Abstract
Diisocyanates, commonly used in the production of polyurethane foams, paints, elastomers, varnishes, and coatings, are considered among the most hazardous inhalation toxicants. The present report describes 2 unusual cases of mortality in pigeon chicks associated with nesting material contaminated by diisocyanates. Case 1 was submitted by a racing pigeon breeder who had lost all the hatchlings (
Diisocyanates are known irritants, carcinogens, and sensitizers that are among the leading causes of occupation-associated asthma in human beings.4,17 Diisocyanates are widely used in industrial manufacturing, especially during production of polyurethane foams and a variety of products such as elastomers and paints, and are considered a significant occupational hazard when exposed by inhalation. Among the diisocyanates, toluene diisocyanate (TDI) is considered highly volatile (vapor pressure: 0.1 mmHg at 37.7°C), whereas methylene diphenyl diisocyanate (MDI) is less volatile (0.00034 mmHg at 37.7°C). Because of this physical property, TDI has a higher inhalation potential and is considered more problematic in an occupational setting. Toxicity information of diisocyanates in avian species is limited, and there are no previous reports of birds being affected by this compound. The current report presents 2 cases in which diisocyanate-contaminated nesting pads were associated with specific clinical signs and death in pigeon chicks.
The first case was presented in January 2010, when all but 1 of the hatchlings of a racing rock pigeon (
Necropsy of 3 of the chicks by an avian veterinarian (J. Smith) showed edematous dark lungs in 2 of the 3 chicks. Examination of intestinal direct smears on 1 chick and crop smears and fecal flotation on 2 adult birds from the flock was negative for parasites. Two sets of fixed tissues (brain, kidney, lung, liver, air sac, bursa, pancreas, intestine, and crop) of 2-day-old chicks and nesting pads in question were submitted to the California Animal Health and Food Safety Laboratory System (CAHFS; Davis, California) for histological examination and chemical analysis, respectively. Two types of nesting pads were submitted; one was a used pad on which a chick had died and another was an unused pad that the owner had washed once with detergent in an unsuccessful attempt to remove possible contaminants. Histological findings included multifocal pneumocyte hyperplasia (Fig. 1), hepatic sinusoidal leukocytosis, and multifocal heterophilic inflammation in the bursa follicles and interfollicular interstitium.
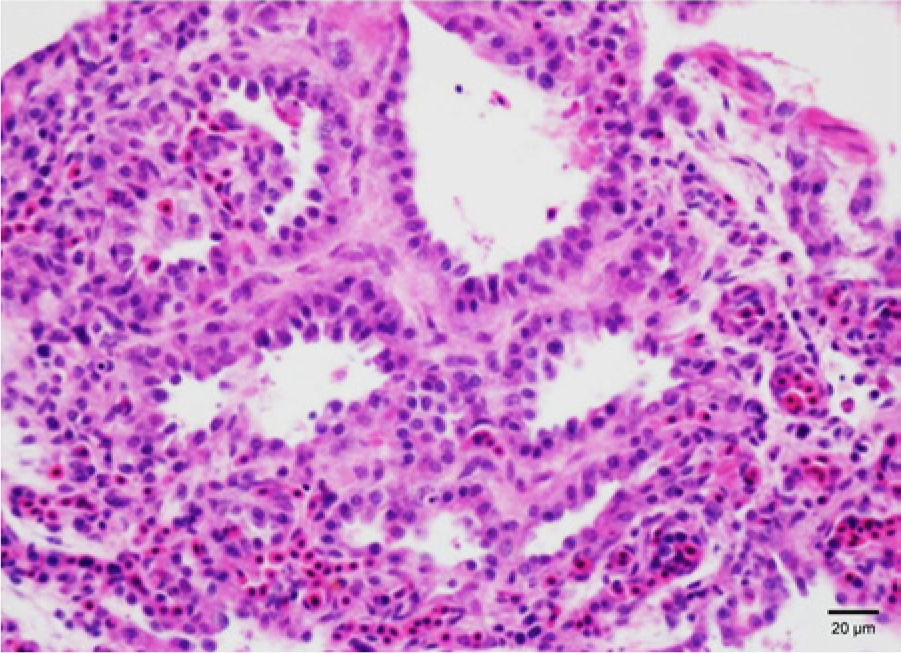
Pneumocyte hyperplasia in lung of pigeon chick from case 1. Hematoxylin and eosin. Bar = 20 µm.
