Abstract
Iridoviruses infect food and ornamental fish species from a wide range of freshwater to marine habitats across the globe. The objective of the current study was to characterize an iridovirus causing systemic infection of wild-caught Pterapogon kauderni Koumans 1933 (Banggai cardinalfish). Freshly frozen and fixed specimens were processed for histopathologic evaluation, transmission electron microscopic examination, virus culture, molecular virologic testing, microbiology, and in situ hybridization (ISH) using riboprobes. Basophilic granular cytoplasmic inclusions were identified in cytomegalic cells often found beneath endothelium, and hexagonal virus particles typical of iridovirus were identified in the cytoplasm of enlarged cells by transmission electron microscopy. Attempts at virus isolation in cell culture were unsuccessful; however, polymerase chain reaction (PCR)-based molecular testing resulted in amplification and sequencing of regions of the DNA polymerase and major capsid protein genes, along with the full-length ATPase gene of the putative iridovirus. Virus gene sequences were then used to infer phylogenetic relationships of the P. kauderni agent to other known systemic iridoviruses from fishes. Riboprobes, which were transcribed from a cloned PCR amplification product from the viral genome generated hybridization signals from inclusions within cytomegalic cells in histologic sections tested in ISH experiments. To the authors' knowledge, this is the first report of a systemic iridovirus from P. kauderni. The pathologic changes induced and the genomic sequence data confirm placement of the Banggai cardinalfish iridovirus in the genus Megalocytivirus family Iridoviridae. The ISH provides an additional molecular diagnostic technique for confirmation of presumptive infections detected in histologic sections from infected fish.
Introduction
Iridoviruses are a frequently encountered group of DNA viruses found in invertebrates, fish, and amphibians, which are associated with a range of disease presentations from skin lesions to systemic infections. 7,9,10,21,30,37,61,63 The family Iridoviridae contains 5 genera, 3 of which are found in fish. 8 One particular genus, Megalocytivirus, contains viruses associated with serious systemic infections resulting in significant mortality (up to 100%) among a growing list of marine and freshwater fishes. 2,25,43,44,50 The Red Sea bream iridovirus (RSIV), which caused major losses in Japan in 1990, was among the first megalocytiviruses described. 31 Similar viruses were circulating in populations of ornamental fish prior to the recognition of RSIV, as revealed by histologic and electron microscopic evidence from diseased orange chromide cichlids exhibiting similar clinical signs. 3 Since these initial observations, megalocytiviruses have been reported among numerous marine and freshwater fish species either endemic or originating in exports from Japan, the South China Sea, and several Southeast Asian countries. 5,8,48,49,54,59 Sequences of the entire genomes of 4 viruses in the genus Megalocytivirus species (i.e., Rock bream iridovirus [RBIV], Infectious spleen and kidney necrosis virus [ISKNV], Orange spotted grouper iridovirus [OS-GIV]), and Ehime-1 strain [RSIV]) are currently known. 12,23,36,40 Sequence data from additional megalocytiviruses indicate a close relationship to the type species ISKNV, sharing up to 97% or greater identity at the deduced amino level for the major capsid protein. 8
Wide host and geographic ranges of this pathogen coupled with the severity of infections caused by these megalocytiviruses have had a major impact on cultured fish and likely have had negative effects on wild fish populations as well. 43,62 Infections of ornamental and farmed food fish in both marine and freshwater environments can have devastating effects on profits of public aquaria, the ornamental hobby fish trade, and food fish industries. Iridoviruses are very stable in aquatic environments, a feature that facilitates their transmission by waterborne routes, as demonstrated in experimental trials. 20,46,60 Natural and experimental infections result in severe disease, as exemplified by morbidity and mortality rates of 50% or greater 41,42 depending on key environmental factors (e.g., water temperature), stressors, or other immunologic challenges often faced by cultured or ornamental fish. 7,25 The stress of transport, acclimation, mating/courtship behaviors, reproduction, and high stocking densities may result in the progression of infections from subclinical to clinical with subsequent and rapid spread through populations. 63
Fish with megalocytivirus infections have exhibited several consistent clinical findings, including lethargy, severe anemia, and branchial hemorrhages, and on gross necropsy, fish have had markedly enlarged spleens. 3,19,25,30,39,41,44 Microscopic examinations of fish with clinical signs revealed large, basophilic, cytomegalic cells in multiple organs including spleen, kidney, intestine, eye, pancreas, liver, heart, gill, and brain, as well as lamina propria and connective tissue throughout the body. 19 Cytomegalic change was seen primarily in cells in subendothelial locations, along with degenerative or necrotic changes in surrounding cells. Ultrastructurally, cytomegalic cells were observed directly below the endothelium, occasionally capped by endothelial cells with large nuclei. Enlarged cells contained electron-dense inclusions consisting of arrays of iridovirus-like particles. 19 During RSIV infection, experimental treatment of infected cells with caspase-3 and caspase-6 inhibitors resulted in cell rounding, but inhibited cell enlargement and formation of apoptotic body-like vesicles, suggesting caspase-3 and caspase-6 may have some role in viral protein synthesis in the latter stages of infection. 29 Histologic detection of the virus with confirmation by electron microscopy was often the primary diagnostic approach employed due to difficulties isolating and propagating these agents in cell culture. 19
Pterapogon kauderni Koumans 1933 (Banggai cardinalfish) is a popular aquarium apogonid endemic to the Banggai Archipelago in Eastern Indonesia. It inhabits a variety of shallow habitats (commonly between 1.5 and 2.5 m) on the margin of about 30 islands, including coral reefs and seagrass beds, with a water temperature range between 29°C and 32°C. It is a sedentary species forming small groups, frequently composed of a few dozen individuals of mixed age classes that prefer to co-inhabit areas with sea urchins, anemones, and branching corals, affording these small fish refuge. Pterapogon kauderni combines a number of biologic characteristics that make it an exceptional species for natural history research (Vagelli A: 2005, Reproductive biology, geographic distribution and ecology of the Banggai cardinalfish Pterapogon kauderni Koumans, 1933 [Perciformes, Apogonidae], with considerations on the conservation status of this species on its natural habitat. PhD Thesis. University of Buenos Aires, Argentina. In Spanish with English abstract). 4,27,55–58 Unfortunately, the human impact on P. kauderni is very significant. Wild fish are collected throughout the Banggai region for the international ornamental fish trade. Currently, at least 100,000 specimens are extracted from the wild every month. This unsustainable harvest, coupled with rampant habitat destruction and pollution, has caused populations to decline further, requiring the P. kauderni to be added to the International Union for Conservation of Nature (IUCN) Red List as an “Endangered” species in 2007 (http://www.iucnredlist.org/search/details.php/63572/summ; Lunn KE, Moreau MA: 2002, Conservation of Banggai cardinalfish populations in Sulawesi, Indonesia: an integrated research and education project. SPC Live Reef Fish Info Bull 10:1–28. Available at: http://www.spc.int/coastfish/News/LRF/10/LRF10.htm#2. Accessed September 26, 2008; Vagelli A: 2005, Reproductive biology, geographic distribution and ecology of the Banggai cardinalfish). 57 Further losses of P. kauderni due to both endemic viral infections in wild populations and unintentional release of infected animals in waters containing naïve fishes may be another factor threatening this unique species.
