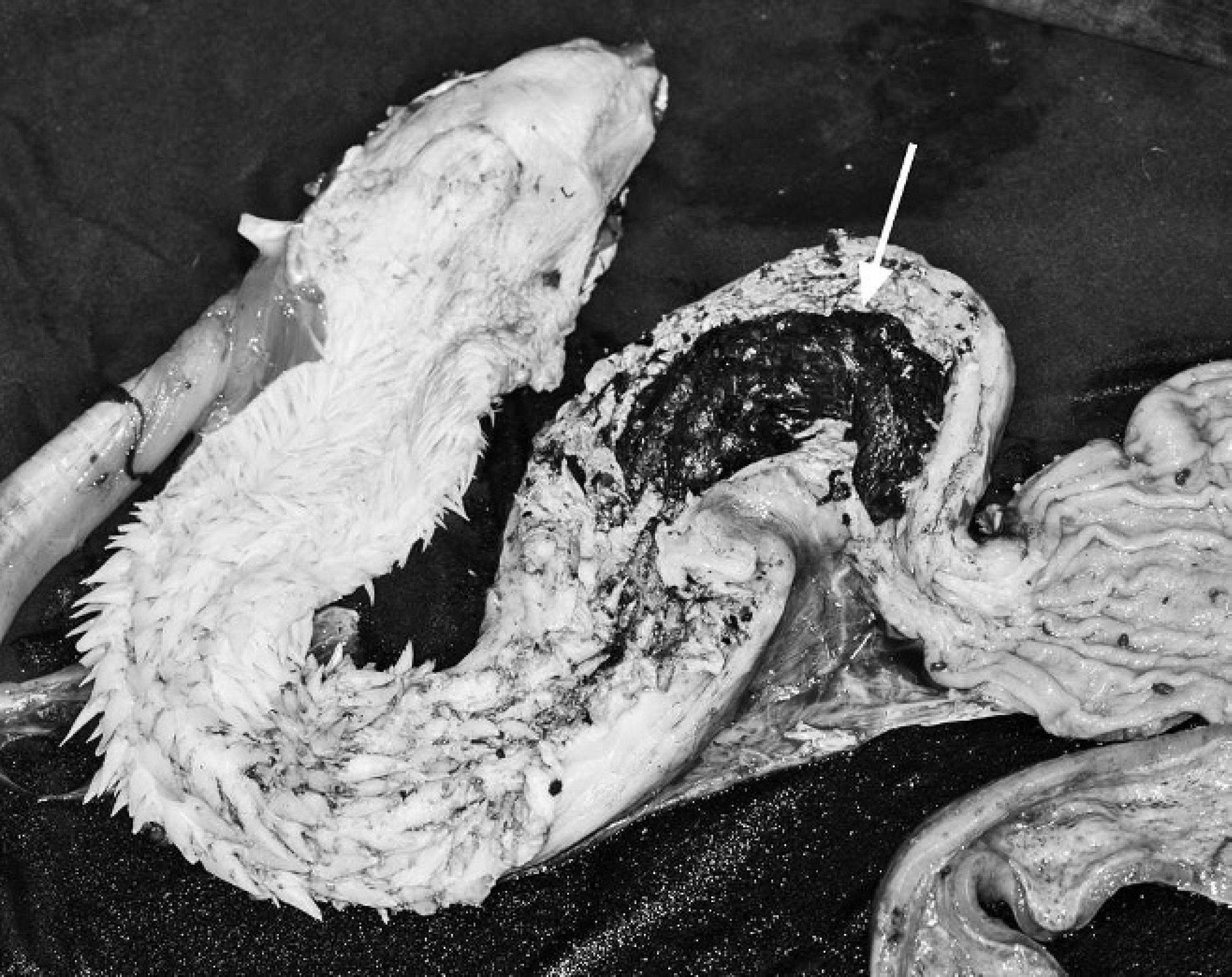
Plastron of an emaciated green turtle, showing bilateral coracoid processes penetration (arrows). The cranial edge of the plastron is at the top of the image.
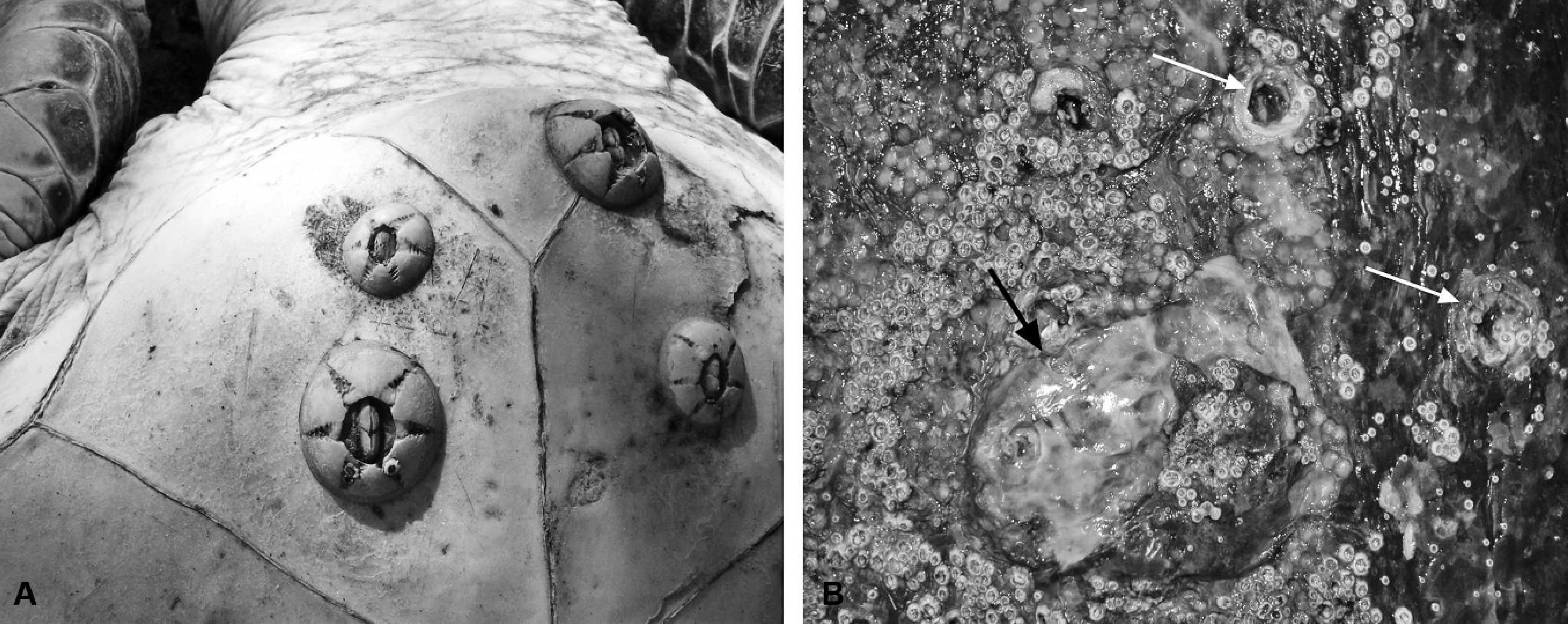
Barnacles.