Gas chromatography–mass spectrometry (GC-MS) screening for unknown compounds was performed on the submitted nesting pads following CAHFS standard operating procedures, with slight modifications. Briefly, 0.5 g of each nesting pad was excised from the central region of the nesting pad using a razor blade. The sample was placed into a 15-ml glass tube with a polytetrafluoroethylene-lined screw cap, 5 ml of ethyl acetate
a
were added, and the sample was sonicated for 15 min in an ultrasonicator. The sample was then mixed in an overhead shaker for 15 min, centrifuged for 5 min at 1,000 ×
Four distinct peaks at retention times (Rt) of 10.02 (peak 1), 17.81 (peak 2), 21.69 (peak 3), and 25.59 (peak 4) min were found in the total ion chromatogram (Fig. 2A). Comparing the spectrum of peak 1 against a known National Institute of Standards and Technology (NIST) spectral library, showed a 98% match with that of 2,4-diisocyanato-1-methyl benzene (2,4-toluene diisocyanate or 2,4-TDI; CAS#584-84-9; Fig. 2B). Peak 2 had a 96% match with 4,4′-methylene diphenyl diisocyanate (4,4′-MDI; CAS#101-68-8; Fig. 2C). Peaks 3 and 4 were identified as di-2-ethylhexyl phthalate (DEHP; CAS#117-81-7) and 5α-cholestan-3-one (CAS#15600-08-5), respectively. Semiquantitative analysis for 2,4-TDI showed unexpectedly high concentrations of 190 ppm in the used sample and 220 ppm in the unused nesting pad sample. A matrix-spike with a known standard of 2,4-TDI
d
was used to estimate the concentration. This analysis was repeated by another analytical chemist in the analytical toxicology section of the Animal Health Diagnostic Center (AHDC) at Cornell University (Ithaca, New York) and the same conclusion regarding the presence of these compounds and similar concentration (290 ppm in the unused sample) was reached. Extraction and analytical