Abstract
The potential use of total sialic acid (TSA) as an acute-phase biomarker in pigs was investigated through clinical and analytic validation of an automated commercial enzymatic-colorimetric assay. Intra- and interassay precision, accuracy, and detection limit were determined for the analytical validation of the method. The intra- and interassay coefficients of variation (%CV) were 2.27% and 3.99%, respectively, whereas the linearity under dilution calculation resulted in a coefficients of correlation of
The acute-phase response refers to a nonspecific and complex reaction of an animal that occurs shortly after a tissue injury. The origin of this reaction can be attributable to infectious, immunologic, neoplastic, traumatic, or other causes, and its purpose is to restore homeostasis and to remove the cause of its disturbance. This response, among other systemic effects, includes the change in serum concentrations of the so-called acute-phase proteins (APP). 11 Testing for these APPs is thought to be a suitable tool for monitoring the health and well-being of pigs. 11 At the herd level, APP might be useful for determining in which part of the production system a disturbance is taking place, 10 and it may also be applied to measure the efficacy of antibiotic treatment. 9
Most APPs are glycoproteins that contain sialic acid. 13 Sialic acid is a 9-carbon monosaccharide mainly attached to the oligosaccharide side-chains of many glycoprotein and glycolipids. 13 In humans, it is known that serum-sialic-acid concentration rises during the acute-phase reaction, because of an increase in serum concentration of sialic-acid-carrying APPs, as well as a higher degree of sialylation of these proteins. 2,13 Hence, sialic acid is considered an acute-phase marker in human medicine. 1 The increase in APP in humans accounts for about 70% of circulating total sialic acid (TSA) concentrations during an acute-phase response. Thus, measuring TSA provides an integrated measure of the inflammatory response, which is less prone to day-to-day variability than individual markers. Based on this fact, it has been suggested that a single measurement of serum TSA may be the most useful estimate of an individual's habitual inflammatory status. 1 In addition to this role as an inflammation marker, TSA levels have been found to increase in humans during various types of cancer, chronic renal failure, cardiovascular diseases, and diabetes. 13 In veterinary medicine, several studies carried out on this marker in cattle, dogs, and horses during inflammatory or neoplastic disorders showed that sialic acid can be used as an acute-phase marker in these species. 3,8,12,15,16 However, to the authors' knowledge, sialic acid has not been evaluated in swine as an acute-phase marker. The purpose of the current study was to evaluate the possible use of serum TSA as an acute-phase marker in pigs by the analytical validation of an automated commercially available method, as well as the estimation of the ability of the assay to differentiate between clinically healthy pigs and pigs with different diseases.
The study sample of animals consisted of 39 conventional Duroc x (Landrace x Large White) pigs from a finishing unit of 1,800 animals. The farm was seropositive to
The group of healthy animals was composed of 14 pigs from the same fattening unit destined for slaughter, aged 27 weeks, seronegative for PRRSV, PCV-2,
The group of diseased animals was composed of 25 pigs aged 27 weeks of age that exhibited various severe clinical signs (i.e., acute dyspnea, cough, fever, lameness, tail bites, and cachexia) and a pronounced weakness, which prohibited shipment to the abattoir; therefore, they were slaughtered at the farm. Blood samples were collected before slaughter by jugular venipuncture by using a single-use blood collection system. b Serum was obtained and preserved as described in the preceding paragraph. After slaughter, postmortem and histologic examinations were carried out. These examinations revealed that all 25 pigs had pulmonary lesions (suppurative/fibrinous bronchopneumonia, embolic/bronchointerstitial pneumonia, or chronic pleurisy), and that 13 of these animals also had other multiorgan lesions (ileitis, polyserositis, tail bites, or chronic arthritis).