In 1996, the New Jersey Academy for Aquatic Sciences (NJAAS; Camden, NJ) initiated a comprehensive research endeavor to describe the natural history of this fish, to investigate the biology and physiology of this species, and to survey wild populations and mitigate conservation of P. kauderni if necessary. Both laboratory and field studies were conducted, including the development of a captive breeding program. Since the beginning of this project, live specimens of P. kauderni have been kept for studies on reproductive biology, nutrition, and behavior. 56–58 Purchase of about 10–15 wild-caught specimens for laboratory broodstock are made periodically to avoid inbreeding. In the early 2000s, reports of sudden mass mortality of newly purchased wild-caught specimens of P. kauderni were spreading within the fish hobbyist community. In both 2003 (10 fish) and 2004 (20 fish), groups of newly acquired specimens at NJAAS showed abnormal behavior 2 to 3 days after arrival, and in 2003 all 10 fish died within a week after shipment. Necropsies were performed on a few specimens, and macroscopic observations led to suspicion of a potential viral infection. On the west coast of the United States, similar observations were made in 2005 (30 fish) after a breeding facility reported nearly 100% mortality of wild-caught P. kauderni recently obtained from wholesalers.
The present study reports the diagnosis and characterization of a systemic iridovirus infection of P. kauderni responsible for morbidity and mortality in wild-caught specimens commercially acquired by the NJAAS on 2 separate occasions, and by an aquaculture facility in California. In this study, histologic, ultrastructural, and molecular data for a virus observed among P. kauderni were assembled and compared to understand both the pathologic features of infection by this agent and the phylogenetic relationship of this pathogen to other previously described systemic iridoviruses identified in other food and ornamental fishes.
Materials and methods
Sample collection
A total of 12 specimens of P. kauderni were received by the Connecticut Veterinary Medical Diagnostic Laboratory from the NJAAS in 2003 (4 fish) and 2004 (8 fish) to investigate the cause of mortalities of multiple cohorts after transport and apparent acclimation. Specimens from 2003 were fixed in neutral buffered 10% formalin or 70% alcohol. Specimens from 2004 were frozen immediately after death or fixed in McDowell-Trump fixative 38 for histopathologic and electron microscopic examination. Frozen fish were necropsied upon thawing, and samples of kidney, spleen, liver, and heart were submitted for bacteriologic culture and virus isolation at a referral laboratory. Specimens from a commercial source were submitted in 2005 (5 fish) to the Fish Health Laboratory (University of California, Davis, CA) for health evaluation and were fixed in neutral buffered 10% formalin.
Histopathology
Fixed specimens from 2003 and 2004 were either decalcified in 0.5 M ethylenediamine tetra-acetic acid (EDTA) and then cut into serial transverse sections, or dissected upon arrival. Fixed specimens from 2005 submissions were midline-sagittally sliced. Slices of whole fish and dissected tissue samples were trimmed to fit plastic cassettes, embedded in paraffin, sectioned at 4 μm, mounted onto glass slides, and stained with hematoxylin and eosin.
Transmission electron microscopy
Tissue samples of spleen, gill, and heart from specimens of the 2004 cohorts were trimmed to fit flat-bottomed BEEM capsules, a then immersed in McDowell-Trump fixative and refrigerated until processing. Tissue samples were prepared using a microwave protocol based on previous methods. 17,18 All procedures were performed in a commercial microwave system with variable wattage b on a tempered-glass surface b using specimen holders. b In brief, tissue samples were postfixed in 1% OsO4 in 0.1 M N-2-hydroxyethylpiperazone-N-2-ethanesulfonic acid (HEPES) buffer under a vacuum of 25 mmHg, rinsed with 0.1 M maleate buffer (pH 5.2), en bloc stained with 1% uranyl acetate in 0.1 M maleate buffer, dehydrated in a graded series of ethanol, then infiltrated under a vacuum of 25 mmHg and embedded using LR (London Resin) White resin. c Thin sections of embedded tissues were cut on a diamond knife, d placed onto mesh grids, stained with uranyl acetate and lead citrate, then viewed in an electron microscope operated at 80 kV. e
Bacteriologic testing and virus isolation
Tissue sample sets consisting of either cranial kidney and spleen or cranial kidney, spleen, heart, and liver were collected from the frozen specimens of the 2004 cohort and were sent to the Washington Animal Disease Diagnostic Laboratory (WADDL; Pullman, WA) for aerobic bacterial cultures and virus isolation following standard procedures. 15 For virus isolation, tissue homogenates were inoculated onto several cell lines, which were maintained for a minimum of 3 passages. The cell lines included: EPC (epithelioma papulosum cyprini, derived from fathead minnow, Pimephales promelas), FHM (derived from fathead minnow, Pimephales promelas), BF-2 (derived from bluegill sunfish, Lepomis macrochirus), and CHSE-214 (derived from Chinook salmon embryo, Oncorhynchus tshawytscha).
DNA extraction, polymerase chain reaction amplification, cloning, and sequencing
Upon completion of the attempts at virus isolation, materials were forwarded to the Fish Health Laboratory at University of California, Davis (UC Davis). DNA was extracted from samples of viscera using a commercial extraction kit f following the protocol for animal tissues. Extracted DNA was stored in buffer at 4°C until tested by polymerase chain reaction (PCR).
Degenerate primers were used to amplify a fragment of the DNA polymerase gene of the Banggai cardinalfish iridovirus (BCIV), following a PCR protocol previously described. 22,31 Genomic sequence alignments of the ATPase gene and flanking regions from ISKNV (ORF 122R) 14 and OSGIV (ORF 119R) 14 facilitated the design of primers to amplify the full length ATPase gene of BCIV (e.g., ATPase-F2: 5′-GTGTGTATTTGTCTTTGTGCAC-3′ and ATPase-R2: 5′-GCTGGGCGACCCCATTCG-3′). The ATPase amplicon was generated using the PCR conditions previously described. 34 A second primer pair previously developed 49 was used to obtain further sequence downstream of the 3′ end of the ATPase gene (e.g., P3-IRB5: 5′-GTACGGGGCTT-GATGATGACGT-3′ and P4-IRB5: 5′-CCTTGTGTCGTGTCTGGCCGAG-3′).
Amplicons were purified from 10 μl of the PCR product by excision of bands from agarose gels and then cloned using a topoisomerase I (TOPO) Taq-amplified (TA) cloning kit and plasmid pCR2.1-TOPO. g Transformed bacterial colonies were screened using PCR as described by the cloning kit using the M13 primers, and plasmids containing relevant inserts were purified using a commercial kit. f Purified plasmids were sequenced by the dideoxynucleotide chain termination method 45 using M13 forward and reverse primers and an automated sequencer. h Purified plasmids containing either the P3-IRB5/P4-IRB5 or DNA polymerase amplicons were forwarded to the Department of Pathobiology and Veterinary Science, University of Connecticut (Storrs, CT), for riboprobe transcription and subsequent in situ hybridization (ISH) experiments.
Primers used in the current study to amplify fragments of major capsid protein gene from iridoviruses.
