Abstract
A gemistocytic astrocytoma is described in the spinal cord of a 3-year-old, spayed, female, Domestic Shorthair cat. The neoplasm was tested for glial fibrillar acidic protein (GFAP), epidermal growth factor receptor (EGFR), and p53 protein expression. On histologic exanimation, the neoplastic cells were pleomorphic, with distinct cell borders and abundant cytoplasm that frequently extended into variably sized fibrillar processes. Neoplastic cells were strongly positive for GFAP and negative for EGFR. Eight percent (mean percentage) of neoplastic cells were p53 positive. Only 1 p53-positive cell was found in the normal spinal cord. The increased expression of p53 in the neoplasm was suggestive of an abnormal biological behavior of the p53 protein and its possible involvement in the neoplastic process of feline astrocytoma.
Dogs are the domestic species most frequently affected by astrocytomas. 13 In contrast, astrocytomas are rare in cats 9 and represent only 2.8% of feline intracranial neoplasms and 3.5% of spinal-cord neoplasms. 7,14 Among the different types of astrocytomas, the gemistocytic astrocytoma is the least common, but, like the other variants, it occurs most frequently in the cerebrum. 2,10,12,16 A retrospective study of 31 canine astrocytomas included only 2 cases of gemistocytic astrocytoma. 13 Cerebral gemistocytic astrocytoma has rarely been reported in cats, 2,5 and only 1 gemistocytic astrocytoma has been reported in the feline spinal cord, in which lymphosarcoma and osteosarcoma are the most common neoplasms. 7
In humans, the transformation of normal astrocytes into neoplastic astrocytes is frequently associated with mutation of the p53 tumor suppressor gene. Neoplastic progression to anaplastic astrocytoma and glioblastoma over time also is associated with accumulation of other genetic abnormalities, such as overexpression of epidermal growth factor receptor (EGFR). 3 Although the genetic characterization of astrocytic tumors has been investigated as a potential indicator of prognosis in human medicine, in veterinary medicine, little data are available on the genetic characteristics of these neoplasms.
The p53 protein promotes DNA repair processes by arresting cells with DNA damage in the G1 phase of the cell cycle or induces cell death by activating apoptosis when irreparable genomic injuries occur. Consequently, mutations that lead to loss of function of this protein induce uninhibited growth, genetic instability, and immortalization of the cell, thus contributing to neoplastic transformation. 8 In the current study, the gross, histologic, and immunohistochemical features of a gemistocytic astrocytoma in the spinal cord of a cat are described.
A 3-year-old, spayed, female Domestic Shorthair cat was presented to Texas A&M University Veterinary Teaching Hospital (College Station, TX) with a 2-week history of difficulty walking. Neurologic examination revealed ambulatory paraparesis, with general proprioceptive ataxia, pelvic limb postural reaction deficits, clonic patellar tendon reflexes, pelvic limb crossed-extensor reflexes, and probable hyperesthesia over the cranial lumbar aspect of the vertebral column. The lesion was localized to the T3—L3 spinal-cord segments. By using computed tomography, a poorly demarcated, isoattenuating, moderately contrast-enhancing, intramedullary spinal-cord mass was seen at the T12-T13 articulation. Analysis of cerebrospinal fluid from the cisterna magna showed albuminocytologic dissociation (protein: 52 mg/dl, reference interval: <25 mg/dl). The cat was treated empirically with prednisone (0.3 mg/kg PO [by mouth] BID [twice a day]) and clindamycin (12.5 mg/kg PO BID), with minimal response to therapy. Because of the poor prognosis, euthanasia was elected.
