Abstract
Camelpox is a contagious viral skin disease that is mostly seen in young camels. The disease is caused by the Camelpox virus (CMLV). In the present study, a polymerase chain reaction (PCR) assay based on the C18L gene (encoding ankyrin repeat protein) and a duplex PCR based on the C18L and DNA polymerase (DNA pol) genes were developed. The former assay yields a specific amplicon of 243 bp of the C18L gene, whereas the duplex PCR yields 243- and 96-bp products of the C18L and DNA pol genes, respectively, in CMLV, and only a 96-bp product of the DNA pol gene in other orthopoxviruses. The limit of detection was as low as 0.4 ng of viral DNA. Both PCR assays were employed successfully for the direct detection and differentiation of CMLV from other orthopoxviruses, capripoxviruses, and parapoxviruses in both cell culture samples and clinical material. Furthermore, a highly sensitive SYBR Green dye–based, realtime PCR was optimized for quantitation of CMLV DNA. In the standard curve of the quantitative assay, the melting temperature of the specific amplicon at 77.6°C with peak measured fluorescence in dissociation plot was observed with an efficiency of 102%. To the authors' knowledge, this is the first report to describe a C18L gene–based PCR for specific diagnosis of camelpox infection.
Details of cell culture–adapted camelpox and other poxviruses used in the current study.*
CMLV = Camelpox virus; BPXV = Buffalopox virus; SPPV = Sheeppox virus; GTPV = Goatpox virus; ORFV = Orf virus; P = passage level.
Camelpox is a highly contagious viral skin disease affecting mostly young camels (2–3 years old). The disease is characterized by papular pustular eruptions on the skin and mucous membranes. Camelpox outbreaks have occurred sporadically among camels in India (first described in Punjab in 1909), Pakistan, Afghanistan, Turkmenistan, 3 the Middle East, north and east Africa, and the United Arab Emirates. 14 The causative agent, Camelpox virus (CMLV; family Poxviridae, subfamily Chordopoxvirinae, genus Orthopoxvirus), 17 is closely related to Variola virus, 1,8 and may also be a zoonotic agent. 6,10,12 The other members of the genus include several pathogens of veterinary and zoonotic importance; namely, Monkeypox virus, Vaccinia virus (VACV), Buffalopox virus (BPXV, a variant of VACV), 17 Cowpox virus (CPXV), Ectromelia virus, Rabbitpox virus (RPXV), Taterapox virus, Raccoonpox virus, Volepox virus, Skunkpox virus, and Uasin Gishu disease virus. 24
Parapoxviruses and papillomaviruses have also been associated with skin infections of camel similar to camelpox lesions. 4,10,16,19,21,25 The clinical diseases caused by these viruses cannot be differentiated from each other based on clinical signs. Routinely, camelpox is diagnosed based on clinical signs, epizootiological and pathological findings, 4 isolation of virus in cell lines (Hela, GMK-AH1, BSC-1, and Vero), electron microscopic detection of virus particles in pox lesions, and genus-specific antigen capture enzyme-linked immunosorbent assay. 5,6,9,13,17 Camelpox could be differentiated from other orthopoxvirus (OPXV) and parapox infections by the use of restriction enzyme analysis (REA) 17 and transmission electron microscopy (typical brick-shaped CMLV can be demonstrated in papules or scabs).
Effective control of any disease warrants a prophylactic as well as a rapid, specific, and sensitive assay for diagnosis and molecular epidemiological studies. The differential diagnosis of the camelpox infection from other pox infections/diseases of camel necessitates confirmation in a laboratory. Among the molecular biological techniques, polymerase chain reaction (PCR) is most employed in the diagnosis of various infectious diseases as an alternative to conventional diagnostic assays. For the detection of OPXV infections in camels, Southern blot hybridization, REA, mapping of viral DNA, and A type inclusion (ATI) gene–specific PCR assays have been used. 18 Although the ATI and the hemagglutinin (HA) gene-based PCRs developed for the diagnosis and differentiation of OPXVs 2,15,18,20 have been effective, these assays may not be suitable for rapid (because the length of PCR products is long), routine, specific diagnosis of camelpox infections and their simultaneous differentiation. A specific diagnostic PCR for the detection of CMLV has not yet been reported. Hence, the present study describes the C18L gene–based single PCR or duplex PCR based on the C18L and DNA polymerase (DNA pol) genes for specific and rapid detection and quantitation of CMLV.
