Abstract
Antemortem biopsy of the rectal mucosa was evaluated as a method for the preclinical diagnosis of chronic wasting disease (CWD) in a herd of ranch-raised Rocky Mountain elk (Cervus elaphus nelsoni) quarantined because of exposure to CWD. Biopsy samples were obtained from 41 elk during the winter of 2005–2006 and from 26 elk from that herd still alive and available for testing during the winter of 2006–2007. Samples were examined for PrPCWD, the protein marker for CWD infection, by immunohistochemistry. PrPCWD was detected in follicles of the rectoanal mucosa–associated lymphoid tissue in biopsy samples from 1 elk with clinical signs of chronic wasting disease and 5 clinically normal elk. The diagnosis was confirmed in all 6 animals by postmortem analysis of brain and peripheral lymph nodes. PrPCWD was also observed in the submucosal plexus and myenteric plexus of the enteric nervous system, and in close association with nonmyelinated mucosal and submucosal nerve fibers. In antemortem rectal biopsy samples from positive animals, immunostaining was consistently observed in approximately 60% of the mucosa-associated lymphoid tissue follicles if 10 or more total follicles per biopsy were present for evaluation. Most antemortem biopsy samples obtained from elk younger than 6.5 years contained at least 10 follicles per rectal mucosal biopsy. These findings support the analysis of antemortem biopsy of the rectal mucosa samples as part of an integrated strategy to manage chronic wasting disease in Rocky Mountain elk.
Introduction
Chronic wasting disease (CWD), a transmissible spongiform encephalopathy, has been reported in captive and free-ranging mule deer (Odocoileus hemionus), white-tailed deer (Odocoileus virginianus), Rocky Mountain elk (Cervus elaphus nelsoni), and moose (Alces alces shirasi). 3,22,26,27 Chronic wasting disease has been a devastating disease in the captive elk industry. An estimated 12,000–14,000 captive elk have been killed in the last 8 years in attempts to control CWD. Several thousand free-ranging mule deer, white-tailed deer, and elk also have been killed through liberalized hunting and targeted sharp-shooting in attempts to reduce the disease prevalence in the wild. Since captive cervids cannot be ruled out as a source of CWD infection for free-ranging cervids, eradication of CWD in captive herds is likely to be critical in controlling the spread of this disease. An accurate antemortem diagnostic test to identify preclinical CWD-infected elk is essential to the success of management strategies aiming to control and eradicate CWD.
An abnormal isoform (PrPCWD) of the host prion protein is a reliable marker for CWD in white-tailed deer, 22 mule deer, 23 and Rocky Mountain elk. 20 PrPCWD and the corresponding abnormal prion protein PrPSc in sheep accumulate in lymphoid tissues during the prolonged preclinical stage of disease, providing the basis for antemortem testing in these species. 21,25,28 Preclinical tests for scrapie in domestic sheep include biopsy of lymphoid tissues from the palatine tonsil, 24 third eyelid, 14 and rectal mucosa. 5,6 Similar preclinical tests have been described for mule deer using biopsy of the palatine tonsil and rectal mucosa. 25,28 An immunohistochemical study 21 of rectoanal mucosa-associated lymphoid tissue (RAMALT) collected postmortem from elk suggested that antemortem diagnosis may be possible in cases with preclinical disease. The present study assessed the feasibility of obtaining suitable antemortem rectal biopsies of RAMALT from a mixed-age herd of Rocky Mountain elk with known exposure to CWD.
. Chronic wasting disease (CWD) in a herd of captive Rocky Mountain elk.*
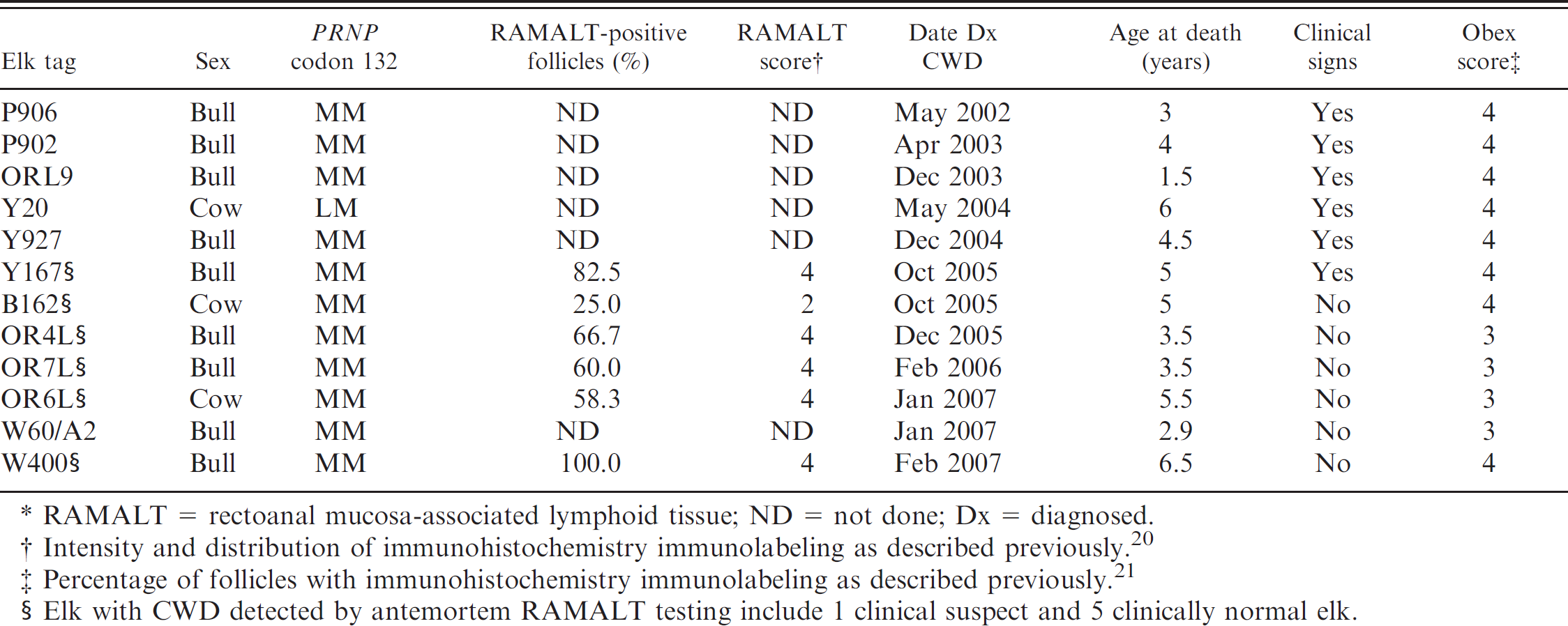
RAMALT = rectoanal mucosa-associated lymphoid tissue; ND = not done; Dx = diagnosed.
