Abstract
Acute phase proteins (APPs) are proteins whose concentrations in serum change after any inflammatory stimulus or tissue damage. The aim of the current study was to evaluate 3 positive APPs (C-reactive protein, serum amyloid A, and haptoglobin) and 1 negative APP (albumin) in female dogs with mammary neoplasia. Acute phase proteins were studied in 70 female dogs aged 8–12 years in the following groups: healthy (n = 10); mammary tumors in stages I (n = 19), II (n = 5), III (n = 6), IV (n = 5), and V (n = 7); and with mammary neoplasia plus a concomitant disease (n = 18). In animals with mammary neoplasia, significant increases of positive APPs were only detected in those that had metastasis or a neoplasm with a diameter greater than 5 cm and ulceration. Dogs with mammary neoplasia and a concomitant disease also had high C-reactive protein concentrations. Albumin concentration was decreased in animals with metastasis and with a concomitant disease. The results of the present study indicate that the acute phase response could be stimulated in female dogs with mammary gland tumors because of different factors, such as metastasis, large size of the primary mass, and ulceration or secondary inflammation of the neoplasm.
Introduction
Mammary tumors are one of the most prevalent types of neoplasm in female dogs, 15 occurring mostly in countries where dogs are not routinely spayed at an early age. 18 Mammary tumors can represent up to 52% of all tumors in female dogs, 3 with 41–53% of tumors diagnosed as malignant. Malignant neoplasms may metastasize to local lymph nodes and internal organs, such as the lungs. 15 Clinically, canine mammary tumors are classified using the TNM (tumor-node-metastasis) system devised by the World Health Organization. 13,15 The TNM system also can be used to achieve offstage the severity of the neoplasm. Both the TNM system classification and staging provide valuable prognostic information that can be used to determine whether tumors can be adequately treated with local therapy or if they require an adjuvant treatment. 16
Acute phase proteins (APPs) are serum proteins whose concentrations change as a part of an innate host defense system called acute phase response. Positive APPs increase in concentration after inflammation and can be divided into major and moderate APPs. Major APPs have an early, marked high rise in concentration followed by a rapid decline in concentration. Moderate APPs require more time to increase in concentration, usually show increases of less magnitude, and then return to reference intervals. In contrast, negative APPs decrease in concentration after an inflammatory stimulus. 6 In dogs, causes of changes in APP serum concentrations include infectious, immunologic, traumatic, or neoplastic diseases. 5 Increased concentrations of positive APPs, such as C-reactive protein (CRP) and serum amyloid A (SAA), have been found in dogs with lymphoma and other hematologic malignancies. 7,8,17 Measurements of APPs can be used as a screening test for systemic response to an inflammatory stimulus and may be helpful to assess clinical progression and to monitor treatment of some neoplastic diseases. 17
Many reports can be found in human literature where APPs have been extensively studied in breast cancer at different stages and clinical presentations. 11,12,18,21 However, similar information is not available regarding APPs and mammary tumors in female dogs. The purpose of this study was to analyze the serum concentrations of the main positive and negative APPs (CRP, SAA, haptoglobin [Hp], and albumin) in female dogs with mammary neoplasia to better understand the concentrations of APPs with regard to the clinical stage of mammary neoplasia.
TNM = tumor-node-metastasis system; T1 = <3cm in diameter; T2 = 3–5 cm in diameter; T3 = >5 cm in diameter; N0 = no metastasis; N1 = metastasis; M0 = no distant metastasis; M1 = distant metastasis.