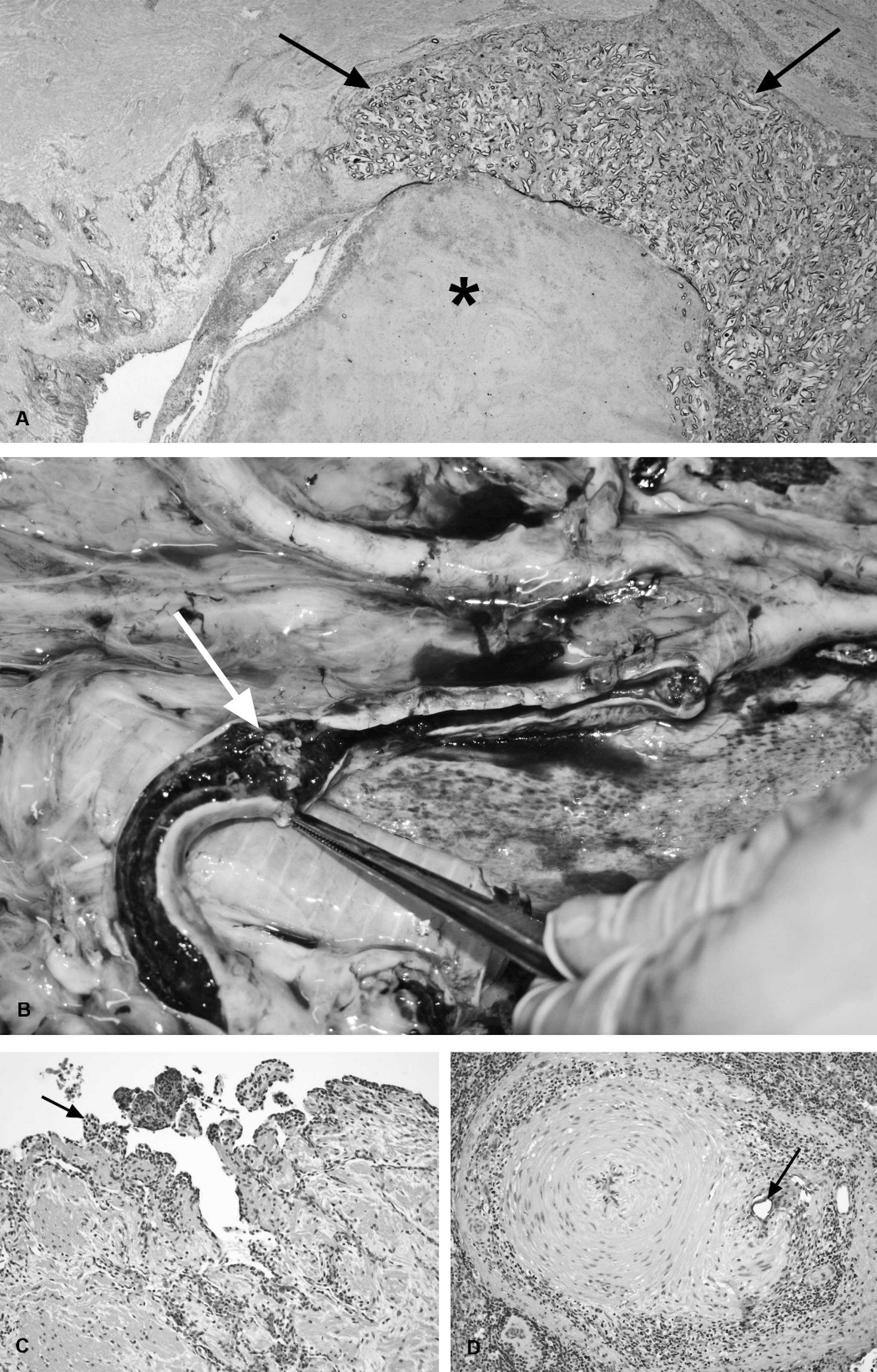
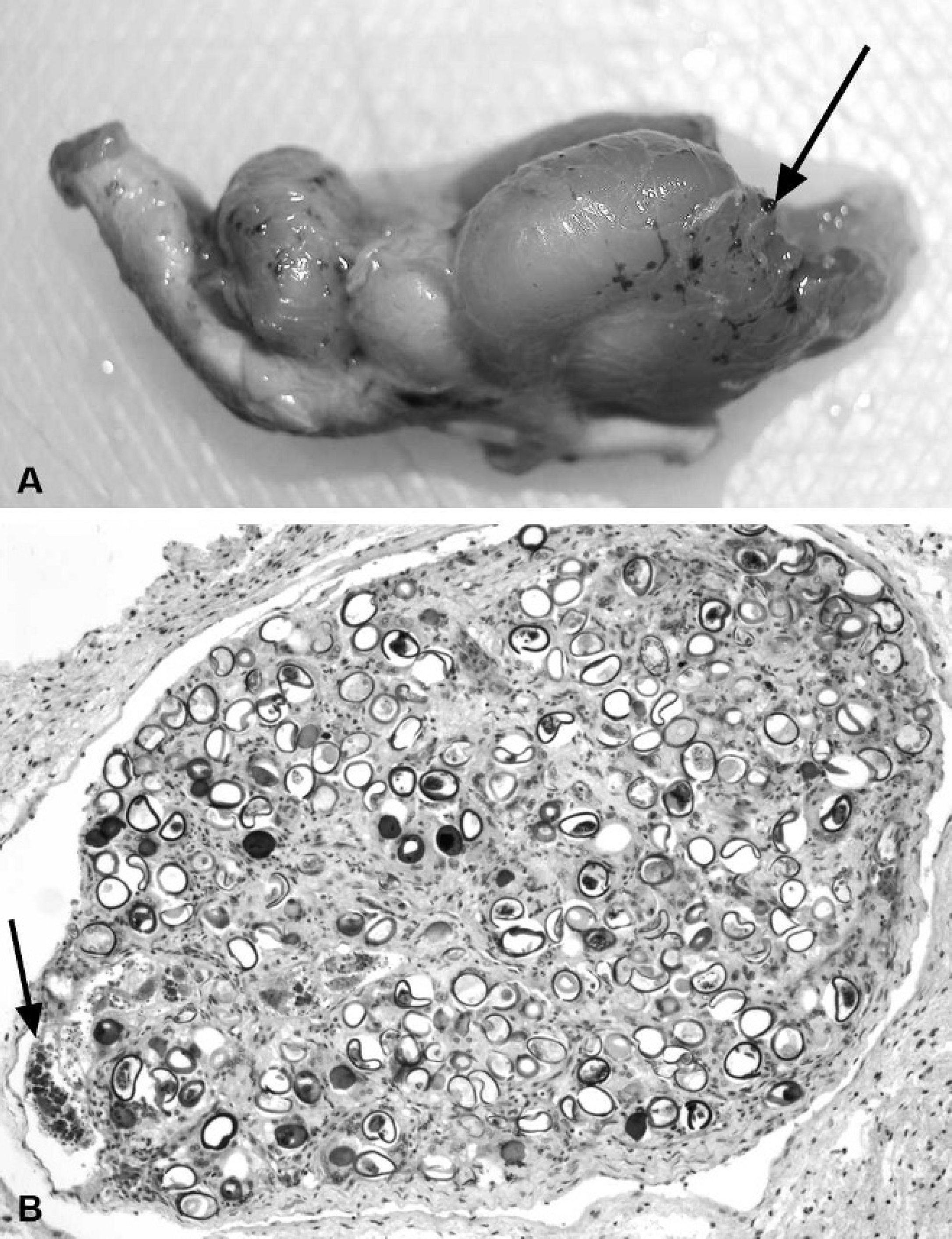
Fibropapillomatosis. Fibropapillomatosis (FP) is a common cutaneous and apparently infectious condition in all species of marine turtles, except the leatherback turtle. Marine turtle FP was first reported in Key West, Florida, in 1938, 127 followed by Hawaii, in 1958, 24 and has now caused numerous epizootics in green turtles worldwide, with significant public and scientific attention. 58 Juvenile turtles are most often affected with low levels of disease in adults and no reported lesions in individuals during or immediately after the pelagic life phase, which occurs after turtles hatch and before they become residents of shallow coastal waters as juveniles or subadults. 38,85 Papillary or smooth, flat, or nodular masses that range in size from <1 cm to >30 cm diameter develop mostly in soft tissues, including the mouth, eye orbits, neck, limbs, and tail; however, they may also arise on the plastron and carapace (Fig. 9A). 150,154 Fibropapillomas are usually gray to black to white, depending on location, and their color may correspond with local skin pigmentation. Tumors, including fibromas, myxosarcomas, and fibrosarcomas, may also be found in internal organs, including the lung, kidney, heart, intestines, and spleen (Fig. 9B). 58 The severity of disease in terms of numbers of turtles affected, and sizes and numbers of tumor masses, varies with geographic location, 47,49 although, even in heavily infected areas such as Hawaii, overall population survivorship has not been negatively affected. 17 Fibropapillomas are benign and do not usually cause death directly, but they may cause severe debilitation because of space-occupying effects or functional interference. 3 For example, tumors around the mouth and eyes can impede the ability to feed and locate food, respectively, and this may contribute to malnutrition; fibropapillomas in ocular sites may be locally invasive. Finally, internal tumors can compromise organ function or predispose to secondary diseases. In live turtles, FP has been linked to lymphopenia, chronic inflammation, immunosuppression, and systemic bacterial infections. 149,152
The etiology and pathogenesis of FP in marine turtles is only partly understood. DNA from an alphaherpesvirus (Chelonid fibropapilloma-associated herpesvirus [CFPHV]) has been detected in naturally occurring tumors, with identification of 5 closely related location-dependant variants. 59,117 Experimental inoculation of cell-free homogenates consistently resulted in horizontal transmission of the disease, suggesting a viral etiology, but the virus has not been cultured, and, therefore, Koch's postulates are not fulfilled. 58,62,63 In addition, genomic analysis has indicated that CFPHV was established in marine turtle populations before the disease was first described, and low levels of viral genetic material have on occasion been isolated from turtles without external tumors. 47,116 During localized outbreaks, turtles are thought to be infected with specific CFPHV variants after they arrive as juveniles in near-shore (neuritic) habitats. 34 The marine leech Ozobranchus margoi and the saddleback wrasse Thalassoma duperrey (a cleaner fish) have been suspected to act as mechanical vectors. 48,93,94 Progression to tumor development with outbreaks of the disease is likely to be multifactorial and involve environmental cofactors, such as seasonal elevation of water temperature, 58 high levels of anthropogenic activity in the immediate environment, 58 or exposure to potential tumor-promoting compounds, including okadaic acid produced by the benthic dinoflagellate Prorocentrum spp. 82 or products of Lyngbya majuscula (toxic cyanobacterium) blooms. 5 The immune status and nutritional status of the turtle populations also may be important. 4,9,152
Each tumor in an FP case should be measured by using a fiberglass or steel tape, and the location should be recorded. Total tumor burden should be scored as outlined by Work and Balazs. 149 Photographs of individual tumors allow for mapping of the progression or regression of the disease if the animal has previously been examined antemortem. Spontaneous regression of tumors was observed in turtles in Hawaii, Florida, and Queensland, Australia, 32,91 although the duration of FP and the frequency of regression still remains to be established for most locations. In Hawaii, the average length of time for a green turtle to have FP is 2–4 years, 9 but regression is uncommon in some geographic locations 147 and may be dependant on the degree of burden if immunosuppression is occurring. Resolved fibropapilloma masses may leave areas of scar tissue, the locations and dimensions of which should be recorded.