Intensity and distribution of immunohistochemistry immunolabeling as described previously. 20
Percentage of follicles with immunohistochemistry immunolabeling as described previously. 21
Elk with CWD detected by antemortem RAMALT testing include 1 clinical suspect and 5 clinically normal elk.
The study included consideration of prion protein genotype in the study animals because a naturally occurring polymorphism in the gene PRNP, encoding the normal prion protein, is associated with relative incubation time in experimentally infected elk. 16 The study also investigated the relationship of a positive antemortem rectal biopsy with disease stage as assessed by clinical signs and a previously described brain immunohistochemical scoring system. 20 Although fewer RAMALT follicles were typically present in biopsy samples obtained from Rocky Mountain elk older than 5 years, the test was used to identify live elk aged 3.5–6.5 years with late-stage clinical, early clinical, and preclinical CWD. Antemortem testing by biopsy of the rectal mucosa should be a suitable component of an integrated CWD management plan for captive elk.
Materials and methods
Samples
Biopsy of the rectal mucosa was performed in a herd of ranch-raised Rocky Mountain elk located in an area of north-central Colorado (the study ranch), where CWD is enzootic in free-ranging elk and deer. The study ranch was started in 2000 on 15 hectares of land with 21 elk (sources: 2 elk ranches in Colorado and an auction). Seven of these elk were purchased from a herd that 4 months later was depopulated because of a single confirmed case of CWD. The study ranch was placed under quarantine and was double fenced; however, the perimeter fence was occasionally visited by free-ranging elk, especially during rutting season. The first case of CWD on the study ranch was diagnosed in 2002 in a 3-year-old bull that had been purchased as a calf from the infected source ranch. As of March 2008, 12 cases of CWD had been diagnosed on the study ranch (Table 1).
In the first year of the study (October 2005–January 2006), antemortem rectal biopsies were obtained from each elk (n = 41) on the study ranch. Animals available for testing included 1 bull elk with evidence of clinical disease, 1 cow with mild weight loss and some behavioral changes, and 39 clinically normal elk (12 cows aged 3–14 years, 14 bulls aged 3–8 years, and 13 long yearlings aged 15–18 months, including 2 females and 11 males). Fifteen of these elk were sampled 2 or 3 times over this 4-month period to develop a preferred method for visualization of the tissue and optimal sample size and location. There was no evidence from examination of the animals on subsequent testing that the repeat biopsy sampling induced inflammation lasting more than a few days, and complete healing of the biopsy site occurred over 2–3 weeks, as evidenced by analysis of the tissue at necropsy. Four elk were removed following detection of PrPCWD in RAMALT samples, 9 elk were euthanized for management reasons, and 1 elk was not available for testing in the second year of the study. The remaining 26 elk were tested using a single rectal biopsy collected between September 2006 and February 2007. Two elk with negative PrPCWD RAMALT samples in the first year of the study had positive RAMALT findings in the second year; these cases are described below. One RAMALT-negative, clinically normal elk was culled for management reasons at the end of this sampling period, and 1 bull that had not been tested in either year of the program died from a fall. Elk with positive immunolabeling in biopsy samples and elk culled for management reasons were euthanized with an overdose of pentobarbital sodium and phenytoin sodium. a Brain, retropharyngeal lymph node, palatine tonsil, and RAMALT were collected at the time of necropsy from all elk in the study.
Antemortem biopsy of the rectal mucosa
Elk were restrained in a hydraulic squeeze chute and positioned so that the rectum could be reached easily and the danger of being kicked was minimized. To relax the rectal wall and enhance exposure of the rectal mucosa, a gloved hand covered with large animal obstetrical lubricating gel compounded with a topical anesthetic b was inserted into the rectum approximately 10 cm and held stationary for 4–5 sec. The hand was then withdrawn. The index and middle fingers of the gloved hand were then used to vertically dilate the anus, thus exposing the lateral walls for biopsy. A second person using sterile, stainless-steel, curved blunt scissors (14 cm) and rat-toothed tissue forceps obtained an approximately 1.0 × 1.5 cm biopsy sample of the rectal mucosa starting at a distance approximately 1.02.0 cm anterior to the mucocutaneous junction. A small degree of bleeding occurred in some cases but was easily controlled by applying local digital pressure. No other adverse side effects were noted. Use of a head lamp by the surgeon greatly facilitated the procedure. All instruments were used once and then soaked for 1 hr in 50% household bleach (3.0% sodium hypochlorite) before discarding.
Immunohistochemical immunolabeling technique
Medulla at the level of the obex (referred to as obex), palatine tonsil, retropharyngeal lymph node, and rectal mucosa were assayed for PrPCWD by immunohistochemistry (IHC) as described previously. 21 Briefly, samples were routinely fixed in buffered formalin, processed, and embedded in paraffin blocks. Antigen retrieval included treatment with formic acid and hydrated autoclaving in citrate buffer. Immunohistochemistry immunolabeling was performed on an automated platform using anti–prion protein monoclonal antibody F99/97.6.1, alkaline phosphatase detection, and hematoxylin counterstaining. c Positive control tissues (lymphoid tissue and obex) from confirmed cases of CWD were included with each run of 19 test slides. Negative control tissues were collected from elk with no known CWD exposure. PrPCWD in the brain appeared as coarse, red, granular, or particulate deposits surrounding neurons and scattered in the neuropil. No deposits were observed in negative control brain. Lymphoid tissues were considered positive if follicles contained coarse, bright red, granular material. Follicles with a pale blue background and no coarse granular deposits were considered negative. No deposits were observed in the absence of primary antibody.