Three Indian CMLV isolates (Table 1) were used to evaluate the PCR assays. Two isolates (CMLV-1 and CMLV-2) were associated with 2 outbreaks in camels aged 2–3 years in a village near Bikaner, Rajasthan, India. The 2 outbreaks occurred at 2 different times (1 month apart) during 1997, leading to high morbidity and productivity losses in camels. The third isolate, CMLV-Hyd, was from a sporadic infection of a 10-year-old camel in an outbreak at Bhutan-ka bass, Bharou, Bikaner, Rajasthan, India, during December 2002 (isolate was adapted to Vero [African green monkey kidney] cells at Indian Immunologicals Pvt. Ltd., Hyderabad, India). All of these viruses were propagated in Vero cells using Eagle minimum essential medium a following standard procedures.
Infected Vero cells in a 25-cm 2 tissue culture flask showing greater than 80–90% cytopathic effect were harvested in 250 μl of phosphate buffered saline (PBS; pH 7.2) and used for extraction of total genomic DNA (gDNA) using a commercial kit b according to the manufacturer's instructions. The propagated vaccine strain CMLV-1 was concentrated using a 30–66% discontinuous sucrose density gradient and further used for DNA extraction using a commercial kit. b The concentrated DNA was used for standardization and quantification of the assay. The purity and quantitation of DNA was assessed by use of restriction fragment length polymorphism and spectrophotometry techniques (data not shown). Vero cells infected with Goatpox virus, Sheeppox virus, BPXV, and Orf virus isolates (Table 1), and clinical samples suspected of pox infections available in the laboratory were also used for assessing the specificity of the assay. Scab material was triturated in PBS, and a 10% (w/v) suspension was made for extraction of gDNA.
The details of oligonucleotide primers and PCR cycling conditions used in the study along with expected amplicon sizes are shown in Table 2. All CMLV isolates were initially confirmed by previously described 7,15 OPXV-specific, ATI gene–based PCR technique. Furthermore, for sequencing of the partial fragment of the C18L gene, extracted gDNA from all the CMLV isolates was subjected to amplification using 10 pmol of each primer (OPXV-specific C18L F and C18L R), 5 μl of 10× PCR buffer, 10 mmol/l each deoxyribonucleotide triphosphate (dNTP), and 0.5 IU of Taq DNA polymerase c in a thermal cycler. d An aliquot (5 μl) of PCR product was analyzed in 1.5% agarose gel to visualize the amplicons after staining with ethidium bromide. The amplicons were directly cloned into pGEM-T Easy vector e after purification using a commercial kit. f The recombinant plasmid DNA was extracted, confirmed by PCR, and sequenced in an automated DNA sequencer. g