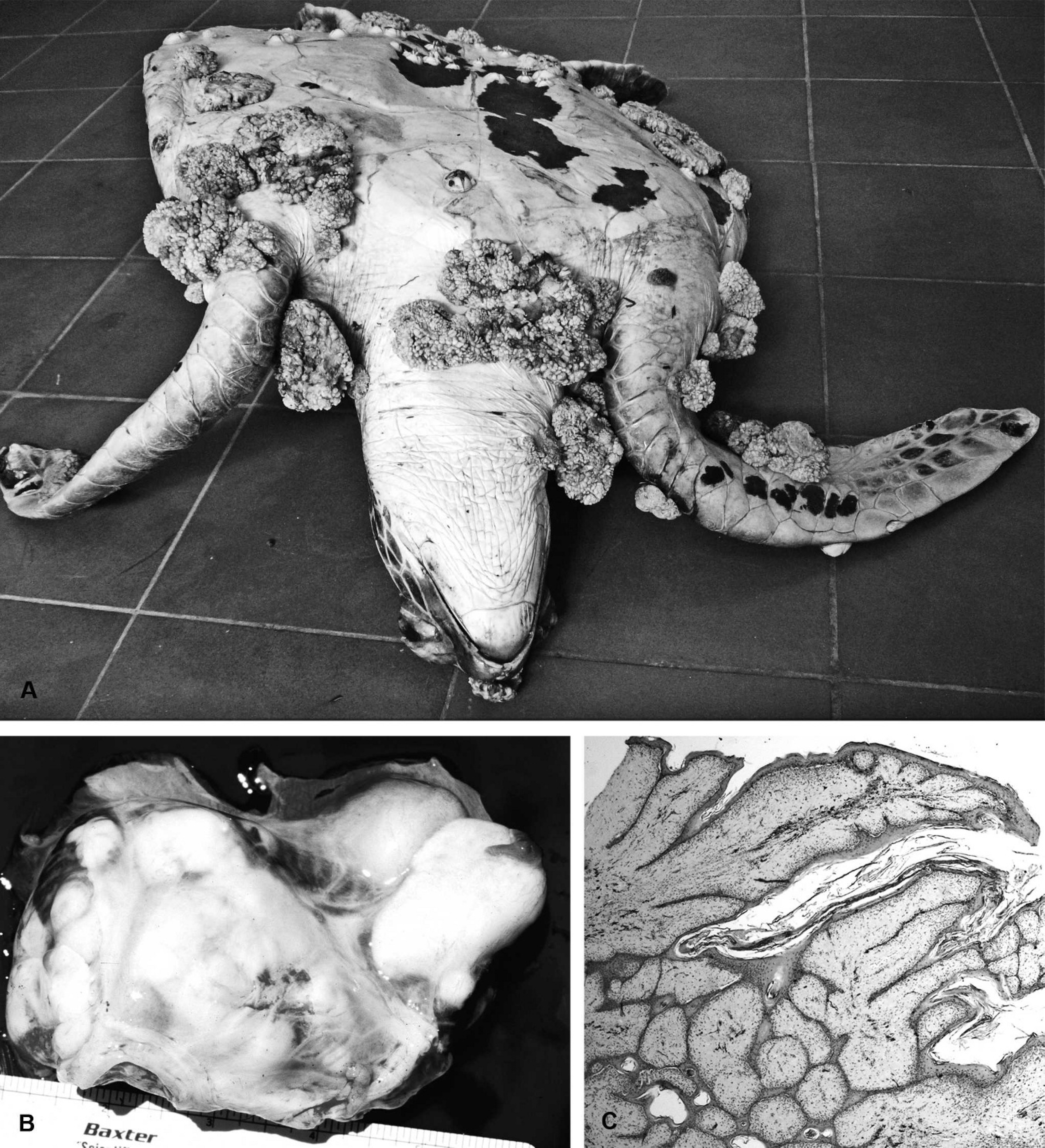
Chelonid fibropapillomatosis.
On histologic examination, the epidermis of the skin tumors is hyperplastic (up to 30 cells thick), often with orthokeratotic hyperkeratosis, and sometimes with cornified inclusion cysts. 58 Hyperkeratosis is not always noted if tumors arise from noncornified epidermal sites, including the cloaca and conjunctiva. 58 The epidermis is often thrown into papillary projections, with anastomosing rete ridges extending into the dermis (Fig. 9C). The basal cells are frequently vacuolated, with individual cell necrosis, and there can be dermal-epidermal cleft formation, with epidermal necrosis and ulceration. 58 Single-cell or more extensive vacuolation in the stratum spinosum may be associated with acantholysis and epidermal pustule formation. Areas of epidermal ballooning degeneration, with eosinophilic intranuclear (herpesviral) inclusions, were identified in some cases. 58,74 In ocular sites, corneal fibropapillomas were described as sessile and multinodular, with less arborization and more variable severity of epithelial hyperplasia than those arising from the limbus, conjunctiva, or eyelid. 16 In all sites, the underlying, thickened dermis contains numerous well-differentiated proliferating fibroblasts and abundant fine collagenous tissue, which may be myxomatous. 61,74 In pigmented tumors, dendritic cells that contain black pigment are noted in the dermis (Fig. 9C) and deeper layers of the epidermis (stratum basale and spinosum), with free epidermal and dermal pigment granules in the most heavily pigmented masses. 61 Many tumors contain granulocytic or lymphocytic dermal inflammatory cell infiltrates. Eggs of trematodes of the family Spirorchiidae and associated inflammatory changes have been noted in cutaneous and visceral tumors, and a causal relationship has been suggested but not proven by some researchers. 61,76,103,127
Recently, other herpesvirus sequences associated with ulcerative lesions in the oral cavity, respiratory system, and genital tract have been isolated from moribund wild loggerhead sea turtles 131 ; the lesions are described in the appropriate systems below.
Cardiovascular system
Turtles have a 4-compartmented heart composed of 3 true chambers and a functional sinus: 2 large atria, 1 ventricle, and a sinus venosus. The thick-walled ventricle is internally subdivided into 3 compartments: the cavum venosum, cavum arteriosum, and cavum pulmonale. The degree of separation of the systemic and pulmonary circulations varies with species. 153 The left and right dorsal aortas may anastomose at any point, from immediately caudal to the heart to just cranial to the kidneys, to form a single dorsal aorta. Documented pathologic changes to the heart include inflammatory lesions caused by spirorchiid trematode parasitism (see below) and fibropapillomas. In a necropsy study of 255 Hawaiian green sea turtles with FP, 11% had heart tumors. 150 In that study, the tumors were predominantly low-grade fibrosarcomas, with a predilection for the right atrium. On gross examination, the tumor masses were firm, white, and well circumscribed. 150 Necrotizing inflammatory foci caused by chlamydial infection have been reported in the heart and other organs of mariculture-reared green sea turtles in the British West Indies but have yet to be reported in free-living populations. 65 Diagnosis can be determined by histochemical staining and immunohistochemical labeling if necessary, with potential confirmation by using molecular methods. Iatrogenic crystalline deposition after euthanasia with barbiturates may be noted in some turtles and should not be misinterpreted.
Spirorchiid trematode infections. The most common lesions involving the heart and vasculature are associated with these parasites. Marine turtle spirorchiids have frequently been reported from Australia, 22,39,42,118 Hawaii, 23,151 and the Caribbean. 31,123 In one study, conducted in Moreton Bay, Queensland, Australia, 98% of green turtles were found to have spirorchiid trematode infections at postmortem. 42 Frequencies in other regions of the world are slightly lower. 23,39 Spirorchiids are related to schistosomes, the blood flukes of humans and other vertebrates. No life cycle is known for any marine spirorchiid and their biology, in general, is remarkably poorly understood. It is almost certain that the worms cycle between marine gastropods and turtles, with motile cercariae exiting the snail host and penetrating directly into the definitive host. At least 10 widely distributed genera of spirorchiids are known to occur in sea turtles, 114 but considerable taxonomic confusion remains. It is common to find spirorchiid eggs in many organs, even when adult worms escape detection either grossly or histologically. The adults infect blood vessels of various organs and grossly can often be found in the heart chambers and distal aorta. 83 Associated gross and histologic lesions can include mural endocarditis, arteritis (Fig. 10A), thrombosis (frequently accompanied by aneurysm formation) (Fig. 10A, 10B), nodules that contain parasites, papillary proliferations into the lumens of blood vessels (Fig. 10C), luminal calcium deposits, perivascular edema, and infarcts. 4,42,118 Walls of muscular arteries may be thickened in certain organs, including the lungs, intestine, and spleen, which may potentially result in hypertension in the antemortem patient, although limited studies on marine turtle blood flow have been conducted. 140 The spleen is a very common site of perivascular spirorchiid egg deposition, with frequently associated hypertrophy of the muscular walls of arteries and arterioles (Fig. 10D); the latter changes are not invariably noted in other organs in the same animal. Thrombi appear to resolve by exteriorization of the granulomatous inflammatory response through the vessel wall, resulting in nodules on and/or surrounding the adventitial surfaces. 42 On microscopic examination, the eggs are noted in highly variable numbers adjacent to blood vessels in virtually any organ within variably sized granulomas that may be observable grossly (up to several millimeters in diameter), particularly on intestinal serosal surfaces (Fig. 11A, 11B). 31 Because lesions associated with spirorchiid trematode eggs will be seen in most turtles, it is a matter of experience in terms of estimating whether they are more numerous, associated with more severe inflammatory lesions, or altered in distribution versus “normal background pathology” for that particular population or subpopulation. On serologic examination, 71–100% of green turtles in Hawaii were found to be positive for antibodies against adult worm antigens but that did not indicate the intensity of infection or the species involved. 45,151 Tissue egg burdens were found to differ regionally in green turtles in the Hawaiian Islands and to increase with deteriorating body condition. 151 Severe lesions associated with spirorchiid trematode infections are often spatially associated with other disease processes, for example, FP, coccidiosis (Caryospora sp.), and disseminated Gram-negative bacterial infections (with or without septic thrombosis), including Escherichia coli, Citrobacter freundii, and Moraxella sp. 42,118 There is no information on whether these infections potentiate each other or simply represent an overall increase in infectious disease as a result of other (possibly environmental) factors.
