Abstract
The current article describes a spontaneous case of old dog encephalitis (ODE) in a 7–year-old, intact, female Miniature Schnauzer dog from Londrina, Paraná, southern Brazil. Unlike conventional distemper encephalomyelitis, ODE is a poorly understood and extremely rare manifestation of Canine distemper virus (CDV) infection. The dog was presented with progressive clinical manifestations consistent with cerebral dysfunction. Briefly, histopathologic lesions were restricted to the forebrain and included chronic multifocal lymphoplasmacytic encephalitis with extensive perivascular cuffing, astrocytosis, and intranuclear inclusions within astrocytes and giant cells, with both intracytoplasmic and intranuclear inclusions. Immunohistochemistry (IHC) was used to identify the antigens of the nucleoprotein (NP) of CDV and to detect cluster of differentiation (CD)3, CD79a, macrophage (MAC) 387, glial fibrillary acidic protein, and vimentin to characterize the neuroparenchymal lesions. By IHC, CDV NP was demonstrated predominantly within neurons and astrocytes. Cells that formed perivascular cuffs and some astrocyte-like cells reacted intensely to vimentin. Reverse transcription polymerase chain reaction assay from brain sections further confirmed a role for CDV in this disease by the amplification and partial sequence analysis of the NP gene. These findings confirmed simultaneous detection of CDV in ODE by IHC and molecular assays. In addition, results of the current study could contribute to the neuropathologic characterization of this rare manifestation of CDV.
Keywords
Introduction
Canine distemper virus (CDV; order Mononegavirales, family Paramyxoviridae, subfamily Paramyxovirinae, genus Morbillivirus) induces encephalomyelitis in dogs and other susceptible animals. 21 Neurologic syndromes associated with CDV include canine distemper encephalomyelitis in immature dogs, multi-focal distemper encephalomyelitis in mature animals, old dog encephalitis (ODE), chronic relapsing encephalomyelitis, 5,22 and post-vaccinal distemper encephalitis. 6,9 These syndromes are directly related to the virus strain, age of the affected animal, and the neuroanatomic location of the neurologic lesions. 11,23
ODE is an extremely rare manifestation of CDV-induced encephalitis, with only 5 descriptions of the naturally occurring disease in the veterinary literature. 1,5,14,15,24 Initially, CDV was proposed as the etiologic agent of this unusual neuropathologic syndrome in 1942. 5 However, etiologic investigations that confirm the participation of CDV have been few because of the paucity of spontaneous cases for study. Most of the neuropathologic manifestations of CDV were reproduced experimentally, but, despite several attempts, 5,15,21,24 there was only 1 successful experimental reproduction of ODE. 3 Thus, the naturally occurring incidence of ODE is low, the pathogenesis is uncertain, and the precise host-CDV relationship is not well elucidated. Spontaneous cases of this disease have not been recorded since 1970, 21,22,24 CDV antigens in brain sections from naturally occurring cases of ODE have only been demonstrated once, 15 and molecular characterization of CDV from ODE cases has not been described previously. Accordingly, the purpose of the current report is to describe the molecular, pathologic, and immunohistochemical (IHC) findings of a naturally occurring case of ODE.
Material and methods
Clinical history and necropsy
A 7-year-old, intact, female Miniature Schnauzer was admitted to the Veterinary Teaching Hospital, Londrina, Paraná, in southern Brazil, in September 2006. This dog had a history of progressive behavioral changes, motor incoordination, compulsive walking, and circling for approximately 5 weeks. The owner reported that the dog had received 3 doses of a modified-live vaccine against canine distemper as a pup; the owner had observed 1 seizure-like episode before examination. Clinical neurologic evaluation led to the anatomic diagnosis of cerebral dysfunction. Medical therapy was initiated, but neurologic signs were progressive, severe seizures could not be successfully controlled by therapy, and the dog died after the onset of cluster seizures 60 days after the initial examination.
Routine necropsy was performed soon after death and selected tissues (brain, tonsils, lung, urinary bladder, kidneys, liver, heart, and spleen) were fixed in 10% buffered neutral formalin solution and routinely processed for histologic evaluation. Tissue sections were routinely stained with hematoxylin and eosin (HE) as well as with special stains to demonstrate possible infectious agents (periodic acid-Schiff, Gomori's methenamine silver, Ziehl-Nielsen, and Giemsa stains).
IHC
Selected paraffin-embedded tissue sections from the nervous tissue lesions were prepared for IHC to detect the nucleoprotein (NP) antigen of CDV a and Toxoplasma gondii P30 antigen. b Replicate tissue sections were also processed to identify glial fibrillary acidic protein (GFAP), c vimentin, d macrophages (MAC 387), e and T and B lymphocytes (cluster of differentiation [CD]3 and CD79α). f For IHC, the streptavidin-biotin technique was used in an automated staining system. g The intensity and distribution of immunoreactivity of the panel of antibodies were subjectively evaluated and combined into grades of 0, absent; +, discrete; ++, moderate; and +++, marked. Tissue sections from a previous case of distemper 10 and toxoplasmosis (Headley, unpublished data) were used as positive controls. For negative controls, phosphate buffered saline solution was substituted for the primary antibody. Known positive controls (authors’ laboratory at University of Helsinki, Finland) were used during automated staining for other immunologic cell markers.
