Abstract
The aim of the current study was to compare feline hematologic variables in blood collected in microcapillary tubes (20 μl) and conventional blood tubes with the Medonic CA620/530 Vet in-house hematologic analyzer. A comparison of results obtained in 60 cats presented at the clinics of the veterinary school showed that the correlations between the 2 methods were 0.97 for white blood cell, 0.95 for red blood cell, and 0.93 for platelet counts; 0.92 for hemoglobin concentration; and 0.99 for mean corpuscular volume. No clinically relevant differences between the 2 blood sampling techniques were observed for any variable, which suggests that both techniques are interchangeable in cats. Moreover, microcapillary tubes would allow easier repeated sampling in the same cat and would likely be useful in other small species.
Feline blood sampling can be difficult in routine practice. One way to make it easier is to take minimally in vasive blood samples (e.g., by using microcapillary devices). 1 The validity of microcapillary blood sampling was discussed in a previous study, and no clinically relevant differences were demonstrated for major biochemical and hematologic analytes between blood collected in conventional vacuum tubes from the jugular vein and blood sampled in 20-μ1 microcapillary tubes from the cephalic vein. 11 A 20-μl microcapillary adapter was recently developed for the Medonic CA620/ 530 Vet impedance-based in-clinic veterinary analyzer a (CA620/530 analyzer, hereafter), which is an updated version of a previous analyzer validated for equine, canine, and feline conventional blood specimens. 12 In cats, the previous analyzer was reported to overestimate the white blood cell (WBC) count within or below the reference interval and to be inaccurate for the platelet count (PLT), which, according to the authors, may be caused by platelet aggregation (Gröndahl G, Bottema B, Lilliehöök I: 2004, Comparison of standard hematologic parameters determined on an impedance-based point-of-care hematology analyser using floating discriminators and an automated hematology analyser commonly used in veterinary reference laboratories. In: Proceedings of the 47th Annual British Small Animal Veterinary Association Congress, p. 568. Birmingham, UK). To the authors' knowledge, this analyzer has not been tested with 20-μl microcapillary blood samples, which reduce the volume needed for complete blood counts, thus making blood sampling less invasive and/or sparing more blood volume for other analyses. Therefore, the aim of the present study was to determine if the results obtained with the CA620/530 analyzer were the same for feline conventional and microcapillary blood samples.
Repeatability and within-laboratory imprecision of white blood cell (WBC), red blood cell (RBC), and platelet (PLT) counts; hemoglobin concentration (HGB); and mean corpuscular volume (MCV) with the Medonic CA620/530 Vet analyzer.*
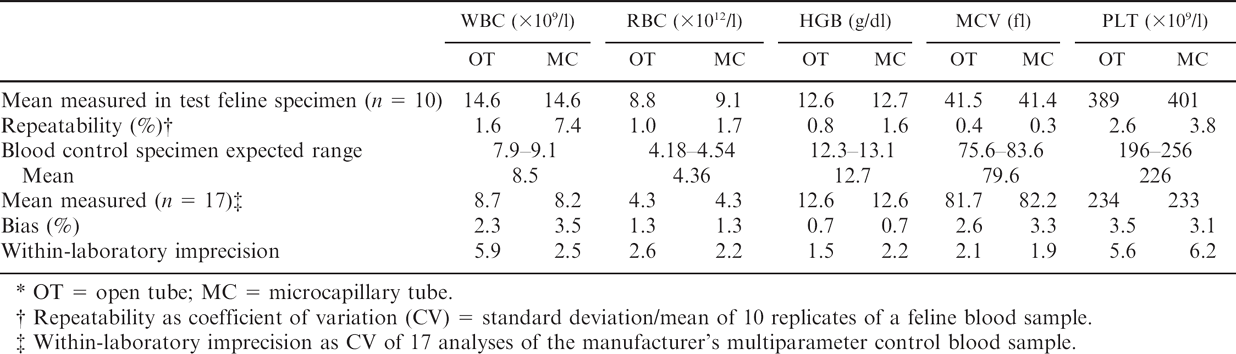
OT = open tube; MC = microcapillary tube.
