Abstract
A 13-year-old female llama was presented to the referring veterinarian for swelling and firmness of the right rear mammary gland, for a duration of 2 months, which had been unresponsive to antibiotics. A formalin-fixed wedge biopsy specimen from the affected quarter was submitted to Purdue University Animal Disease Diagnostic Laboratory for histopathology. Histopathologic examination revealed tubulopapillary acinar or solid nest-like clusters of neoplastic epithelial cells surrounded by whorls and sheets of proliferative myoepithelial cells. Histologic criteria for malignancy observed in neoplastic epithelial cells included marked cellular and nuclear atypia, high mitotic index, and numerous bizarre mitoses. The presence of osseous metaplasia in the proliferative mesenchymal component justified classification as a mixed tumor. Positive immunohistochemical staining of neoplastic epithelial cells with anticytokeratin antibody, and proliferative spindloid cells with antiviemtin and antismooth muscle actin antibodies supported the histopathologic diagnosis. The llama was in good health after about 1 year of initial presentation, and metastasis to regional lymph nodes was not reported. Mammary neoplasia is rare in camelids. To the authors' knowledge, this is the first report of a carcinoma in a mixed mammary tumor in a llama.
Neoplastic diseases are uncommon in camelids, including llamas (Lama glama). With the increasing popularity of New World camelids for commercial and recreational purposes, there has been an increase in reports of various neoplastic conditions, including mammary adenocarcino- ma 3,11,13 in these animals. The incidence of neoplasia in llama and alpaca has been reviewed. 2,10,12 In a recent survey of camelid biopsy and necropsy submissions during a 5-year period, the authors report that cutaneous and mucocutaneous fibroma and fibropapilloma were most common, followed by cutaneous and mucocutaneous squamous-cell carcinoma and disseminated lymphoma. 12 Although mixed mammary tumors are common in dogs 6 and have been reported in humans, 8 these tumors have been rarely reported in other domestic animals. To the authors' knowledge, this is the first report describing the histologic and immunohistochemical features of a carcinoma in a mixed mammary tumor in a 13-year-old llama.
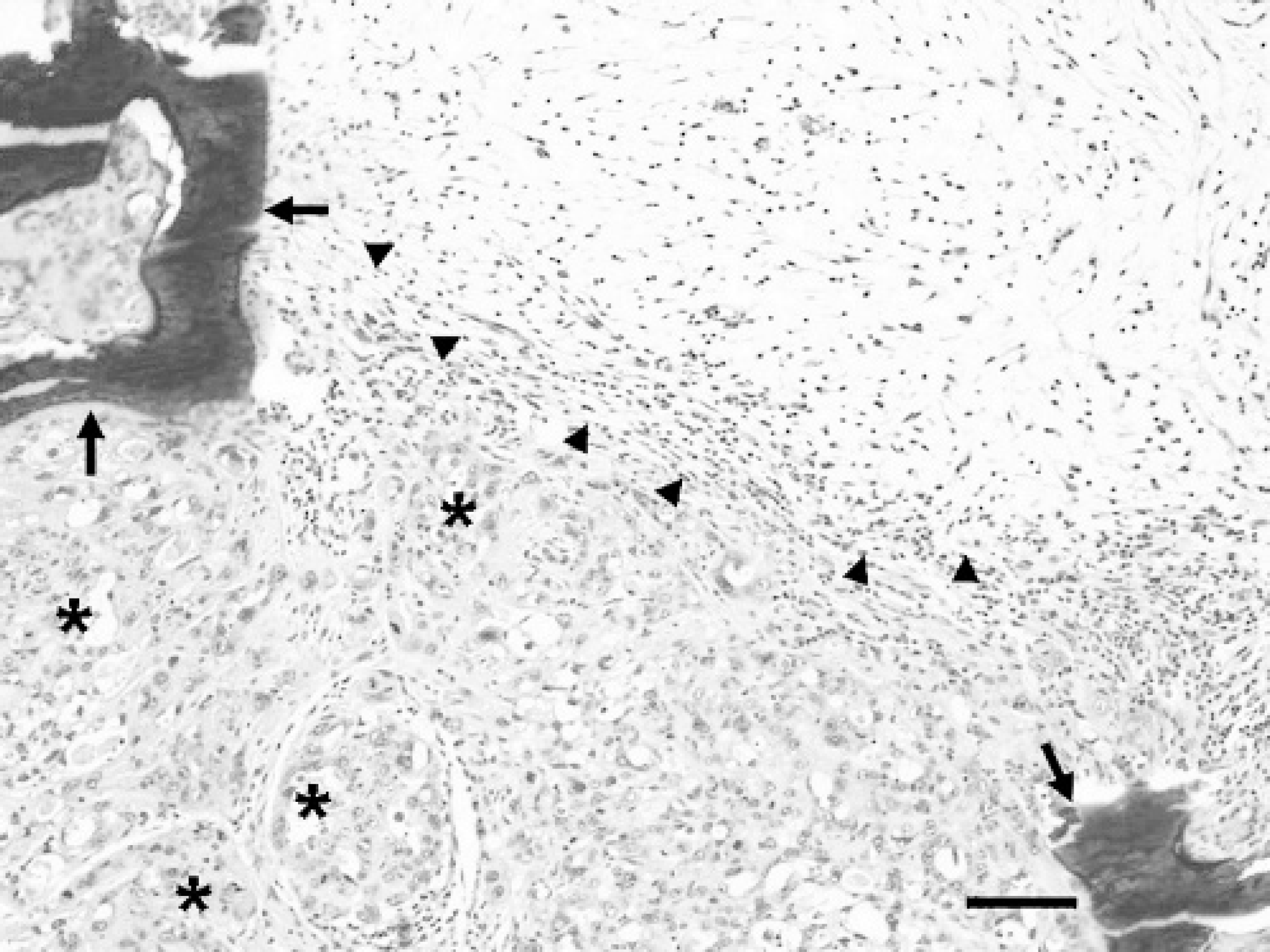