Materials and methods
Animals
Sixty female dogs between 8 and 12 years of age with a clinical history of mammary masses were studied. These animals were patients from the San Marco Veterinary Hospital (Padova, Italy), different veterinary clinics in southern Spain, or the Veterinary Teaching Hospital of the University of Murcia (Murcia, Spain). Each patient had a physical examination with additional evaluation of the size of the neoplasm and the presence or absence of ulceration. A complete blood cell count, serum biochemical profile, serum protein electrophoresis, blood gas analysis, and urinalysis (with bacterial culture if needed) were performed to detect or exclude any concurrent disease. Inguinal and axillary lymph nodes also were examined, and biopsies were taken during tumor surgery if lymph node metastases were suspected. If distant metastases were suspected, radiography, ultrasonography, and multidetector computerized tomography were used to detect these lesions. Biopsies of the neoplastic masses were obtained prior to or after surgery for a definitive diagnosis of mammary neoplasia. Biopsy specimens were fixed in 10% neutral buffered formalin solution, processed routinely, embedded in paraffin sections, stained with hematoxylin and eosin, and coverslipped. Tissue sections were evaluated by pathologists to determine the histologic type and degree of malignancy of all tumors. Histologic type was determined following World Health Organization criteria. 9
Recorded data regarding tumor size, lymph node involvement, and metastasis were used to classify tumors according to the TNM system. Subsequent staging (stages I–V) of the neoplasms was based on the criteria in Table 1. Concurrent diseases, such as pyometra, mastitis, or other inflammatory diseases, were observed in 18 dogs that were grouped separately (concomitant disease group).
The control group consisted of ten 8–12-year-old female German Shorthaired Pointers that were housed in the Animal Resources Centre Service of the University of Murcia. These dogs were determined to be healthy after physical examination and laboratory testing. All hematologic, serum biochemical, and urinalysis test results were within the reference intervals established at the authors' laboratory (Murcia Laboratory).
Specimen collection
Blood samples for APP determination were obtained by venipuncture of cephalic vein at the time of clinical diagnosis and prior to any treatment. The blood was divided and placed in tubes containing ethylenediamine tetraacetic acid a (EDTA) and clotting activator. b EDTA-anticoagulated samples were used for complete hematologic analysis on the day of collection. Tubes with clotting activator were used for serum APP determination. These specimens were allowed to clot for 30 min and then centrifuged c at 3,000 × g for 10 min. The serum was collected, placed in a new tube, and frozen. All samples were transported on dry ice to the Animal Medicine and Surgery Department (University of Murcia), where they were stored at −20°C until analysis.
APP analysis
Serum Hp concentration was determined by a commercially available kit d in an automated analyzer. e C-reactive protein and SAA concentrations were determined using a solid-phase sandwich enzyme-linked immunosorbent assay d according to the manufacturer's instructions; absorbance was measured at a wavelength of 450 nm using a microtiter plate reader. f Albumin concentration was determined by the bromocresol green dye–binding method using a commercial kit. g All these methods had previously been validated for use with canine samples (Martínez-Subiela S, Tecles F, Parra MD, Cerón JJ: 2003, Analytical validation of different methods for determination of main acute phase proteins in canine serum samples. In: Proceedings of the 4th European Colloquium on Acute Phase Proteins, p. 142. Segovia, Spain). In order to avoid high interassay coefficients of variation, all samples were analyzed in a single run.
Statistical analyses
Descriptive statistics were calculated using a spread-sheet, h whereas statistical analyses were performed using a statistical computer application. i Kolmogorov–Smirnov's test was performed to assess normality of data, giving a nonparametric distribution. Data was log-transformed to assume Gaussian distribution and variance homogeneity. Concentrations of each APP were statistically compared with different clinical aspects of disease, including 1) the clinical stage of disease (stage I, II, III, IV, or V) and presence of concomitant disease (CD), 2) histologic malignancy in which cases were grouped as benign tumors (B), malignant tumors without metastasis (M), or malignant tumors with metastasis (MT), and 3) tumor size designated as nonulcerated masses less than 3 cm in diameter (T1), nonulcerated masses between 3 and 5 cm in diameter (T2), nonulcerated masses with diameter greater than 5 cm (T3a), or ulcerated masses with diameter greater than 5 cm (T3b). In each statistical approach, the group of healthy dogs served as the control group. Groups were analyzed by analysis of variance, and significant differences between group means were located with Tukey's test. The level of significance tested was P > 0.05.
Dogs with mammary tumors grouped by clinical stage of disease.