Oligonucleotide primers along with polymerase chain reaction (PCR) conditions standardized for different set of primers used in the current study.*
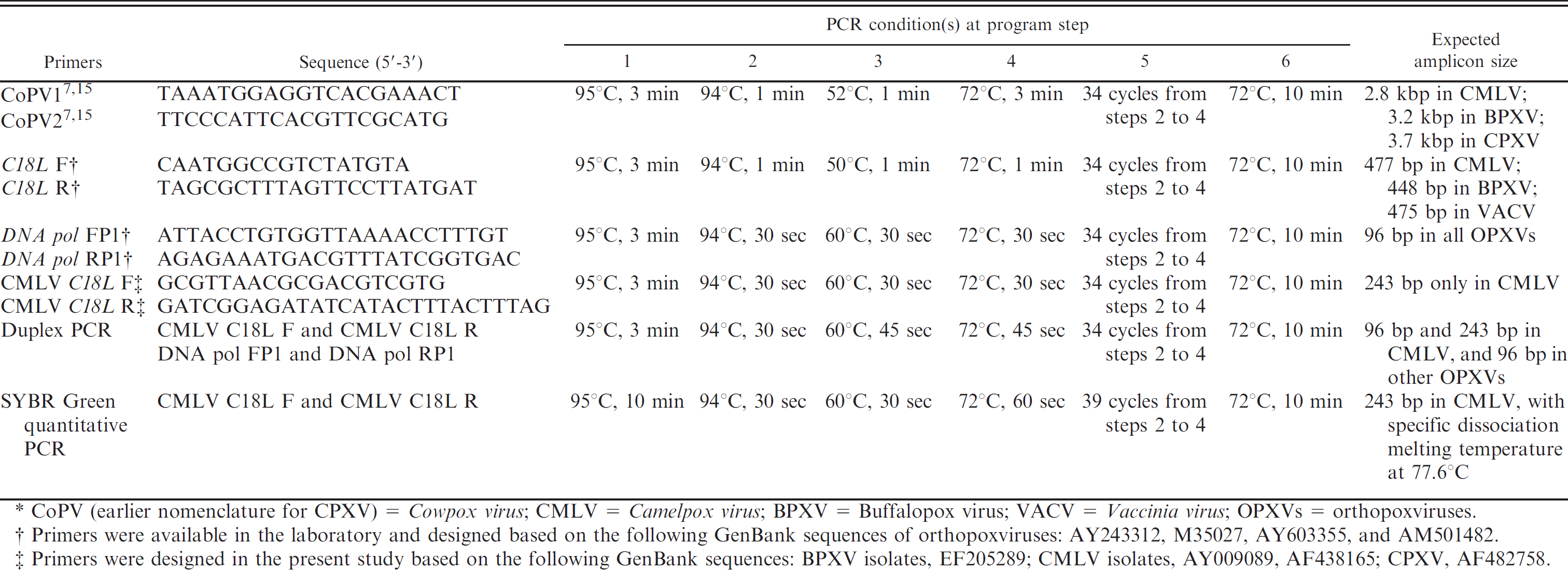
CoPV (earlier nomenclature for CPXV) = Cowpox virus; CMLV = Camelpox virus; BPXV = Buffalopox virus; VACV = Vaccinia virus; OPXVs = orthopoxviruses.
Primers were available in the laboratory and designed based on the following GenBank sequences of orthopoxviruses: AY243312, M35027, AY603355, and AM501482.
Primers were designed in the present study based on the following GenBank sequences: BPXV isolates, EF205289; CMLV isolates, AY009089, AF438165; CPXV, AF482758.
For designing the CMLV-specific primers, sequences of the C18L gene of Indian CMLV were compared with analogous sequences from other CMLV isolates and related OPXVs available in the GenBank database (CMLV-CMS, AY009089; M-96 Kazakhstan, AF438165; BPXV-BP4, EF205289; VACV strains-Copenhagen, M35027; Lister, AY678276; Ankara AM501482; Acambis 3000 Modified Virus Ankara, AY603355; CPXV strains-Brighton Red, AF482758; Germany 91–3, DQ437593; Horsepox virus [HPXV]-MNR-76, DQ792504; RPXV, AY484669). Sequence identity among the CMLV isolates and OPXVs was determined, and the deduced amino acid sequences were assembled into multiple sequence alignment using the ClustalW program of Lasergene 6.0. h A phylogenetic tree was constructed based on the amino acid sequences using the neighbor-joining method in Molecular Evolutionary Genetics Analysis (MEGA) version 4 software program, 23 and the tree topologies were evaluated using 1,000 replicates of the data set.
For standardization of the PCR, extracted DNA from the purified virus was used to amplify 96-bp (DNA pol FP1 and RP1 primers) and 243-bp (CMLV C18L F and CMLV C18L R primers) products from the DNA pol and C18L genes of the CMLV genome, respectively, in separate reactions. Polymerase chain reaction was performed in a 50-μl reaction containing 2 μl of DNA (approximately 400 ng), 10 pmol of each primer, 5 μl of the 10× buffer, 10 mmol/l each dNTP, and 0.25–0.5 IU of Taq DNA polymerase. c Similarly, duplex PCR was performed with 2 sets of primers (FP1 and RP1, and CMLV C18L F and R) together using 1 IU of Taq DNA polymerase. c The amplicons were analyzed on a 2% agarose gel after staining with ethidium bromide.