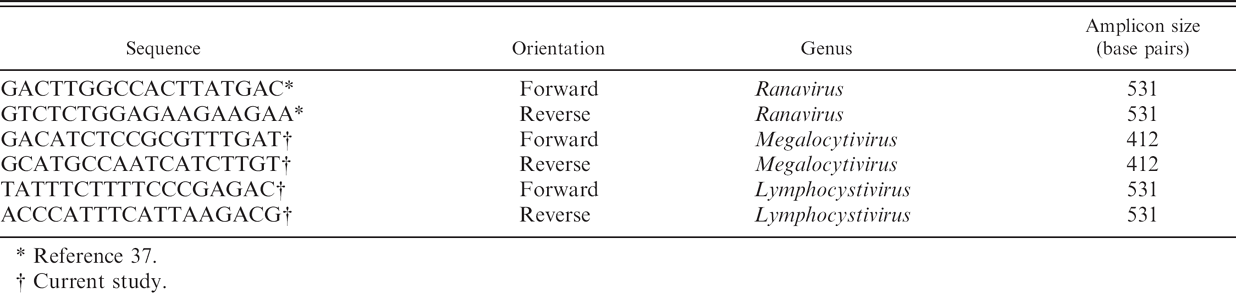
Reference 37.
Current study.
Amplification of a segment of the major capsid protein (MCP) gene from BCIV was obtained from DNA extracted from 20-μm sections of formalin-fixed, paraffin-embedded tissues of specimens of the 2004 cohort confirmed by previous histopathologic evaluations as having iridovirus inclusions. DNA was extracted using a commercial extraction kit f according to the manufacturer's instructions. A 50-μ1 PCR mixture was prepared containing 200 μmol of each deoxyribonucleotide triphosphate (dNTP), 200 pmol of each primer (Table 1), 5 μl of Advantage2 10X PCR buffer, i 2 U of Advantage2 DNA polymerase, i and 50 ng of total DNA. Cycling conditions included a holding step at 95°C for 1 min followed by 35 cycles at 95°C for 30 sec, 55°C for 30 sec, and 68°C for 1 min. Amplicons were resolved in 1% wt/vol agarose-TAE (Tris-acetate 0.4 M, 0.01 M EDTA Na 2 ), and gels were stained with ethidium bromide for visualization under ultraviolet (UV) light. Amplicons of expected molecular size, 412 base pairs (bp; primers included), were excised from agarose gels and purified using a commercial gel extraction kit f according to the manufacturer's instructions. Purified DNA fragments were then cloned into pCR1 TOPO TA vector using a TOPO TA cloning kit g following the manufacturer's instructions. Plasmid DNA was purified using a commercial kit f from selected clones, after a blue/white screening of bacterial colonies on lysogeny broth (LB) Xgal (5-bromo-4-chloro-3-indolyl-beta-D-galacto-pyranoside)/ampicillin plates. DNA clones were sequenced using the dideoxynu-cleotide chain termination method. 45 , j
BLASTP, molecular dataset, sequence alignment, and phylogenetic analysis
Following cloning and sequencing, general BLASTP searches (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi) using the amino acid sequences deduced from the partial DNA polymerase, MCP, and full-length ATPase genes of the BCIV were performed in order to acquire appropriate outgroups for phylogenetic analysis. 1
The deduced amino acid sequences of the partial DNA polymerase, ATPase, and MCP genes were aligned using Mafft 5.8 33 followed by minor manual adjustments in ClustalW. 52 The E-INS-I alignment strategy was used with the following parameters: scoring matrix (BLOSUM62), gap open penalty (1.53), and offset value (0).
To assess gene concordance, the primers were removed from the sequences and Bayesian analyses were performed independently for each gene. Phylogenetic trees were constructed using Bayesian analyses (MrBayes version 3.1.2). 28 A mixed prior was used on amino acid models and default priors for topology (uniform) and branch lengths (Exp and 10). The Markov chain was run for a maximum of 10 million generations, with a stopping rule implemented so that the analysis would halt when the average deviation of the split frequencies was less than 0.001%. Four independent analyses were conducted, each with 1 cold and 3 heated chains with the default heating parameter (temperature = 0.2). Every 50 generations were sampled and the first 25% of mcmc samples discarded as burn-in.
In vitro transcription of riboprobes
For the purpose of transcribing sense- and antisenseoriented riboprobes, primers and protocols previously described were used to amplify DNA polymerase and P3-IRB5/P4-IRB5 amplicon inserts from original plasmids. Polymerase chain reaction products were separated in 1.5% agarose gels, stained with ethidium bromide, and visualized by UV transillumination alongside a pGEM molecular weight marker k to verify amplification of appropriately sized products. Amplicons were purified from agarose gels using reagents and protocol provided by a commercial gel DNA recovery kit l and cloned using the TOPO TA cloning kit and pCR2.1–TOPO plasmid. g Plasmids were purified using a commercial purification kit f and were quantified by UV spectrometry and fluorometry m using Hoechst dye.
Plasmid inserts were sequenced by dideoxynucleotide chain termination sequencing method 45 using the M13 forward and M13 reverse primers. j Sequence data was examined using Sequencher version 4.0 n and DNAMAN, o then compared with original sequence data and used to determine the orientation of the PCR product insert in each plasmid and to determine appropriate restriction enzymes for linearization. Clones with inserts in the sense and antisense orientations of each PCR product were selected and reproduced in larger numbers using the manufacturer's reagents and the protocols provided, f then quantified by fluorometry, UV spectrometry, and estimation by comparison with molecular weight standards using a pGEM marker p after separation in 1.5% agarose gels.
Riboprobes were generated by transcription of cloned PCR products. Select plasmids were linearized with HindIII. g Linearized plasmids were the template for transcription reactions using a commercial T7 DIG (digoxigenin) RNA labeling kit q as described by the manufacturer. Concentrations and appropriate DIG-labeling of resultant transcribed riboprobes were estimated by dot blot analyses using alkaline phosphatase-conjugated anti-DIG Fab fragments q and 5-bromo-4-chloro-3-indolyl phosphate/4-nitro blue tetrazolium (BCIP-NBT) q color substrate system.