Repeatability as coefficient of variation (CV) = standard deviation/mean of 10 replicates of a feline blood sample.
Within-laboratory imprecision as CV of 17 analyses of the manufacturer's multiparameter control blood sample.
Blood samples were obtained from 60 cats presented at the clinics of the veterinary school of Toulouse (France) from January to March 2007. No selection was made for breed, age, sex, or health status. Two samples were collected from each cat within less than 3 min by using the following methods: 1) a conventional 3.0-ml K3 ethylenediamine tetraacetic acid (EDTA) tube, b with a 22-gauge needle, by jugular venipuncture; 2) a 20-μl K2 EDTA microcapillary tube, c with a 25-gauge needle, by sampling from the cephalic vein. The order of sampling was randomized. All analyses were performed within 5 min by using the same CA620/530 analyzer. Before the analyses were done, a 15-day period of training with the new system was accomplished to ensure working competence. 5 The calibration of the analyzer was done before each series of measurements with the manufacturer's multiparameter veterinary hematology control d by using both the open tube (OT) and microcapillary (MC) modes. The calibration was repeated if the controls were outside the manufacturer's acceptability range, which occurred in 5 cases of 17. The results obtained forcalibration were used to calculate within-laboratory imprecision (n = 17). Repeatability was evaluated from 10 replicates of the same feline blood sample with both OT and MC modes. For each animal, duplicate measurements of the OT and measurements of the duplicate MC tubes were obtained for the following analytes: WBC, red blood cell (RBC), and PLT counts; hemoglobin concentration (HGB); and mean corpuscular volume (MCV).
After applying descriptive statistics to each set of data, the OT and MC results were compared by using the Student's paired t-test (after testing for the homogeneity of variances), correlation coefficients, Passing–Bablok regression analysis, Bland–Altman diagrams of difference with an Excel spreadsheet, and the Analyse-It e set of macroinstructions. 5,6 Medical decision (MEDx) charts were also used based on the clinical laboratory improvement amendments (CLIA) values of total allowable error (TAE) in human clinical pathology for all analytes 5 except MCV, which is not reported. For MCV, the TAE was determined according to Tonks 13 by using the reference interval for feline MCV in Schalm's textbook 2 (i.e., 8.5%). Possible clinical misinterpretations were based on the reference intervals of all analytes in Schalm's textbook. 2
Coefficients of variation of repeatability were <2%, except for the PLT count in OT and MC and WBC counts in MC (Table 1). Within-laboratory imprecision was <2.6%, except for the PLT count with both tubes and the WBC count in OT (Table 1). Accuracy could not be evaluated from control feline samples because these samples are not available. With the manufacturer's control specimen, results were close to the expected value within the range of acceptability (Table 1). Based on MEDx charts (data not shown), the overall agreement of results was excellent for the WBC count and MCV, good for the PLT count, and marginal for the RBC count and HGB concentration.
The means of all duplicates were used in the comparison of OT and MC (Table 2). The difference between OT and MC was not statistically significant for the WBC count, RBC count, and HGB concentration, whereas it was statistically significant for MCV and PLT count (Student's paired t-test, P < 0.05), even if differences were minimal.
