Abstract
Bovine viral diarrhea (BVD) viruses are pestiviruses that have been isolated from domestic and wild ruminants. There is serologic evidence of pestiviral infection in more than 40 species of free-range and captive mammals. Vertical transmission can produce persistently infected animals that are immunotolerant to the infecting strain of Bovine viral diarrhea virus (BVDV) and shed virus throughout their lives. Seven species (white-tailed deer, mouse deer, eland, domestic cattle, alpaca, sheep, and pigs) have been definitively identified as persistently infected with BVDV. This study provides serological, molecular, immunohistochemical, and histological evidence for BVDV infection in 2 captive mountain goats from a zoological park in Idaho. The study was triggered by isolation of BVDV from tissues and immunohistochemical identification of viral antigen within lesions of a 7-month-old male mountain goat (goat 1). Blood was collected from other mountain goats and white-tailed and mule deer on the premises for BVDV serum neutralization, viral isolation, and reverse transcription polymerase chain reaction. One 3-month-old mountain goat (goat 2) was antibody negative and BVDV positive in serum samples collected 3 months apart. This goat subsequently died, and though still antibody negative, BVDV was isolated from tissues and identified by immunohistochemistry within lesions. Sequencing and phylogenetic analysis identified the isolates as BVDV-2. These findings provide evidence of persistent infection in a mountain goat, underscoring the need for pestivirus control strategies for wild ruminants in zoological collections.
Introduction
Pestiviruses within the family Flaviviridae have been historically classified into 4 main phylogenetic groups: Bovine viral diarrhea virus 1 (BVDV-1), Bovine viral diarrhea virus 2 (BVDV-2), Classical swine fever virus (CSFV), and Border disease virus (BDV) of sheep. 22 Affected species are primarily within the order Artiodactyla and include Cervidae, Antilocapridae, and Bovidae. Specific antibody has been detected in more than 40 species of free-range and captive mammals. 13 , 45
While serologic surveys indicate that exposure of wild ruminants to BVDV is common, 18 , 41 , 44 isolation of the virus from these species is only rarely reported. Specifically, BVDV has been isolated from many artiodactylids including mule deer (Odocoileus hemionus), 45 white-tailed deer (Odocoileus virginianus), 31 Japanese serow (Capricornis crispus), 19 Canadian bison (Bison bison bison), 12 water buffalo (Bubalis bubalis), 10 roe deer (Capreolus capreolus), 3 mouse deer (Tragulus javanicus), red deer (Cervus elaphus), 27 bongo (Tragelaphus eurycerus), 3 eland (Taurotragus oryx), 48 wildebeest (Connochaetes sp.), 13 nilgai (Boselaphus tragocamelus), 13 axis deer (Axis axis), 13 and barasingha deer (Cervus duvaucelii). 13 In addition, BVDV has been isolated from alpacas, 25 sheep, and goats following natural infections. 26,33,37
Because BVDV infections result in significant economic losses in beef cattle, this virus has been the subject of intense research. Two viral genotypes, BVDV-1 and BVDV-2, have been identified, and 2 biotypes are described according to their effects in cell culture (cytopathic and noncytopathic). Horizontal transmission can produce an array of clinical presentations, including subclinical infections, immunosuppression, hemorrhagic syndrome, secondary infections, peracute death, reproductive disease, and respiratory disease including pneumonia. 7 , 45 Vertical transmission in domestic cattle can cause infertility, abortion, stillbirths, weak calves, and persistent infection.
Persistent infection of the bovine fetus occurs when an immunologically naive cow is infected with a noncytopathic viral strain before 125 days of gestation. The fetus develops immunotolerance to the infecting virus, and these persistently infected (PI) calves shed virus throughout their lives. 7 Consequently, PI calves are significant reservoirs for viral transmission. Persistently infected calves do not develop neutralizing antibody specific to the virus they carry; these lifelong infections may be associated with immunosuppression, runting, and secondary infections. 32 Superinfection of PI animals with a cytopathic strain may trigger mucosal disease (MD). In a herd, MD is characterized by low morbidity and high case fatality in cattle between the ages of 6 months and 2 years, and typical gross lesions include extensive mucosal ulceration primarily within the gastrointestinal tract. 38
Both acutely and persistently infected cattle may introduce BVDV into herds, and control strategies should attempt to identify and address both modes of infection. 32 Currently, there are only a few reports describing acute disease in wild ruminants, specifically in white-tailed deer. 35 , 46 In addition to cattle, persistent BVDV infection has been definitively identified in domestic sheep, 37 alpaca, 8 , 25 and pigs. 39 Although there are reports of definitive natural and experimental persistent infection in wild ruminants including mouse deer, 42 , 44 white-tailed deer, 29 and eland, 48 there are suspected persistent infections in other artiodactylids. Histological lesions have been described in experimentally induced, PI white-tailed deer fawns, and antigen was identified within many tissues and cell types. 14 The current study provides serological, molecular, immunohistochemical, and histological evidence for persistent infection with BVDV-2 in captive mountain goats (Oreamnos americanus). These findings underscore the need for pestivirus control strategies for wild ruminants in high-density, open populations.