Mammary mass; llama. Mammary mass consists of atypical tubulopapillary acinar structures (asterisks) interspersed with abundant spindloid cell proliferation (outlined by arrowheads). Also note the areas of osseous metaplasia (arrows) within the neoplasm. HE. Bar = 100 μm.
An alert and otherwise healthy, intact female llama was presented with swelling and firmness of the right rear quarter of the udder for a 2-month duration that was unresponsive to antibiotic therapy. Physical examination revealed a distinct, firm mass that contained multiple foci with a bone-like consistency. A 3 × 1.5 × 1-cm wedge biopsy specimen from the mass was submitted to Purdue University Animal Disease Diagnostic Laboratory for histopathologic evaluation.
Microscopic examination revealed that the parenchyma of the mammary gland was obscured and obliterated by a multinodular, unencapsulated neoplastic proliferation of moderately atypical epithelium that formed single to multiple cell layers in tubulopapillary acinar or solid nestlike patterns, separated by a moderate amount of pale basophilic mesenchymal ground substance (Figs. 1 and Figs 2). Interspersed around the neoplastic epithelial structures were foci of spindloid cell proliferation, which were frequently mineralized, with occasional osseous metaplasia. Neoplastic epithelial cells were cuboidal to low columnar, had distinct cell borders, and contained moderate pale eosinophilic cytoplasm and large round to oval, vesicular nuclei, with 1 to 3 prominent nucleoli (Fig. 2A, Fig. 2B). Nuclear polarity was frequently lost. Mitotic figures were abundant and bizarre, and ranged from 4 to 15 per high power field. There was moderate-to-marked cellular and nuclear atypia. Scattered foci of necrosis, hemorrhage, and infiltration by hemosiderin-laden macrophages were frequently noted within the neoplasm. Proliferative spindloid cells formed whorls or haphazard sheets and had indistinct cell boundaries, moderate pale eosinophilic cytoplasm, and an oval-to-elongate nucleus, with open, vesicular chromatin, and usually a distinct nucleolus. The epidermis overlying the neoplastic mammary tissue was ulcerated. There was multifocal periductal infiltration of lymphocytes and fewer golden-brown pigment-laden macrophages. Multifocally, in apparently non-neoplastic mammary tissue, acini, and a few ducts were distended with amorphous pink secretory material. Other ducts were moderately distended, lined with attenuated epithelial cells, and contained foamy macrophages in the lumens.