For assessing the analytical sensitivity of the assays, 10-fold serial dilutions of viral DNA containing 400 ng to 0.4 pg in 2 μl were prepared in nuclease-free water and subjected to assays as described earlier. The sensitivity was defined as the least amount of viral DNA giving an amplification signal. Further, SYBR Green quantitative PCR (qPCR) was optimized for rapid detection and quantitation of CMLV, as well as to determine the efficiency of conventional PCR, using viral DNA and CMLV-specific diagnostic PCR primers. The reaction was carried out in a 25-μl volume using 10 pmol of primers and QuantiTect SYBR Green PCR Master Mix i containing HotStarTaq DNA polymerase i in a Mx 3000p machine j for 40 cycles, with a final dissociation curve program of 95°C for 1 min, ramping down (55–95°C) at a rate of 0.2°C/sec and collecting fluorescence data continuously on the 55–95°C ramp. Mean cycle threshold values of duplicate samples were used for analysis. Standard curve was also performed for absolute quantification as well as to determine efficiency of the assay using serial 10-fold dilutions of viral DNA.
The diagnostic methods previously described for skin infections of camels are more cumbersome, and they either require equipment not available in many diagnostic laboratories (such as electron microscopy) or consist of generic PCR relying upon restriction enzyme or sequence analysis to identify CMLV. The current study describes a new, simple, diagnostic PCR approach based on the C18L gene sequences to identify camelpox in a single assay and to differentiate from other poxvirus infections.
Initially, the identity of the 3 isolates as CMLV was confirmed using previously described techniques. 7,15 Second, to identify a target nucleotide sequence in the CMLV genome for the development of a novel CMLV-specific PCR, the sequencing of a partial fragment of the C18L gene was performed based on the authors' earlier observations. Conserved signature sequences were sought in the host range ankyrin repeat protein coding C18L gene of BPXV, and sequence analysis was subsequently used to devise a PCR strategy for the detection and differentiation of BPXV from other OPXVs. 22 The C18L gene of OPXVs is located in the terminal region of the genome, which encodes ankyrin repeat protein and is a determinant of host range. As per the published sequence data, the open reading frame (ORF) of the C18L gene is 756, 759, 453, and 150 nucleotides in length in CMLV, 1 CPXV, VACV (Copenhagen), and BPXV, respectively.
The PCR amplification of a partial fragment of C18L gene using the OPXV-specific C18L primer set (C18L F and R) resulted in a 477-bp product (data not shown) in all CMLV isolates, which were subsequently sequenced following cloning into pGEMT Easy Vector, and the sequences were submitted to GenBank (accession nos. EF205282, EF205283, and EF592574). Comparative analysis of the C18L gene sequences of Indian CMLV isolates with other representative members of the OPXV genus revealed that the targeted gene was relatively divergent. CMLV isolates showed 99.3–99.6% and 98.7–99.3% identity among themselves at the nucleotide and amino acid levels, respectively. The sequence identity of CMLV was only 59–59.3% with BPXV, whereas with other OPXVs, considerable variations in the identity (92.1–96.2%) were observed. At the amino acid level, CMLV showed sequence identity of 35.4–36.8% with BPXV and 85.3–93.4% with other OPXVs. The detected identities were 90.15–93.4% (CPXV), 87.3–88% (RPXV), 85.3–86% (HPXV), and 86.7–87.3% (VACV). In the multiple aligned amino acid sequences of ORF (data not shown), many substitutions were common to both CMLV and OPXVs. However, a unique change in the Indian CMLV isolates at amino acid position G147D was noticed. Phylogenetic analysis showed that CMLV sequences cluster in a separate group and that these differ from other OPXV members.