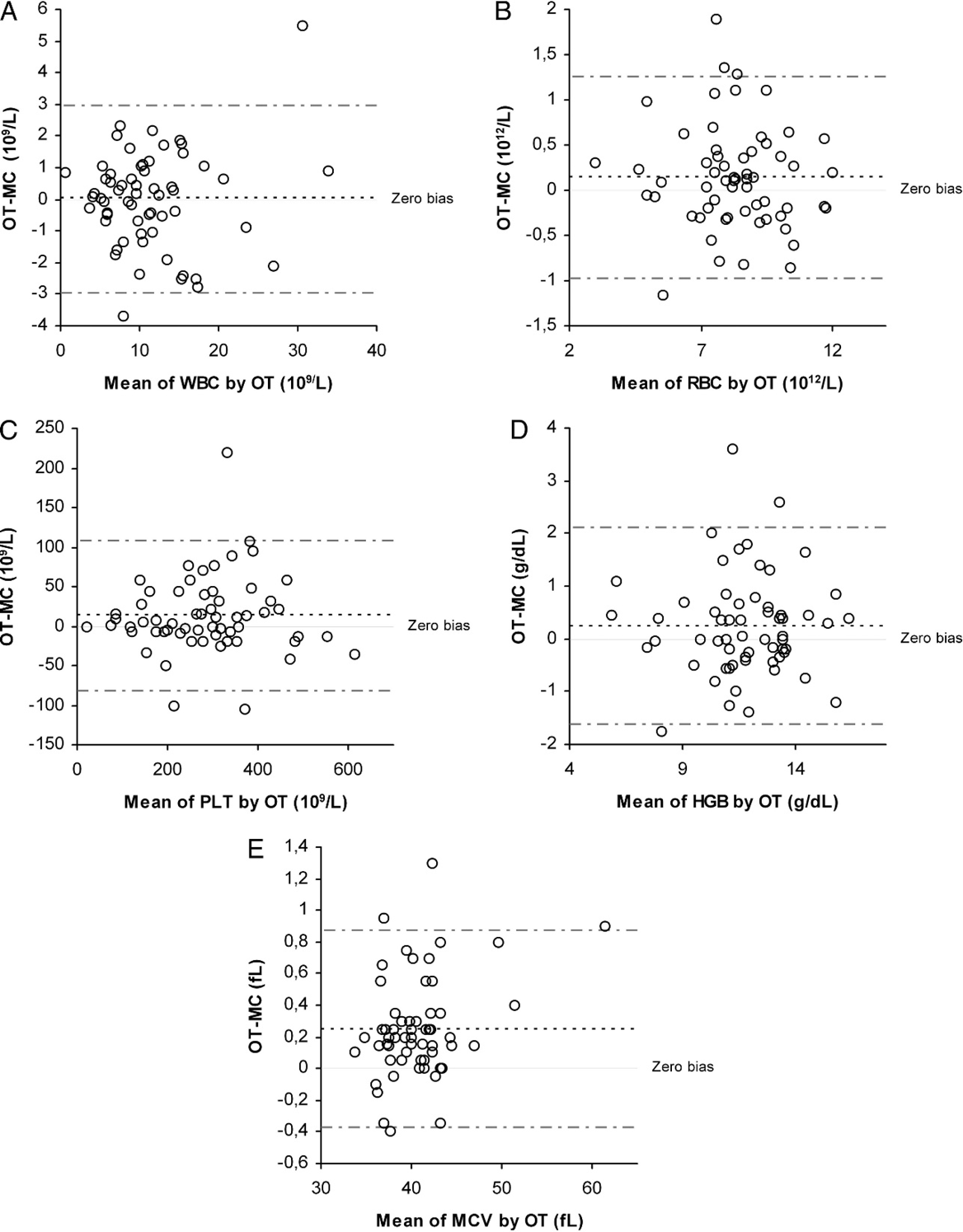
Diagrams of differences between results obtained with conventional open tubes (OT) and microcapillary tubes (MC) in 60 feline blood samples. A, white blood cell count (WBC); B, red blood cell count (RBC); C, platelet count (PLT); D, hemoglobin concentration (HGB); and E, mean corpuscular volume (MCV). Dotted lines are mean bias and 95% confidence interval.
Results for white blood cell (WBC), red blood cell (RBC), and platelet (PLT) counts; hemoglobin concentration (HGB); and mean corpuscular volume (MCV) in 60 feline blood samples collected with conventional open tubes (OT) and microcapillary tubes (MC).