Respiratory system
Documented pathologic processes that involve the trachea and lungs of free-living marine turtles include viral, bacterial, or fungal infection, parasitism, neoplasia, and obstruction. Loggerhead genital-respiratory herpesvirus and Loggerhead orocutaneous herpesvirus have been associated with airway lesions in 2 animals from Florida waters, with histologic epithelial lesions, including hyperplasia, syncytial cell formation, intranuclear eosinophilic inclusion bodies, and heterophilic inflammation. These are the first herpesviruses other than CFPHV to be diagnosed in wild marine turtles and are considered to be opportunistic pathogens in already debilitated animals. 32 Bacterial infections (usually Gram negative) may occur in the lung in association with lesions due to spirorchiid trematode infection, as in other organs; such parasitic lesions are common in lung tissue and are of highly variable severity. Fungal pneumonia has occasionally been reported 29,107 ; systemic mycoses are thought to be common sequelae of cold stunning but have also been seen in the absence of such events. 112 Tumors, when they occur in the lungs of turtles with FP, tend to be fibromas or myxofibromas. In one Hawaiian study, 40% of green turtles with FP had oral tumors that often seriously impacted the glottis 7 and were associated with pulmonary necrosis, suggested to be because of aspiration of foreign material. 150 Metastatic neoplastic masses (unrelated to FP) were reported in rare cases, including squamous cell carcinoma and lymphoma. 109,110 Obstruction of the cranial respiratory tract has also been caused by attempted ingestion of novel food sources, such as porcupine fish, which has resulted in deaths in several incidences. 92
Lymphoid system
Primary lymphoid organs in reptiles are the bone marrow, spleen, and thymus, with secondary lymphoid tissue in the gastrointestinal tract and airways as in other vertebrates, in addition to some other lymphoid aggregates. It should be noted that bone marrow in turtles can be found in the carapace, plastron, and pelvis, in addition to long bones. In the spleen, arterioles in the white pulp are surrounded by periarteriolar lymphoid sheaths and capillaries by periellipsoidal lymphoid sheaths; germinal centers do not occur. 106 Granulomas centered on spirorchiid trematode eggs are often noted in these sites, with highly variable severity (as stated above). The reptilian thymus is similar to that in mammals, being a site of maturation of T lymphocytes. Thymic cysts may be noted; they are associated with epithelial cells, may be intracellular or extracellular, and are of unknown origin and function. 106
There is a strong seasonal influence on the immune status of marine turtles, caused by both temperature changes and the breeding cycle; decreased thymic mass has been reported during winter torpor (decreased activity), with regeneration in the spring. In males, lymphoid mass is greatest after breeding activities have been completed and testosterone levels decline. 95 In chronically sick marine turtles, the thymus becomes thin and less well demarcated, as it does in aged animals (i.e., involution is progressive). There has been no systematic examination of the relationships between acute and long-term stress on immune function in marine turtles, however, tools that were previously not available to measure the immune status of sea turtles, including complete blood counts and lymphocyte stimulation tests, may now be used.
Gastrointestinal system and liver
Identification of foreign bodies in marine turtle gastrointestinal tracts may be useful in identifying the point source (originating location and/or element) of the foreign body. For example, in the case of fishing hook and line, experts may view a photograph to determine the type of hook and line from which they may accurately identify the specific recreational or commercial fishery industry. In Queensland, this type of point-source investigation has resulted in successful negotiations to protect both the turtle populations and the fishing industry. With respect to plastic ingestion, it is still not known where (urbanized areas vs. open ocean) and how (mistaken vs. byproduct ingestion) fatal or incidental plastic ingestion occurs, so identifying location and types of plastics with accurate records is vital.
Vascular lesions associated with spirorchiid trematode infections.
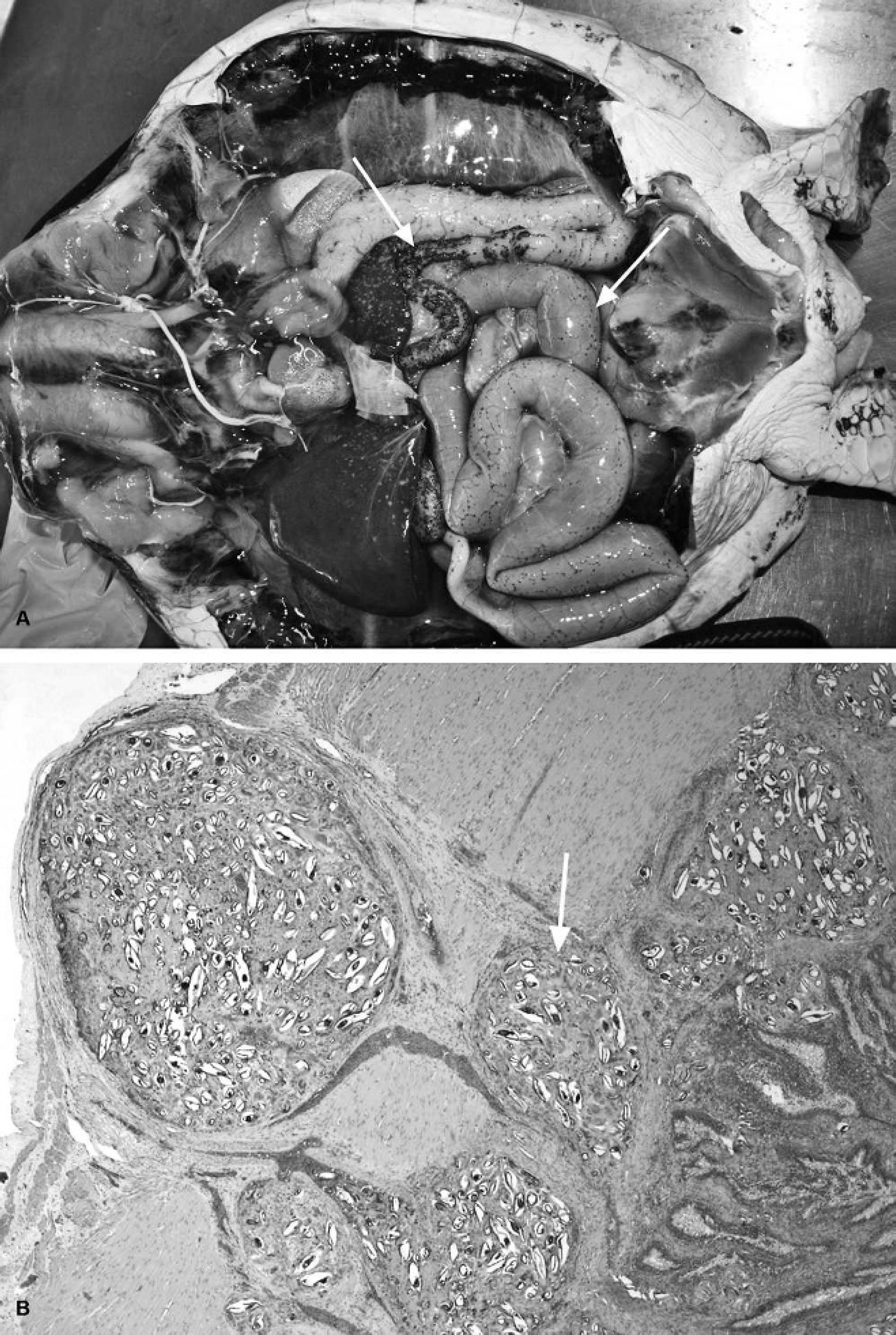
Lesions associated with spirorchiid trematode egg deposition in the intestinal tract.
As marine turtle digestive rates are slow in comparison to mammals, collecting digesta and/or fecal content from each region of the intestinal tract is warranted, because it may indicate changes that have occurred in dietary composition over a long period of time. It is not uncommon for an emaciated turtle that is known to not have eaten for months to have undigested seagrass in its crop and a progression of digested food to fully formed feces in its large intestine. Gut fill should be subjectively estimated as a percentage (of an entirely filled gut) on removal.