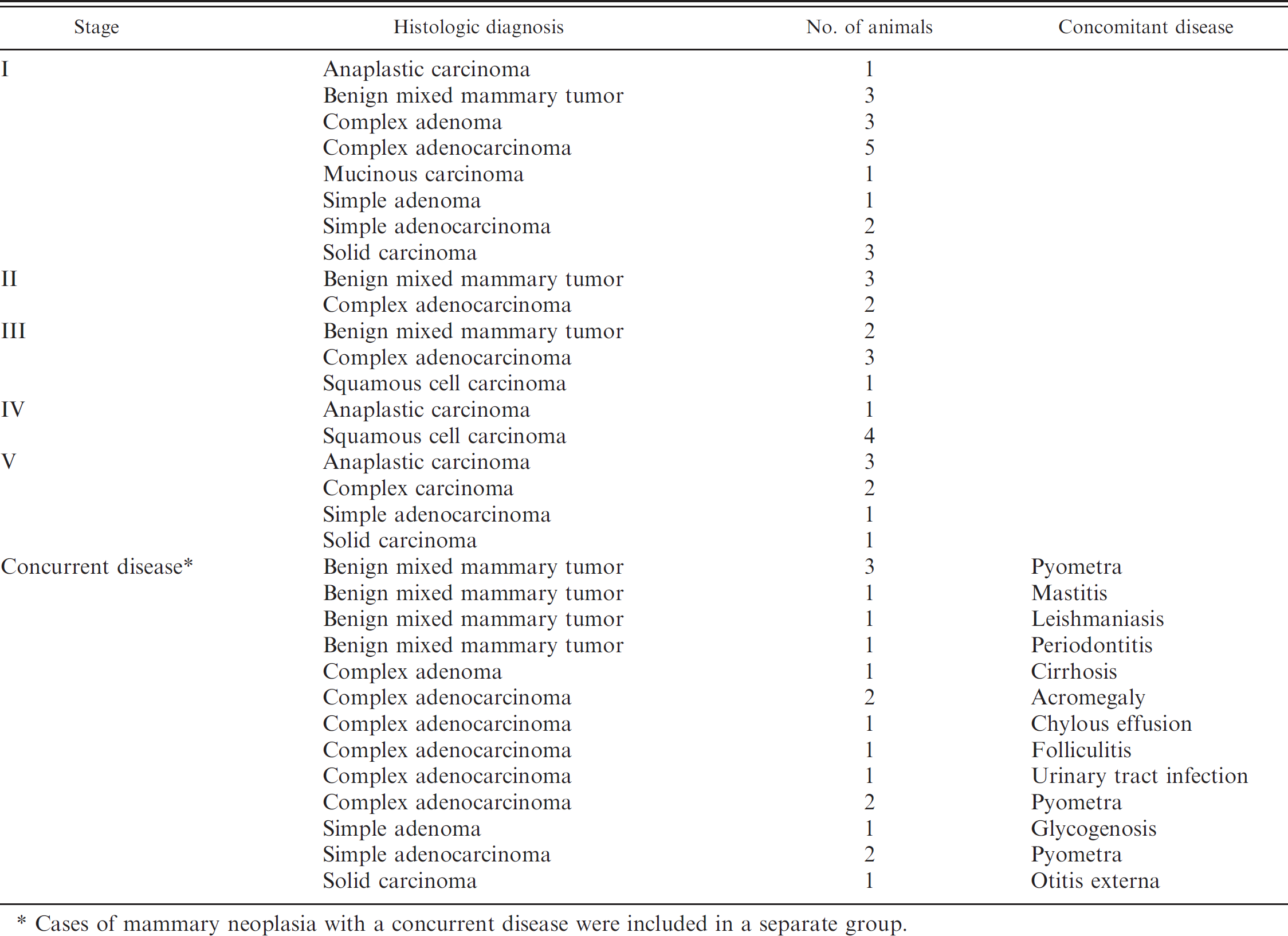
Cases of mammary neoplasia with a concurrent disease were included in a separate group.
Results
Clinical data
Tumor staging, histologic diagnosis, and concurrent diseases detected are shown in Table 2. For the group of 42 dogs with mammary masses and no concurrent disease, 12 dogs had evidence of metastasis (stages IV and V), and 10 of these individuals had ulcerated masses.
Relationship between serum APP concentrations and the clinical stage of the disease
The APP concentrations obtained for each group are shown in Table 3. No differences in CRP concentration were detected between healthy animals and dogs with nonmetastatic mammary neoplasia. Dogs with mammary tumors at stages IV and V and a CD had a significant increase in serum CRP concentration when compared with healthy animals and dogs with stage I and stage II neoplasia. Serum amyloid A concentration was significantly increased in dogs with stage IV mammary neoplasia when compared with healthy animals and dogs with stages I, II, and III neoplasia. Serum amyloid A concentration also was significantly increased in dogs with stage V mammary neoplasia when compared with dogs with stage I or II neoplasia. No statistically significant differences were found between groups for Hp concentration. Albumin concentration decreased significantly in dogs with inflammatory conditions when compared with dogs that had stage I or II mammary neoplasia.