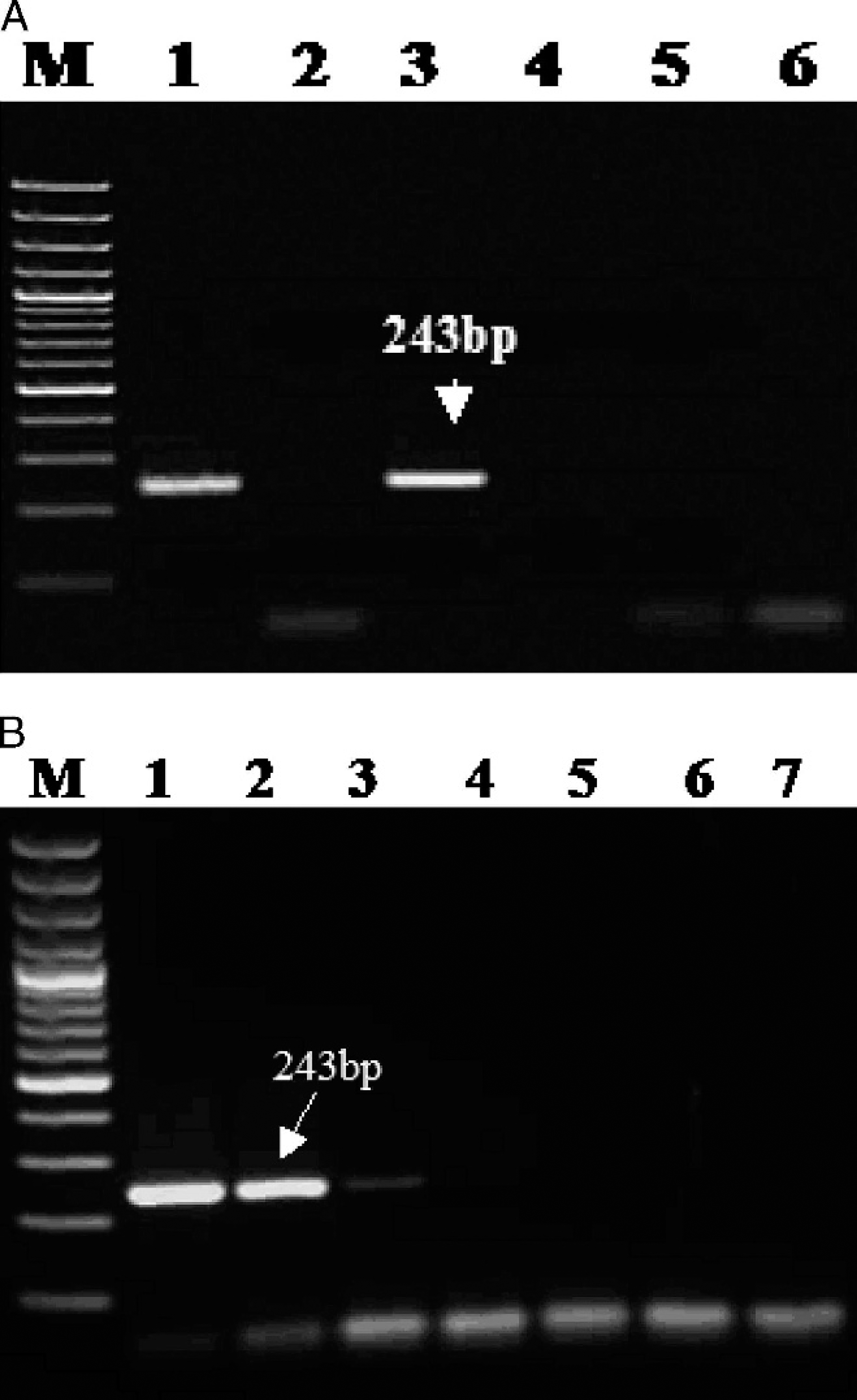
Specificity and sensitivity of diagnostic polymerase chain reaction.
These features were exploited to identify the conserved sequences in the C18L gene of the CMLV genome in order to develop a specific PCR for detection of CMLV and its differentiation from other OPXVs. Therefore, CMLV-specific internal primers (CMLV C18L F and R) were designed using the primer select program of Lasergene 6.0 h for the development of diagnostic PCR. For standardization of the PCR, total gDNAs extracted from purified CMLV-1 and healthy Vero cells were used as positive and negative controls, respectively. The PCR resulted in the specific amplification of a 243-bp product, as anticipated (data not shown), from all CMLV isolates tested. The assay showed specific amplification in CMLV only and did not cross-react with orthopoxviruses, capripoxviruses, and parapoxviruses (Fig. 1A), thus highlighting the specificity of the designed CMLV primers. The specificity of the amplicons was analyzed in the agarose gel and by sequence analysis. The detection limit of the assay was determined to be 0.4 ng of DNA (Fig. 1B).