Disease stage determination
In addition to recording clinical signs and assessing the obex for spongiform change as described previously, 20 the stage of hindbrain pathology was assessed by scoring the obex (0–4) for the distribution and intensity of IHC immunolabeling as described previously. 20 Briefly, the obex was scored 0 if no PrPCWD was detected in a single section. The obex was scored 1 if PrPCWD deposits were restricted to the lower half of the dorsal motor nucleus of the vagus nerve (DMNV). PrPCWD deposits filling the entire DMNV and the nucleus of the solitary tract with no immunolabeling of adjacent nuclei were considered grade 2. Relatively heavy immunolabeling of the DMNV and moderate immunolabeling in surrounding nuclei (the caudal portion of the nucleus of the spinal tract of the trigeminal nerve, the ambiguus nucleus, the reticular formation, the hypoglossal nucleus, the olivary nuclei, and the accessory cuneate nucleus) was considered grade 3. Intense immunolabeling of the DMNV and the surrounding nuclei was considered grade 4. The total number of lymphoid follicles observed and the number considered positive were recorded for a single section made from each biopsy sample of rectal mucosa. Samples in which fewer than 10% of the follicles contained immunolabeling were considered RAMALT intensity score 1; samples with immunolabeling of ≥10% and <30% were considered score 2, ≥30% and <50% were considered score 3, and samples with ≥50% of the follicles containing PrPCWD were considered RAMALT intensity score 4.
Genotype analysis
The open reading frame of the PRNP gene, encoding the prion protein precursor, was sequenced as described previously. 20 The deduced amino acid sequence 15 for codon 132 is shown as methionine homozygote (MM), leucine homozygote (LL), or leucine/methionine heterozygote (LM).
Statistical analysis
SAS/INSIGHT for Windows d was used to explore and analyze the observations made on antemortem rectal biopsies. Graphs for presentation were produced using the program Origin e version 6.0.
Results
Study farm history
The study farm was established in 2000, and the first case of CWD was diagnosed 2 years later. Chronic wasting disease was diagnosed in 4 more elk before this biopsy study was initiated. These 5 cases included four 132MM homozygotes (3 purchased and 1 born on the ranch) and one 132LM purchased heterozygote. The heterozygous animal was 6 years of age at the time of death; age at death for the 132MM homozygotes ranged from 1.5 to 4.5 years. These cases are shown in Table 1. Of the 41 elk in the current study, 27 were 132MM, 12 were 132LM, and 1 was 132LL. One elk with clinical signs suggestive of CWD and 5 clinically normal elk were identified as CWD suspects based on RAMALT testing; these cases are described below. One 2.9-year-old 132MM elk died from a fall and was diagnosed with CWD by postmortem analysis; this animal had not been tested by RAMALT sampling.
Antemortem testing and disease staging of elk
Case 1. Case 1 was a 5-year-old bull (Y167; Table 1) with clinical signs consistent with late-stage CWD. Clinical signs included those associated with respiratory distress, weight loss, grinding of the teeth, and excessive salivation. Antemortem biopsies of the lymphoid tissues were collected from the nictitating membrane (Fig. 1A) and rectal mucosa (Fig. 1B); the tissues had similar IHC immunolabeling patterns, consistent with those reported for samples collected from necropsy specimens. 21 Because of the relative ease of collecting rectal tissue biopsies, only RAMALT was collected from the other elk in the present study. The bull was euthanized. At necropsy, gross lesions included emaciation, serous atrophy of fat, and bronchopneumonia. Immunohistochemistry immunolabeling in the obex was scored as 4. Spongiform degeneration was evident within the DMNV and surrounding nuclei, confirming the diagnosis of CWD. Immunohistochemistry immunolabeling was observed in follicles of the palatine tonsil and retropharyngeal lymph node, and also in 52 (82.5%) of 63 RAMALT follicles.
Case 2. A 5-year-old cow (B162; Table 1) displayed subtle signs suggestive of CWD, including mild weight loss and behavioral changes, such as seeking solitude and mild aggression. The behavioral changes were noticed by the owners but could not be confirmed by the attending author (T. R. S.). In the absence of unequivocal signs, the cow was considered to be in the preclinical phase of the disease for the purposes of this study. Immunohistochemistry immunolabeling was observed in 1 (25%) of 4 RAMALT follicles in an antemortem biopsy of the rectal mucosa. At postmortem examination, the only gross finding was the minimal presence of adipose tissue. Examination of the rectum revealed a small healing area where the antemortem biopsy had been taken 10 days previously. Immunohistochemistry immunolabeling of the obex was similar to the distribution and intensity seen in case 1 and was assigned a score of 4. Spongiform degeneration was found in the DMNV and adjacent nuclei of the brain stem. Immunohistochemistry immunolabeling was observed in follicles of the palatine tonsil and retropharyngeal lymph node, and in 23 (71.9%) of 32 RAMALT follicles.
Histopathological examination of the site of the biopsy revealed a small area of the rectum characterized by loss of rectal crypt mucosa covered by a single layer of rectal epithelial cells. A moderate degree of fibroplasia was present in the bed of this defect. Only a few neutrophils were observed in the healing region of the rectal mucosa. The RAMALT follicles were not found under the healing defect of rectal mucosa.
Case 3. Immunohistochemistry immunolabeling was detected in 10 (66.7%) of 15 RAMALT follicles from a biopsy of the rectal mucosa collected from a clinically normal 4.5-year-old bull (OR4L; Table 1). At postmortem examination, the bull was in good body condition. Immunohistochemistry immunolabeling of the obex was scored a 3. Mild spongiform degeneration was found in the DMNV but not in the adjacent nuclei. Immunohistochemistry immunolabeling was observed in follicles of the palatine tonsil and retropharyngeal lymph node, and in a total of 184 (68.4%) of 269 RAMALT follicles as the sum of 3 sections collected from different areas of the rectal tissue. Examination of the rectum revealed no lesion where the biopsy had been taken 3 weeks prior to euthanasia.