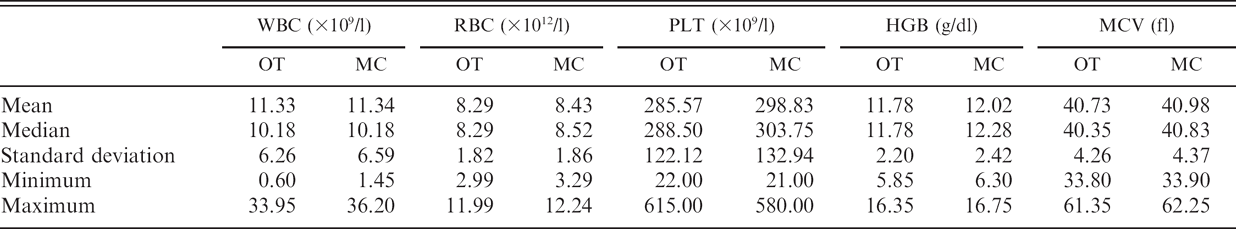
The differences between the means were 13.109/1 and less than 1 fl for the PLT count and MCV, respectively. The slopes of the Passing–Bablok equations (Table 3) were close to 1, except for HGB concentration, and their confidence intervals all included 1. The intercepts were close to 0, except for the HGB concentration and the PLT count, and their confidence intervals all included 0. All correlation coefficients of measurements made in OT and MC were >0.92. Bias was constant for all analytes (Fig. 1). The means of differences (MC – OT) were 0.01 10 9 /1, 0.14 1012/l, 13.26 109/1, 0.24 g/dl, and 0.25 fl for the WBC count, RBC count, PLT count, HGB concentration, and MCV, respectively. Most individual differences between OT and MC were lower than would be expected from analytical variability. There were very few clinical misinterpretations (i.e., results that would be classified differently as physiologic vs. pathologic according to Clinkenbeard's reference limits). 2 Misinterpretations were observed in 2, 4, 3, 4, and 0 cases for the WBC count, RBC count, PLT count, HGB concentration, and MCV, respectively.
The current study compared feline hematologic data obtained with conventional tubes and microcapillary tubes. Because no clinically relevant differences in hematologic variables between blood samples collected from different sites in cats was previously reported, 3,4,11 no effects of the site of blood collection were expected. The clinical impression of the current study was that the cats were less stressed by MC than OT sampling. Sixty cats were sampled, which was assumed to represent a sufficient number of animals, as recommended for comparisons of methods. 5,9 In the present study, most analytical variations were controlled, because the blood specimens were analyzed by both methods within 5 min after venipuncture, thus avoiding changes because of instability of samples. Moreover, the order of samples was randomized to prevent systematic error that results from possible analyte instability.
The quality of the results was warranted by working competence because of preliminary training with the system, quality control according to manufacturer's recommendations, and duplications of analyses to identify possible analytical outliers and to reduce analytical variation. Moreover, if the quality control proved inadequate, then the feline samples were not analyzed. Repeatability was almost the same as reported by the manufacturer for humans (i.e., 2.0% for the WBC count, 0.85% for the RBC count, 0.8% for the HGB concentration, 0.5% for MCV, and 3.2% for the PLT count). Within-laboratory imprecision was higher with MC than OT, except for a paradoxically higher value for WBC count in OT, which could not be explained. However, this imprecision was within the limits for TAE according to CLIA for human clinical pathology. 8 High within-laboratory imprecision was also observed for PLT counts in both tubes. Repeatability was better for OT than MC. This might be explained by the easier use of OT compared with MC, both at the sampling step for ensuring perfect filling of the tube and at the analytical step when inserting the MC into the adapter.