Relationship between serum APP concentrations and malignancy
The results obtained for each group are shown in Table 4. Dogs with metastasis had a significant increase in CRP, SAA, and Hp concentrations compared with healthy animals and dogs with mammary tumors without metastasis. Using the reference interval from the authors' laboratory (0.15–12 mg/l for CRP, 0.79–5 mg/l for SAA, and 0.02–3 g/l for Hp; unpublished results from Murcia Laboratory), serum concentrations of CRP, SAA, and Hp were within the reference interval in 73.3%, 96.6%, and 72.0% of patients with limited disease, respectively. In animals with metastatic disease, serum concentrations of CRP, SAA, and Hp were within the reference interval for 23.0%, 27.3%, and 27.3% of patients, respectively. Albumin concentration was significantly decreased in dogs with metastasis compared with dogs without metastasis. No differences in APPs were found between benign and malignant tumors without metastasis.
Median serum concentrations (25th–75th percentiles) of acute phase proteins of healthy dogs and dogs with mammary tumors in different clinical stages of disease.*
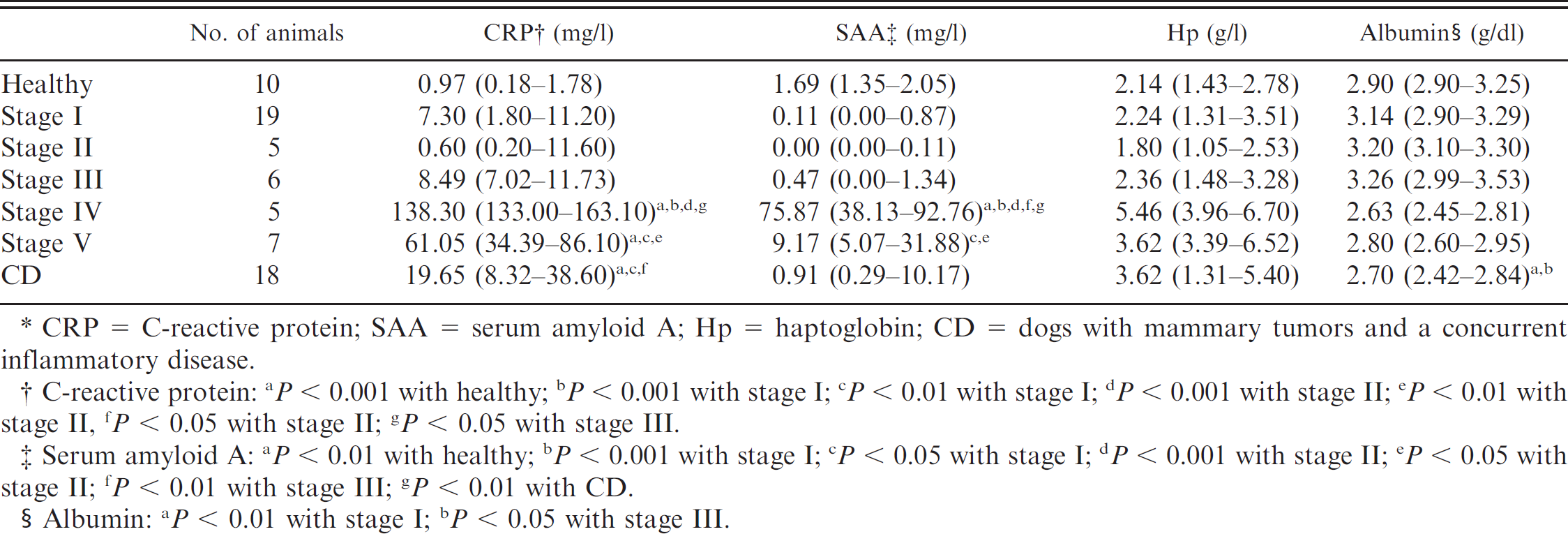
CRP = C-reactive protein; SAA = serum amyloid A; Hp = haptoglobin; CD = dogs with mammary tumors and a concurrent inflammatory disease.