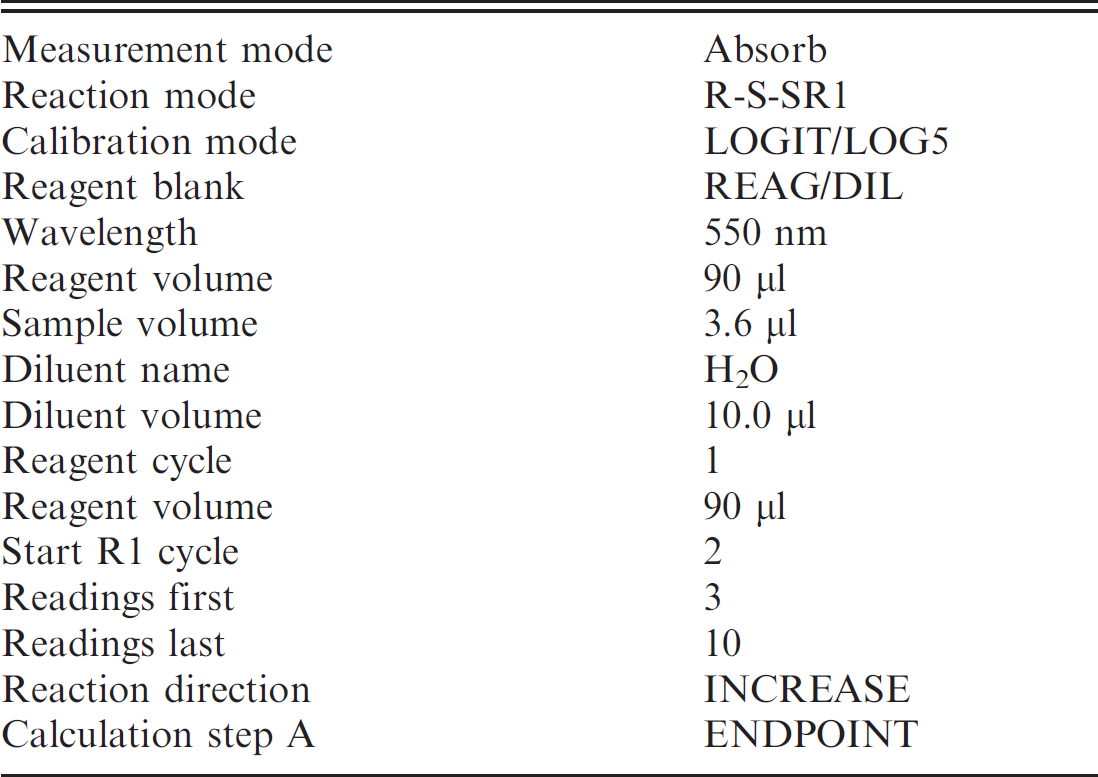
Settings for the Cobas Mira® Plus analyzer for determining porcine serum total sialic acid concentration.
The TSA concentrations were determined by using the sialic-acid test kit, c a colorimetric assay for enzymatic measurement of sialic acid in serum samples. The assay starts with the release of sialic-acid molecules bound to the sample glycoconjugates; this results in free sialic acid, which, after several enzymatic steps, is converted into a red chromophore. The absorbance of this chromophore is measured at 550 nm and is then related to the sample's original sialic-acid concentration, with a calibration curve established by using the standard serum included in the kit.
Because the sialic-acid kit was originally designed for manual use, it was necessary to adapt the assay to a Cobas Mira Plus Analyzer. d The adaptation was performed as previously described for canine serum samples, 16 with slight modifications. Settings for the Cobas Mira Plus are summarized in Table 1. This automation has several advantages when compared with the manual method, such as higher throughput, use of a lower sample volume per determination, and a significant decrease in the volume of reagents used, which leads to a reduction in the cost of the test. 4
Analytical validation of the sialic-acid test was carried out by using 6 porcine serum samples: 3 samples were from healthy animals (samples 1, 2, and 3), which were expected to have low sialic-acid content, and 3 acute-phase serum samples were from diseased animals (samples 4, 5, and 6), which were expected to have high sialic-acid content. Precision was expressed as a coefficient of variation (CV). Intra-assay CVs were determined by analyzing the 6 samples 5 times in a single analytical series. Interassay CVs were determined by analyzing duplicates of the 6 samples on 3 different successive days. Samples were frozen in aliquots, and only vials needed for each analytical run were thawed to prevent variation from repetitive freeze-thaw cycles. The accuracy of the assay was evaluated indirectly by the linearity under the dilution method. For this study, 2 samples with known amounts of TSA were serially diluted (50%,25%, 12.5%, and 6.25%) with distilled water, and the sialic-acid content was determined. The obtained concentration results were compared with the expected ones by a linear regression analysis, and the ability of the method to measure sialic acid in a proportional and linear way was established. 