Anthropogenic marine debris is a significant cause of impaction and entanglement in marine animals. 81 Ingestion of foreign material is believed to be common in marine turtles. Although this is speculated to be greater in pelagic than in benthic dwelling turtles, the extent of pathology and mortality caused by this condition is largely unknown. Studies in Australia indicated synthetic materials, including milk bottle containers, balloons, and fishing lines and nets, contribute over half of the measured marine debris along its coastline. 143 Marine debris was found in the gut of 9 of 78 green and loggerhead turtles examined during a postmortem survey in southern Queensland. In this study, plastics, when present in high levels in the gut, were found to contribute to intestinal impaction, and fishing lines and hooks (as linear foreign bodies) were found to cause intestinal plication and/or rupture. Two additional animals of the 78 turtles examined were suspected to have asphyxiated because of fishing net entanglement (Flint, unpublished observation). Similarly, synthetic flotsam was reported in more than half of the examined green and loggerhead posthatchling turtles stranded in southern Queensland. 15
Esophagus, crop, and stomach. In the leatherback turtle, the esophagus extends half the length of the body before returning cranially to the level of the axilla. In the other marine turtle species, the esophagus descends to a point just inside the celomic cavity before joining the stomach. 153 In adult green turtles, the esophagus comprises 3% of the gastrointestinal tract. 137 The mucosa has an unusual appearance, with numerous sharp, keratinized papillae (spines) that should be rigid and directed caudally in the cranial portion (Fig. 12); on histologic examination, they are covered by stratified squamous epithelium, and no glandular tissue is present. 137 In turtles in poor condition, these spines may be flaccid. Foreign bodies, including fishing line and hooks embedded into the mucosa, are common findings; although, depending on the location, they may not necessarily cause a significant problem. Ulcerative and fibrinous esophagitis, esophageal penetration, and impaction with crude oil balls are further lesions that were observed at necropsy. 108 A muscular pouch termed the crop is found at the base of the esophagus of green turtles from the Pacific Ocean (Fig. 12); the mucosa has a stratified squamous epithelium and contains small or large mucous glands in herbivorous and carnivorous species, respectively. As in birds, the crop is not an organ for chemical or mechanical breakdown of food, functioning solely for retention of food. In adult green turtles, the crop comprises 2.4% of the gastrointestinal tract and as little as 0.9% in loggerhead turtles. 137 The stomach may be divided into cardiac, fundic, and pyloric regions, as in other species. In adult green turtles, the stomach comprises 6% of the gastrointestinal tract and 3.8% in loggerhead turtles. 137 The gastric mucosa has transverse rugae and may contain ulcers, inflamed areas, and foreign bodies. Larval nematodes of the family Anisakidae were reported in the stomachs of turtles in the Canary Islands, with associated transmural granulomatous inflammation and mucosal ulceration, 108 as were the larval ascaridoid nematodes Sulcascaris sulcata in the stomachs of loggerhead turtles in Western Australia, also noted to be causing ulceration. 84
The opened esophagus, showing numerous caudally directed, sharp keratinized spines on the mucosal surface. The esophagus leads into a crop (white arrow) in green turtles from the Pacific Ocean that functions to retain ingesta.
Small and large intestine. As in other reptiles, the transitions from duodenum to jejunum to ileum are difficult to discern grossly or histologically. In adult green turtles, the small intestine comprises 26.2% of the gastrointestinal tract and 50% in loggerhead turtles. 137 The duodenal mucosa has a honeycomblike appearance grossly and produces significant amounts of mucus; the cranial duodenum contains the ampulla of Vater that is often discolored by bile in healthy animals. In leatherback and green turtles, there are overlapping crypts to increase surface area along the length of the duodenum and into the jejunum that are lined by mucous epithelial cells. 153 The ileum ends in a sphincter (the ileocecal valve), after which is the bulging caecum and then the colon; the caecum is more prominent in the green turtle compared with other species. The colon narrows past the caecum to be constricted weakly by segmentally arranged muscle bands. In adult green turtles, the colon comprises 65% of the gastrointestinal tract and 44% in loggerhead turtles. 137 Distally, the colon tapers to form a thickened and folded muscular rectum, which may be pigmented. 153 The rectum, urinary bladder, and gonadal duct (via the urogenital papilla) enter the muscular cloaca. Common grossly observable lesions in the intestinal tract include intussusceptions, strictures, blockages, plication, torsion, rupture, and ileus; many such lesions will be associated with ingested foreign material, including fishing line and hooks, and byproduct material of forage feeding behavior, including sharp skeletal fragments of fish, crabs, and mollusks. There may be associated diarrhea, constipation, localized infection, or generalized septicemia. 102 Fibrinous, catarrhal, and necrotizing enteritis are frequently diagnosed, and may be associated with the above lesions or with bacterial infection and/or multisystemic septicemic lesions. Where chronic and nonspecific intestinal inflammation is noted, and there is also gross evidence of intestinal impaction, blockage, or stasis, it is usually difficult to determine what the primary process was. Histologic lesions in those cases can include mucosal erosion, edema, small intestinal villous blunting and fusion, crypt hyperplasia, and increased numbers of intramucosal inflammatory cells (diagnosis of the latter requires the pathologist to have experience with these species).
Some specific infectious agents may be noted in the gastrointestinal tract: ulcerative gastrointestinal lesions have been described in cases of Loggerhead orocutaneous herpesvirus and Loggerhead genital-respiratory herpesvirus (herpesvirus infection; described above in the respiratory system) 131 ; granulomas in the intestinal wall, as in other sites, may contain bacteria; granulomas associated with eggs deposited by spirorchiid trematodes are common findings and may be seen grossly in variable (sometimes very large) numbers as dark raised masses several millimeters in diameter on serosal surfaces (Fig. 11A, 11B); and various protozoa may be noted either histologically or in fecal specimens, the presence of which is not necessarily correlated with (clinically) significant disease (Gordon AN: 2005, A necropsy-based study of green turtles (Chelonia mydas) in South-East Queensland. PhD dissertation. School of Veterinary Science, p. 234. The University of Queensland, Brisbane, Australia). 43,46,139 Protozoal intestinal parasites in marine turtles include the genera of coccidia Caryospora cheloniae, Emeria caretta, and Cryptosporidium spp. 43,46,139 Caryospora cheloniae has been documented as the cause of epizootic mortality of green turtles in the Moreton Bay area of Queensland, Australia, 43 and is a continuing cause of acute disease. Gross lesions include exudative enteritis that is fibrinous or necrotizing in some cases, and a fluid-filled atonic stomach and intestinal tract with mucosal hemorrhage (Gordon AN: 2005, A necropsy-based study of green turtles). On histologic examination, hyper-plastic enterocytes contain gametogenous coccidial stages and schizonts (Fig. 13A); protozoa may also be noted in renal tubular epithelial cells (Fig. 13B), the thyroid gland, and in perivascular locations in the brain. 43
Liver. On gross examination, discoloration of the liver may be noted. Melanomacrophages (or other pigmented macrophage centers) are normal cellular populations in the liver, as in other reptiles, where they phagocytose erythrocytes and foreign material. Black discoloration may be discernible when sizes and/or numbers of the melanomacrophage centers increase (melanomacrophage hypertrophy or hyper-plasia). 129 On histologic examination, there is a wide range of severity in this nonspecific response that is assessed subjectively by the pathologist (Fig. 14A-C) and has been associated with emaciation, stress, chronic inflammation, and chronic bacterial infection. 20,129 The liver may alternatively be pale and swollen because of increased lipid deposition, as in other species, that on histologic examination may be macro- or microvesicular. Hepatic lipidosis is poorly understood in reptiles but can occur because of or after a number of insults, including cachexia, toxicosis, and metabolic abnormalities. Inflammatory lesions, including necrotizing and granulomatous processes, are also frequently noted in the liver, 108 including reactions to spirorchiid eggs and/or bacterial or fungal infection. In turtles that are emaciated, hepatocytes are usually atrophic, with resultant loss of liver parenchyma, and the liver as a whole may be reduced in size. Distension of the gall bladder may indicate cachexia, as in other species. Normal bile color is similar to that of mammals, being dark green.