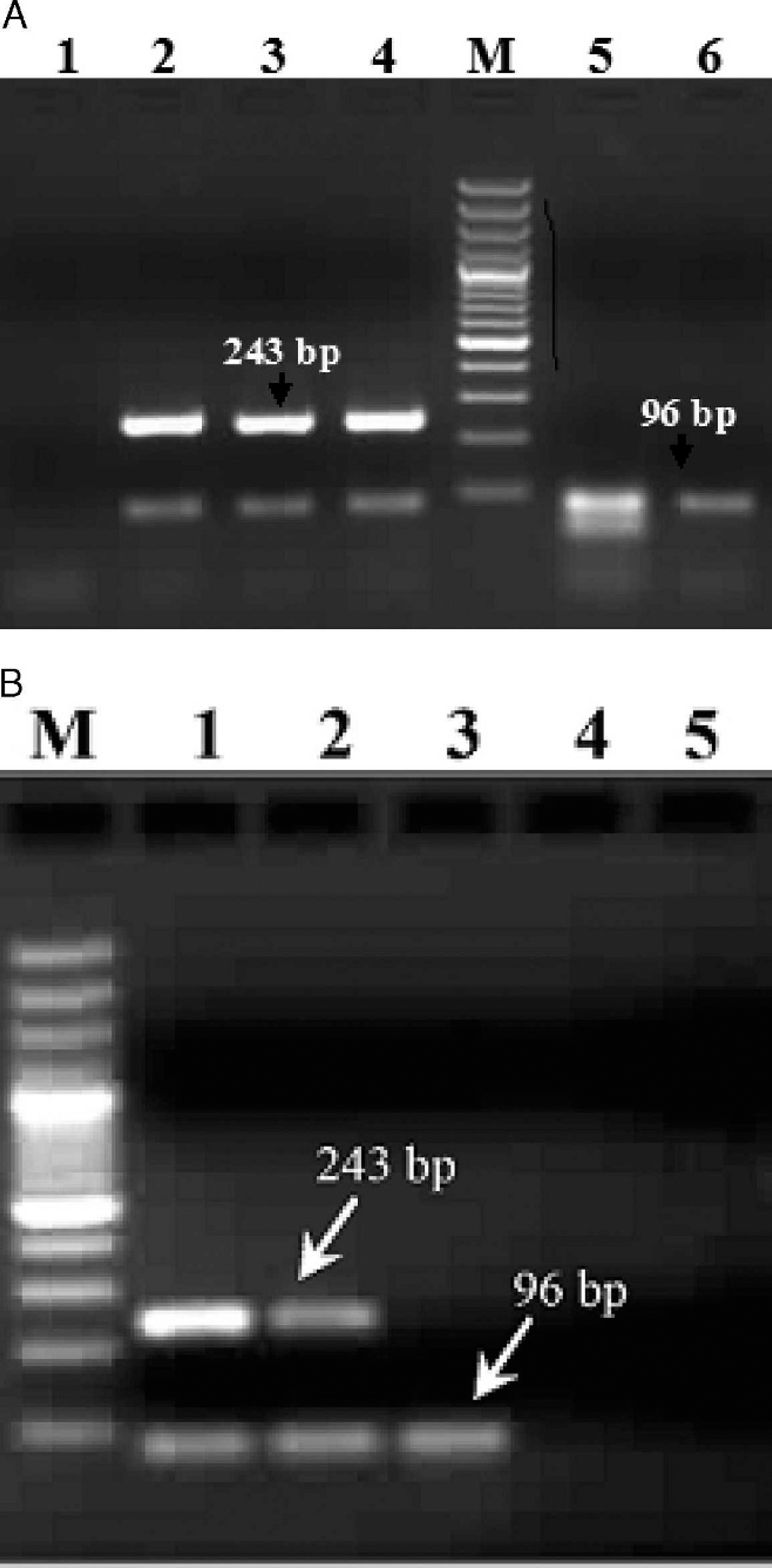
Agarose gel electrophoresis of duplex polymerase chain reaction products of different orthopoxviruses.