Molecular analysis
For RNA extraction, 1–mm x 1–mm slices of formalin-fixed paraffin-embedded sections of the cerebrum and lung were used. Paraffin was removed by washing twice in xylene at 56°C for 30 min and thereafter in 100% ethylene. Paraffin block-derived samples were then treated with 50 μl of 20 mg/ml proteinase K h and 200 μl of lysis buffer (pH 8.0; 100 mM NaCl, 100 mM ethylenediamine tetraacetic acid, and 0.5% sodium dodecyl sulfate) for 1–3 hr at 55°C. RNA extraction was performed as described 4 on the previously obtained lysate, and elution was done in diethylpyrocarbonate-treated water.
Reverse transcription polymerase chain reaction (RT-PCR) was done by using the oligonucleotide primers CDV1 (forward) and CDV2 (reverse), specifically designed to amplify a 287 base pair [bp] segment of the CDV NP gene. 7 Amplification reactions were performed as previously described. 2 Formalin-fixed paraffin-embedded brain samples from a case of conventional distemper encephalomyelitis 2 served as positive controls, and brain tissue from a healthy dog was used as negative control.
Sequence determination, phylogenetic analyses, and GenBank accession numbers
The specificity of the RT-PCR was confirmed by direct sequencing of the amplicons in an automated capillary DNA sequencer i by using the forward and reverse primers. The RT-PCR amplicons were sequenced, and the quality of each sequence was analyzed with the Phred/Phrap/ Consed Analysis program (http://www.phrap.org). The partial nucleotide sequences of the CDV NP gene were compared with those deposited in GenBank. Initially, the CDV sequences were aligned and compared by the CLUSTAL W method, 13 and the degree of similarity between nucleotide sequences was determined by using BIOEDIT version 5.0.9 software. 8 The alignments subsequently were used as input for phylogenetic analysis by using Mega v.3.1 software. 12 The phylogenetic tree was constructed by the neighbor-joining method and based on 1,000 bootstrapped data sets. Distances values were calculated by using the Kimura 2 parameter model. The GenBank accession numbers of the CDV strains used for phylogenetic analyses during this study are presented in Table 1.
Results
Clinical and neurologic examinations
Clinical evaluation demonstrated no systemic abnormalities. Neurologic examination revealed postural reactions deficits, compulsive circling, and head pressing. Cranial nerve deficits were not observed initially. However, dorsal strabismus of the right eye occurred on the 42nd day after initial presentation at the veterinary hospital along with the other previously described neurologic manifestations.
Gross and histopathologic findings
Serial transverse sections of brain revealed significant gross lesions (Fig. 1) at the forebrain (frontal cortex, basal nucleus, pyriform lobe, rostral thalamus, and rostral subcortical white matter [corpus callosum, corona radiata, and internal capsule]; Fig. 1A), with sparing of the hindbrain (cerebellum and caudal brain stem) and occipital cortex (gray and white matter). Gross changes were manifested by the loss of distinct demarcation between the gray and white matter (Fig. 1B, C),
GenBank accession numbers, viral strain, and geographical origin of Canine distemper virus nucleotide sequences.

irregular surface of affected tissue relative to control, and areas of gray-to-brown discoloration of the neural tissue. Tracheal edema and wet, heavy lungs with hemorrhagic foci characterized the respiratory lesions.
Histologic alterations occurred primarily in the brain and, to some extent, the lungs; all other tissues were normal. Neurologic lesions were restricted to the forebrain; the cerebellum and caudal brain stem were not affected. Diffuse forebrain lesions occurred within the cerebral cortex, basal nucleus, rostral thalamus, and rostral subcortical white matter (corpus callosum, corona radiata, and internal capsule). Within the cerebral cortex, the sensorimotor and paleocortex were the mainly affected with sparing of the visual areas. Lesions consisted of severe parenchymal white and gray matter rarefaction associated with extensive perivascular cuffs, severe influx of lymphoplasmacytic inflammatory cells, and proliferation of glial components (Fig. 2A). More than 6 layers of mononuclear inflammatory cells formed these angiocentric lesions (Fig. 2B), and lymphocytes and plasma cells were the predominant cell type in perivascular cuffs as observed in HE-stained tissue sections. Several irregularly shaped multinucleated giant cells were observed within areas of perivascular cuffing, and some of these cells contained intranuclear and intracytoplasmic inclusions (Fig. 2C). Several eosinophilic intranuclear inclusions were present within astrocytes (Fig. 2D). Most neurons in the cerebral cortex adjacent to the areas of severe parenchymal destruction were necrotic. Furthermore, severe astrocytosis and discrete areas of white matter demyelination were observed in HE-stained tissue sections. Pulmonary lesions included edema, congestion, hemorrhage, and emphysema. Infectious agents were not identified in nervous or pulmonary tissues by periodic acid-Schiff, Gomori's methenamine silver, Ziehl-Nielsen, and Giemsa staining.