procedures performed at AHDC were similar to that of CAHFS with small differences in oven temperatures (held at 80°C for 2 min, ramped at 15°C/min to 290°C, held for 10 min) and capillary column used (HP-5MS, 30 m × 0.25 mm × 0.25 µm), which generated a slightly different Rt of 2,4-TDI at 8.26 min. Matrix-matched calibration curve method (
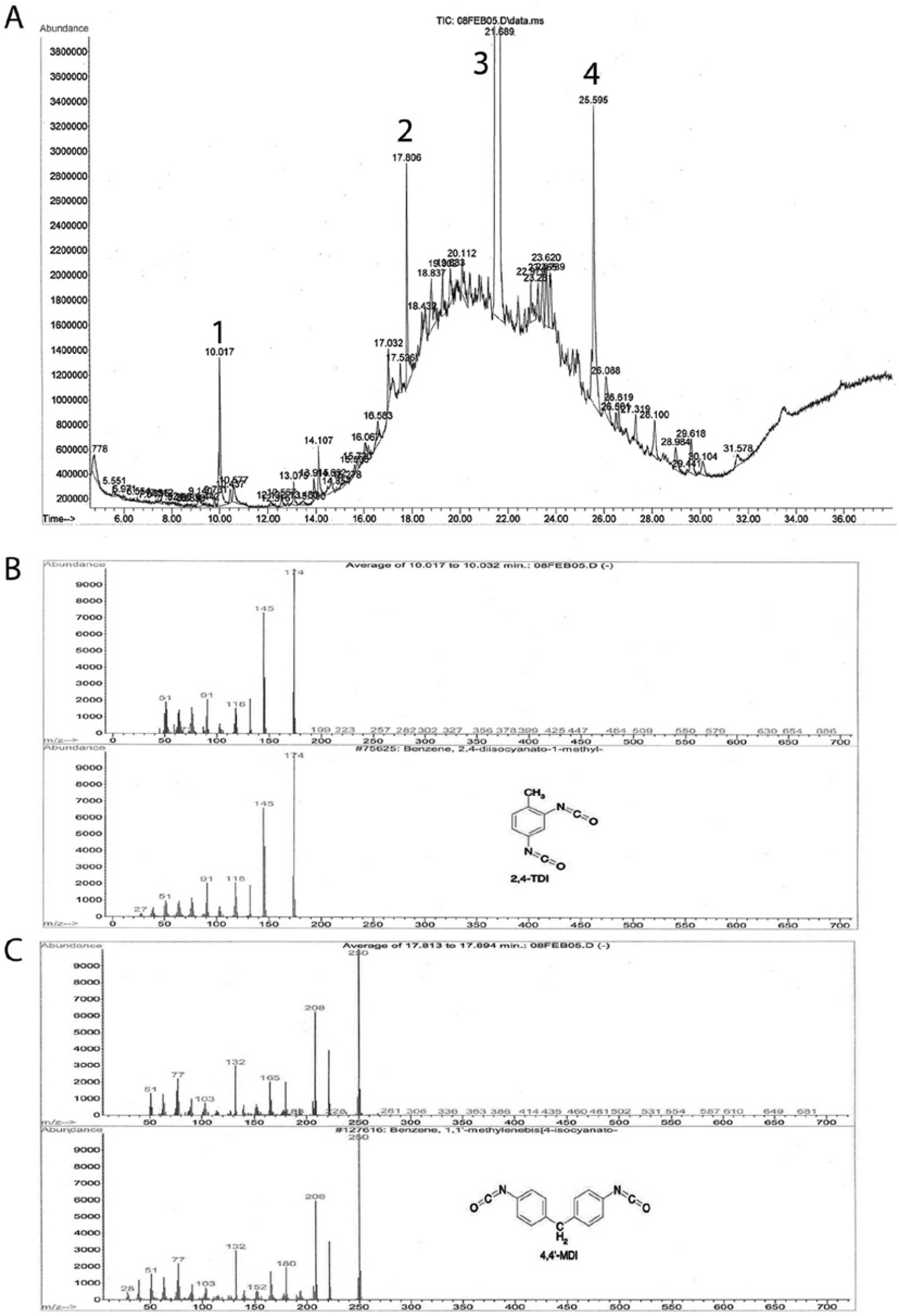
Gas chromatography–mass spectrometry (GC-MS) chromatograms of used nesting pad from case 1.
After realizing the nesting pads could be contaminated, the owner stopped the use of the nesting pad but the chicks born from pigeon pairs that were on the nesting pad prior to removal continued to die for the third round of breeding. However, when the eggs were fostered to unaffected pumper pigeons immediately after they were laid, all eggs hatched, and the chicks survived and developed normally. By March 2010, the owner reported that 24 new pairs bred in his flock, which had never been on nesting pads, produced healthy chicks, and hatchability was back to 95% with 0% mortality.
The second case was presented in April of the same year by a show-roller pigeon breeder. Show-rollers are pigeons specifically bred for their ability to do backward somersaults during flight. In contrast to the first case, this owner reported low hatchability and, of those that hatched, all died within 2 days with signs of lethargy and respiratory difficulty. A total of approximately 100 hatchlings were lost; approximately 30 were lost during the April–August breeding season in 2009 and the rest between the January–April breeding season in 2010. The owner reported that there was no mortality in the hatchlings in previous years and changing the pads to those made out of natural fiber stopped the illness and death in the hatchlings almost immediately.