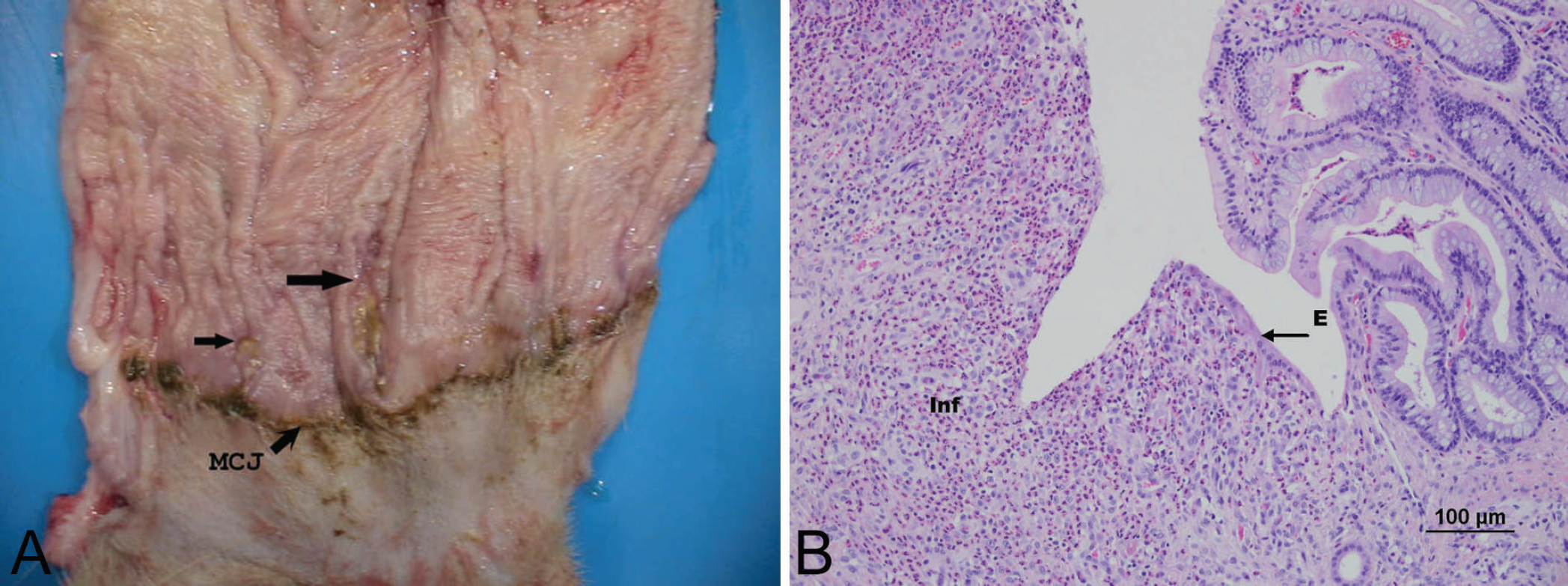
Case 4. Immunohistochemistry immunolabeling was detected in 27 (60%) of 45 RAMALT follicles in a biopsy sample of rectal mucosa from a clinically normal 4.5-year-old bull (OR7L; Table 1). The owners requested a second biopsy because of the apparent health of this bull. Immunohistochemistry immunolabeling in the second biopsy sample was observed in 21 (55.3%) of 38 RAMALT follicles. The bull was found to be in good body condition at postmortem examination. Based on IHC immunolabeling, the obex was scored 3. Mild spongiform degeneration was found in the DMNV but not in adjacent nuclei. Immunohistochemistry immunolabeling was observed in follicles of the palatine tonsil and retropharyngeal lymph node, and in 29 (85.3%) of 34 RAMALT follicles.
Examination of the rectum revealed 2 small healing areas where the biopsies had been taken 7 and 10 days prior to euthanasia (Fig. 2A). These areas were characterized by total loss of mucosa with a moderate degree of fibroplasia within the submucosa. A moderate number of neutrophils were present on the surface and within the fibroplastic bed of the defect (Fig. 2B).
Case 5. Cow OR6L (Table 1) was a large animal with no apparent clinical signs when sampled at 5.5 years of age in November 2005 and when sampled again at 6.5 years of age in January 2007. Immunohistochemistry immunolabeling was not observed in any of the 50 RAMALT follicles examined in the November 2005 biopsy samples (1 sample with 20 follicles and 1 sample with 30 follicles), but was observed 14 months later in 14 (58.3%) of 24 RAMALT follicles in the biopsy sample collected in January 2007. A fourth biopsy was requested by the owners because of the apparent health of the cow; IHC immunolabeling was observed in 8 (53.3%) of 15 RAMALT follicles. At postmortem examination, this cow was in excellent body condition. The obex was scored a 3, based on the widespread but moderate deposits of PrPCWD. Mild spongiform degeneration was found in the DMNV but not in adjacent nuclei. The palatine tonsil and retropharyngeal lymph node contained PrPCWD deposits. Examination of the rectum revealed 2 small healing defects where the biopsies had been taken 7 and 14 days prior to euthanasia; these appeared histologically similar to those described above.
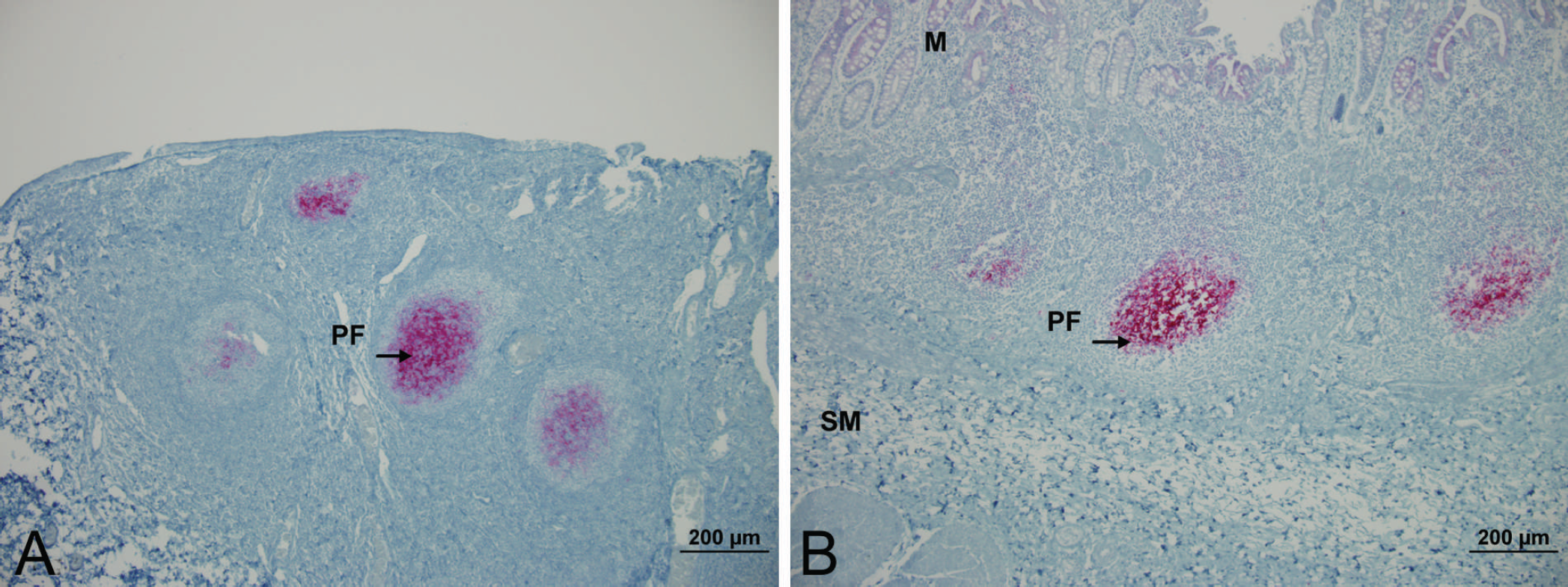
Photomicrograph of lymphoid follicles in the third eyelid of an elk with CWD.