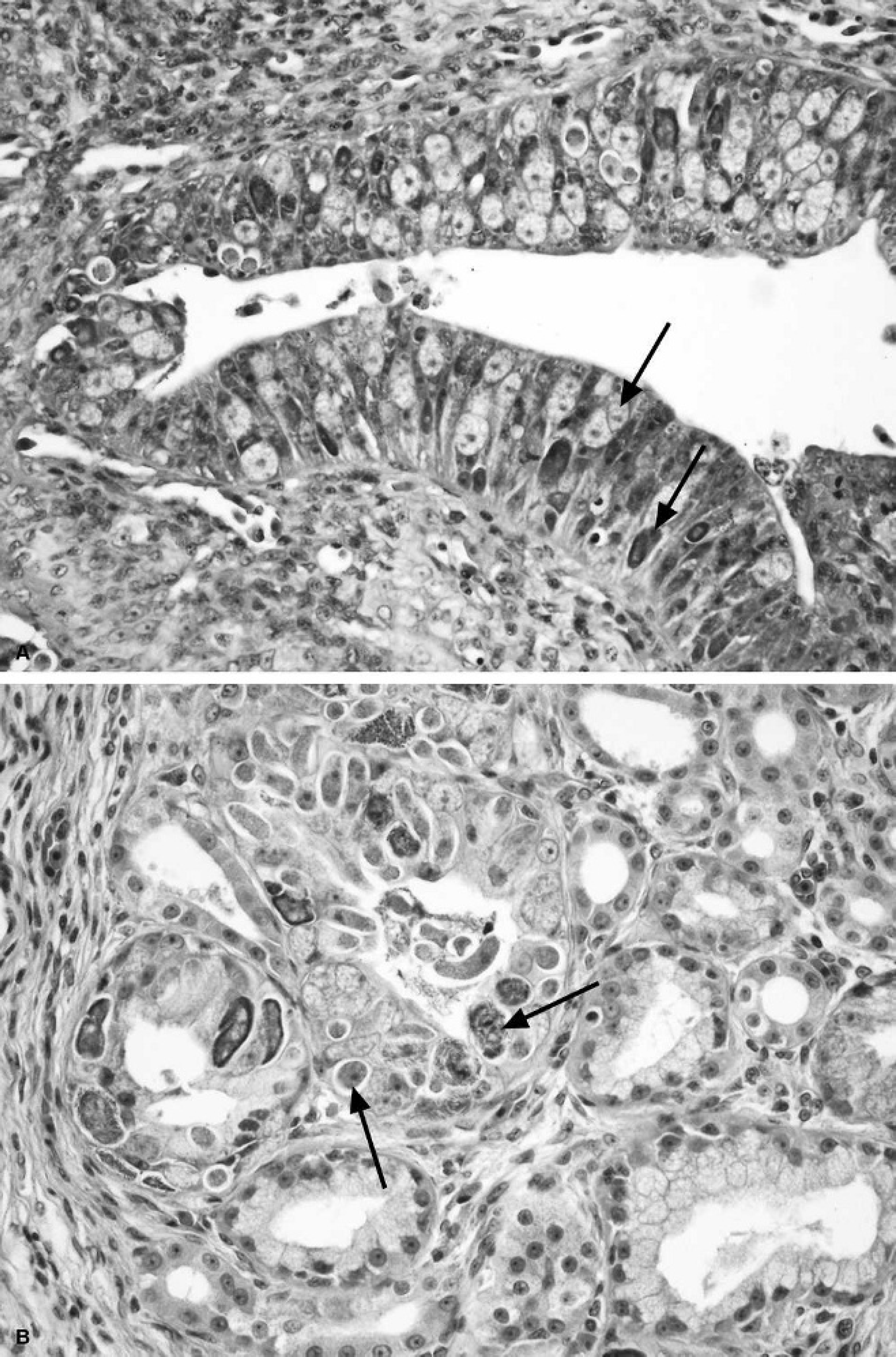
Caryospora cheloniae infection in a green turtle from Moreton Bay in Queensland, Australia.
Abnormal bile may be crystalline or contain parasites. The pathologic capacity of spirorchiid gall bladder parasites has been reported to include proliferative multifocal endoarteritis of the small peripheral vessels, potentially resulting in localized vessel rupture and release of the parasites into the bile. 124
Genitourinary system
Kidneys and urinary tract. The kidneys of marine turtles are retroperitoneal and lobular, and lack a grossly distinct cortex and medulla. The lungs extend to the cranial poles of the kidneys near the adrenal glands; adrenal glands are small, elongate yellow-orange structures that, as in other reptiles, do not have outer cortical and inner medullary regions. Renal lesions reported in marine turtles include chronic, fibrosing and lymphocytic interstitial nephritis, granulomatous nephritis (including that associated with spirorchiid eggs), perinephric abscesses, tumors, and renal calculi. 96,108,130 Fibropapilloma tumors of the kidney were noted to occur in 9% of the 255 necropsied stranded green turtles in Hawaii. They were reported to displace the interstitium of the kidney, causing atrophy of the renal tubules. 150 They were described as white firm nodules that may contain Spirorchiid parasite eggs. 103 Granulomatous inflammation associated with renal calcium oxalate deposition was reported in 18 green turtles in Florida and Costa Rica but was considered to be subclinical and of dietary origin 131 and was also noted in green turtles in Queensland, again at levels not considered to be clinically significant (Patterson-Kane, unpublished observation).
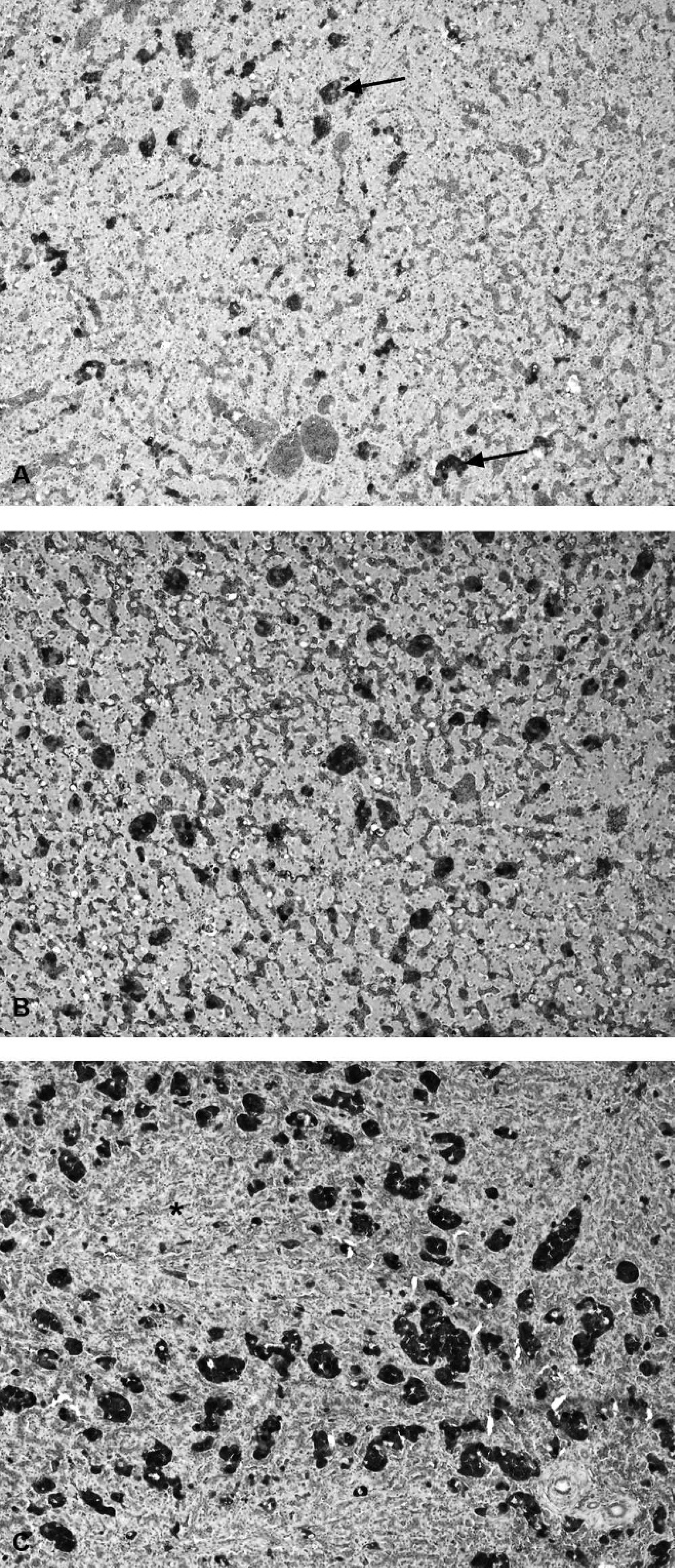
Degrees of melanomacrophage hypertrophy and hyperplasia in the liver (as assessed subjectively), a nonspecific response associated with numerous factors, including emaciation, stress, and inflammatory disease.
The urinary bladder in normal animals may contain both urine and mucus; the mucus should be clear. Irregular dark mottling of the tan mucosa is a normal finding. In cases of intestinal impaction, retrograde flow of digesta can occur from the cloaca. Also, the bladder is a potential site for calculus formation, 96 and trematode infection (Pyelosomum spp.), which usually does not lead to significant pathology. 122
Genital systems. The gonads of turtles are extensive and may extend from a point cranial to the kidney to the level of the urinary bladder. They tend to be located along the dorsal surface of the celomic cavity but become pendulous at maturity. In small immature marine turtles, gonads have a similar appearance, irrespective of sex, but sex may be identified either by gross or microscopic examination. Paired ovaries contain numerous follicles that range in size from 2 mm to several centimeters, depending on breeding status and age. In immature female turtles, the ovaries are tan to pink, with a grainy structure because of numerous tiny follicles, and white straight oviducts lying laterally to each ovary. At sexual maturity, mature (bright yellow) follicles will tend to cluster on the cranial aspect of the ovary. Adult females can be identified by the presence of a tortuous enlarged oviduct as they prepare for breeding. In addition, mature female turtles that have ovulated will have large follicles and scar tissue in the ovaries. Scar tissue is classed as corpora luteum (yellow body) from recent ovulations, becoming corpora albicans (white body). In small immature males, the epididymis tracks within the body wall (retroperitoneal). It may be grossly discerned from female reproductive organs, because it has a convoluted vas deferens. At sexual maturity the testes, epididymis, and ductus deferens change in shape and size. The epididymis becomes pendulous and distends from the body wall, with seminiferous tubules that range in diameter from 1 mm to several millimeters, also depending on breeding status. 53 There are few reports of specific lesions of the genital tract. Ulcers of the phallus were described in one loggerhead turtle with herpesvirus infection, 131 and Norton 102 outlined penile and cloacal prolapses, dystocia, and urolithiasis as potential conditions in various species of turtle and tortoise.