In situ hybridization using riboprobes
Tissue sections of P. kauderni were chosen for use in nonisotopic ISH experiments based on presence of lesions and density of inclusions in affected tissues. Fixed specimens from 2003 and 2004 cohort samplings processed for paraffin embedment as previously described were included. Paraffin blocks from formalin-fixed, paraffin-embedded specimens from 2005 submissions were provided by the Fish Health Laboratory. Procedures were adapted from previous researchers. 6,13,16 Serial sections were mounted unstained on saline-coated glass slides. Slides were heated to 70°C for 20 min to melt paraffin, and then washed in xylene for three 3-min washes. Tissues were allowed to dry, circled with a peroxidase anti-peroxidase (PAP) pen, and then rehydrated using rehydration buffer (phosphate buffered saline, 5 mM MgCl 2 ) for 10 min at room temperature. Tissues were digested by incubation with proteinase K solution (20 mg/ml proteinase K in 10 mM Tris, pH 7.5, 2 mM CaCl2) for 7 min at 37°C in a humidified slide moat. k Enzyme activity was stopped by washing slides in coplin jars filled with 0.2 M Tris pH 7.5 and 0.1 M glycine solution for 2 min. Slides were then returned to the humidified slide moat, and prehybridization solution (5X saline-sodium citrate [SSC; 1X SSC = 0.15 M NaCl and 0.015 M sodium citrate], 5% blocking reagent, 48% formamide, 0.02% sodium dodecyl sulfate [SDS], 0.1% N-lauroylsarcosine) was applied to surfaces of tissue sections. Slides were coverslipped and incubated for 1 hr at 38°C. While slides remained coverslipped, target DNA was denatured by heating slides covered with prehybridization solution to 95°C and incubating at this temperature for 3 min. Coverslips were removed and prehybridization solution was drained from slides. Each slide was then coated with 100 μl of prehybridization buffer containing 35–100 ng of riboprobe in optimization experiments and 85 ng of riboprobe thereafter, then coverslipped, and sealed with rubber cement. Slides were incubated in the humidified slide moat at 38°C overnight. Following overnight incubation, slides were washed in coplin jars filled with 2X SSC with 1% SDS at 50°C for 20 min, followed by 1X SSC with 0.1% SDS at 50°C for 20 min, then 1X SSC at room temperature for 10 min, and 0.1X SSC at room temperature for 15 min. Slides were let stand in buffer 1 (0.1 M Tris, 0.15 M NaCl, pH 7.5) for two 5-min incubations. Slides were placed in the humidified slide moat and covered with 100 μl of buffer 1 containing 2% sheep serum r and alkaline phosphatase-conjugated anti-DIG Fab fragments at a 1:275 dilution, coverslipped, and incubated for 2 hr at 37°C. Following incubation, slides were washed in coplin jars filled with buffer 1 at room temperature for 15 min, then in TBST (0.15 M NaCl, 2.7 mM KCl, 25 mM Tris, 0.05% Tween 20, pH 7.6) for two 5-min washes. Hybridization signal development was achieved using Liquid Permanent Red s with 4.5% 5 mM levamisole. r Slides were counterstained with Gill's modified hematoxylin, air dried, and coverslipped according to the manufacturer's directions. s
The Mycoplasma gallisepticum riboprobe t was used as a nonhomologous riboprobe control in subsequent hybridization experiments testing P. kauderni tissue sections. Tissue sections of skin from a fish with lesions characteristic of Lymphocystis iridovirus (LCIV) infection, obtained from the diagnostic specimen archive of the Connecticut Veterinary Medical Diagnostic Laboratory (University of Connecticut, Storrs, CT), were included to assess the specificity of riboprobe hybridization.
Results
Ethological and macroscopic observations
Specimens acquired by the NJAAS exhibited consistent clinical signs of lethargy, darkening in body pigmentation, and inappetence. Shortly after the occurrence of these signs, animals became listless, had an increase ventilation rate, and continued to darken. Affected specimens produced an unusual white fecal cast and then died within 2 days.
The group acquired in 2003 began to show signs of the infection immediately after arrival. Within 2 days, signs were evident in 3 specimens and 1 died. Other specimens from the same group behaved normally for a few days before showing signs of infection, but mortalities occurred daily. By day 12, there was 100% mortality of the animals acquired in 2003.
The 20 specimens purchased in 2004 followed a similar pattern, but a few individuals did not show similar clinical signs for up to 4 months, and 1 pair successfully hatched a clutch of eggs and began mouth brooding fry after 3 months of cohabitation with infected fish. By 4 months, there was 100% mortality of the 2004 cohort and newly hatched offspring.
Necropsies were performed on 5 and 4 specimens from the 2003 and 2004 groups, respectively. Freshly dead specimens had extensive hemorrhage on renal and ovarian tissue. The spleens were swollen and had serosal hemorrhage, and gall bladders were very enlarged and filled with clear green bile. In a few specimens, the liver was friable.
Histopathology
Microscopic examination of gill sections revealed necrotic cells, pyknotic debris, fibrin, and protein within vascular cores of many lamellae, indicative of necrosis of branchial microvasculature. Along the margins of vascular spaces of filaments and lamellae were numerous enlarged cells that contained granular basophilic intracytoplasmic inclusions together with marginated nuclear or karyorrhectic material (Fig. 1). Cytomegalic cells containing basophilic granular inclusions were also observed in the lamina propria and along the serosa of the stomach and intestine, in the mesentery, liver, cranial and caudal kidney, spleen, hypodermis, brain, capillary network of the choroid, gas gland of the swim bladder, skeletal muscle, and ovary, where these cells were often located adjacent to or along the margins of blood vessels. Cytomegalic change and granular basophilic inclusions were not observed in epithelial cells, such as those of the epidermis, intestinal mucosa, or branchial lamellar epithelium. Cytomegaly of cells that contained basophilic granular inclusions was the only histopathologic finding in most of the viscera and coelomic tissues. In some specimens, however, foci of necrosis were observed in splenic parenchyma and renal hematopoietic interstitium, wherein cytomegalic cells were located amidst pyknotic and cellular debris, often situated subjacent to the endothelium of splenic sinuses or renal sinusoids (Fig. 2). Necrotic cells and pyknotic debris were present in glomeruli of several specimens together with cytomegalic cells containing basophilic granular inclusions (Fig. 3).
Transmission electron microscopy
Transmission electron microscopy focused on cytomegalic cells corresponding to those identified on light microscopic examination of histologic sections. Cytomegalic cells protruded into the lumens of blood spaces, and endothelial cells extended over the cytoplasmic margins of infected cells, indicating infected cells were located subendothelially (Fig. 4). Cytoplasm of affected cells was markedly expanded and contained numerous hexagonal particles consistent with an icosahedral virion. Cytoplasmic and nuclear contents were condensed and marginated to the cell periphery (Fig. 5A, B). Virus particles were nonenveloped, hexagonal, with a central electrondense nucleic acid core surrounded by a pale zone that, in turn, was marginated by an outer, moderately electron-dense nucleocapsid layer of uniform width (Fig. 6). The average width of virus particles in embedded material was 126 nm with a range of 122 to 130 nm. Necrosis of individual cells was apparent in multiple sections.
Bacteriologic testing and virus isolation
Aerobic bacterial cultures of tissue samples of cranial kidney, spleen, liver, or heart from specimens of the 2004 cohort yielded either no growth of bacteria or in one instance mixed bacterial growth, which included Micrococcus sp., coagulase-negative Staphylococcus sp., Alcaligenes sp., and Acinetobacter-like species. Attempts at virus cultures performed on EPC, FHM, BF-2, and CHSE-214 cell cultures failed to isolate virus.