IHC
A summary of the distribution of immunologic markers within the nervous tissue is presented in Table 2. Within the area of severe parenchymal rarefaction of the cerebrum, immunoreactivity to the NP antigen of CDV occurred predominantly within the cytoplasm of neurons (Fig. 3A) but also in some astrocytes. By estimate, approximately 30% of neurons contained antigen of CDV. Weak intranuclear immunoreactivity to CDV antigens occurred within peripheral nuclei of some syncytial cells (Fig. 3B). Plump GFAP-positive astrocytes were predominantly within areas of severe glial proliferation. Subjectively, GFAP- and vimentin-positive astrocytes (Fig. 3C) were more evident in well-preserved areas of the cerebrum compared with those that contained severe angiocentric lesions and marked tissue rarefaction. However, areas of tissue rarefaction contained comparatively more immunostained astrocytic processes than unaffected regions. The multinucleated giant cells were negative for GFAP, CD3, and CD79a, but strongly immunoreactive for vimentin and MAC 387 (Fig. 3D). Immunopositive macrophages were more abundant within the region of severe perivascular cuffing. Intense immunoreactivity to vimentin occurred within most mononuclear cells of perivascular cuffs (Fig. 3E). Inflammatory cells of perivascular cuffs were more immunoreactive to vimentin than to specific cell markers. Subjectively, approximately 60% of the inflammatory cells were positive for CD3, 30% for CD79, and less than 10% for MAC 387 (Fig. 3F–H). Where GFAP staining was weak, in duplicate sections astrocytes within
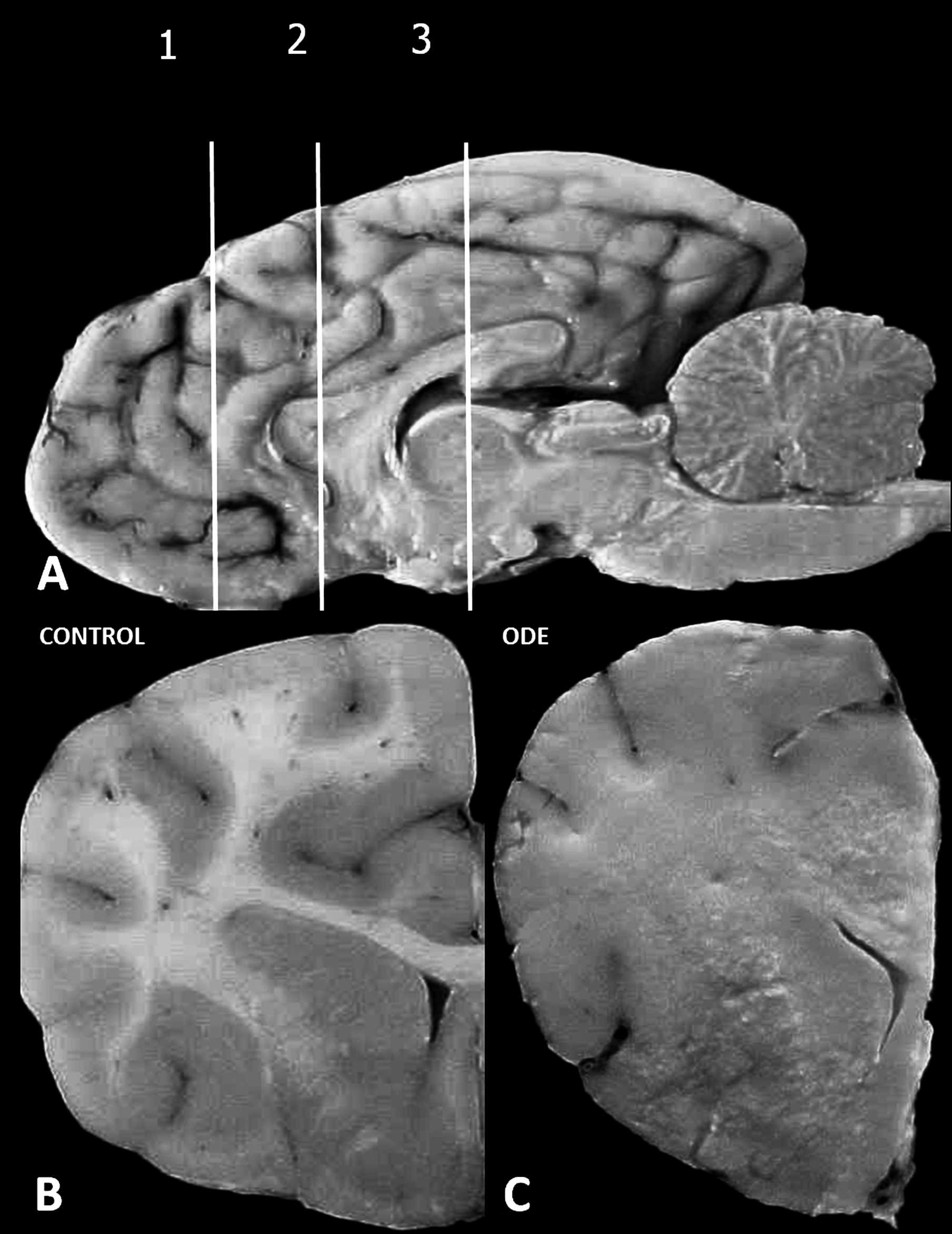
Dog; gross demonstration of forebrain lesion.