Case 6. Immunohistochemistry immunolabeling was observed in 3 (100%) of 3 RAMALT follicles in a biopsy sample of rectal mucosa obtained from a clinically normal 6.5-year-old bull (W400; Table 1) in February 2007. This bull had been sampled in January 2006, at which time IHC immunolabeling was not observed in 10 RAMALT follicles examined. The bull was not euthanized following the positive biopsy sample; the animal died in July 2007. At postmortem examination, this bull was emaciated. Intense widespread deposits of PrPCWD were observed in the obex (grade 4). Spongiform degeneration was found in the DMNV and adjacent nuclei. In addition to the rectal biopsy findings, IHC immunolabeling was also observed in the palatine tonsil and retropharyngeal lymph node. Postmortem examination of the rectum revealed no evidence of the biopsy.
Elk with no detectable PrPCWD in RAMALT samples. Immunohistochemistry immunolabeling was not observed in RAMALT follicles obtained by antemortem rectal biopsy from any of the other elk during the study period. Eleven elk with negative RAMALT findings were culled to reduce the size of the herd. These elk were found to be PrPCWD negative in samples of obex, retropharyngeal lymph node, and palatine tonsil; this group included nine 132MM and two 132LM elk between the ages of 4.5 and 10.5 years. Twenty-four elk remained alive at the end of the study period. The CWD status of these elk will not be known until they are culled; thus, sensitivity and specificity of antemortem biopsy of the rectal mucosa for detection of PrPCWD cannot be determined for this herd at this time.
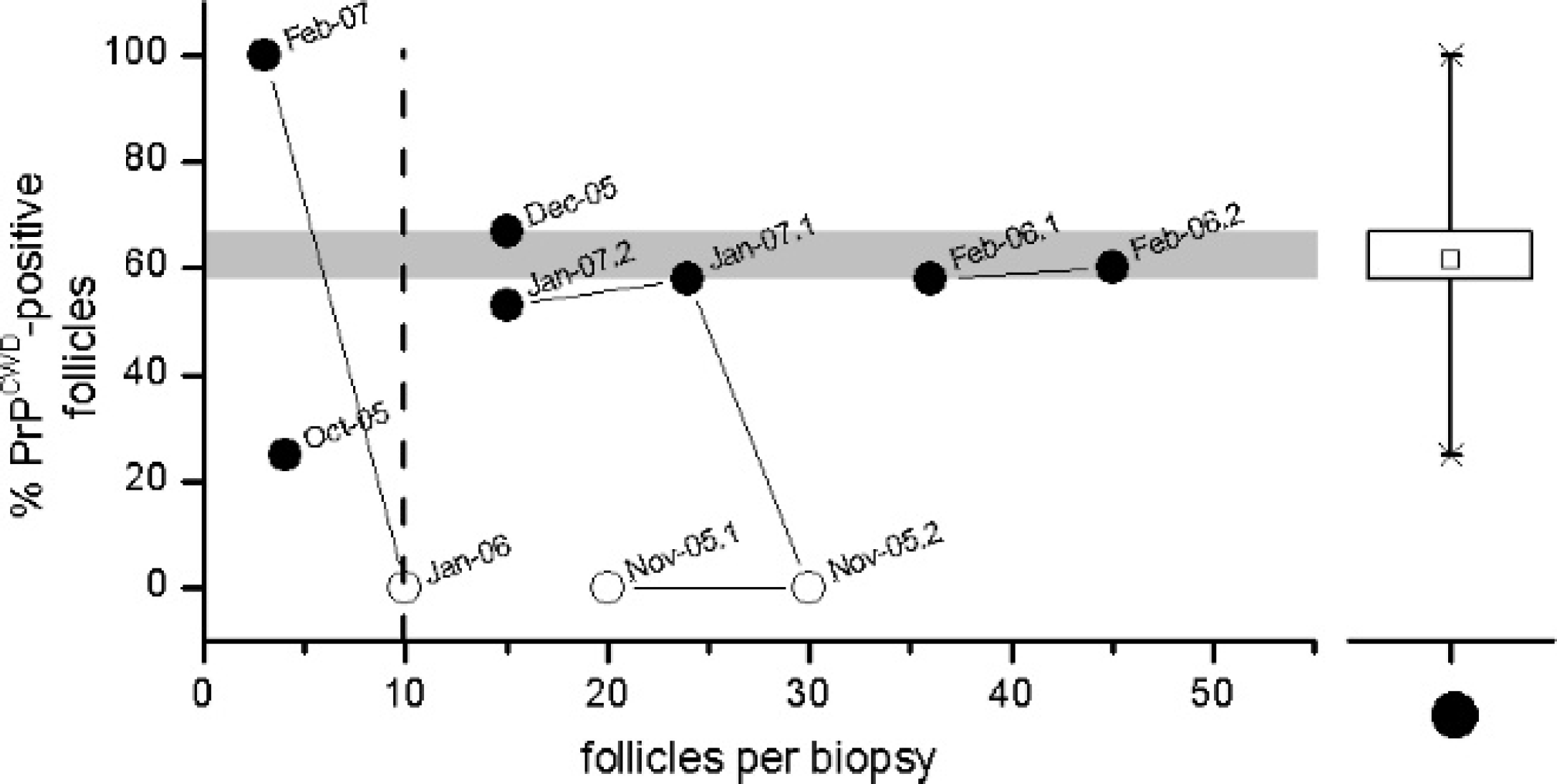
The percent of follicles in which PrPCWD was detected varied with the number of follicles present per rectal biopsy in 5 cull-positive elk. Data shown are the antemortem detection (•) or nondetection (○) of PrPCWD in rectal biopsies obtained from cull-positive elk; all elk in this group were MM132 for the PRNP genotype at codon 132. Lines sequentially connect biopsies obtained from the same animal, as indicated by the sampling times (month-year.sample; sample indicates order if more than 1 biopsy collected per time period). The vertical dashed line offsets data outside of the interquartile range (gray horizontal box) from those within the interquartile range, as determined by the box-and-whisker plot of the first positive biopsy for 5 elk.