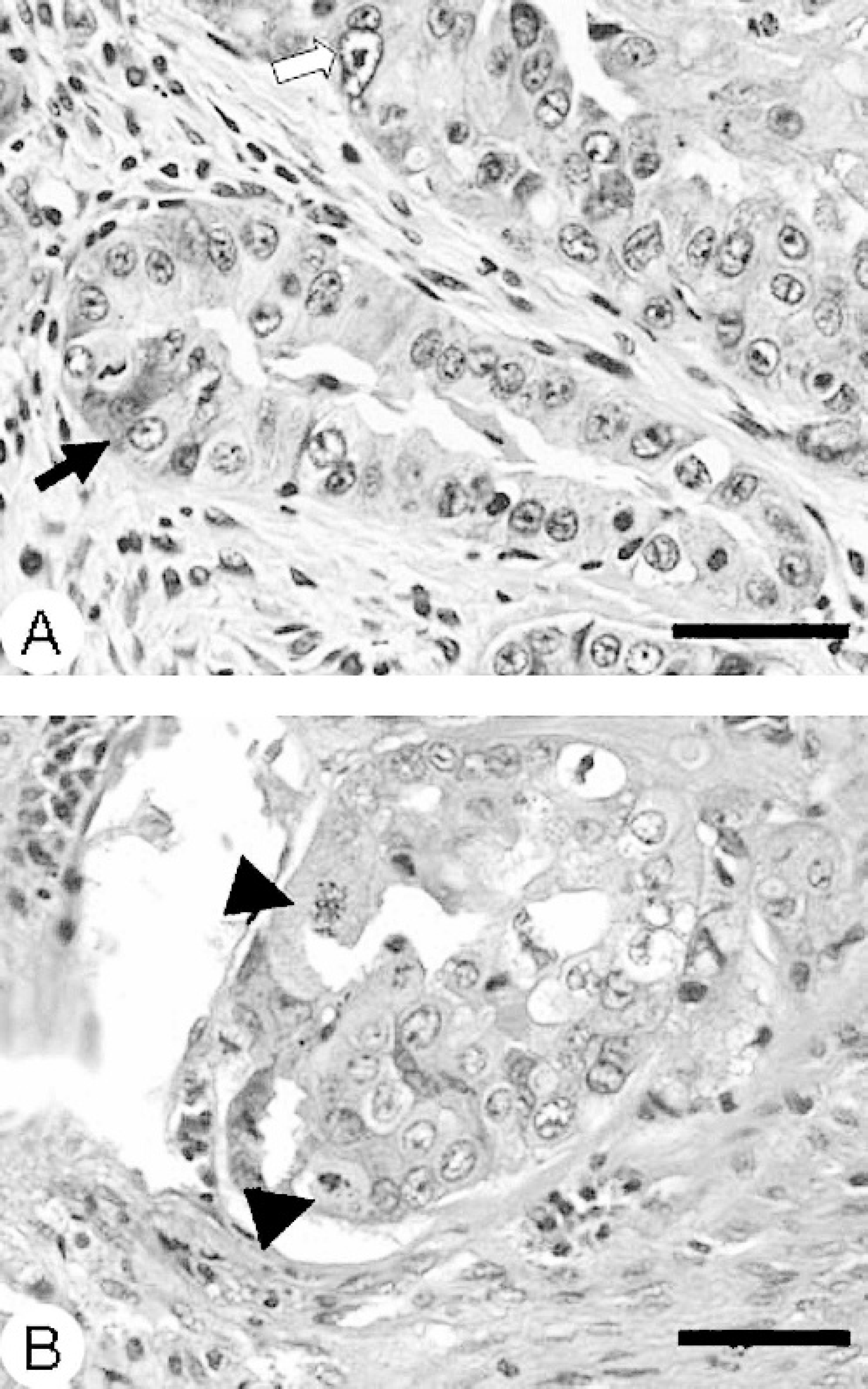
Mammary mass; llama.
By immunohistochemistry (IHC), the cytoplasm of the cuboidal-to-columnar neoplastic cells stained diffusely positive for cytokeratin, indicating epithelial cell origin (Fig. 3). Immunohistochemical staining for the cytoplasm of the proliferating spindloid cells was diffusely positive for vimentin and smooth muscle actin, indicating myoepithelial origin (Fig. 3).