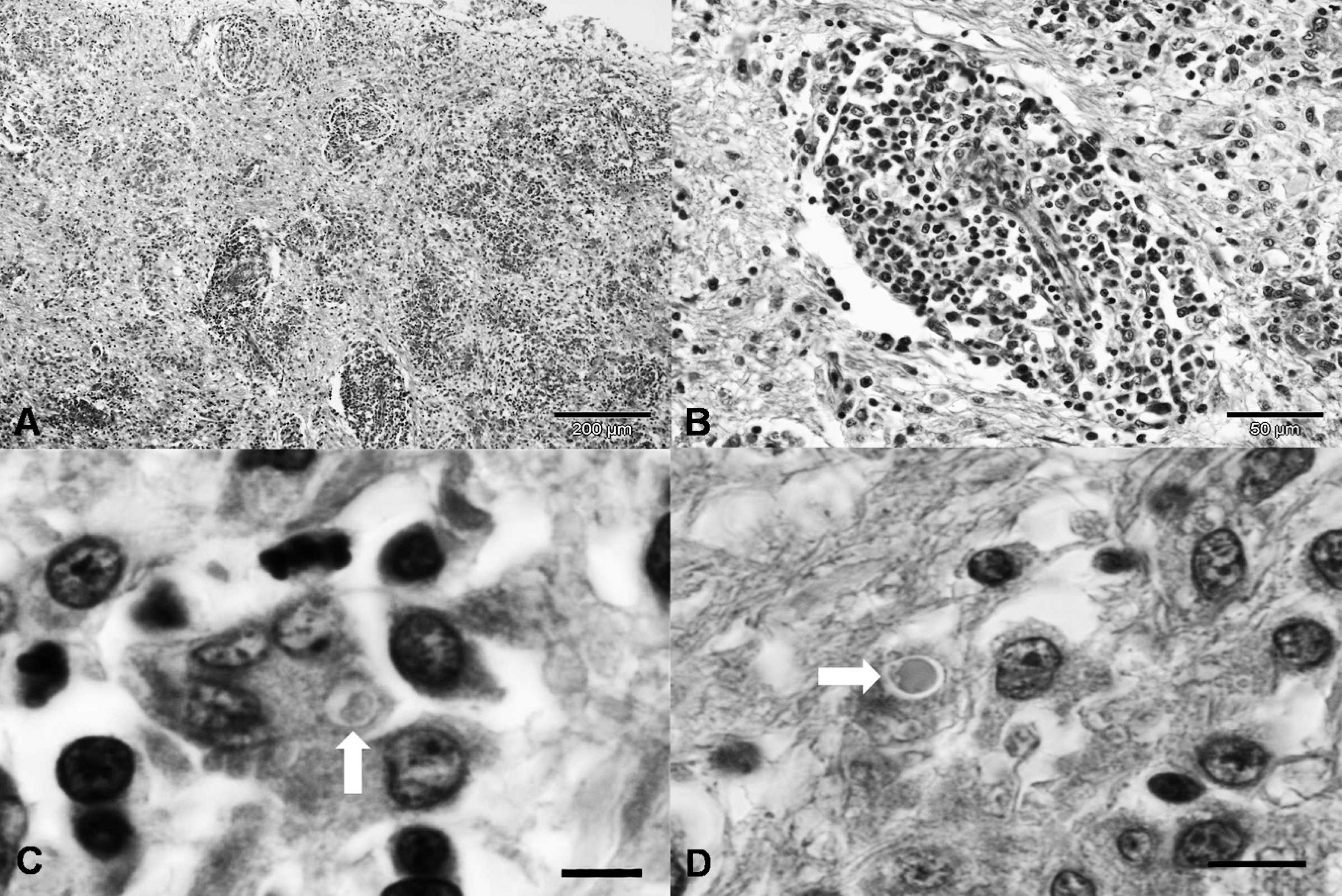
Dog; old dog encephalitis; photomicrograph of the brain.
areas of extensive glial proliferation and those in normal nervous tissue adjacent to these areas reacted intensely for vimentin. Toxoplasma gondii organisms were not demonstrated by IHC in any tissues evaluated and CDV antigens were not observed within the lung. The positive controls stained similarly to the target slides, and the negative controls did not stain.