Polymerase chain reaction amplification, BLASTP, molecular dataset, and phylogenetic analysis
The degenerate DNA polymerase primers generated a 629-bp PCR product (including primers) of the BCIV DNA dependent DNA polymerase (type-B family; 100% amino acid similarity to ISKNV ORF 19R). 14 The ATPase-F2 and ATPase-R2 primer pair yielded a 987-bp PCR product (including primers) containing the full length BCIV ATPase gene (100% amino acid similarity to ISKNV ORF 122R). 14 The P3-IRB5 and P4-IRB5 primer pair yielded a 701-bp PCR product encoding the partial sequence of a putative protein of unknown function (95% amino acid similarity to ISKNV ORF 123R) 14 and the partial sequence of a putative ankyrin repeat protein (97% amino acid similarity to ISKNV ORF 124L). 14 Assembly of the ATPase-F2/ATPase-R2 and P3-IRB5/P4-IRB5 amplicons generated 1,643 bp of continuous sequence (including primers) for BCIV containing the homologous sequence to ISKNV ORFs 122R and 123R and partial sequence for ORF 124L. The MCP primers (Table 1) generated a 412-bp PCR fragment of the BCIV major capsid protein (100% amino acid similarity to ISKNV ORF 6L). 14
Overall, general BLASTP searches for the BCIV DNA polymerase, ATPase, and MCP sequences revealed strongest similarity with iridoviruses belonging to the genus Megalocytivirus, lower similarity to viruses belonging to the genera Lymphocystivirus and Ranavirus, and even lower similarity to members of the genera Chloriridovirus and Iridovirus (data not shown). BLASTP searches revealed 14 additional iridoviruses each for which the 3 homologous sequences could be obtained.
Preliminary phylogenetic analysis revealed that there was no significant incongruence among individual gene trees (defined by the presence of incompatible bipartitions that received a posterior probability of > 90%, respectively) allowing concatenation of the sequence for the 3 genes into 1 final dataset. This dataset contained 676 amino acid characters (including gaps) for 15 viral taxa with at least 1 representative of each of the 5 genera within the family Iridoviridae (Table 2).
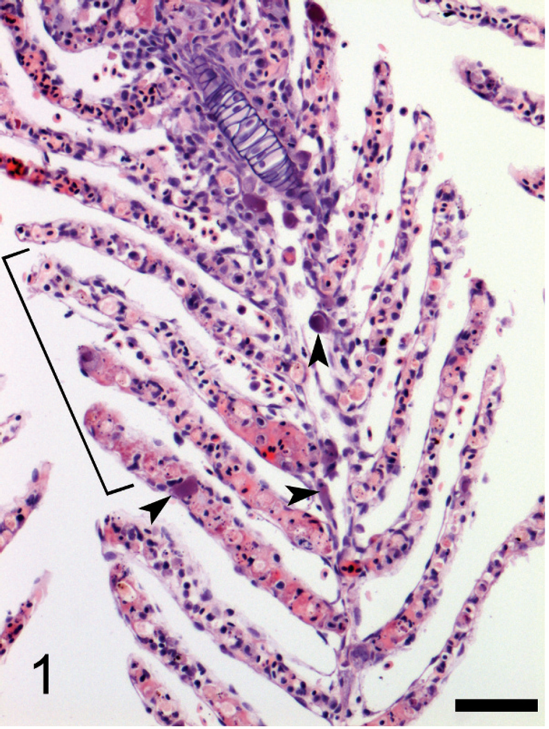
Photomicrograph of gill from a Banggai cardinalfish (Pterapogon kauderni) naturally infected with iridovirus. There is necrosis of the microvasculature of several lamellae with fibrin, protein, and pyknotic debris within vascular cores (bracket). Cytomegalic cells with basophilic cytoplasmic inclusions are present along the margin of the central venous sinus of the filament and within some lamellae (arrowheads). Hematoxylin and eosin. Bar = 50 μm.

Photomicrograph of kidney from a Banggai cardinalfish (Pterapogon kauderni) naturally infected with iridovirus. Cytomegalic cells with basophilic inclusions (arrows) are present together with pyknotic and cellular debris along the margins of sinusoids and in the hematopoietic interstitium. Hematoxylin and eosin. Bar = 25 μm.

Photomicrograph of a glomerulus in the kidney of a Banggai cardinalfish (Pterapogon kauderni) naturally infected with iridovirus. There is a loss of cellular detail and scattered pyknotic debris in the mesangium together with granular basophilic inclusions (arrows) along the periphery of the glomerular tuft. Hematoxylin and eosin. Bar = 25 μm.

Transmission electron photomicrograph of an iridovirus-infected cell located subjacent to the endothelium along a segment of a sinus in the spleen. The cytoplasm of an endothelial cell (en) extends along the cytoplasmic margin (arrows) of a cell wherein a cluster of virus particles (*) is present in the cytoplasm. In the lumen of the sinus are red blood cells (rbc). The nucleus (nu) of the endothelial cell is evident. Lead citrate and uranyl acetate. Bar = 1 μm.
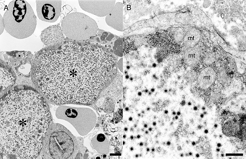
Transmission electron photomicrographs of iridovirus-infected cells protruding into a sinus of the spleen.
The concatenated 3-gene Bayesian analysis demonstrated with a high level of confidence that the BCIV is an iridovirus belonging to the genus Megalocytivirus (Fig. 7). Infectious spleen and kidney necrosis virus was found to be the sister species to BCIV, but this result was not strongly supported. All the systemic iridoviruses included in the present study belonging to the genus Megalocytivirus were found to be monophyletic. In addition, the lymphocystivirus and ranavirus isolates used also represented monophyletic genera and sister clades, and together formed a sister group to the viruses in genus Megalocytivirus. Finally, the 3 genera Lymphocystivirus, Megalocytivirus, and Ranavirus formed a well-supported monophyletic group.
In situ hybridization
In situ hybridization experiments using the antisense DIG-labeled riboprobe of the 701-bp P3-IRB5/P4-IRB5 amplicon resulted in specific labeling of cytoplasmic inclusions in cytomegalic cells. No hybridization signal was detected in comparable tissue sections to which a nonhomologous riboprobe was applied. Tissue sections from at least 1 specimen representing the 2004 samplings and 2005 submissions demonstrated hybridization signals. No hybridization signal was detected in tissue sections of fish skin with lesions characteristic of LCIV infection (Fig. 8).