C-reactive protein: a P < 0.001 with healthy; b P = 0.001 with stage I; c P = 0.01 with stage I; d P < 0.001 with stage II; e P = 0.01 with stage II, f P < 0.05 with stage II; g P < 0.05 with stage III.
Serum amyloid A: a P < 0.01 with healthy; b P = 0.001 with stage I; c P = 0.05 with stage I; d P < 0.001 with stage II; e P = 0.05 with stage II; f P = 0.01 with stage III; g P = 0.01 with CD.
Albumin: a P < 0.01 with stage I; b P = 0.05 with stage III.
Relationship between serum APP concentrations and tumor size
The results obtained for each group are shown in Table 5. C-reactive protein concentration was increased in dogs with tumors greater than 5 cm in diameter when compared with healthy animals. The highest CRP concentrations were found in dogs with ulcerated tumors (T3b group). Serum amyloid A concentration was only significantly increased in dogs with ulcerated tumors. No differences were observed in Hp concentration between the groups. Lower albumin concentrations were observed in the CD group when compared with T1.
Discussion
An analytic profile of different APPs was used in the current study, including 2 major positive APPs (CRP and SAA), 1 moderate positive APP (Hp), and 1 negative APP (albumin). Measurement of an APP profile or a serum APP profile may be more meaningful than making observations of a single protein. Specifically, the use of APP profiles that quantitate at least 1 major and 1 moderate APP are preferred to the determination of individual APPs. 5
In animals with mammary neoplasia, significant increases of CRP and SAA concentration were only detected in those dogs that had metastasis or a tumor diameter greater than 5 cm and ulceration. Although it is unknown why stage IV tumors had higher APP concentrations than stage V tumors, factors that could influence the magnitude of the acute phase response of tumors in stages IV and V (e.g., individual variation) should be further studied. Female dogs with stages I–III neoplasia had a poor acute phase response. Similar findings have been described in human patients with breast carcinoma. 12,14 Results would suggest that, as described in humans, mammary tumors are poor stimuli for the production of APPs in the absence of metastasis and secondary inflammation or ulceration.
Median serum concentrations (25th–75th percentiles) of acute phase proteins in healthy dogs and dogs with mammary tumors classified by histologic evidence of malignancy.*
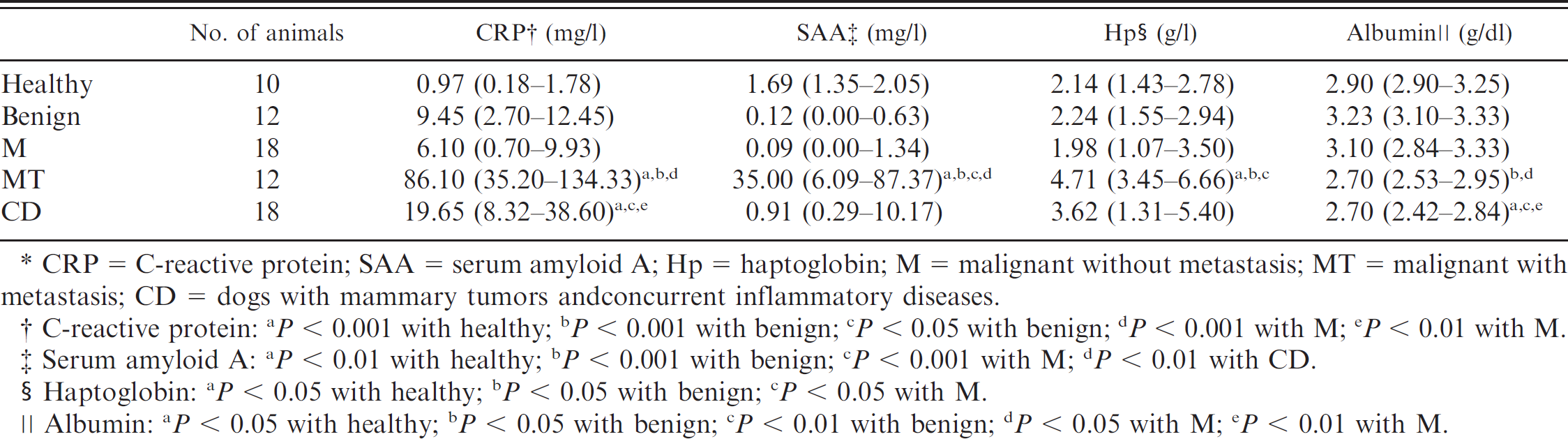
CRP = C-reactive protein; SAA = serum amyloid A; Hp = haptoglobin; M = malignant without metastasis; MT = malignant with metastasis; CD = dogs with mammary tumors andconcurrent inflammatory diseases.