A total of 6 eggs, 1 hatchling, and 2 nesting pads (1 used and 1 unused) were submitted to the San Bernardino branch of CAHFS. Out of the 6 submitted eggs, 5 had fully developed embryos (2 alive and 3 dead) and 1 was unfertilized. No gross lesions were found in the examined hatchling and embryos. Lung, liver, skeletal muscle, kidney, trachea, skin, proventriculus, gizzard, brain, and heart of embryos as well as hatchling were evaluated histologically. Focally extensive myositis and pyogranulomatous omphalitis were found in the 1 hatchling that was submitted. No histological lesions were observed in the submitted embryos.
Gas chromatography–mass spectrometry screen was performed on the submitted nesting pads. The used pad was positive for 2,4-TDI (Rt: 9.96 min), 4,4′-MDI (Rt: 17.73 min), and mono-(2-ethylhexyl) phthalate (MEHP; CAS#101-68-8; Rt: 21.58 min). The new nesting pad was positive for 2,4-TDI (Rt: 9.98 min), MEHP (Rt: 21.57 min), and
In an attempt to reproduce the condition, 1-day-old chicken hatchlings (
In order to understand a potential reason for high residual 2,4-TDI, scanning electron microscopy e (SEM) and a differential scanning calorimeter f (DSC) were used to identify materials used in the submitted nesting pads. The DSC heating curve (Fig. 3D) indicated small features at 150°C and 219°C and a large melting peak with onset at 240°C. All 3 of these thermal transitions were reproduced on repeated heating and cooling of the sample. The melting peak was consistent with nylon-6. 10 The small features at 150°C and 219°C represent melting points for minor components in the nest pad potentially including polypropylene, other nylons, or polyurethanes. The bulk of the polymeric fibers were determined as nylon-6, although other types of fibers, including metallic fibers were observed in SEM images and by visual observations and by an endoscope magnifier (Fig. 3A, 3B). This type of felt would typically be produced using scrap fibers from other textile processes including carpet manufacture. Adhesive-like substances were observed attached to the nylon fibers under SEM (Fig. 3C).
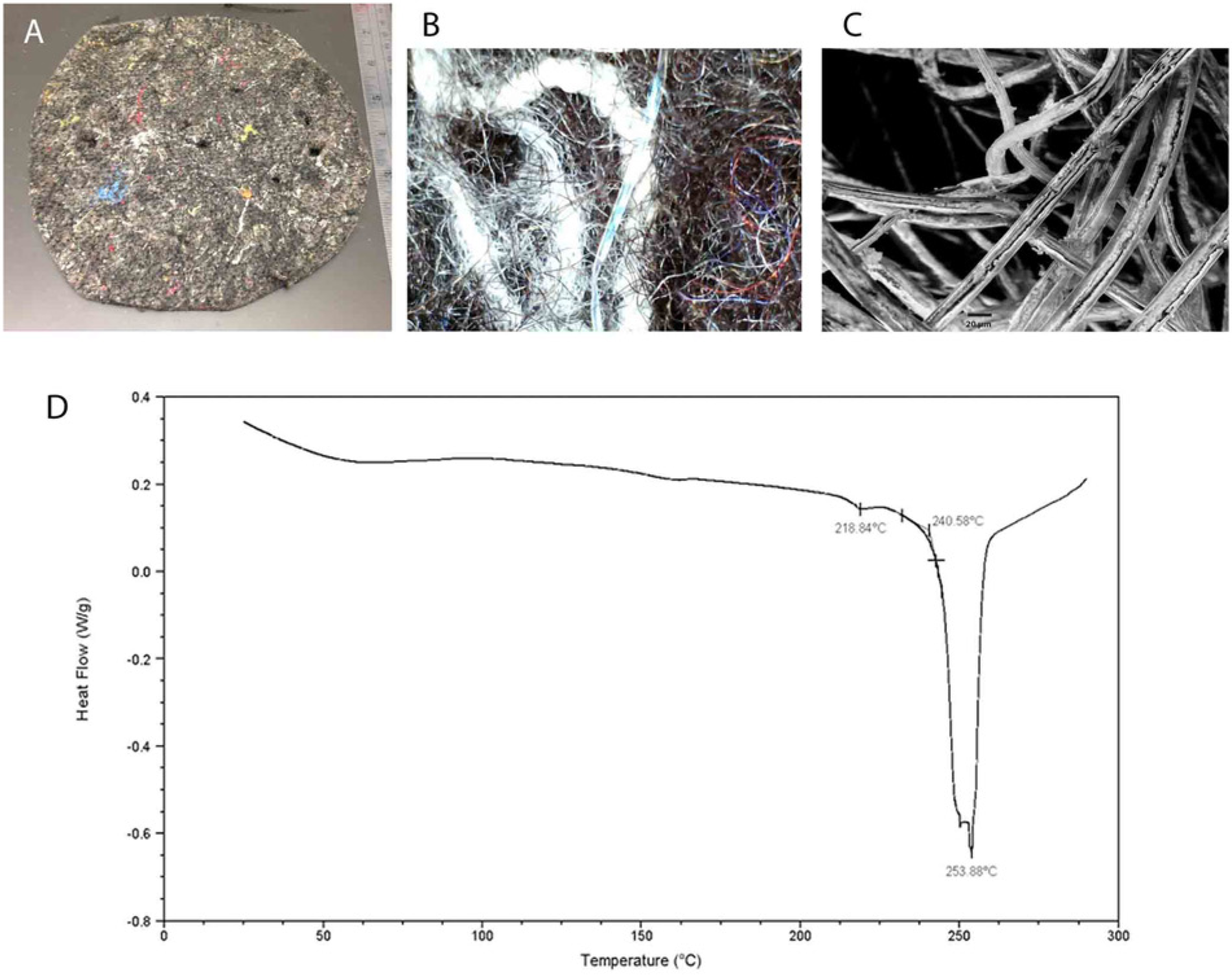
Physical properties of submitted nesting pads.