RT-PCR assay and sequence analysis of the CDV NP gene
The RT-PCR produced a 287-bp amplicon from formalin-fixed, paraffin-embedded sections of the
Comparative distribution of cell markers and Canine distemper virus in old dog encephalitis. *
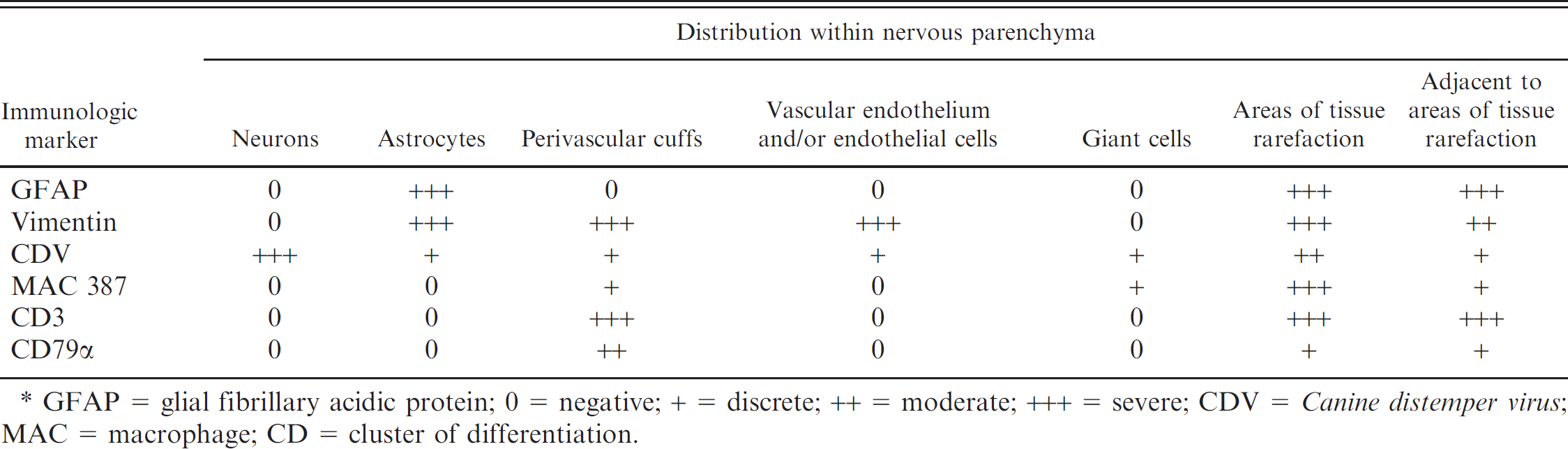
GFAP = glial fibrillary acidic protein; 0 = negative; + = discrete; ++ = moderate; +++ = severe; CDV = Canine distemper virus; MAC = macrophage; CD = cluster of differentiation.
brain and phylogenetic analyses indicated that the amplicon was CDV specific. In contrast, RT-PCR failed to produce the 287-bp amplicon from sections of pulmonary tissue. The nucleotide sequences obtained from brain tissue in the current study were deposited in GenBank (accession no. EF197736). The phylogenetic tree, based on a 244-bp nucleotide sequence, is shown in Figure 4. Phylogenetic analysis demonstrated 2 definite clusters, with corresponding subclusters. The sequence derived from the current study was more antigenetically related (98%) to one North American, one European, and a previously described South American strain of CDV. Furthermore, a similarity of 95–98% was observed between the CDV NP gene of the present case and other CDV NP sequences deposited in GenBank when the identity table for the nucleotide sequences was evaluated (Table 3).
Discussion
The diagnosis of ODE was based on a combination of clinical findings, typical histologic lesions within the cerebrum, with marked sparing of the cerebellum and caudal brain stem, and the absence of CDV-induced alterations in other tissues. These findings were consistent with those of naturally occurring 1,5,14,15,24 and experimentally induced cases of ODE. 3 Virus isolation was not attempted in the current case but is often difficult because the persisting virus may be defective. 15 Involvement of CDV within the lesions of the forebrain was confirmed by the concomitant identification of CDV antigen by IHC staining and nucleic acid by RT-PCR and sequence analysis. In addition, the CDV NP antigen was detected by IHC, whereas RT-PCR amplified the expected nucleotide sequence of the NP gene. Thus, the present study demonstrated CDV within typical ODE histologic lesions and confirmed the first case of ODE from Brazil and South America. Further, the current case contributes to the documentation of this extremely rare manifestation of CDV and suggests that this disease can occur naturally in canine populations as originally described. 5 In addition, the IHC results of this study indicate the inflammatory and reactive lesions in naturally occurring ODE, thereby contributing to the neuropathologic characterization of this rare manifestation of CDV. The morphology of the pulmonary lesions observed in the current case was not consistent with a typical CDV infection because CDV antigens were not demonstrated by IHC and the NP gene was not identified by RT-PCR. Edema, congestion, and hemorrhage were interpreted as manifestations of pulmonary shock because of recurrent seizures before death. In addition, a T. gondii-induced infection was excluded by negative immunoreactivity to the P30 antigen, and mycotic and bacterial agents were not observed by special stains.
Phylogenetic analyses demonstrated that the viral strain of CDV involved in the present case of ODE is antigenically related to other previously described wild-type strains of this virus in northern and southern America and in Europe. These findings suggest that all strains of CDV are closely related antigenically, although isolated in distinct geographic locations. In addition, because the virus associated with ODE is almost identical to other CDV strains deposited in the GenBank, this close relationship might suggest that an unusual event occurs after infection with a common strain of CDV to induce this uncommon manifestation of distemper encephalitis. Two vaccine strains of CDV (Rockborn and Onderstepoort) are commercially available in Brazil. Further, the nucleotide sequences of the Rockborn and Onderstepoort CDV strains available at GenBank were included in the phylogenetic analysis. However, the NP gene sequence of CDV obtained from the present case of ODE was different from those of these vaccine strains, which suggests that the virus involved in this case of ODE was a wild type rather than a vaccine strain of CDV.