Central nervous system and eyes
Central nervous system. Prosectors often neglect to obtain the brain, although removal is easily achieved by the above mentioned method (Fig. 5A, 5B), even in the field, and it is an essential organ to examine as a common site of significant pathologic change in marine turtles found stranded or dead. 113 The entire brain should be fixed appropriately, as for other species, and sectioned transversely, with processing of all sections for histologic examination. It is useful to note that a segment of the proximal spinal cord may be frozen for future examination as a representative of the central nervous system. This reserves the brain for histopathologic examination. Traumatic injury to the skull and spine with meningeal hemorrhage have been associated with a boat strike 108 and concussive injuries caused by blunt objects. Granulomatous inflammation associated with intravascular and perivascular spirorchiid trematode eggs may be noted grossly in the meninges (Fig. 15A) and histologically in both the meninges and parenchyma, often associated with small intravascular adult parasites (Fig. 15B). Clinical neurologic disease associated with spirorchiid infection was reported in small numbers of stranded green turtles in south-eastern Queensland, Australia, and in subadult loggerhead marine turtles in south Florida. 42,75 Adult trematodes were dissected from the meningeal vessels of both turtle species and identified as Neospirochis sp. 42,75 ; it was suspected but not proven in the loggerhead turtles that chronic exposure to a novel toxin present in the diet may also have contributed to the clinical signs and pathologic lesions. 75 Abscesses or heterophilic granulomas associated with gram-negative bacterial infection (with or without association with spirorchiid egg lesions) may be noted in the brain as in other organs. In green turtles with Caryospora cheloniae infection, protozoa may be found in brain tissue associated with marked meningoencephalitis; inflammation is particularly intense where schizonts have ruptured. 43 Such infections have been associated with significant clinical neurologic signs that necessitated euthanasia.
Eyes. Few publications discuss the range of eye lesions that marine turtles may experience. These range from the simple, such as sunken eyes in a fresh carcass (D1 or D2 carcass), which indicate dehydration, 102 to the more complex. A previous study described yellow deposits on the cornea that were most likely keratoconjunctivitis and ulcerative blepharitis, which were thought to be from trauma and secondary bacterial infection in green turtles farmed on the Torres Strait Islands in the late 1970s. 40 Fibrinous deposits on the cornea, infiltration by the marine leech Ozobranchus margoi, and fibropapilloma tumors growing on the cornea in green turtles have been described as a rare but emerging manifestation of FP. 16,35,74
Spirorchiid trematode-associated lesions in the meninges of the brain.
Osmoregulatory system
Salt glands (Fig. 5C) are large in all marine turtle species but are particularly prominent in leatherback turtles. They assist in osmoregulation by excreting solutes via the lacrimal ducts. Active transport via sodium-potassium pumps moves salt from the blood into the glandular tissue. This process is required because reptilian kidneys are significantly less efficient than those of mammals. Salt glands should be firm, lobular, and pink to brown. Common abnormalities include pale spots or calculi, which indicate malfunction in solute dissolution, which may occur in severely dehydrated marine turtles. This manifests as hard rugose deposits associated with necrosis of surrounding tissues. Granulomas associated with spirorchiid eggs, of variable severity, are common in stromal tissue that surrounds central canals of lobules and may sometimes extend into and disrupt the glands themselves.
Musculoskeletal system
Muscle. Cachectic muscular atrophy has been reported in marine turtles. It is frequently associated with concurrent diseases, including gastrointestinal disorders, immunosuppression (FP), or traumatic lesions. 40 Pale skeletal muscle is also often seen secondary to anemia and cachexia.
Bone. Fibrous osteodystrophy, 40 stress fractures, remodeling osteoarthritis, 119 and FP 150 were reported in green and loggerhead turtles. Diseases of the skeletal system have been reported to be uncommon 40 ; however, trauma-related skeletal conditions, such as fractures and amputation are frequently seen during postmortem. Osteoarthritis was reported in a stranded Kemp's ridley sea turtle at a rehabilitation center in Baltimore, Maryland, associated with systemic Mycobacterium cheloniae infection. 51 FP tumors were noted to invade bone in both firm and cystic forms in 4% of green sea turtles in a retrospective necropsy study in Hawaii. 150 Bone marrow in fresh samples should be examined. Splitting the distal femur should provide an adequate sample for immediate fixation to detect changes in immune status. 106
Ear. Although not externally apparent, the ear of aquatic chelonians has a thin tympanic membrane, and, in terrestrial chelonians, the ear is often susceptible to abscess formation. 71 In contrast to terrestrial turtles, lesions of the ear in marine turtles have not been documented, and prosectors should remember to examine these structures when possible. Transverse sections of the skull can be made by using a band saw after removal of the brain to access the middle and inner ear. This also facilitates access to the nasal cavity for examination and sampling.
Sampling protocols
Sex determination in immature turtles
As stated above, sex is best determined via internal examination of the gonads. However, blood samples collected immediately after death and assayed for testosterone show males have higher concentrations (>20 pg/ml) than females (<10 pg/ml). 13 This method is expensive, and the accuracy is variable. A blood sample may be collected via cardiac puncture if the carcass is fresh, however, if the turtle is not being euthanized, it is preferable to collect it antemortem via the cervical dorsal sinus (Fig. 16), external jugular vein, or popliteal vein. 30,111,154 Under these circumstances, blood biochemistry and hematology analyses should also be performed whenever possible, because this may contribute valuable information to population disease investigations. 54,73,144
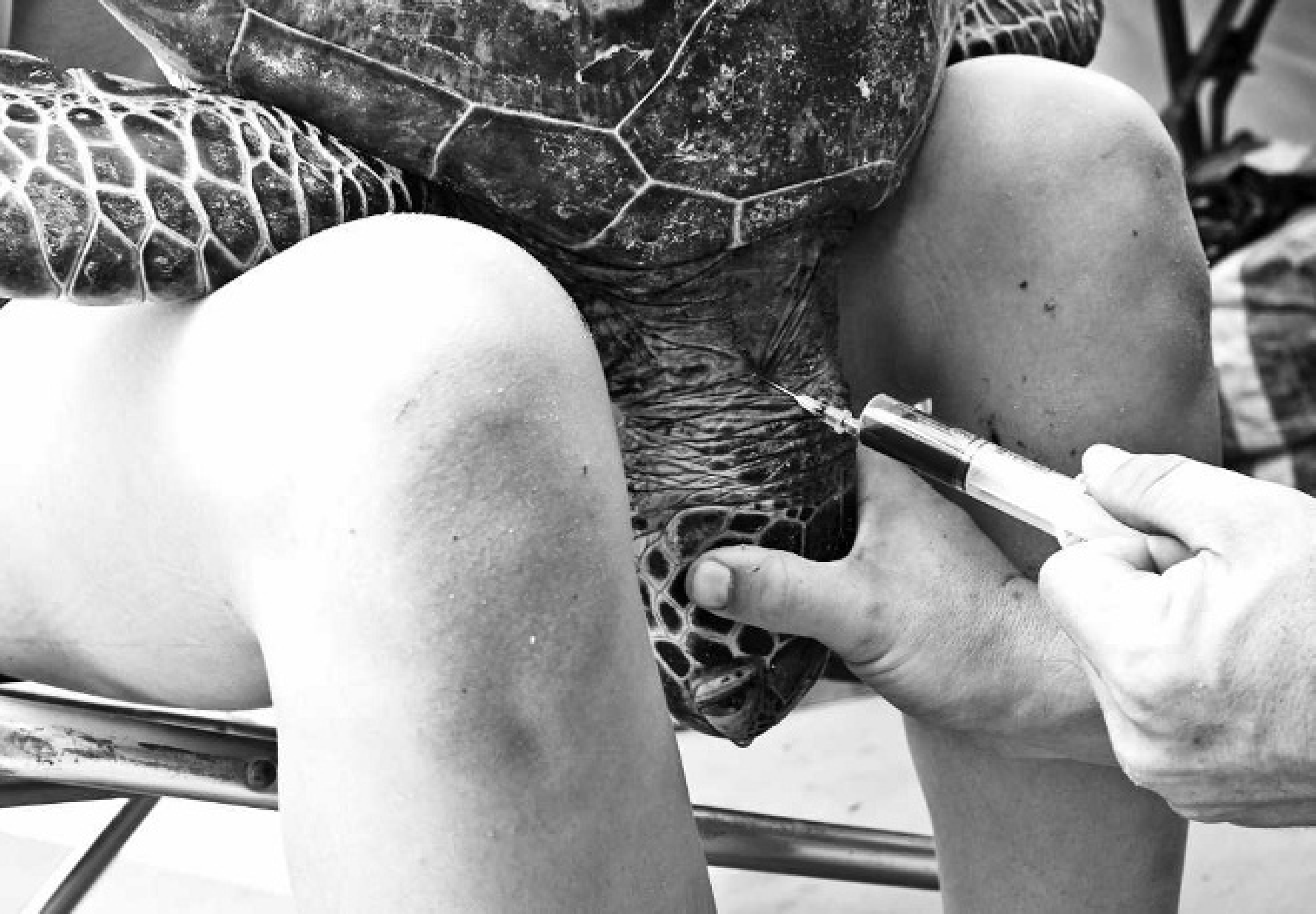
Collection of blood from the cervical dorsal sinus, antemortem. The sinus is located deep to the biventor cervical muscle when a needle is inserted on the medial aspect of muscles. Because of low circulatory pressure, inverting the turtle achieves increased sinus pressure.