Nesting pads in both cases were made from a recycled textile material, manufactured by a company in Belgium, and sold as “Black European Felt Nesting Pads” at different retail stores in the United States. The 2 cases presented herein were very similar in terms of absence of clinical signs in adult birds and mortality of most hatchlings soon after hatching. The timeline for the use of specific nesting pads in different locations, onset of death, and the recovery after changing to new source pad in both cases strongly suggested that use of this specific nesting pad was associated with the clinical signs and mortality in these birds. Finding of permethrin in 1 sample suggested potential contamination of recycled material with this insecticide, but levels were not high enough to cause clinical signs in this case. Detection of DEHP and MEHP, a common plasticizer and its metabolite often detected in GC-MS screens, was also likely to be incidental in this case. Leaching of phthalates from plastic bags used to wrap the bedding material was the likely source. The finding of 5α-cholestan-3-one was also insignificant because, as a cholesterol derivative, it is likely to be of pigeon origin (e.g., feces); moreover, it was only found in the used sample of case 1.
The most significant gross finding was dark wet lungs found in case 1. Microscopically there was a proliferative interstitial pneumonia, which may suggest a response to pneumocyte necrosis in the air capillaries. Pathological effects of TDI by inhalation in various animal species (rabbits, guinea pigs, rats, and mice) have been investigated, and subepithelial edema, necrosis, ulceration, and acute inflammatory response in the tracheal/bronchial epithelium have been reported although there were interspecies variability. 7 Moreover, the detection of these lesions did not necessarily correlate with the species susceptibility to the compound evaluated by LC50. 7 Bronchial epithelium was not necrotic in the lung tissue submitted in case 1, and trachea was not submitted. Differences in target cells between experimentally exposed animals and the pigeons in case 1 may be due to differences in species. These histological changes were not observed in case 2. However, submitted samples in case 2 were mostly eggs (embryos) instead of hatchlings. Thus, lack of lesion in respiratory organs of case 2 could simply be because exposure to 2,4-TDI through inhalation had not occurred in those embryos although there could have been other routes of exposure, such as penetration through the eggshell. Lack of lesions in respiratory organs of 1 submitted hatchling might have been due to individual variability.