C-reactive protein: a P = 0.001 with healthy; b P = 0.001 with benign; c P = 0.05 with benign; d P = 0.001 with M; e P = 0.01 with M.
Serum amyloid A: a P = 0.01 with healthy; b P = 0.001 with benign; c P = 0.001 with M; d P = 0.01 with CD.
Haptoglobin: a P = 0.05 with healthy; b P = 0.05 with benign; c P = 0.05 with M.
Albumin: a P = 0.05 with healthy; b P = 0.05 with benign; c P = 0.01 with benign; d P = 0.05 with M; e P = 0.01 with M.
Median serum concentrations (25th–75th percentiles) of acute phase proteins in dogs with mammary tumors compared with the size of the neoplasm.*
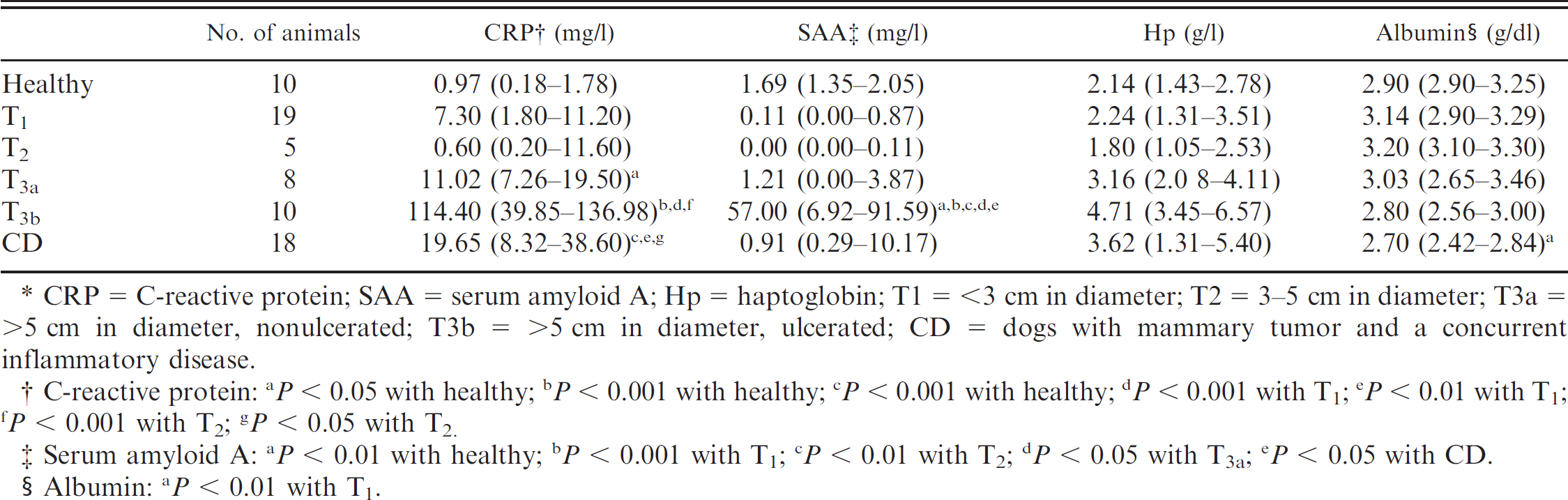
CRP = C-reactive protein; SAA = serum amyloid A; Hp = haptoglobin; T1 = <3 cm in diameter; T2 = 3–5 cm in diameter; T3a = >5 cm in diameter, nonulcerated; T3b = >5 cm in diameter, ulcerated; CD = dogs with mammary tumor and a concurrent inflammatory disease.
C-reactive protein: a P < 0.05 with healthy; b P < 0.001 with healthy; c P < 0.001 with healthy; d P < 0.001 with T1; e P < 0.01 with T1; f P < 0.001 with T2; g P < 0.05 with T2.