By light microscopy, the inflammatory cells of perivascular cuffs consisted predominantly of lymphocytes and plasma cells. However, IHC revealed that most perivascular cells were vimentin positive, which corroborated previous studies that identified vimentin immunoreactivity within lymphocytes in cases of chronic lymphocytic leukemia 20 and within cells of perivascular cuffs of rats with experimentally induced autoimmune encephalitis, 19 which suggests that leukocytes might express this intermediate filament in some cases. In addition, the expression of vimentin by lymphocytes plays an important role in adhesion and transcellular migration. 16 Further, the perivascular cells initially identified predominantly as lymphocytes and plasma cells by HE reacted with different degrees of positivity to CD3, CD79a, and MAC 387, which indicated that not all perivascular cells in cases of ODE are exclusively of lymphocytic origin. However, the macrophage-positive cells of perivascular cuffs in the current case might possibly be transient after recruitment from the vascular lumen and migration into areas of intense chemotactic activity in addition to intrinsic perivascular microglia resident in the vessel wall. This transmigration might be because of the presence of CDV antigen within the injured nervous parenchyma.
Several multinucleated (syncytial) giant cells were identified by HE. Further, IHC revealed weak immunoreactivity to CDV antigens within syncytial
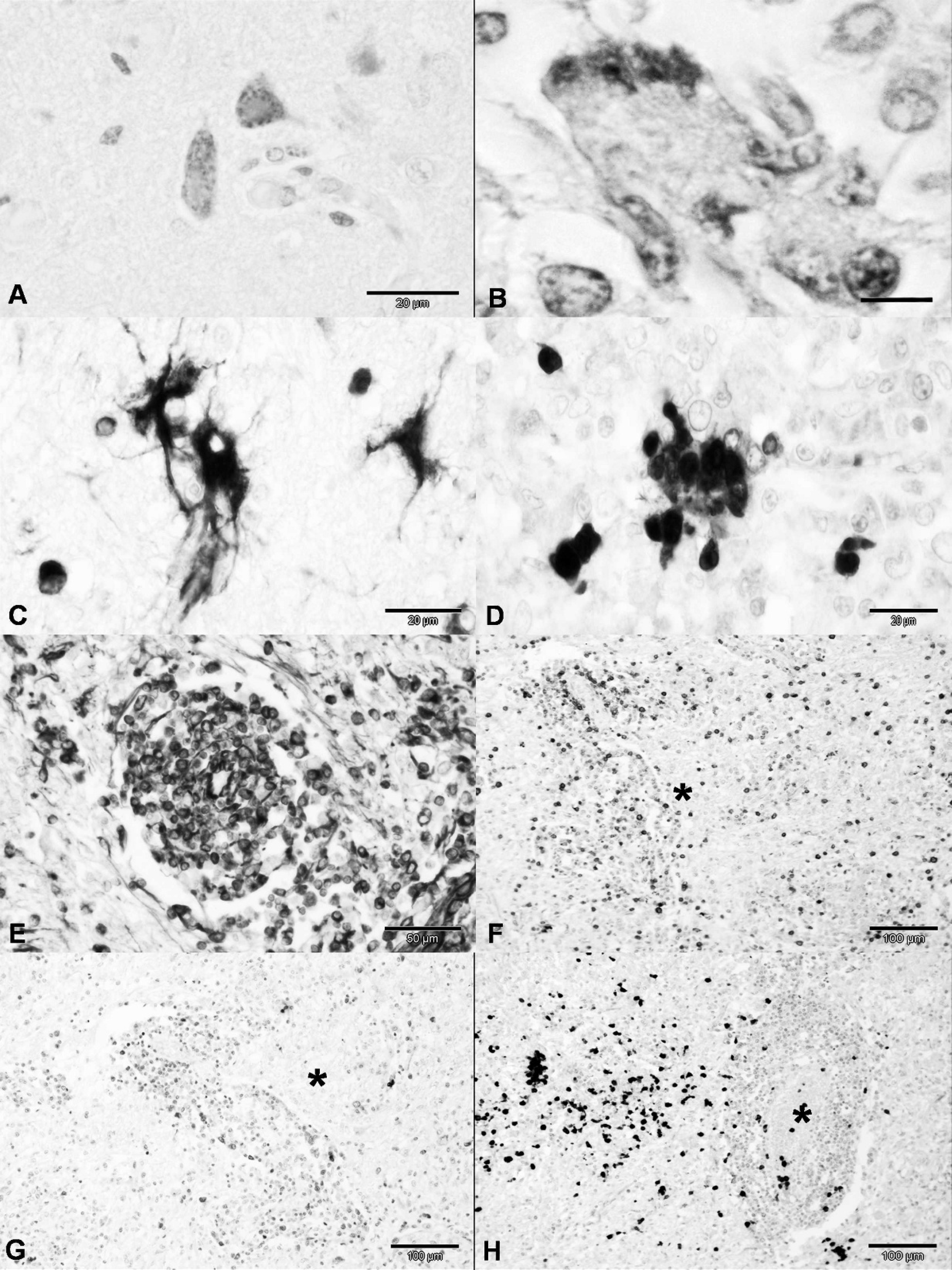
Dog, brain; old dog encephalitis; immunohistochemical (IHC) characterization of neurologic lesions.