Other potential causes of the mortalities were also investigated by auxiliary testing. Elevated vitamin E and low vitamin A in the liver were additional diagnostic results in case 2. Elevated vitamin E has been reported to cause abnormal clotting and bone mineralization 13 as well as decreased pigmentation and waxy appearing feathers. 15 Clinical signs associated with vitamin A deficiency in poultry include decreased hatchability, impaired development, reduced growth rate and egg production, lethargy, ataxia, and increased lacrimal/nasal discharge. 5 However, there were no gross or microscopic lesions to suggest vitamin E toxicosis or vitamin A deficiency. Reference ranges for these vitamins are not available for this specific species at this age, and the fact that all adult birds appeared healthy made their significance unclear. Fresh tissues were not submitted for case 1, therefore these auxiliary tests could not be performed.
There are multiple potential reasons why the condition could not be reproduced experimentally, including differences in species susceptibility, behavior, and/or differences in exposure period (as the eggs were not incubated on the pads). It is possible that pigeons are particularly susceptible to the diisocyanates. Because pigeon chicks feed on crop milk, which is a regurgitated secretion product from the crop of adult birds, it is possible beak handling of nesting material by the adults resulted in oral exposure of diisocyanates in the chicks. Further investigation using pigeons during earlier stages of life may be warranted.
Toluene diisocyanate is used commonly during production of polyurethane foams. The reported residue of TDI in postproduction polyurethane foams, which are ubiquitously found in households and automobiles, for example, are usually lower, in the range of 3–20 ppb.8,11,20 Considering this and the high toxicity, finding 2,4-TDI >190 ppm in this case was unexpected. Nesting pads were made of primarily nylon-6. TDI is used for polymerization on nylon-6, 6 thus it is possible that residual TDI remained in the sample due to incomplete cross-linking or excess use of the compound. Presence of adhesive-like substances suggests that it is also possible that TDI was used in the adhesives 18 of these pads and resulted in high concentration of TDI. TDI is also used in the dyeing processes 9 and this could be an alternative reason for excess TDI in the pads. Although the results of SEM and DSC do not confirm the use of TDI, it suggests that multiple reasons may exist for high TDI residues.
It is of significant concern that high levels of TDI were found in the pads used for pigeon nesting considering that birds are generally more susceptible to respiratory toxicants. Toluene diisocyanate is considered one of the most hazardous occupational respiratory toxicants 1 and its effect on the ventilatory capacity can be observed even as low as 0.014 ppm in human beings. 16 The LC50 for single 4-hr exposure through inhalation assessed at 14 days postexposure ranges from 10 to 14 ppm in mammals but can be less toxic when exposed dermally or orally with LD50 ranging from 3 to 10 g/kg body weight (World Health Organization, International Programme on Chemical Safety: 1987, Environmental Health Criteria 75: toluene diisocyanate. http://www.inchem.org/documents/ehc/ehc/ehc75.htm). 21 Inhalation exposure limits have been set by several regulatory agencies such as Occupational Safety and Health Administration (OSHA) and American Conference of Governmental Industrial Hygienists (ACGIH) in 0.005–0.02 ppm ranges (ACGIH: 2004, Toluene-2,4 or 2,6-diisocyanate: TLV chemical substances. https://www.acgih.org/; OSHA: Occupational Safety and Health Standards—Table Z-1 limits for air contaminants, standard no. CFR 1910.1000 Table Z-1. https://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=STANDARDS&p_id=9992).