7 To check the minimum measure of TSA that can be detected by the assay with reasonable certainty, the analytical limit of detection (DL) was determined. To do this, the mean and standard deviation (SD) for the concentration of sialic acid of 13 replicates of zero standard (distilled water) were calculated; DL was determined as 3 SDs plus the mean value for the zero control samples. e
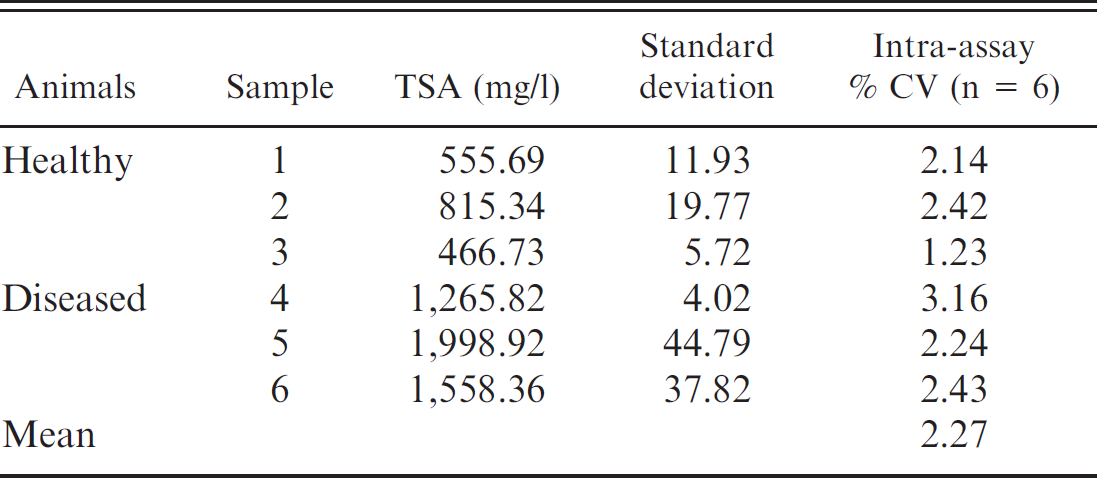
Intra-assay coefficients of variation (CV) of determinations of total sialic acid (TSA) in porcine serum samples with low (1, 2, and 3) and high (4, 5, and 6) TSA content, and intra-assay CVs mean.
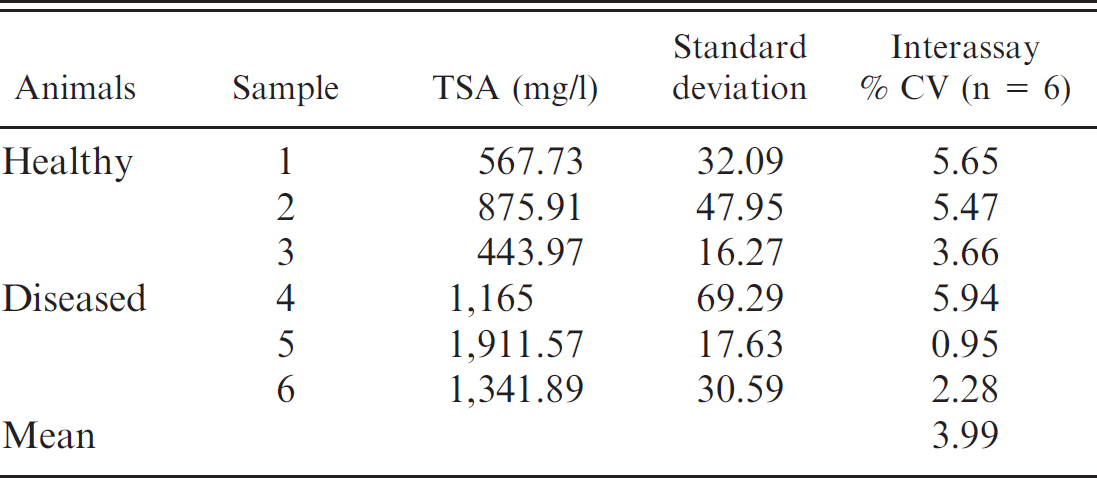
Interassay coefficients of variation (CV) of determinations of total sialic acid (TSA) in porcine serum samples with low (1, 2, and 3) and high (4, 5, and 6) TSA content, and interassay CVs mean.
Serum TSA concentrations from healthy and diseased animals described above were determined and compared. For clinical validation, all results were evaluated for normality of distribution by using a Kolmogorov—Smirnov test, and variance homogeneity was tested by a Levene test. When it was determined that the data did not follow a normal distribution (
Mean values, SDs, and precision results obtained in the analytical study are shown in Tables 2 and 3. The intra- and interassay CVs were lower than 6% and were similar to those reported in humans 4,6 and dogs. 16 The imprecision of this assay is lower than that of assays commercially available for the determination of other acute-phase markers in pigs, such as pig major acute protein, serum amyloid A, and C reactive protein, which ranged between 6.7% and 37.5% for the intra-assay CVs and between 9.5% and 39.4% for the interassay CVs. 14 The main reason for these differences is that most of the assays used to determine APPs in pigs are manual enzyme-linked immunosorbent assays that involve manual sample dilution, which can be a source of imprecision.