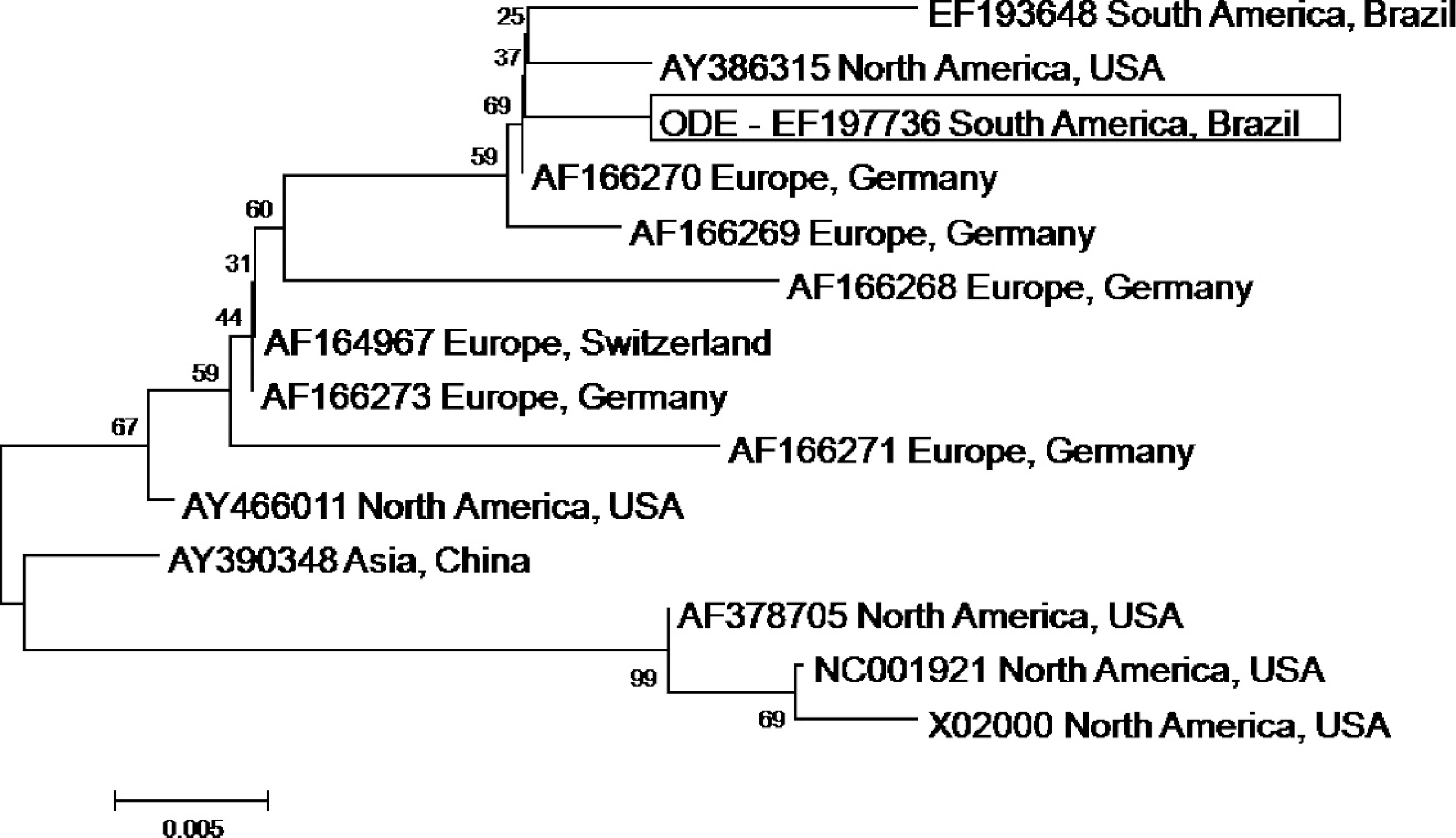
Phylogenetic tree constructed from 244 nucleotides of Canine distemper virus nucleoprotein gene by the neighbor-joining method. The tree was prepared based on 1,000 bootstrapped data sets. Distances values were calculated by using the Kimura 2 parameter model. The result of phylogenetic analysis is shown as a radial (bootstrap consensus tree) pattern.
cells, which confirmed their participation in ODE. The multinucleated giant cells observed in the present case of ODE were also described in experimental 3 and spontaneous 15 cases of ODE, and in common forms of distemper encephalitis. 22 Further, similar findings were described in human patients with multiple sclerosis and subacute sclerosing pancencephalitis. 1 In previous studies, multinucleated giant cells were considered as syncytial cells of glial origin 3 or originated from other central nervous system cells. 22 However, IHC characterization of multinucleated giant cells in ODE was never performed to identify the origin of these cells. In the present case, IHC demonstrated that these cells were GFAP-negative but were strongly immunoreactive to MAC 387 and vimentin, indicating they are likely monocytic.
Characteristic CDV inclusions were identified within astrocytes of the cerebral cortex of the dog in the present case and were similar to previous cases of ODE. 1 ODE-related inclusion bodies were described in astrocytes and neurons of the cerebral cortex, thalamus, and brain stem, 1,5,15,25 but were not observed in other cases of ODE. 3,5 In addition, inclusion bodies in the current case were also demonstrated within multinucleated giant cells. Paradoxically, neurons did not show identifiable inclusions but were often IHC positive.