Observations on factors affecting the number of RAMALT follicles present for evaluation in a biopsy of rectal mucosa
Antemortem RAMALT evaluation. Rectal biopsies were collected antemortem from 40 elk (25 bulls and 15 cows) with no unequivocal clinical signs suggestive of CWD during the winter of 2005–2006. Bull elk had a narrower age range of 1.5–8.5 years (median: 3.5 years) in comparison with the age range of the cow elk (1.5–14.5 years; median: 6.0 years). Bull elk represented 62.5% of the population examined at this time. The frequencies of PRNP codon 132 genotypes (MM/LM/LL) within the bull elk population were 19:6:0, and in the cow elk population were 8:6:1. The single 132LL elk, a 14.5-year-old cow, was the oldest animal examined.
Of the 5 preclinical RAMALT-positive elk cases described above, PrPCWD had been detected in antemortem rectal biopsy samples (Fig. 3, filled circles) but not detected (open circles) in the initial rectal biopsy samples collected 1 year earlier from 2 of these animals, even though a similar or larger number of follicles were present for evaluation in each biopsy sample. A first approximation of the percent of observed RAMALT follicles per rectal biopsy in which PrPCWD could be detected in the culled preclinical elk was 62% ± 27% (mean ± SD; n = 5 elk; Fig. 3, box plot), the variation from which was minimal in biopsies having more than 10 follicles available for examination.
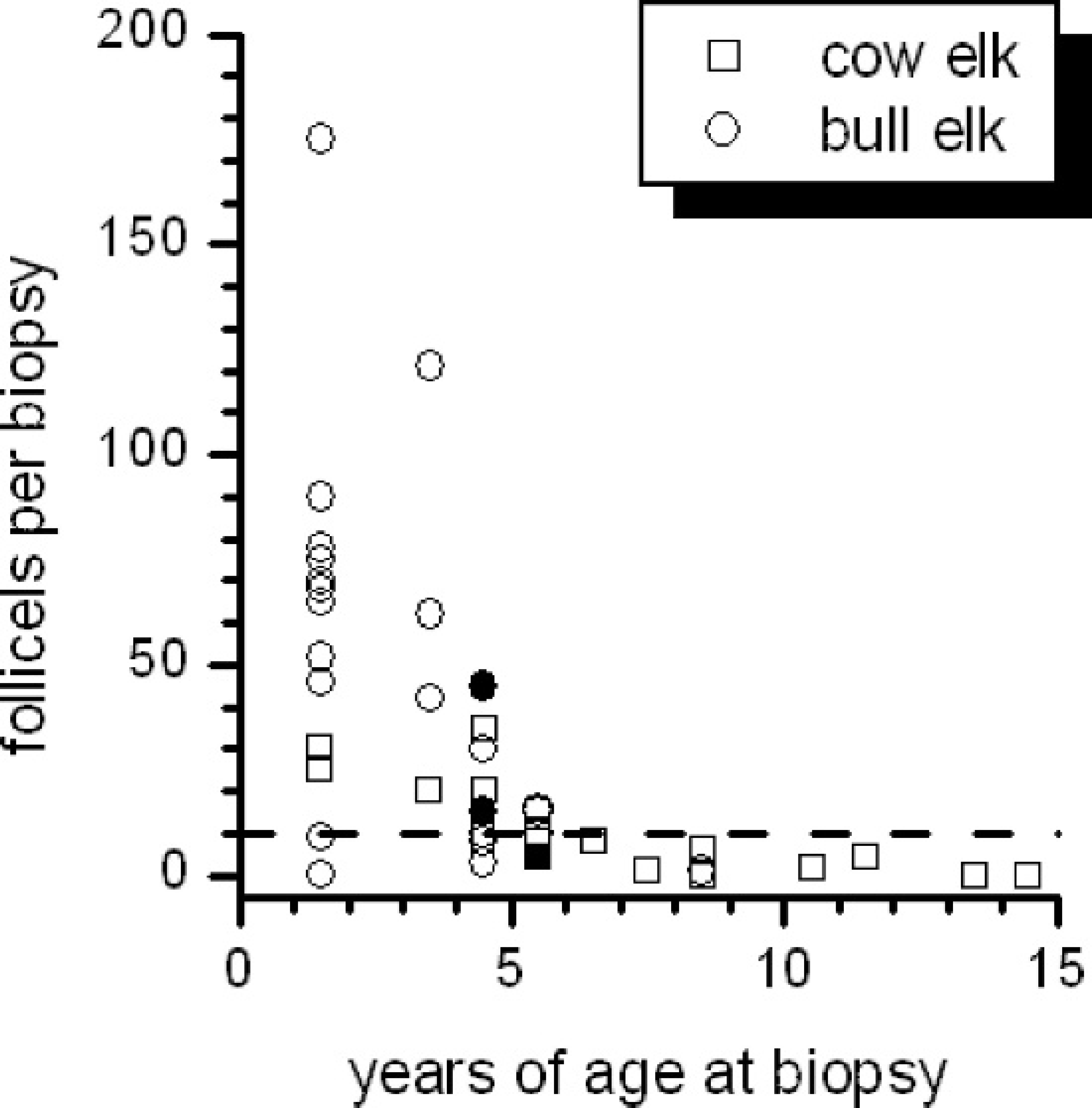
Relationship of the number of RAMALT follicles observed per rectal biopsy to an animal's age in a captive herd of elk. Data are presented as the antemortem detection (closed symbols) or nondetection (open symbols) of PrPCWD in the initial rectal biopsy obtained from 15 cow elk (squares) and 25 bull elk (circles) during the winter of 2005–2006. Note the inverse relationship of follicle number per biopsy and animal age. Most rectal biopsies obtained from animals younger than 5 years contained at least 10 follicles (horizontal dashed line), whereas the number of follicles per biopsy was fewer than 10 for most animals older than 5 years.
The relationship between the number of RAMALT follicles present within a biopsy section (follicles per biopsy) and an elk's age is shown in Figure 4 for the first rectal biopsy sample obtained from 40 clinically normal elk during the winter of 2005–2006 (the bull with clinical signs of CWD was excluded from this analysis). Under the field conditions of this study, fewer follicles were present for evaluation within rectal biopsies obtained from older elk; in fact, most rectal biopsies from animals older than 5 years had fewer than 10 RAMALT follicles present for evaluation. The appearance of this inverse relationship of follicles per biopsy with animal age was the same for the results from biopsies obtained from the elk remaining in the winter of 2006–2007 (data not shown). Furthermore, there was no obvious difference between 132MM and 132LM genotypes (data not shown).
