Abstract
The current study demonstrates the ability of an indirect enzyme-linked immunosorbent assay (iELISA) to detect antibodies against Theiler's murine encephalomyelitis virus in mice colonies. The antigen was produced from infected baby hamster kidney (BHK)-21 cells and treated with 1% Nonidet P40 in saline buffer. Control antigen was prepared following the same procedure using uninfected BHK-21 cells. The optimal antigen and serum dilutions were established. The reaction was revealed using an anti-mouse-horseradish peroxidase conjugate and 2,2′-Azino-bis(3-ethylbenzthiazoline-6-sulfonic acid). Optimized iELISA was validated by detection of antibodies in known positive and negative serum samples before testing the samples of unknown status. Performance of the iELISA was compared with the indirect fluorescent antibody test, and the cutoff value was determined by receiver operating curve. Indirect ELISA showed 100% sensitivity, 99.38% specificity, and 97.78% predictive positive value. The antigen used is easy to produce, and no special equipment is required. The iELISA developed is simple and provides a rapid and less costly tool for diagnosis and research.
Theiler's murine encephalomyelitis virus (TMEV), an enteric pathogen of mice that belongs to the Cardiovirus genus in the family Picornaviridae, is a common cause of asymptomatic enteric infections in mice housed in non-barrier animal colonies. 1 In addition, TMEV spreads to the central nervous system (CNS) to cause poliomyelitis (flaccid paralysis) and, more rarely, encephalitis. 11 Theiler's murine encephalomyelitis virus can be divided into 2 groups on the basis of biological activities and the disease produced after intracerebral inoculation of mice. The first group consists of 2 highly virulent strains (GDVII and FA), which produce an acute fatal encephalitis in adult mice.
Members of the Theiler's original group are less virulent and cause CNS disease in the form of poliomyelitis followed by a chronic inflammatory demyelinating disease, which is due to viral persistence. Worldwide spreading is indicated. 2,8 This demyelinating disease has immune parameters and histopathology similar to those of chronic progressive multiple sclerosis (MS) and thus is extensively studied as an infectious model for MS. 4,6,10 The 2 most commonly used serological tests for diagnosis of TMEV-infected mice are the indirect fluorescent antibody test (IFAT) and the enzyme-linked immunosorbent assay (ELISA). The IFAT is the test of choice for detection of infection because it is rapid, simple to perform, and can be used for a small sample size. The ELISA has now been recognized as an excellent alternative to the conventional IFAT, one with high sensitivity and specificity. Other recognized advantages of the ELISA are the simple and automated procedures. 12 Some ELISA procedures employing partially purified virus or synthetic peptides have been used for the detection of TMEV, but they require special technology for antigen (Ag) production. 5,7 In the current report, a simple indirect ELISA (iELISA) performed with a solubilized Ag was developed to analyze a large sample size and to use it for screening mice colonies.
For Ag preparation, the GDVII strain of TMEV was propagated on baby hamster kidney (BHK)-21 cells grown in Eagle minimum essential medium supplemented with 10% fetal calf serum, 0.3 mg/ml of glutamine, 200 IU/ml of penicillin, 0.5 mg/ml of streptomycin, and 10% tryptose phosphate broth. The infected cultures were harvested when 90% of the cells exhibited cytopathic effect, clarified by centrifugation, and were washed twice with phosphate-buffered saline (PBS). One percent of Nonidet P40 a in Tris (0.01 mol) and ethylenediamine tetra-acetic acid (EDTA; 0.001 mol, pH 7.4) saline buffer was used to lyse and solubilize the pellet of TMEV-infected cells at 1:100 of original volume. The lysate was centrifuged at 8,000 x g for 30 min, and the supernatant was used as a source of Ag for the iELISA. Control Ag was prepared following the same procedure, but with uninfected BHK-21 cells. The optimal Ag dilution was established by titrating an Ag dilution series against a varying dilution of a reference positive serum. The optimal serum dilution was chosen based on the lowest dilution of the positive sera tested with control Ag, which showed optical density (OD) values of less or equal to those presented by negative reference serum tested with viral Ag at the same concentration. Plate coating was carried out using 0.1 ml of the predetermined Ag dilution (1:1,000) in carbonate/bicarbonate buffer (0.05 mol, pH 9.6) in all wells of flat-bottomed, polystyrene microtiter plates (protein concentration, 0.5 μg/well). The plates were incubated at 4°C overnight in a humid chamber and then blocked with 100 μl of blocking buffer (0.05% Tween-20 in PBS [PBST] containing 5% skim milk powder). After 1 hr of incubation at 37°C, the wells were washed 5 times (for 5 min each wash) with PBST. Sera diluted 1:40 in blocking buffer were added to a 50-μl volume and incubated for 1 hr at 37°C. After repeating the 5 washes for 5 min with PBST, the antimouse-horseradish peroxidase conjugate diluted in blocking buffer (1:1,000) was added (50 μl/well) and incubated for 1 hr at 37°C. The plates were washed as previously described, and substrate solution containing 0.1 mol of citric acid, 0.2 mol of sodium phosphate, 0.003% H2O2, and 0.3 mg/ml of 2,2′-Azino-bis(3-ethylbenzthiazoline-6-sulfonic acid) a was added in each well in 0.1-ml aliquots. The color reaction developed at 20 min and was read on a microplate spectrophotometer. b To minimize day-to-day variation, all serum samples were tested in duplicate on 2 different days, and the results were expressed as the average of 4 OD values. Optimized iELISA was validated by detection of antibody (Ab) in known positive (experimentally infected mice: n = 20) and negative (reference specific pathogen-free: n = 20) serum samples (based on IFAT status) before testing the samples of unknown status (n = 208) provided by 10 conventional facilities.