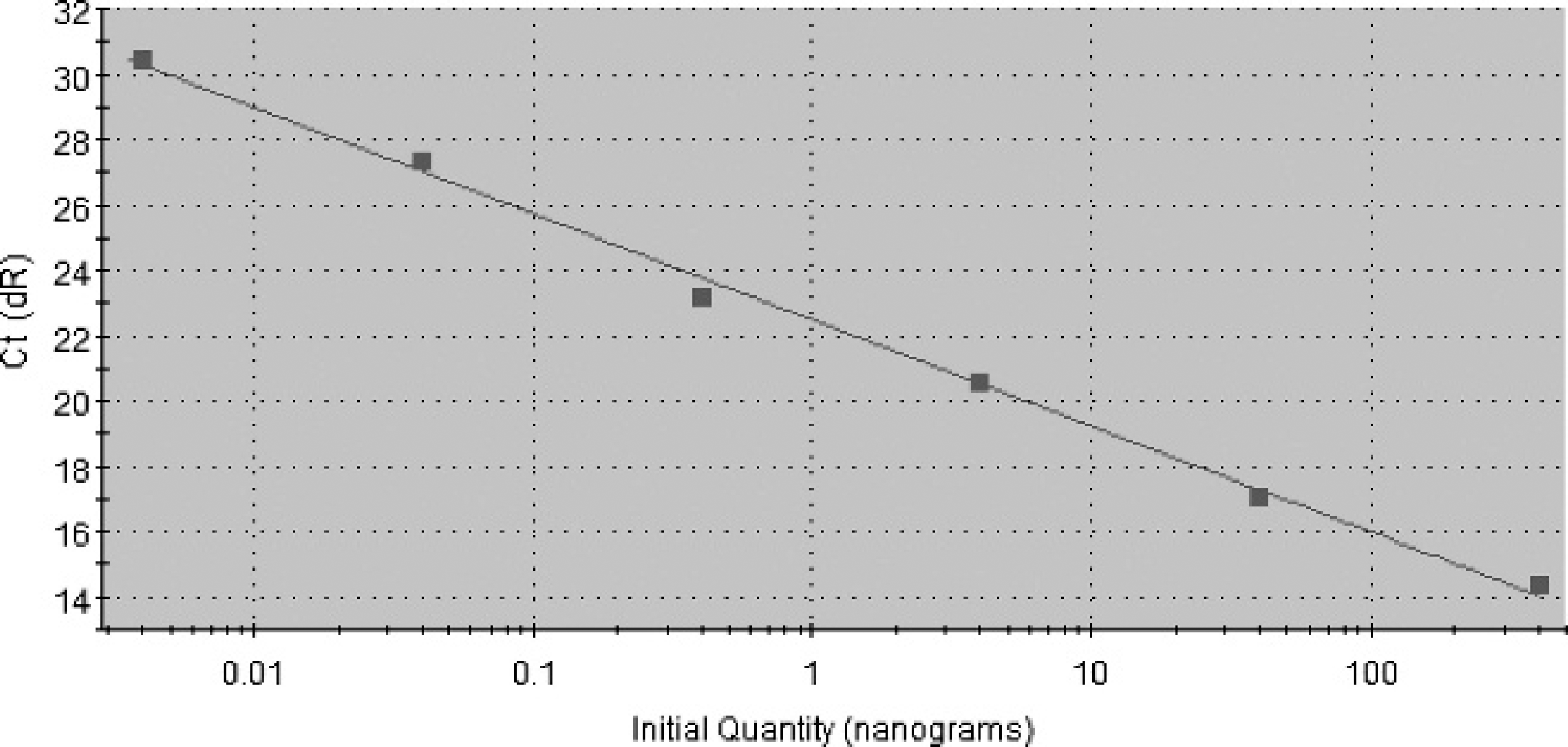
SYBR Green diagnostic quantitative polymerase chain reaction (qPCR) for specific detection and quantification of Camelpox virus (CMLV) DNA. Standard curve obtained with different concentrations of viral DNA ranging from 400 ng to 4 pg.
Diagnostic PCR based on highly conserved regions of a particular gene(s) of a virus can be of immense value for differential diagnosis. Therefore, for routine diagnosis of a virus of diverse origin, PCR using specific primers designed on the basis of 2 different genes to avoid false-negative results are highly preferable. Hence, in the present study, already designed primers targeting the DNA pol gene were also used for the development of a duplex PCR. DNA pol gene is the most conserved nonstructural protein among the viruses in OPXV genus and is group specific (Sahay B: 2006, Evaluation of gene silencing by RNA interference (RNAi) in control of animal virus infections. PhD Thesis, Deemed University, IVRI, Izatnagar, India). A duplex diagnostic PCR based on C18L and DNA pol genes is a much more appropriate assay than an assay based solely on OPXV-specific ATI 18 or HA genes 2 for the simultaneous detection and differentiation of CMLV.