Postmortem RAMALT evaluation. A total of 3 to 6 full-thickness sections of rectum, starting at the mucocutaneous junction and progressing anterior for a distance of 10 cm, were prepared from tissue collected at necropsy from 1 clinical case (Y167) and 2 preclinical cases (B162 and OR7L) in order to compare the number of RAMALT follicles present within a biopsy between sampling locations. The number of RAMALT follicles observed within 1–3 cm of the mucocutaneous junction ranged from 34–67 per biopsy but were rarely present in samples more than 3 cm anterior to the mucocutaneous junction. Within the initial antemortem rectal biopsies from these elk, 4, 15, and 45 RAMALT follicles per biopsy were observed. The increased number of follicles in these 3 antemortem biopsies probably represents improved technique over the course of the project, underscoring the impact of biopsy collection technique (location, size, and depth) on the number of RAMALT follicles likely to be present for evaluation.
Additional observations of IHC immunolabeling in biopsies of the rectal mucosa
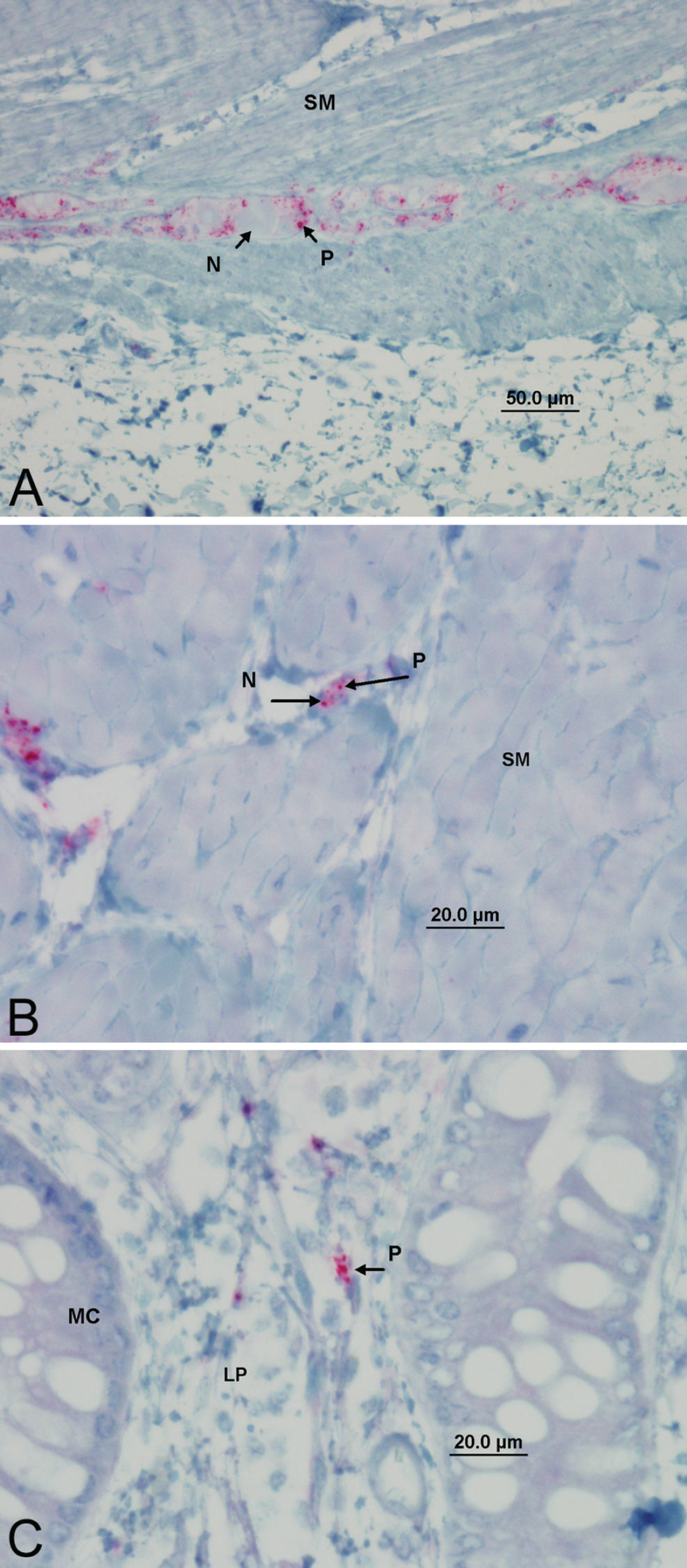
In addition to the presence of IHC immunolabeling within RAMALT follicles, bright red chromogen deposits were also observed near the periphery of neurons (Fig. 5A). A few small, bright red chromogen deposits could be found in nonmyelinated nerves (Fig. 5B). In addition, bright red chromogen deposits could be found in the loose connective tissue of the lamina propria near rectal mucosal cells (Fig. 5C). Some or all of these immunolabeling patterns were observed in the clinical elk and the 5 preclinical elk in this study.
Discussion
Antemortem diagnosis of ovine scrapie is performed by biopsy of lymphoid tissue from palatine tonsil, 13 nictitating membrane, 12 and rectal mucosa. 3,4 The disease-associated isoform of the ovine prion protein is typically detectable in lymphoid tissue by IHC immunolabeling at approximately one third to one half of the incubation period, and thus is considered a reasonable marker for preclinical scrapie disease in sheep. 24
Similarly, preclinical accumulation of PrPCWD in palatine tonsil and retropharyngeal lymph node is considered a reliable marker for preclinical CWD disease in mule deer. 12,13,20,22,23 Experimental oral infection of mule deer resulted in PrPCWD accumulation in alimentary tract–associated lymphoid tissues (retropharyngeal lymph node, palatine tonsil, Peyer patches of the small intestine, and mesenteric lymph nodes) as early as 42 days after inoculation. 19
The pathogenesis of CWD in elk is less well defined. PrPCWD was detected in brain and lymph nodes (68%), nodes only (19%), and brain only (13%) in a sample of 226 naturally infected captive elk. 20 If PrPCWD accumulates in RAMALT follicles at a frequency similar to that in the retropharyngeal lymph node, the relatively high number of CWD-infected elk with PrPCWD restricted to the central nervous system will limit the sensitivity of RAMALT testing as an antemortem test for CWD diagnosis in preclinical animals. In the current study, biopsy-negative animals culled for herd management reasons were also negative by postmortem examination of obex and retropharyngeal lymph node. The remaining elk in this facility will be monitored by annual RAMALT testing and complete necropsies when culled. Larger studies of hunter-harvested elk and elk removed from infected herds will be performed to determine the sensitivity of RAMALT testing in elk.