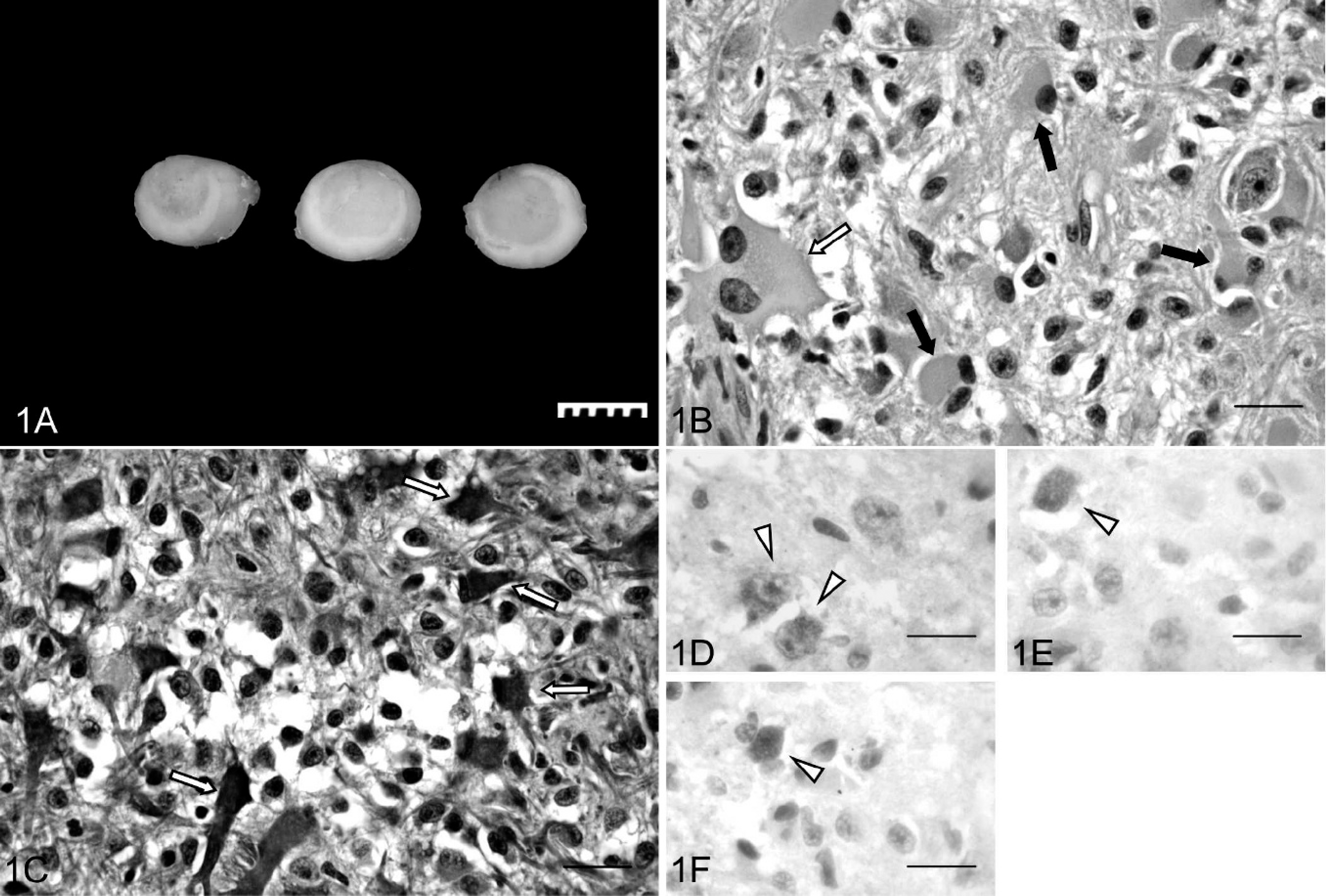
At necropsy, 1 cm of spinal cord between T12 and T13 was swollen and pale pink. Cross sections of the spinal cord examined after fixation in 10% neutral buffered formalin contained a well-demarcated, homogeneous, grayish mass that ranged from 0.4 to 0.5 cm in diameter and that displaced approximately 70% of the normal spinal-cord parenchyma in the affected segment (Fig. 1A). After fixation, the neoplasm was routinely paraffin embedded, and 4-μm sections were stained with hematoxylin and eosin. Five-micron sections were mounted on silanized slides, deparaffinized in xylene, and rehydrated in graded alcohols. Immunostaining was performed by using a polymer-based detection kit. a Slides were incubated with 1 of 3 primary antibodies: glial fibrillary acidic protein b (GFAP; 1:2000), p53 c (1:100), or EGFR d (1:100) for 1 hr at room temperature. Color reaction to detect the site of the antigen was obtained with 3,3′-diaminobenzidine. b A universal negative control b was substituted for the primary antibody in the negative control.
Cells positive for p53 protein were manually counted, and the positivity was expressed as the mean percentage of the number of positively stained cells in 30 microscopic fields (100X) within the neoplasm. Positive cells were further subgrouped according to the cytoplasmic or nuclear location of the protein. To provide comparative data on p53 expression, 4 representative cross sections of spinal cord from an adult, neutered, male Domestic Shorthair cat were immunostained for p53 protein as described above.
Spinal cord gemistocytic astrocytoma; cat.