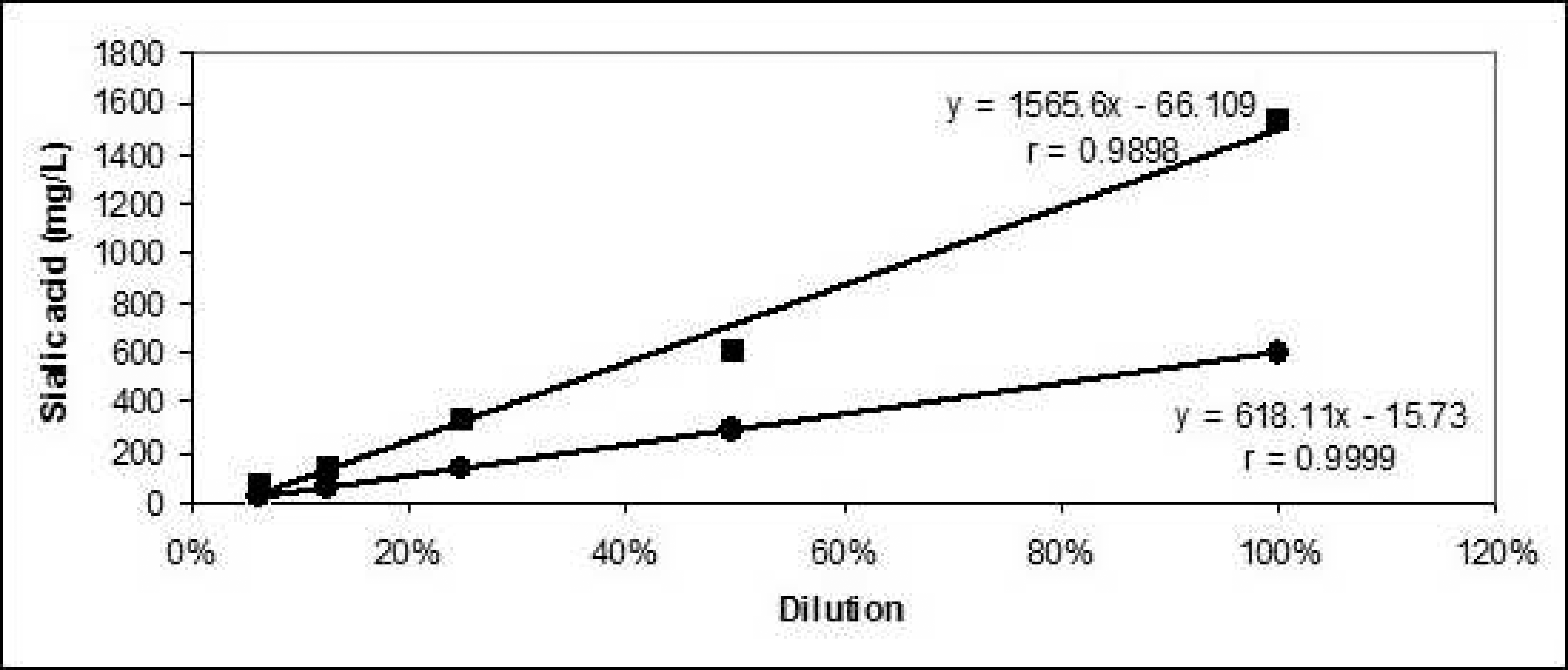
Linearity under dilution of porcine serum samples with known concentrations of total sialic acid.