The age of the animal might also affect the design of diagnostic testing protocols. In this study, numerous RAMALT follicles were present in the rectal biopsy samples obtained antemortem from elk ≤5 years old, but fewer follicles were present in biopsy samples obtained from elk ≥6 years old. Thus, the number of follicles present per biopsy sample appeared to decline with age in this herd of elk and is similar to the findings of a previous study on postmortem samples. 21 Although the minimum number of follicles necessary for an accurate test is not known, the RAMALT follicle number in this study was adequate for detection of PrPCWD in 1 clinical elk and 5 preclinical elk between 4.5 and 6.5 years of age, which is the typical age of ranch-raised elk with CWD.
The low numbers of follicles found occasionally in young animals were believed to be attributable to the experience of the person taking the biopsy samples. Even though the biopsy technique is relatively easy, there was a learning curve. For example, one problem encountered initially was animal straining with mechanical anal dilation. This caused protrusion of the rectal mucosa, increasing the chance that the biopsy would be collected further anterior to the mucocutaneous junction than intended.
In this study, approximately 60% of the RAMALT follicles were PrPCWD positive in biopsies containing at least 10 follicles. The extent to which this approximation depends on the epidemiology of this particular outbreak is not known; CWD was observed 5 years after introduction of elk from an infected herd into this presumed naïve herd, and antemortem rectal biopsy sampling was performed 3 years after the first of the purchased animals developed clinical disease. Herds with more recent introduction of infected animals or long-term housing of infected animals may have other characteristics. Nonetheless, the proportions of positive RAMALT follicles observed in the present antemortem study in which at least 10 follicles per biopsy were present for evaluation are quite similar to the proportions previously reported for RAMALT samples collected at necropsy from 2 other ranch-raised herds of elk. 21
The interval between infection and the appearance of PrPCWD in the RAMALT of elk is not known, but disease progression appears to be highly variable and can be rapid in some cases. In this herd, 1 animal was diagnosed with CWD as early as 1.5 years, and 1 animal progressed from having no apparent clinical signs of CWD but a positive rectal biopsy in January 2007 to having clinical disease and an obex IHC score of 4 by July of that year. PrPCWD was not observed within rectal biopsy samples from 2 of the 5 cull-positive cases collected in the previous year, even though 10 or more follicles were evaluated in those samples. The observation that 2 of these animals had obex IHC scores of 3 or 4 within a year after converting to a positive rectal biopsy suggests that involvement of the RAMALT occurs relatively late in disease progression within elk, especially when compared with scrapie in sheep.
No PRNP genotype is protective in experimental disease, 16 and CWD was diagnosed in wild elk of genotype frequencies in proportion to their abundance in the sampled population. 17 However, PRNP genotype was associated with a prolonged incubation period in experimentally infected elk. 7 If a lag time in development of clinical signs is associated with a delay in the onset of lymphoid tissue PrPCWD accumulation in natural disease, PRNP genotype will need to be considered when developing testing protocols for Rocky Mountain elk.
In spite of the potential limitations, identification of infected herds by whole-herd screening using biopsy of the rectal mucosa should be a useful adjunct to federal and state control programs. Rectal biopsies can be performed in elk under restraint conditions currently used for artificial insemination, and the procedure can be done with disposable instrumentation. This technique may prove to be useful in preventing movement of infected clinically normal animals from herds of unknown CWD status if they are tested prior to movement. Risk factors for transmission of CWD in farmed herds in Canada included exposure to elk that died of CWD or developed clinical signs 2 ; if identification and prompt removal of elk earlier in disease reduce transmission efficiency, rectal biopsies may be useful in management of infected herds. It will be imperative for producers to understand that a negative test cannot be interpreted to mean that the elk is not infected; a biopsy-negative elk may be in an early stage of infection with the CWD agent before the detectable accumulation of PrPCWD in RAMALT and other lymphoid tissues, 21 or it may represent one of the 13% of animals in which detection of peripheral PrPCWD prior to accumulation in the central nervous system is not possible. 20
In addition to their value in regulatory programs, antemortem tests using minimally invasive methods have value in experimental studies of CWD pathogenesis and transmission in elk. The distribution of PrPCWD in the neural and extraneural tissues of naturally infected elk at early stages of disease can be examined in animals identified through live animal testing. In this study, the finding of bright red chromogen deposits indicative of PrPCWD on the periphery of enteric neurons, nonmyelinated nerves located within the intestinal submucosa, and loose connective tissue of the lamina propria in the clinical and preclinical elk examined in this study is extremely interesting. The accumulation of PrPCWD on the periphery of enteric neurons is similar to the deposition observed around neurons of the central nervous system, suggesting that PrPCWD also accumulates in and around these peripheral neurons of the intestinal tract early in disease. Similar findings have been described in studies of the pathogenesis in sheep and mule deer. 1,4,8 11,18,19
Prospective studies on captive and free-ranging elk using this technique are in progress. These studies will delineate the diagnostic sensitivity and specificity of RAMALT screening as a method for identification of infected elk herds as part of an integrated management program to reduce the incidence of CWD in Rocky Mountain elk.
Acknowledgements
The authors thank Dennis and Stephanie White (owners of Velvet Ridge Elk Ranch, Fort Collins, CO) for allowing us to biopsy their elk and for providing assistance with handling the elk and recording data. The authors also thank Denise Spraker, Troy Spraker, Sandra Spraker, Michael Lavalle, Justin Fischer, Patrick Burke, Tara Rudy, Heidi VanRackel, and Scott Leach for assisting with biopsy collection and data recording, and Robert Zink for embedding and cutting biopsy samples. Bruce Cummings provided immunohistochemical immunolabeling of the tissues. This project was supported in part by USDA ARS/ADRU Specific Cooperative Agreement 58–5348–2–0678 and the Colorado State University Diagnostic Laboratory, College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins, CO. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Footnotes
a.
Beuthanasia-D Special®, Schering-Plough Animal Health Corp., Union, NJ.
b.
3% methylcellulose base with 2% lidocaine, Vetricare Pharmaceuticals, Templeton, CA.
c.
Ventana Medical Systems Inc., Tucson, AZ.
d.
SAS 9.1.3, SAS Institute Inc., Cary, NC.
e.
Microcal Software Inc., Northampton, MA.