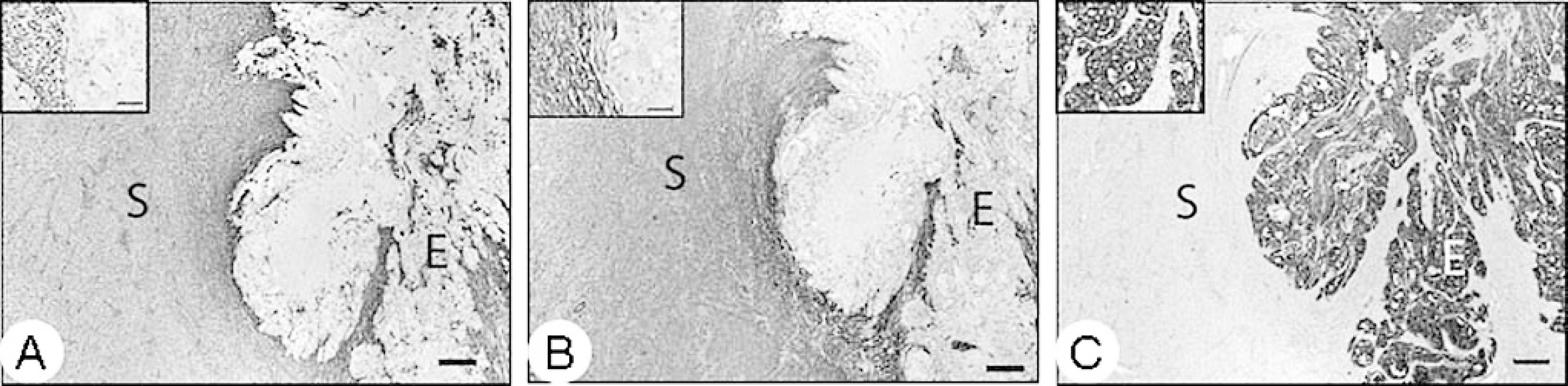
Mammary mass; llama. Immunohistochemical staining of the mammary mass with
Based on histologic findings, a diagnosis of carcinoma in a mixed mammary tumor was made in this llama. The mammary tumor was diagnosed as a “mixed tumor,” based on the presence of proliferative epithelial and mesenchymal cells with osseous metaplasia. It was histologically interpreted as “malignant,” based on the lack of a capsule and multifocal necrosis, as well as the marked cellular and nuclear atypia, a high mitotic index, and numerous bizarre mitotic figures in neoplastic epithelial cells. The nomenclature of mixed mammary tumors with features of malignancy is fraught with confusion. According to the classification scheme of canine and feline mammary tumors by the World Health Organization, mixed mammary tumors are usually benign and are composed of benign neoplastic epithelial cells and proliferating mesenchymal cells that have produced cartilage and/or bone. 7 However, mixed tumors may contain “malignant-appearing” cells and are termed “sarcoma” or “carcinoma in a mixed tumor,” depending on the malignant component. Likewise, according to a more recent classification scheme for canine mammary neoplasms, 1 malignancies involving mixed tumors are designated as either carcinomas or sarcomas in a mixed tumor.
The presence of histologic criteria in mammary biopsy samples consistent with a diagnosis of a malignant mixed mammary tumor in dogs does not invariably imply a clinically malignant course. 6 In this case, although histologic criteria of malignancy are present, the llama was in excellent body condition, without evidence of metastasis of the mammary mass at the time of preparation of this manuscript, more than a year after the biopsy specimen was examined. The referring veterinarian reported no additional surgery of the affected mammary quarter. Regional lymph nodes were not examined histologically, and there was no gross or clinical evidence of regional or distant metastases. It is possible that mammary mixed tumors in llamas behave similarly to canine mammary tumors, with slow progression and local invasion.
Increased expression of steroid hormone (estrogen and progesterone) receptors has been found to provide reliable prognostic information in human breast cancer, as well as canine mammary tumors. 4,5 Human breast cancers, as well as canine breast tumors expressing steroid hormone receptors are most likely to be responsive to antihormone therapy and have been associated with better prognosis. 5,9 An attempt was made to determine the expression of estrogen receptor alpha (ERα) and progesterone receptor (PR) in this mammary tumor. Because antibodies specific for llama ERα and PR were not available, anti-ERα antibody a and anti-PR antibody b that are used in the authors' laboratory for routine IHC were used. Formalin-fixed, paraffin-embedded tissue was processed for IHC by using the EnVision system. c Unfortunately, the anti-ERα antibody exhibited nonspecific staining and could not be reliably interpreted. Staining with anti-PR antibody, however, was specific, as indicated by intense nuclear staining of normal mammary epithelial cells (Fig. 4A). A majority of neoplastic epithelial cells and a few myoepithelial cells in the tumor stained with intense nuclear staining with this antibody (Fig. 4B).