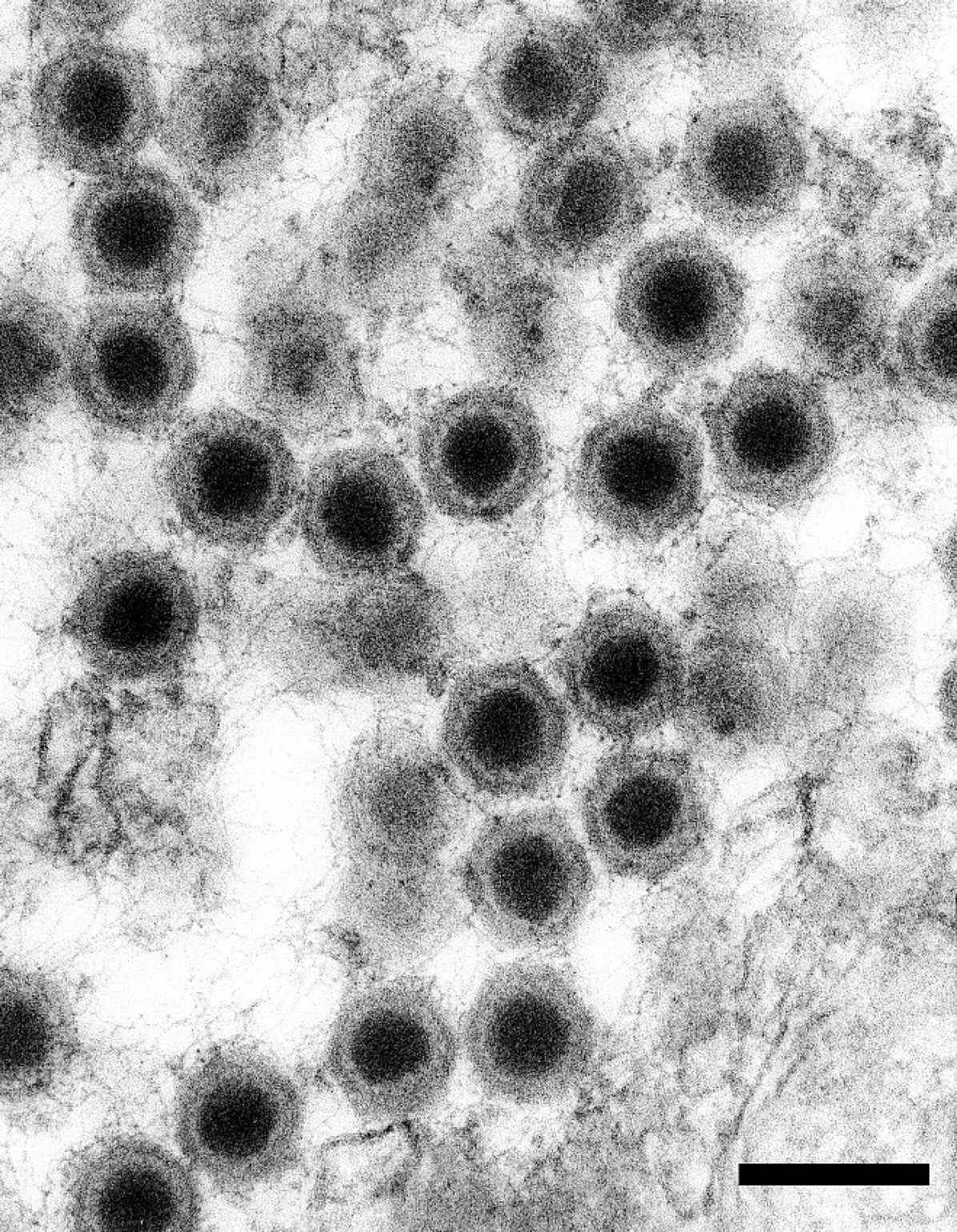
Viral particles are hexagonal and nonenveloped with an electron-dense nucleic acid core surrounded by a pale zone and an outer nucleocapsid layer that is moderately electron-dense and of uniform width. Lead citrate and uranyl acetate. Bar = 200 nm.
Discussion
Banggai cardinalfish iridovirus joins a growing list of megalocytiviruses identified from fish that are associated with epidemic mortality episodes in their respective marine and freshwater hosts. 48 The disease associated with BCIV infection in P. kauderni with the presence and systemic distribution of enlarged virus-infected cells resembles that described for ISKNV, the type species for the genus Megalocytivirus. Phylogenetic analyses using sequences from a portion of the DNA polymerase, and MCP and ATPase genes of BCIV, demonstrate a close relationship with ISKNV. Banggai cardinalfish iridovirus was found to be the sister group to ISKNV, related but distinct from the 4 other megalocytiviruses from red sea bream, rock bream, large yellow croaker, and orange spotted grouper (RSIV, RBIV, LYCIV, and OSGIV, respectively). The disease associated with BCIV shares several clinical characteristics in common with diseases reported in other fish species infected with megalocytiviruses. Pterapogon kauderni develops clinical signs such as darkening, lethargy, and inappetence, similar to those reported in other infected species in both freshwater 20,43,44,50 and marine 25,59 environments.
Histologically, inclusions identified in P. kauderni examined from all 3 submissions from 2003–2005 resembled those of previously published studies describing the presence of systemic iridoviral infections in tissue. 19 Characteristic cytomegalic cells containing basophilic granular cytoplasmic inclusions were identified in large numbers in multiple organs in these cardinalfish. Cytoplasmic inclusions shared features with those noted in previous studies, and the predominant pathologic change in some tissues was the presence of cytomegalic cells only. 19 Necrosis of splenic and renal parenchyma was present in some P. kauderni specimens, and has been reported as a feature of histopathologic findings in other systemic iridovirus infections, as were necrotic changes in glomeruli. 19 In P. kauderni specimens, necrosis together with exudation of serum protein and fibrin was present in branchial microvasculature, which could have contributed to the morbidity and mortality observed with infection. It is important to note that other studies describing the pathology of systemic iridoviral disease in fish examined large numbers of fish by histology in most instances. 19
Ultrastructural studies performed on specimens from the 2004 cohort revealed infected cells in subendothelial locations and that those cells had cytoplasmic inclusions, which in turn contained virions that were hexagonal in shape with an average diameter in embedded material of 126 nm. The intracellular location of virus together with the symmetry and size of virions are consistent with reports of systemic iridoviruses causing disease in 3 tropical marine and freshwater ornamental fish. 3,19,30,43,44,46,47 Positive hybridization signals were obtained from iridoviral inclusions in tissues of 2004 and 2005 P. kauderni specimens using the riboprobe generated from the P3-IRB5/P4-IRB5 PCR product in ISH experiments, which provides correlation indicating that the virus inclusions in histologic sections and the virus particles identified by electron microscopy likely do represent the iridovirus from which the genetic sequence data was obtained.
Iridovirus samples and sequences used in the current study.*

Pol. = polymerase.