The purpose of the present experiment was to compare results obtained with conventional and microcapillary tubes. Analyses were all done within 5 min after blood collection, which could be considered as a much shorter time than usual, whereas routine analyses are typically performed within 30–60 min. However, it is likely that practitioners willing to use MC samples would not allow them to stay unanalyzed more than a few minutes. This justifies the short delay used in the current study. When differences were observed, it was not possible to determine which of the results was the more accurate because no reference method was used. Correlations for the WBC count, RBC count, and MCV were 0.97, 0.95, and 0.99, respectively, with slopes close to 1 and intercepts close to 0. This suggests that the 2 techniques are interchangeable in cats. For HGB concentration, the correlation was only 0.92, and there was a bias that could result in a total error of about 10%, which is higher than the 7% TAE recommended by CLIA in human clinical pathology. 8 The discrepancy in the PLT count between OT and MC probably resulted from suboptimal separation between platelets and RBCs. This could be expected in cats, which normally have large platelets and small RBC and are prone to platelet clumping. 2,7 Moreover, in the current study, all samples with an “abnormal” PLT count showed thrombocytopenia. Similar difficulties in PLT counting and the high prevalence of thrombocytopenia in automated PLT counts were already reported and were attributed to a combination of platelet aggregation and the impedance method of cell differentiation by size. 10 Furthermore, in a previous study that used the Medonic CA530 Vet analyzer, which involved the same technology as the CA620 Vet, PLT counts were also found to be abnormally low, with no real explanation other than PLT aggregates. 12 Nevertheless, most differences between OT and MC were <50.109/1, which was below the 25% TAE recommended in human clinical pathology. 8
Passing–Bablok equations* and correlation coefficient (r) relating measurement of white blood cell (WBC), red blood cell (RBC), and platelet (PLT) counts; hemoglobin concentration (HGB); and mean corpuscular volume (MCV) in feline blood samples (n = 60).
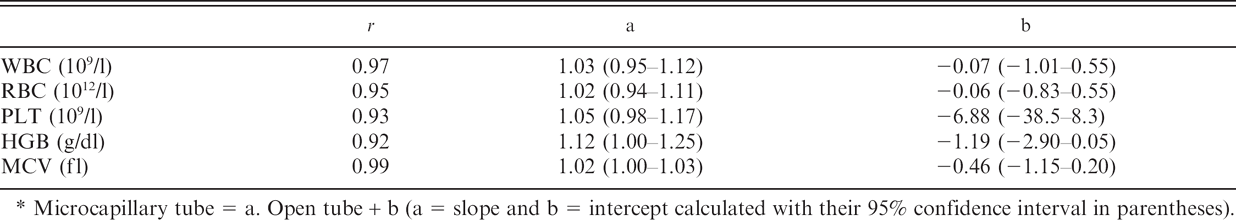
Microcapillary tube = a. Open tube + b(a = slope and b = intercept calculated with their 95% confidence interval in parentheses).
Based on the generally very high correlations for all analytes, even the PLT count and HGB concentration, it was not surprising that very few clinical misinterpretations between OT and MC were observed. The reference intervals chosen were from Schalm's textbook, 2 because no reference interval has been determined with the CA620 Vet analyzer. However, the accuracy of the reference limits has little relevance in the present study, because its aim was to investigate whether possible analytical differences between OT and MC may result in clinical misinterpretation. It is likely that reference intervals taken from other sources would have resulted in similar observations. In routine practice, these differences would not have changed the clinical decision threshold except possibly for the approximately 1 g/d1 bias in HGB concentration. Nevertheless, when clinical differences between OT and MC were found, it was not possible to determine which of the 2 methods was the more accurate, because no reference method was used. With the Cell-Dyn 3500 as a reference, it was shown that both the CA620 Vet and CA530 Vet 12 analyzers could be used for routine feline blood cell analyses, except for PLT and total WBC counts in OT with the CA530 Vet (Gröndahl G, et al.: 2004, Comparison of standard hematologic parameters determined on an impedance-based point-of-care hematology analyzer).
This first test of MC demonstrated that the results obtained in MC and OT were similar, except for the HGB concentration. It also confirmed that there were no clinically significant differences between blood sampled from the jugular and from the cephalic veins. 11 Moreover, the very small volume collected and the moderate restraint required during microsamplings were suitably adapted to cats, which are typically very reluctant to be restrained.
Such MC would also allow easier repetition of sampling in the same cat and could be useful in other small species.
Footnotes
a.
Medonic CA620/530 Vet analyzer, Boule Medical AB, Stockholm, Sweden.
b.
Venoject® EDTA (K3) K3E, Terumo Europe NV, Leuven, Belgium.
c.
MPA micropipette plastic, Boule Medical AB, Stockholm, Sweden.
d.
Boule Vet-CON, Boule Medical AB, Stockholm, Sweden.
e.
Analyse-it Software Ltd., Leeds, UK.