Materials and methods
Animals and histopathological and immunohistochemical analysis
The current study was initiated by submission of field necropsy tissues from a 7-month-old, intact, male mountain goat (goat 1: 1831, WADDL 05–320) to the Washington Animal Disease Diagnostic Laboratory (WADDL; Pullman, WA) on January 12, 2005. This goat had been introduced with 3 other mountain goats (including goat 2, described below) during the previous fall into a zoological collection in eastern Idaho. All 4 mountain goats at this facility were obtained at the same time from the same source in Montana. The animals housed in contact or immediately adjacent to these mountain goats included 3 white-tailed deer, 1 mule deer, 10 elk, 2 bison, 1 domestic sheep, and 3 domestic goats. The mountain goat (goat 1) had not been growing adequately and was found dead on January 10, 2005. On the same day, formalin-fixed (lung, heart, kidney, liver, and small intestines) and fresh (lung, liver, spleen, kidney, and small intestine) tissues were collected by the submitting veterinarian. On May 14, 2005, a 6-month-old, intact, male mountain goat (goat 2: 1830, WADDL 05–5440, 05–5265, 05–1761) from the same facility was found dead and was refrigerated for 3 days before collection and submission of formalin-fixed (lungs, heart, liver, spleen, and kidney) and fresh (lung, liver, spleen, kidney, intestine, and mesenteric lymph node) tissues to WADDL for evaluation. No skin samples were submitted for evaluation.
Formalin-fixed tissues were embedded in paraffin, sectioned, and stained with hematoxylin and eosin. Select 5-μm sections of tissues were mounted on charged slides for indirect immunohistochemical chromogen staining using the avidin-biotin peroxidase complex method for all antibodies. Samples were deparaffinized and rehydrated, and antigen retrieval was performed using 0.06% pronase a for 10 min. Slides were incubated with 5% normal goat serum for 10 min at room temperature as a protein block and incubated with diluted primary monoclonal antibodies (BVDV monoclonal antibody [mAb] 15c-5, IgG2b) b against the highly conserved BVDV gp 43 antigen 1 for 30 min at room temperature. Control slides were stained concurrently and included bovine tissue sections known to be infected with BVDV (positive control), serial sections lacking primary antibody (negative control), and serial sections using a primary antibody specific for scrapie prion protein (IgG2b mAb 89/193.1 as an isotype control). c Slides were rinsed and then labeled with a streptavidin detection kit using undiluted, biotinylated secondary antibodies d for 30 min at room temperature, and immunostaining was visualized with an amino-ethyl carbazole substrate chromogen e (2 × 4 min at room temperature). Following washes, slides were counterstained with hematoxylin, dehydrated, and permanently mounted.
Etiological analysis
To identify potential causative agents, select necropsy tissues were submitted for virus isolation and bacterial cultures. Samples for viral isolation were processed as previously described. 15 Briefly, bovine turbinate (BT) cells free of BVDV were maintained in Minimal Essential Medium (MEM) f supplemented with 10% fetal bovine serum free of BVDV antibodies and BVDV with 100 IU/ml of penicillin, 100 IU/ml of streptomycin, 50 μg/ml of gentamicin, and 2.5 μg/ml of amphotericin B deoxycholate. The cells were monitored weekly for BVDV contamination by BVDV-specific, reverse transcription quantitative polymerase chain reaction (RT-qPCR) using a duplex, duallabeled hydrolysis probe g reaction for BVDV-1 and BVDV-2 as previously described. 4 Tissue samples were homogenized in MEM and centrifuged at 1,207 x g for 10 min, and the supernatant was inoculated onto BT cells seeded in 24-well plastic trays. Cells were monitored daily for cytopathic effect. Cells and supernatant from the 24-well plastic trays were passaged 3 times at 3-day intervals by freezing and thawing the cells and using the lysate to infect fresh cultures. Then the cultures were tested for the presence of BVDV by the BVDV-specific RT-qPCR.
Isolation of a pestivirus from goat 1 prompted a serosurvey of the ruminants in the collection. Intravenous whole blood (ethylenediamine tetra-acetic acid [EDTA]) and serum samples were collected over a 4-month time period from mountain goats, white-tail deer, and mule deer within the same pen and submitted for analysis. Whole blood was centrifuged at 839 × g for 20 min, the buffy coat was removed and added to 1 ml of MEM and shaken, and the dispersed white blood cells were inoculated as the tissue sample homogenate for virus isolation as described above and RT-qPCR. Serum was processed and frozen at −20°C until the BVDV (Singer strain, BVDV-1) serum neutralization microtiter assay was run (56°C for 30 min). 5 Fresh tissue samples (heart, lung, liver, spleen, kidney, and small intestine) submitted for bacteriology were cultured for aerobic growth.