Astrocytes within areas of severe parenchymal lesions also reacted strongly to vimentin, which is common in chronically activated astrocytes in which GFAP expression might diminish. In 2007, dogs with naturally occurring distemper encephalitis were shown to have comparatively more intense cellular expression of vimentin by astrocytes than normal dogs. 17 In conventional acute distemper lesions, the astrocytic response is predominantly composed of GFAP- and CDV-positive cells. Alternatively, vimentin-positive astrocytes are present in advanced chronic inflammatory lesions and represent the principal cell type that harbor the pathogen. 18
In conclusion, CDV inclusion bodies were observed within astrocytes and multinucleated giant cells in a
Nucleotide identities of 244-bp reverse transcription polymerase chain reaction amplicons of Canine distemper virus (CDV) nucleoprotein (NP) gene (nucleotide 683–926 of the Onderstepoort NP gene). *
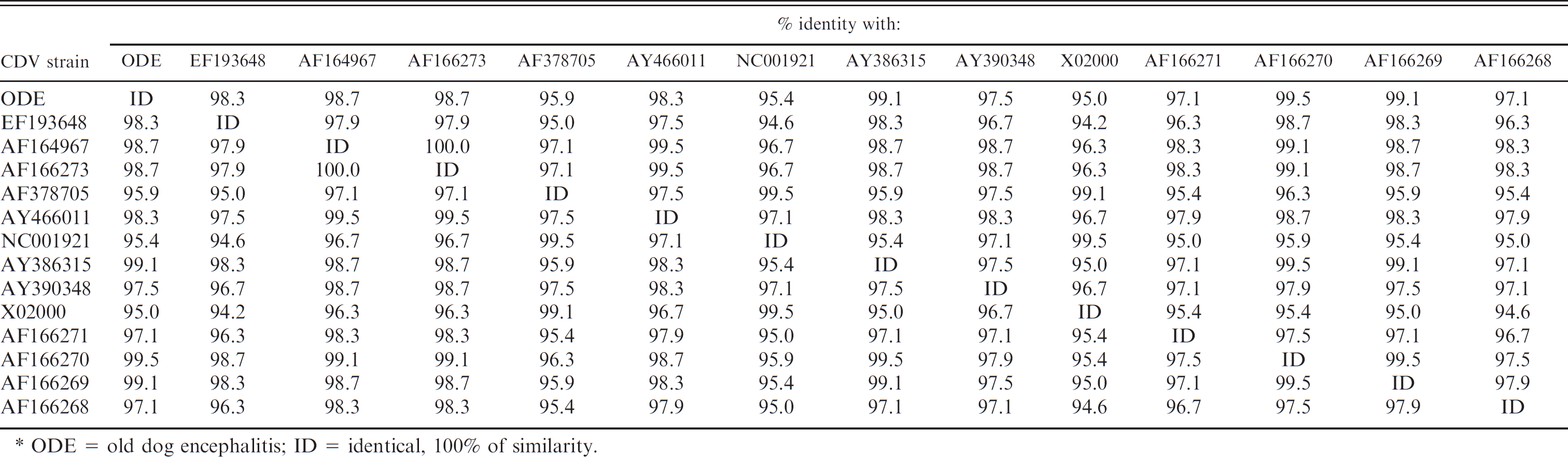
ODE = old dog encephalitis; ID = identical, 100% of similarity.
case of ODE characterized by severe chronic lymphoplasmacytic encephalitis with extensive perivascular cuffing within the cerebrum. The presence of CDV antigen was confirmed by IHC because of strong immunoreactivity to antigens of the NP gene within neurons and astrocytes and weak reactivity in syncytial cells. In addition, RT-PCR and sequence analyses identified a portion of the CDV NP gene from cerebral sections.
Acknowledgements
The authors thank Kati Holmsten, University of Helsinki, Finland, for the realization of the histologic and IHC techniques used during this study. The authors are grateful to Brazilian agencies (CNPq, Fundação Araucária [FAP/PR], CAPES, and FINEP) for financial support. A. A. Alfieri and A. F. Alfieri are recipients of CNPq fellowship; A. M. Amude is a recipient of a CAPES fellowship. S. A. Headley and A. M. Amude contributed equally to the preparation of this manuscript.
Footnotes
a.
Clone DV2–12, AbD Serotec, MorphoSys UK Ltd., Kidlington, Oxford, U.K.
b.
NCL-TG, Novocastra Laboratories Ltd., Newcastle upon Tyne, U.K.
c.
Clone DP46.10, AbD Serotec, MorphoSys UK Ltd., Kidlington, Oxford, U.K.
d.
Clone V9, Dako North America Inc., Carpinteria, CA.
e.
Clone MAC387, AbD Serotec, MorphoSys UK Ltd., Kidlington, Oxford, U.K.
f.
Clone HM57, Dako North America Inc., Carpinteria, CA.
g.
Lab Vision Corp., Fremont, CA.
h.
Invitrogen Corp., Carlsbad, CA.
i.
MegaBACETM1000 DNA Analysis System, GE Healthcare Technologies, Piscataway, NJ.