Methylene diphenyl diisocyanate (MDI) is considered less toxic but is also commonly used; therefore, regulated similarly as TDI. To date, there are no regulations of how much diisocyanates can be left as residues in consumer products, although there has been an action plan set forth by the U.S. Environmental Protection Agency (EPA: 2011, Methylene diphenyl diisocyanate and related compounds action plan, RIN 2070-ZA15. http://www.epa.gov/oppt/existingchemicals/pubs/actionplans/mdi.pdf; EPA: 2011, Toluene diisocyanate and related compounds action plan, RIN 2070-ZA14. http://www.epa.gov/oppt/existingchemicals/pubs/actionplans/tdi.pdf) to protect self-employed workers, consumers, and the general public using or unintentionally being exposed to products containing diisocyanates.
Clinical signs of respiratory difficulty and pulmonary lesions observed in the current case were consistent with reports of animals experimentally exposed to TDI. Clinical signs in many mammalian species (mice, rats, guinea pigs, rabbits, and dogs) include mouth breathing, lacrimation, salivation, restlessness, and hyperactivity, with terminal response being gasping for air and the cause of death is usually pulmonary edema and hemorrhage (World Health Organization, International Programme on Chemical Safety: 1987). 7 In human beings, clinical signs of TDI exposure by inhalation are severe irritation and burning of mucous membranes (eye, nose, throat), cough, laryngitis, chest pain, bronchitis, emphysema, and pulmonary heart disease as well as gastrointestinal symptoms such as nausea, vomiting, abdominal discomfort, 3 and neurologic signs in acute exposures. 12 Low and chronic exposures are also of concern for development of asthma, and TDI is one of the leading causes of occupational asthma among many industrialized countries.2,14,17 Therefore it is likely that exposure to TDI led to the respiratory problems and death in these chicks.
Toxicity information of diisocyanates in avian species is limited. According to a previous report, the oral LD50 of TDI in red-winged blackbirds is 100 mg/kg body weight, 19 which is much lower than the LD50 reported for rodents (>3,000 mg/kg body weight), suggesting higher susceptibility of avian species to this toxicant. Information on toxicity through inhalation and eggshells, or for exposures in chicks, is not available in any avian species. Although other potential causes, such as contamination of nesting pads with other toxic compounds but not detected by the analytical method cannot be completely ruled out, the presence of these highly toxic diisocyanates at high concentration is of concern.
Contamination of nesting material with volatile toxic compounds should be considered as differential diagnoses when there is high mortality in avian chicks. Although little is known on inhalation toxicity of diisocyanates in birds, care must be taken to prevent inhalation exposure, as avian species are particularly susceptible to inhalation toxicity. Additionally, a more rigorous quality control of nesting and bedding products should be established to prevent toxic residues, particularly because these products are used for neonatal animals and avian species, considering their susceptibility to toxicants.
Footnotes
Acknowledgements
The authors thank Mike Filigenzi for reviewing analytical results and providing suggestions for the article.
a.
Ethyl acetate (HPLC grade), Fisher, Waltham, MA.
b.
Agilent 6890/5973 gas chromatography/mass selective detector, Agilent Technologies, Santa Clara, CA.
c.
HP-1 capillary column, Agilent Technologies, Santa Clara, CA.
d.
2,4-toluene diisocyanate for standards (T39853), Sigma-Aldrich, St. Louis, MO.
e.
LEO 1550 field emission-scanning electron microscopy, Carl Zeiss AG, Oberkochen, Germany.
f.
Q2000 differential scanning calorimeter, TA instruments, New Castle, DE.
Authors’ note
Part of this work was presented during the oral session of 54th Annual Meeting of the American Association of Veterinary Laboratory Diagnosticians, October 1, 2011, Buffalo, New York.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