Serum amyloid A: a P < 0.01 with healthy; b P < 0.001 with T1; c P < 0.01 with T2; d P < 0.05 with T3a; e P < 5 with CD.
Albumin: a P < 0.01 with T1.
When female dogs with mammary tumors were grouped according to the degree of malignancy of the neoplasm, a significant increase in concentration of all positive APPs studied, especially in CRP and SAA, was found in dogs with metastasis. The relationship between the presence of metastasis and increased CRP concentration has been reported. 4 C-reactive protein and SAA have greater increases in concentration in human patients with metastasis of breast carcinoma 1,12,19 and in other neoplastic diseases. 2 Tumor dissemination is described as a series of complex biologic processes that move tumor cells from the primary neoplasm to distant locations. 20 Therefore, the increased concentrations of APPs observed in cases of metastasis could be due to the direct invasion and damage of tissues by neoplastic cells or due to inflammatory mediators released during this process. Another hypothesis is that dogs with metastasis could have a higher degree of immunosuppression, as has been previously described in humans. 10 This would result in an increased incidence of secondary infections that would stimulate an acute phase response. Two dogs with metastasis that lacked ulceration had high concentrations of the 3 positive APPs and a low albumin concentration. This would support the hypothesis that an inflammatory reaction can be produced in dogs with metastasis without having a direct relationship to ulceration of the mass.
More than 70% of animals with metastasis had high APP concentrations when compared with dogs without metastasis (less than 30%). In humans with cancer, SAA concentration was within the reference interval in 70% of patients with limited disease compared with 44% of patients with metastasis. 19 In addition, increases in SAA concentration have been associated with more advanced disease with other tumors. 1,14 According to the present study, high CRP and/or SAA serum concentrations in a female dogs with mammary neoplasia that lack clinical signs of a concurrent inflammation could be clinically relevant. Further studies should be made to determine the sensitivity and specificity of SAA and CRP quantitation to detect possible metastasis in dogs with mammary neoplasia. It must be pointed out that the current APP study was not designed to detect early metastases or micrometastases. This should be accomplished by histologic examination of intraneoplastic or perineoplastic lymphatic vessels and immunohistochemical detection of tumor markers. Future studies of APPs in mammary neoplasia should clarify whether the presence of micrometastases could be responsible for high APP concentrations.
In the current study, the highest concentrations of CRP and SAA were observed in patients with ulcerated tumors. In humans, it has been reported that high CRP values were observed in response to mammary neoplasms that infiltrated skin or muscle. Furthermore, increased CRP concentrations were more pronounced if the mass was ulcerated. 12 These observations could lead to the hypothesis that in cases of neoplasia, the magnitude of the acute phase response is related to the activity or mass of inflamed tissue, reflecting a nonspecific inflammatory response to tumor necrosis or to local tissue damage.
Female dogs with mammary tumor and CD showed significant increases in CRP concentration, even in those animals with stages I, II, and III of neoplasia. These results were expected, because most of the CDs are recognized causes of an acute phase response. 5 The CD animals in the current study had a wide range of APP concentrations that could be explained by differences in the cause and severity of disease.
The results of the present study indicate that the acute phase response is stimulated in female dogs with mammary gland tumors because of factors such as metastasis, large size of the primary mass, and ulceration or secondary inflammation of the neoplasm. Further studies are desirable to assess the value of APP determination in the prognosis, efficacy of therapy, or recurrence of mammary tumors in female dogs.
Footnotes
a.
Vacutainer® EDTA K3 tubes, BD, Franklin Lakes, NJ.
b.
Tapval, Aquisel SL, Barcelona, Spain.
c.
Refrigerated Centrifuge Z 300 K, Hermle Labortechnik GmbH, Wehingen, Germany.
d.
Tridelta Development Ltd., Maynooth, Ireland.
e.
Cobas Mira PLUS, Horiba ABX, Montpellier, France.
f.
PowerWave XS, BioTek Instruments Inc., Winooski, VT.
g.
Spinreact SA, Gerona, Spain.
h.
Microsoft Excel® for Windows, Microsoft Corporation, Silicon Valley, CA.
i.
SPSS 15.0 for Windows, SPSS Inc., Chicago, IL.