The standardized duplex PCR, using a specific set of primers in a single reaction, yields both DNA pol (96 bp) and C18L (243 bp) gene products in CMLV samples, and only a 96-bp amplicon in other OPXVs, such as BPXV, as expected, which can easily be separated in a 2% agarose gel (Fig. 2A). While developing the duplex assay, the procedure was initially carried out only with a single primer pair for either C18L or DNA pol. Initially, when 10 pmol ofeach C18L and DNA pol gene primer, 10 mmol each dNTP, and 1 IU of Taq DNA polymerase in 50 μl of reaction were used, the intensity of the C18L band was faint. However, when the amount of C18L gene primers was increased from 10 to 20 pmol, the intensity of band was increased satisfactorily. The 96-bp band specific for OPXVs is faint when compared with the 243-bp specific band of CMLV, which appears to affect the detection of OPXVs in the duplex PCR format. Therefore, the analytical sensitivity of this duplex PCR format has limitations due to the different sensitivity that is attained for either the C18L or DNA pol gene–based PCR.
For rapid detection, as well as quantitation of CMLV to determine the efficiency of conventional PCR, SYBR Green qPCR was optimized. The sigmoid curve obtained in the standard curve (Fig. 3) confirmed the presence of a specific CMLV C18L gene amplicon at an annealing temperature of 60°C with an efficiency of 102.9% (Y = −3.254 logX + 22.51; R 2 = 0.996). The dissociation melting temperature of the product at 77.6°C with peak-measured fluorescence [d(F)] was noticed. Quantitative PCR was found to be 1,000 times more sensitive than conventional PCR, because it could detect as little as 0.4 pg of DNA.
The developed assays using DNA of purified virus, namely single or duplex PCR formats, were successfully applied for the diagnosis of camelpox infection using suspected camelpox lesions (Fig. 2B) in an outbreak that occurred during March 2008 at the National Research Centre on Camels, Bikaner, India. Because only a limited amount of clinical scab materials from camelpox infections was available in the current study, the assay was validated using cell culture–adapted CMLV as well as non-CMLV isolates. The C18L gene–based, CMLV-specific novel technique either in single or duplex PCR format described in the current study is not only a precise, simple, rapid method, but the first screening tool in lieu of OPXV-specific, PCR-based assay either on the ATI or HA gene for the detection and differentiation of CMLV. Such an assay may be of immense value in situations where camelpox or pustular dermatitis and other OPXV infections occur concurrently.
In India, sporadic cases of camelpox occur mostly in the northern and central regions (Rajasthan, Haryana, Punjab, Uttar Pradesh, and Madhya Pradesh). Detection of antigen or nucleic acid sequences in camelpox-suspected clinical specimens would be of value not only in India, but also globally. Results of the current study suggest that the assay could attain the required sensitivity to detect the virus in scabs for diagnosis of CMLV infections. Although qPCR was not available for the duplex PCR format, the assay is more convenient owing to its higher sensitivity and rapid detection. The duplex PCR also has a limitation in the simultaneous detection of CMLV and OPXVs because of the difference in the sensitivity of the individual assays. The developed assay requires further testing on biological materials from animals suspected of camelpox and other viral and nonviral diseases of camels that would pose problems in the differential diagnosis of camelpox.
Acknowledgements. The authors thank the Director of the Indian Veterinary Research Institute for providing the necessary facility to carry out this work. This study was supported by grants from the Ministry of Forest and Environment of the Indian government, under the All India Coordinated Project on the Taxonomy capacity building of poxviruses.
Footnotes
a.
Sigma-Aldrich, St. Louis, MO.
b.
AuPreP DNA Extraction Kit, Life Technologies Pvt. Ltd., New Delhi, India.
c.
Fermentas Inc., Glen Burnie, MD.
d.
PTC-200 Thermal Cycler, MJ Research Inc., Waltham, MA.
e.
Promega Corp., Madison, WI.
f.
AuPreP PCRGelx Kit, Life Technologies Pvt. Ltd., New Delhi, India.
g.
ABI 3100, PerkinElmer Life and Analytical Sciences Inc., Waltham, MA.
h.
DNASTAR Inc., Madison, WI.
i.
Qiagen Inc., Valencia, CA.
j.
Stratagene Inc., La Jolla, CA.