Comparison of results obtained from 208 mice sera of conventional facilities analyzed by indirect fluorescent antibody test (IFAT) and indirect enzyme-linked immunosorbent assay (iELISA).*
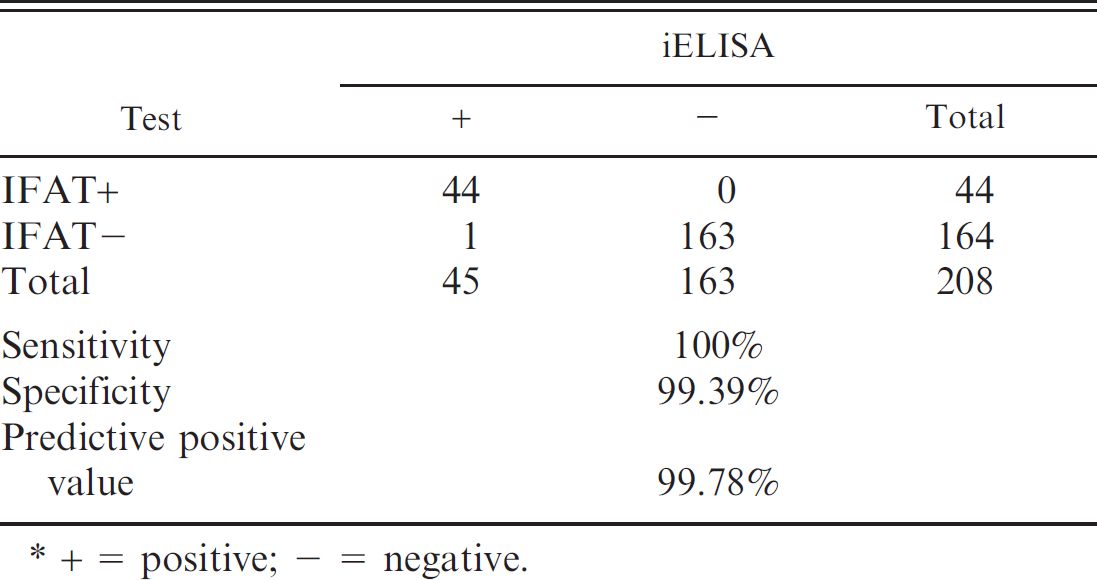
+ 5 positive; − = negative.
The performance of the developed iELISA was compared with that of IFAT. The IFAT was developed with infected cell suspensions dropped on spot slides, fixed, and stored at −20°C until used. Test sera were overlaid onto the spots, incubated for 30 min at 37°C, and washed off with PBS. The anti-mouse antiserum (fluorescein isothiocyanate-conjugated anti-immunoglobulin G) c was added onto the spots, incubated for 30 min at 37°C, and positive fluorescence were observed.
The performance of iELISA in terms of relative sensitivity and specificity was compared to that of the IFAT, and the cutoff value was determined by receiver operating curve. A 2-sided contingency table was constructed with the obtained results. Indirect ELISA showed 100% sensitivity, 99.38% specificity, and 97.78% predictive positive value. All samples with OD values >0.115 were considered positive (Table 1). The 21.15% of the sera analyzed were found to be positive.
Routine screening is needed in all mice colonies used for research, and the IFAT was used for several years with satisfactory results. However, when it is necessary to analyze a large number of samples, the IFAT is more laborious, and subjective results can be present. The present study demonstrates the ability of an iELISA to detect TMEV Ab in mice colonies. The iELISA used in the current study was sensitive, specific, precise, rapid, and easily automated and could represent a good alternative assay for detection of TMEV Ab in routine serodiagnosis of mice colonies. A recent study concerning MS refers to the use of an antigen for ELISA prepared with partially purified virus Theiler. 9 This could also be considered a good alternative, but this method requires a higher centrifugation speed to concentrate virus particles. However, the Ag production method developed in the present study and previously used for another veterinary assay 3 is simple and easily performed. It did not require special equipment and provides a rapid and less costly tool for diagnosis of and research on TMEV. An important finding that emerged from the results of the present study was that the Argentine mice colonies analyzed were found to be seropositive. These data have 2 implications: 1) the virus is circulating in conventional facilities; and 2) the virus could interfere with research that involves the CNS. In addition, data indicates that a complete prevalence study of TMEV Ab in mice colonies of Argentina should be performed.
Acknowledgements. C. M. Galosi is a member of the Scientific Research Commission, Buenos Aires, Argentina. This study was supported by grants from the Department for Science and Technology at the National University of La Plata and the Scientific Research Commission (CIC), Buenos Aires, Argentina. The authors would like to acknowledge the professional assistance of Prof. Marcelo Pecoraro (Department of Virology, Faculty of Veterinary Sciences, National University of La Plata).
Footnotes
a.
Sigma-Aldrich, St. Louis, MO.
b.
Titertek Multiskan Plus, EFLAB, Helsinki, Finland.
c.
Zymed Laboratories Inc., South San Francisco, CA.