One previous report 3 of mammary adenocarcinoma in llama described moderate mixed positive nuclear and cytoplasmic staining for ER in approximately 30% of the neoplastic cells. Staining for PR was not attempted in that case. In another report of llama mammary adenocarcinoma, 13 ER expression was negative in the tumor but positive (nuclear staining) in the normal mammary tissue. Reportedly, the PR antibody failed to stain normal llama mammary tissue. PR staining was specific in both the normal mammary tissue and the tumor in the present case, and its relevance to the clinical course of such tumors in llamas is unknown. Sources of antibodies used for ER and PR IHC differ between our case and other reports, likely explaining difference in reactivities.
Mammary neoplasia is rare in llamas and other South American camelids. There are 3 reported cases of mammary adenocarcinoma, including one that metastasized to the skin. 3,11,13 These mammary tumors in llamas were found to invade locally or to metastasize to regional lymph nodes or the skin. To the authors' knowledge, this is the first reported case of a carcinoma in a mixed mammary tumor in a llama. Although not very common, mammary adenocarcinoma and carcinoma in a mixed mammary tumor should be included as a differential for mammary gland lesions in llamas.
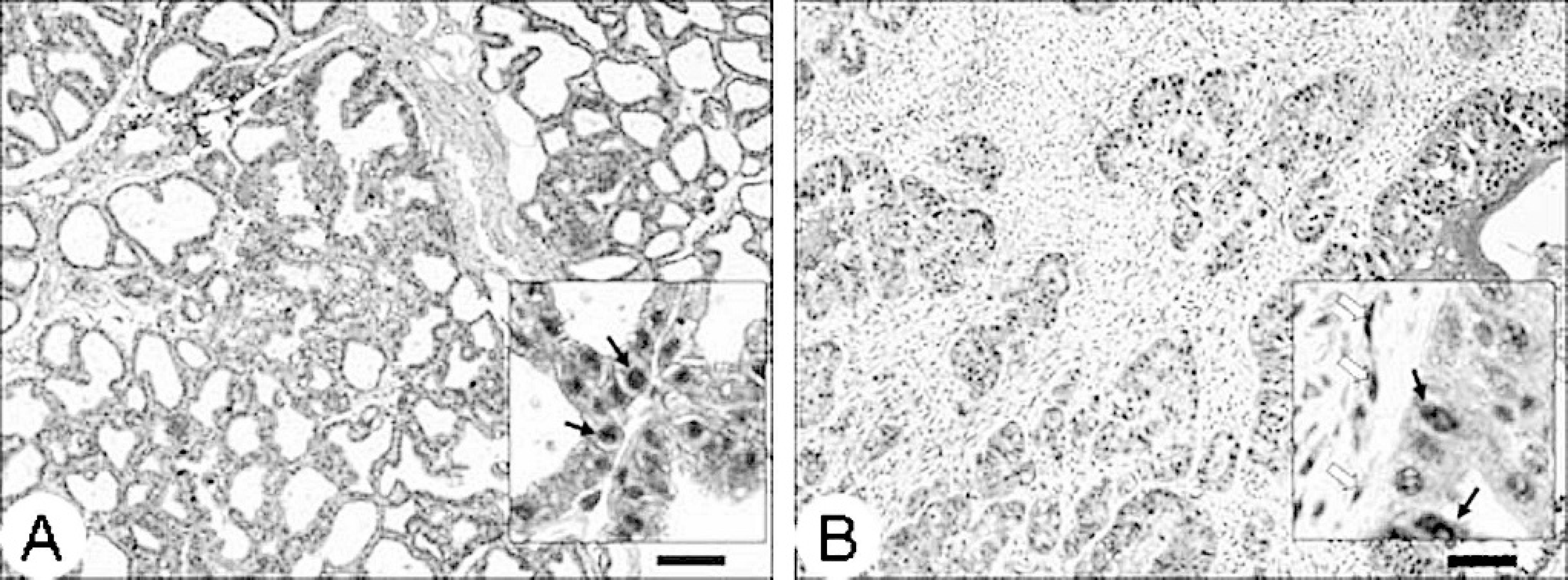
Immunohistochemistry for progesterone receptor (PR) expression in
acknowledgements. The authors wish to thank Dr. Wayne Bayerly for submitting this biopsy specimen and providing the follow-up information on this llama, Dr. Jose Ramos-Vara for his advice with immunohistochemistry, and the staff of the Histopathology Laboratory of Animal Disease Diagnostic Laboratory, Purdue University for technical help.
Footnotes
a.
Novocastra Laboratories Ltd, UK
b.
Lab Vision Corporation, Fremont, CA
c.
Dako North America, Inc., Carpinteria, CA