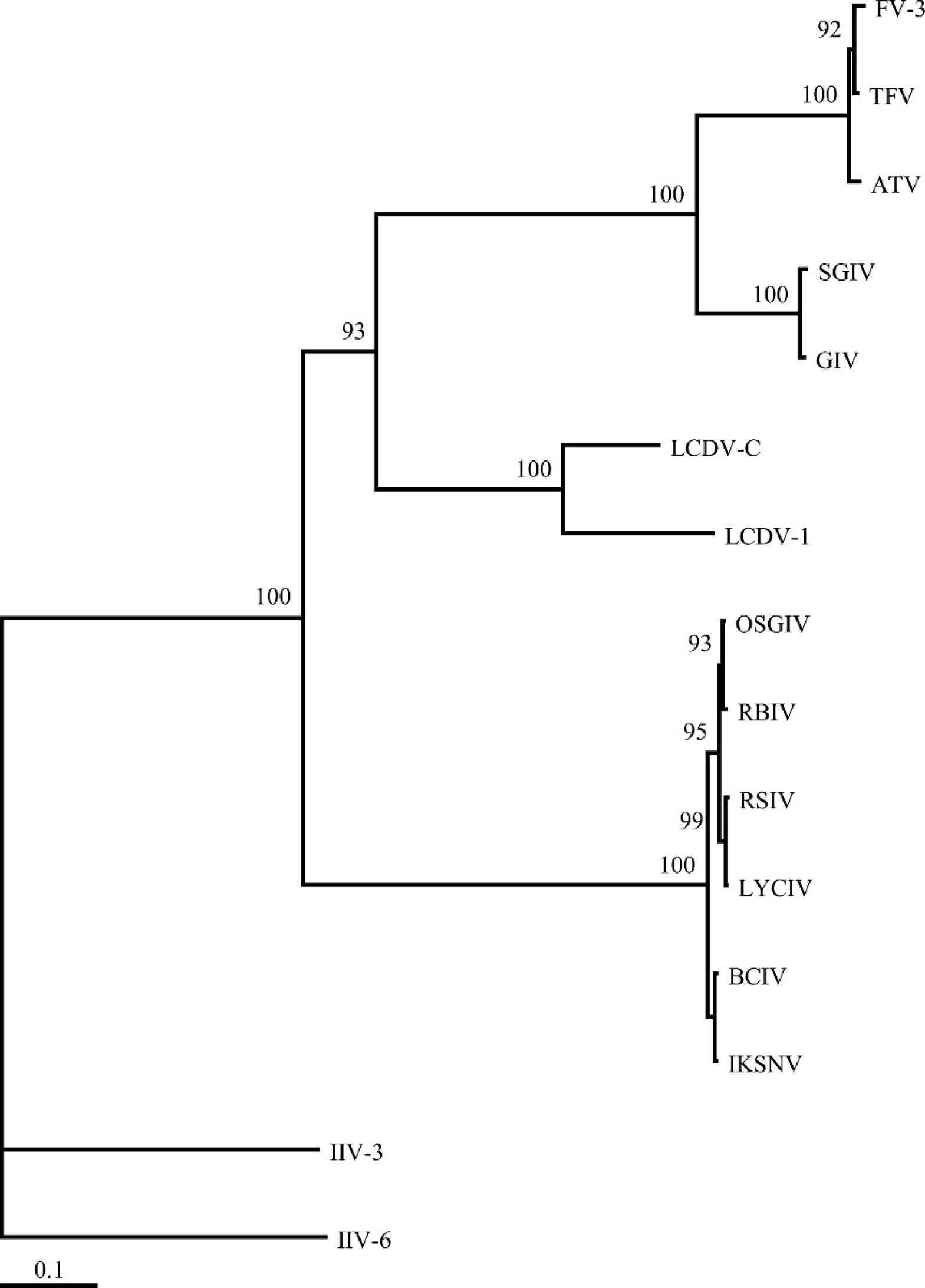
Phylogram generated from the concatenated amino acid sequence of the partial DNA polymerase and major capsid protein and full-length ATPase genes. The tree depicts the relationship of the Banggai cardinalfish iridovirus (BCIV) to representatives from each of the 5 genera of iridoviruses. The numbers at each node represent posterior probabilities (values >90 shown) of the Bayesian analysis.
Positive hybridization signals from tissues of 2004 and 2005 specimens suggest that the same or a closely related megalocytivirus was responsible for infections of multiple cohorts of P. kauderni and that cohorts sampled over different years did not have distinctly different iridovirus infections.
An analysis of the full-length ATPase and partial DNA polymerase and MCP genes for BCIV clearly demonstrated affinities with other described megalocytiviruses (Fig. 7). Megalocytiviruses examined to date share considerable nucleic acid and amino acid identity (>93% and >97%, respectively). 8 More recent studies of megalocytiviruses detected by PCR present in numerous cultured and wild fish species in the South China Sea support a strong homogeneity in the group with evidence for 2 clusters, the most well represented being identical to ISKNV and the second more closely related to the orange-spotted grouper (OSGIV) agent. 59 The phylogenetic analysis in the current study suggests BCIV is more closely related to ISKNV than to OSGIV (Fig. 7). A researcher, studying the genetic variation among 48 Asian megalocytivirus isolates, erected 3 genogroups with RSIV, RBIV, and OSGIV within the first, ISKNV within the second, and the third group infecting mainly Asian flatfish species. 48
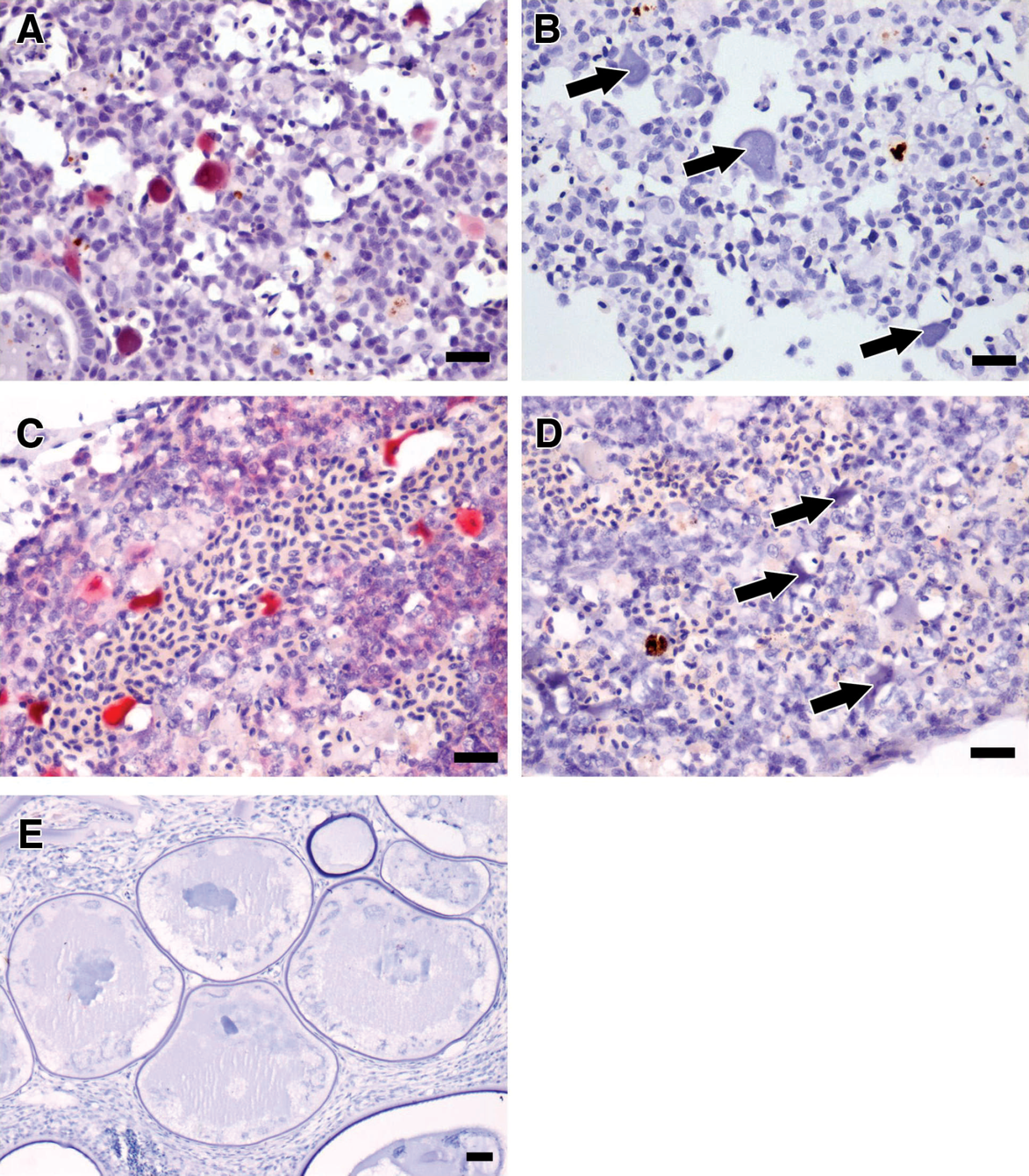
Results of in situ hybridization experiments using the antisense digoxigenin-labeled riboprobe of the cloned 701-bp P3-IRB5/P4-IRB5 amplicon performed on sections of kidney from specimens from 2004 and 2005.