Routine specimen collection and storage for histologic examination
At the conclusion of the postmortem, ensure that a minimum data set of the following tissue samples have been collected: heart, aortic tissue, adrenal glands, thymus, thyroid gland, liver, gall bladder, tongue, esophagus, crop, stomach, duodenum, spleen, pancreas, jejunum, ileum, caecum, colon, trachea, lung, urinary bladder, kidneys, gonads, spinal cord, brain (entire including pituitary gland), salt glands, eyes, skeletal muscle, skin, nasal mucosa, and bone marrow. Tissues should not exceed 3 mm in thickness, except for the brain, which may be preserved whole. All tissues should be stored in 10% formalin at a ratio of 10 parts formalin to 1 part tissue, for at least 24 hr.
Specialized sampling techniques: parasitology
Parasites can be identified based on their morphology and, increasingly, their genetic sequences. The likelihood of positive identification is increased if fresh turtle carcasses are examined, because this maximizes the likelihood of finding live specimens that betray their presence by movement. After gross examination and collection of specimens for histologic examination, each organ should be serially sliced at approximately 10-mm intervals and irrigated with saline solution to flush eggs and worms into a labeled collection vessel. The gut can be opened and cut into short lengths, which are treated in a similar way. The celomic cavity should be irrigated for collection of parasites that have seeped from vessels when organs are removed. Clotted blood from vessels may be carefully dissolved by using heparinized saline solution to collect parasites. Any washings are allowed to settle over 15–30 min, and the supernatant is then decanted. The sediment is placed in a shallow clear dish for scanning under a dissecting microscope, and, by using a pipette, any parasites found are extracted and placed initially into saline solution. The use of sieves and forceps to extract specimens is not recommended, because this may physically damage the worms. If it is desired to identify a trematode to species by using morphology, then fixation is best done while it is being gently compressed to avoid contraction. As an alternative, trematodes can be placed in tap water or saline solution and refrigerated for a few hours before being placed directly into fixative. Ninety-five percent ethanol fixation is preferable if genetic sequencing is to be undertaken, but 70% is preferable if morphologic studies only are to be done, if formalin is not being used to preserve specimens for morphology. 78,135 Current practice favors higher concentrations, 78 but excellent parasite DNA preservation has been shown in concentrations of 70% ethanol. 135,138 Of greater consequence is the selection of fixative, with formalin-buffered solutions preserving less genetic material when compared with ethanol solutions. 33,66
If trematode specimens will be used for morphologic identification only, then 10% neutral buffered formalin is the preferred fixative. Such specimens, after fixation, can be stained by using acetocarmine or Gower's carmine. 44 After staining, specimens are carefully dehydrated through a series of ethanol concentrations; cleared in an agent, such as methyl salicylate; and mounted in a medium, such as Canada balsam. 124 In some cases, worms might need to be embedded in wax and serial sections cut to reveal internal anatomy. Nematodes should be processed and examined in a different way. They can be killed by gently heating (but not overheating) while held in saline solution or a similar fluid. The worms will straighten out as they die and should be fixed in formal acetic fixative (9 parts of formaldehyde to 1 part glacial acetic acid to 90 parts water). Nematodes are often cleared in lactophenol and studied in this medium. Parasites can be identified by reference to the taxonomic literature (CABI Publishing 2001–2008). There is no such resource for nematodes of sea turtles. However, these parasites are rarely encountered in sea turtles.
Increasing use is being made of genetic sequence data to identify parasites. There are some advantages to this approach. Sequence data can be obtained from otherwise unidentifiable larval or egg stages, and these data matched to known species. Sequence data can also reveal the presence of cryptic species. Genome regions commonly used include portions of the large and small nuclear ribosomal subunit genes and the spacers between these. 11 It is also common to include some data from the mitochondrial genome. It is important to ensure that collected samples are contamination free, because otherwise the success of a true sequence is compromised. Preliminary work has begun along these lines on turtle parasites. An idea of the possibilities for future studies can be gleaned from previous studies. 12,128,131
Skeletal chronography
It is important to preserve the humeri if skeletal chronology tests are to be conducted 155 ; this assessment of skeletal age involves morphometric measures of the cross section of the humerus, including diameter, medullary thickness, and number of annuli and spacing. 155 Storage for these types of tests should preserve the integrity of the bone structure and not allow degradation or bone shrinkage. Freezing may provide the best storage method.
Toxicologic analyses
Because of the possible and potentially highly significant associations between water contamination and increasing disease and mortality in marine turtle populations 55,70,142 consideration should be given to taking standard specimens for toxicologic analysis, even if it cannot be conducted immediately. Bioaccumulation of heavy metals (copper, cadmium, lead, mercury), and organochlorine (OC) pesticides (polychlorinated biphenyls [PCB]) predominantly occur in the liver and adipose tissue, 27,100 although vertical transmission of OCs from mother to eggs has also been reported in leatherback turtles 134 and significant OC, PCB, and heavy metal concentrations have been found in green and hawksbill turtle eggs that entered the consumer market in Peninsular Malaysia. 141 Bioaccumulation may be significant, because hormone-mimicking chemicals (dichlorodiphenyltrichloroethane [DDT], dichlorodiphenyldichloroethylene, dieldrin, atrazine, lead, zinc and copper) have been proposed to alter sex-related hormones estradiol and testosterone in nesting green turtles, potentially reducing reproductive capacity, 67 in addition to causing health risks to humans who consume turtle products. 141
Heavy metals, including copper, cadmium, lead, and mercury, have been measured in various marine turtle populations, with concentrations varying significantly, depending on turtle species, diet, region, life stage (i.e., access to certain food types and/or exposures), migratory status, and tissue type sampled but, in some cases, greatly exceeding levels measured in other marine vertebrates. 2,27,100 In one study of loggerhead turtles in U.S. waters extending from South Carolina to Florida, turtles with higher blood mercury levels had lower numbers of circulating lymphocytes and lower ex vivo B-lymphocyte proliferative responses. 26 Anthropogenic contaminants, including polychlorinated biphenyls and OC pesticides (e.g., DDT), may also modulate immune responses and disrupt endocrine and reproductive function in various species, even at low concentrations. OCs accumulate in adipose tissue, as in other animals, and reach higher levels in carnivorous marine turtle species compared with herbivorous species; however, in different studies, their levels have varied relative to other marine animals, including fish, fish-eating birds, and mammals. 1,79,99 In loggerhead turtles from North Carolina, elevated concentrations of various OC contaminants in blood or tissues, although lower than those in other wildlife, were associated with clinical changes in blood analytes and poorer body conditions. 79 Results of this study suggested that, despite OC concentrations being lower relative to other wildlife, they may still significantly influence the susceptibility of marine turtles to disease.
Specimens from skin, adipose, and skeletal muscle tissues and internal organs, including the liver, should be taken for analyses. 75,79,80 In general, samples taken for heavy metal analysis should be placed in separate plastic bags, and those for pesticide analysis should be wrapped in inert packaging, such as aluminium foil or sterile glassware; both types of samples can be stored at −20°C, 136 although some pesticides (e.g., atrazine), are best detected by using fresh kidney samples. It is best to consult the appropriate toxicology laboratory for specific instructions for proper sample preservation and the amount of tissue required. A minimum of 200 g is generally recommended. 136 In addition, small samples of tissue may be preserved for polymerase chain reaction and/or virology. Specific lesions should be excised and stored. All specimens for toxicologic analyses are best stored long term in a −80°C freezer. Similarly, specimens for electron microscopy can be stored in Trump's solution (4% formaldehyde mixed with 1% glutaraldehyde). 77
Summary
The present article reviewed pathology of marine turtles on a systems basis, indicating common and less frequently reported gross and histologic lesions to assist diagnosticians in the comprehensive necropsy examination of these species. Techniques to enable the collection and storage of diagnostic quality samples for common diseases that may be encountered were suggested, in addition to those for advanced testing, such as skeletal chronology, microbiological culturing, and toxicologic analyses. Disease in these species is still poorly understood, and sustained effort by veterinary diagnosticians performing postmortems in the various parts of the world where these populations are found will be required to improve our knowledge of the multiple threats to their sustainability.
Acknowledgements
This review was conducted as part of a joint Department of Environment and Resource Management Queensland Turtle Conservation Project and a University of Queensland Veterinary-Marine Animal Research, Teaching and Investigation project. The authors thank Dr. Judy St Leger, Sea World (San Diego, CA) and Dr. Jon Hanger, Australia Zoo (Beerwah, Australia) for their constructive comments on manuscript drafts. The research that provided the unpublished findings presented in this review was funded by an Australian Government Department of the Environment, Water, Heritage and the Arts Marine Species Recovery and Protection Grants Programme grant and a grant from the Great Barrier Reef Marine Park Authority.