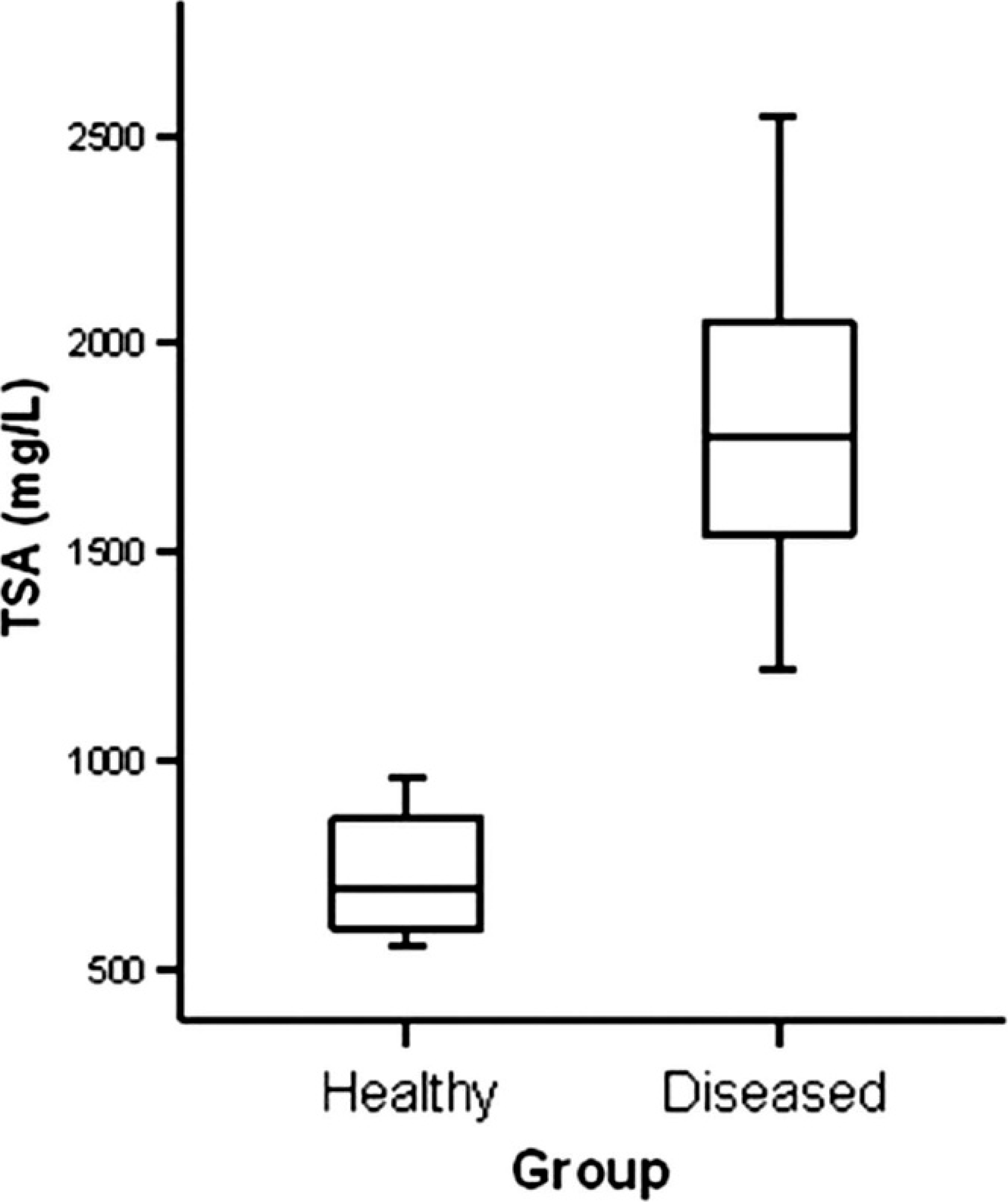
Total sialic acid (TSA) concentrations in serum of healthy (n = 14) and diseased (n = 25) pigs. The plots show the median (line within box), 25th and 75th percentiles (box), and 5th and 95th percentiles (bars).
As can be seen in Figure 1, the analysis of serial dilutions of a known amount of TSA serum samples to check the linearity under dilution of the assay resulted in linear regression equations with correlation coefficients higher than 0.99, which means that the accuracy of the automated method is acceptable. The analytical DL of the method was 6.07 mg/l, slightly lower than the previously reported ones in humans (12 mg/l 6 and 18.55 mg/l 4 ). This discrepancy could be attributable to differences in the methodology and in the DL calculation. Furthermore, the DL is far below the lowest TSA concentrations found in healthy pigs (466.73 mg/l), which demonstrates that the assay is adequate to determine any TSA concentration in porcine serum samples.
Diseased animals showed significantly higher concentrations of serum TSA (median: 1,779.16 mg/l) than healthy animals (median: 692.78 mg/l;
Footnotes
a.
Anitec AB, Malmö, Sweden.
b.
Vacutainer®, BD SA, Madrid, Spain.
c.
Roche Diagnostics GmbH, Mannheim, Germany.
d.
Cobas Mira® Plus Analyzer, Horiba ABX, Montpellier, France.
f.
Microsoft® Excel, Microsoft Corp., Silicon Valley, CA.
g.
SPSS software, version 11.5, SPSS Inc., Chicago, IL.