On histologic examination, the neoplasm was a densely cellular, poorly demarcated, unencapsulated, intramedullary mass that compressed the surrounding neuropil at its borders. The cells were arranged in bundles within a fine vascular and fibrillar stroma. The gemistocytic neoplastic population was characterized by pleomorphic, angular-to-polyhedral cells, with distinct cell borders that often had abundant, homogeneous to finely granular cytoplasm that frequently extended into variably sized fibrillar processes. Nuclei were large, oval to indented, primarily eccentric, with a hyperchromatic nucleolus and finely stippled chromatin. Rare binucleate cells were at the periphery of the lesion. Anisocytosis and anisokaryosis were moderate (Fig. 1B).
Neoplastic cells stained strongly positive for GFAP (Fig. 1C). The degree of differentiation of the neoplastic population along with the strong GFAP reactivity is consistent with a low-grade astrocytoma. Differentials for this category include the pilocytic, 9 fibrillary, protoplasmic, and gemistocytic variants. The lack of predominant anaplasia, mitotic activity, vascular proliferation, and necrosis exclude more malignant astrocytic neoplasms like the anaplastic astrocytoma (medium-grade astrocytoma) and the glioblastoma multiforme (high-grade astrocytoma). The main neoplastic population in this neoplasm was characterized by large astrocytic cells that resembled reactive gemistocytes, which led to the diagnosis of gemistocytic astrocytoma. Eight percent of the neoplastic cells were positive for p53 reactivity. Among the reactive population, 62% had mainly cytoplasmic expression (Fig. 1D), 26% had nuclear and cytoplasmic expression (Fig. 1E), and 12% had a mainly nuclear expression (Fig. 1F). Only 1 glial cell in the gray matter of 1 of the 4 sections of normal spinal cord showed cytoplasmic and nuclear staining for p53. No positivity was observed for EGFR in the neoplastic tissue.
In veterinary medicine, genetic characterization of astrocytic tumors is uncommon, and to the authors' knowledge, investigation of p53 and EGFR expression has not previously been performed in feline astrocytomas. The absence of EGFR expression in this neoplasm and the very low p53 protein expression observed in the normal spinal cord were consistent with previous reports of canine 13 and human gemistocytic astrocytomas 1 and p53 protein expression in normal human brains, 4 respectively.
In humans, wild-type p53 protein has a half-life that ranges from 5 to 20 min, whereas the mutated protein has an extended half-life of 5 to 24 hr. 15 Hence, increased p53 staining is an indicator of overexpression of this protein and may indicate genetic mutation. The overexpression of the p53 protein has been considered significant by several investigators when the mean percentage of positive nuclei in the neoplasm is higher than 10%. 6,13,17 Overexpression of the p53 protein has been reported 1,6 in 50–75% of human gemistocytic astrocytomas and has been shown in 1 of 2 tested canine gemistocytic astrocytomas. 13 However, nuclear expression of p53 is not the only pattern observed in astrocytic neoplasms, and it is not necessarily associated with mutation in the protein. 11 The pattern of expression of p53 protein in human gemistocytic glioblastomas included cytoplasmic, nuclear, or the absence of p53 expression, and the mutated p53 protein seems to accumulate preferentially in the nucleus of neoplastic cells of secondary glioblastomas that evolve from lower-grade astrocytomas. 11 Moreover, in humans, glioblastomas have shown a positive correlation between expression of vimentin and GFAP and cytoplasmic accumulation of wild-type p53. 11 Thus, it was suggested that the p53 protein could be sequestered in the cytoplasm of neoplastic cells that express vimentin and eventually GFAP, which would inhibit the translocation of the protein in the nucleus and activation of antineoplastic transcription factors. 11 In the present case, overexpression of the p53 protein because of possible mutation cannot be compared with what was reported in other studies, because the mean percentage of positive cells was lower than 10%. However, the results indicate that, when compared with the normal spinal cord, the neoplasm is associated with a different biological behavior of the p53 protein. The pattern of expression of the p53 protein observed in the neoplasm in the current study is similar to what was reported in human gemistocytic glioblastomas 11 and suggests that p53 may be involved in the neoplastic process of feline astrocytomas. Whether the expression of the p53 protein in the present case is the result of accumulation of the wild-type protein with ineffective antineoplastic activity or because of a mutation of the protein, it cannot be ultimately assessed without further studies on the protein encoding gene sequence.
Acknowledgements. The authors thank Dr. Andy Ambrus and Ms. Rosemary Vollmar for immunohistochemistry and histology support.
Footnotes
a.
Mach 2 Polymer Detection, Biocare Medical, Concord, CA.
b.
Dako North America Inc., Carpinteria, CA.
c.
Santa Cruz Biotechnology Inc., Santa Cruz, CA.
d.
BioGenex Laboratories Inc., San Ramon, CA.