The repeated presence of a megalocytivirus among groups of wild-caught P. kauderni sold in the United States through the international ornamental fish trade raises the question of whether BCIV occurs among wild populations or is the product of postharvesting transmissions from other species at an export or import center. The former would reflect the combined stresses experienced from the point of capture and the enzootic presence of the virus in native populations, such that systemic infection may manifest as a consequence of collection, crowded containment, and long-distance transport, which are know to occur in Indonesia (Schmidt C, Kunzmann A: 2005, Post-harvest mortality in the marine aquarium trade: a case study of an Indonesian export facility. SPC Live Reef Fish Info Bull 13:3–12. Available at: http://www.spc.int/coastfish/News/LRF/13/. Accessed on September 26, 2008). This would indicate a range extension for the ISKNV megalocytiviruses to the Banggai Archipelago. A 2007 study 59 reported ISKNV-like virus affecting 39 wild marine teleosts representing 20 families in 6 orders, with 12 families of perciformes. In addition, wild caught Lophiomus setigerus, Eopsetta grigorjewi, and Sphyraena pinguis carrying a (less related “genotype I”) megalocytivirus has been detected. 48 However, all these occurrences were restricted to the East and South China Sea, above latitude ∼N18′, and no wild marine species have been reported with megalocytivirus from Southeast Asia, including the Philippines and Indonesia. Moreover, during four 1-month expeditions to the Banggai Archipelago (in 2001, 2002, 2004, and 2007) to conduct ecologic and conservation status surveys of P. kauderni, specimens showing signs of the viral infection were never identified. This fieldwork included samplings of all islands inhabited by P. kauderni to distinguish populations for various ecologic and genetic studies and also included detailed observations of dozens of holding nets containing captured specimens held and observed over several weeks. In addition, no reports from local collectors or buyers indicated disease outbreaks during these times, even though fishermen, after collecting P. kauderni, commonly keep hundreds or even thousands of specimens in nets from several days to a few weeks.
These ecologic and historic data suggest that infection is occurring after capture and transport from the Banggai region. Since the first population and trade surveys were conducted in 2001, the harvest of P. kauderni (currently a minimum of 100,000 specimens/month) is shipped from the Banggai Archipelago to main export centers in Bali and Manado, North Sulawesi. From Manado, most P. kauderni are exported via Bali or Singapore (Vagelli, personal communication; Lunn KE, Moreau MA: 2002, Conservation of Banggai cardinalfish populations in Sulawesi, Indonesia). 58 Spreading of disease through the international ornamental fish trade is well known, 5,20,62 as previously noted with iridovirus infections traced to several species of fishes imported from Singapore. 2,3,50 There is a high probability that shipments of P. kauderni could have been infected at some export (or import) centers. Virus transmission could have occurred by keeping P. kauderni specimens in holding systems with other (infected) species, or by exposing them to water carrying the virus, as megalocytivirus have been shown to be easily transmitted through the water between individuals of the same species 26 or between different species sharing holding systems. 21 In addition, all 3 cohorts began showing clinical signs postcollection in only 2–3 days after shipment receipt at their final destinations, which suggests that these animals were infected prior to arrival at their respective facilities. Laboratory experiments have shown that clinical signs of virus infection and mortality began about 7–10 days after transmission, in either inoculation or bath exposure trials. 20,25,26,60
Pterapogon kauderni represents 1 of about 1,400 species of marine fishes that actively move as part of a large worldwide trade in ornamental fishes, which is a trade that is poorly regulated or unregulated with regard to consistent aquatic animal health standards. 62 The unsustainable harvest of P. kauderni led to its inclusion in 2007 in the IUCN Red List in the “Endangered” category. Pterapogon kauderni is especially vulnerable to indiscriminate collecting due to its highly restricted geographic distribution, low productivity, and ease of capture. Conservation concerns over the exploitation of this coral reef species are further exacerbated by the identification of a significant viral pathogen originating from either the point of capture or, more likely, from one of its export centers. Continued exploitation of this species heightens not only conservation concerns but also the potential spread of serious viral pathogens in the international ornamental fish trade. 5,43,62
Acknowledgements
The authors wish to thank Stephen Daniels and the Electron Microscopy Laboratory, University of Connecticut for assistance with transmission electron microscopy, Ione Jackman and Denise Woodward for histotechnological services, Daniel Buttrey for assistance with image processing, Dr. Hugh W. Ferguson for comparative histopathologic insights, and the Center of Excellence for Vaccine Research, University of Connecticut, for the donation of in situ hybridization tissues and Mycoplasma gallisepticum riboprobe.
Footnotes
a.
BEEM®, Electron Microscopy Sciences, Fort Washington, PA.
b.
PELCO® 3440 microwave, PELCO ColdSpot®, PELCO Prep-Eze™ specimen holders; Ted Pella Inc., Redding, CA.
c.
Medium grade LR White resin, London Resin Company Ltd., Reading, Berkshire, United Kingdom.
d.
Diatome AG, Biel, Switzerland.
e.
Phillips EM 300 electron microscope, Pye Unicam, Cambridge, UK.
f.
Qiagen DNeasy® Blood and Tissue Kit, QIAprep® Spin Miniprep Kit, Qiagen Gel Extraction Kit, Qiagen Maxi Kit; Qiagen Inc., Valencia, CA.
g.
Invitrogen Corp, Carlsbad, CA.
h.
ABI 377, Applied Biosystems, Foster City, CA.
i.
Clontech Laboratories Inc., Mountain View, CA.
j.
MWG Biotech, Huntsville, AL.
k.
Fisher Scientific, Pittsburgh, PA.
l.
Zymoclean™, Zymo Research Corp., Orange, CA.
m.
VersaFluor™ fluorometer, Bio-Rad Inc., Hercules, CA.
n.
Gene Codes Corp., Ann Arbor, MI.
o.
DNAMAN, Lynnon Corp., Vaudreuil-Dorion, Quebec, Canada.
p.
Promega Corp., Madison, WI.
q.
T7 DIG RNA labeling kit, Roche Diagnostics Corp., Indianapolis, IN.
r.
Sigma-Aldrich, St. Louis, MO.
s.
Dako North America Inc., Carpinteria, CA.
t.
M. gallisepticum riboprobe, Center of Excellence for Vaccine Research, University of Connecticut, Storrs, CT.