Molecular analysis of the virus
RNA was extracted from EDTA blood samples using viral RNA purification kits. h Extracted RNA was stored briefly at 4°C and tested for BVDV using RT-qPCR. Although this duplex hydrolysis probe assay distinguishes between BVDV-1 and BVDV- 2 , 4 it is not able to discriminate between BVDV-2 and BDV (D. Bradway, personal communication, May 2005). For this reason, a separate RT-qPCR reaction was run, using recombinant Thermus thermophilus DNA polymerase i to amplify a highly conserved 290-bp portion of the 5′ untranslated region (5′UTR) of the BVDV genome, as previously described. 49 To discriminate between BVDV-2 and BDV, a longer sequence was obtained using the same RT-qPCR conditions with primers BVD372F (5′-GCTGTACATGGCACATGGAGT-3′) and BVD973R (5′-TGGGGCTATTTTCATCTTGC-3′) and following a method previously described. 49
To distinguish BVDV-2 from BDV for diagnostic purposes, amplicons were visualized on a 1.5% agarose gel j containing ethidium bromide, k excised using a sterile scalpel blade under ultraviolet illumination and purified using a DNA gel extraction spin column 1 according to the manufacturer's directions. Amplicon DNA was sequenced directly on both strands by Amplicon Express m using the dye terminator cycle-sequencing ready reaction kit, n with analysis on an ABI 373 DNA sequencer. o Forward and reverse sequences were aligned using the ClustalW algorithm. 40 Each sequence was compared to the nucleotide sequence database for similarity using BLASTn (nucleotide Basic Local Alignment Search Tool; http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). 30
Phylogenetic analysis
To identify the particular BVDV strains involved, RNA templates were prepared from isolated virus as described previously. 34 RT-qPCR was performed on the RNA templates amplifying a longer portion of the 5′UTR. 36 The 5′UTR sequences were derived by direct sequencing of amplification products. 34 All sequencing reactions were done in duplicate, and all sequences were confirmed by sequencing of both strands. Sequences were aligned and compared with type viruses from BVDV-1, BVDV-2, BDV, and CSFV species of the pestivirus genus using Align Plus p and MacDNASIS. q Phylogenetic trees were generated using the Higgins-Sharp alignment algorithm of Mac-DNASIS.
Results
Pathology (goat 1)
In goat 1, the tissues were mildly to moderately autolyzed, with the intestines being the most affected. The hepatic lobules were multifocally and randomly disrupted by small aggregates of degenerate and necrotic hepatocytes admixed with small numbers of macrophages and neutrophils. Within the small intestines, the apical villi were sloughed, and the remaining lamina propria was slightly expanded by scattered macrophages and neutrophils. No significant lesions were observed in the lungs, heart, and kidney. The following cells had cytoplasmic immunoreactivity when stained for BVDV antigen (Fig. 1): monocytes and endothelial cells within the hepatic sinusoids, kidneys, heart, and lamina propria of the small intestines; mesangial cells of the renal glomeruli; and cardiomyocytes. The necrotic cells within the liver and intestines did not have immunoreactivity for BVDV antigen. Bovine viral diarrhea virus was isolated from the lung and abdominal tissue pool. Mixed bacterial aerobes consistent with postmortem contamination or overgrowth were cultured from the lung and tissue pool. Campylobacter spp. were not visualized within Victoria blue-stained intestinal contents, and no Salmonella spp. were cultured from the intestinal contents.
Survey of herd mates
One mountain goat (goat 2) was negative for antibody against BVDV yet positive for BVDV by both virus isolation and RT-qPCR of repeated blood samples (Table 1). The 2 other mountain goats, 2 white-tailed deer, and 1 mule deer were positive for antibody against BVDV and negative for BVDV virus by both virus isolation and RT-qPCR (Table 1). The virus neutralization assay used for the serum samples from these cases targeted BVDV-1a rather than BVDV-2. Although this particular neutralization assay has cross-reactivity for BVDV-2, the reactivity is relatively higher within a genotype than between genotypes. 2 , 23 Therefore, the reported titers may be falsely depressed. The 4 mountain goats were newly purchased from the same source and introduced to the pen during October 2004. The mountain goats, white-tailed deer, and mule deer were housed together with a domestic lamb, pot-bellied pig, and 3 domestic goats in a petting pen on a drive-through wildlife preserve.
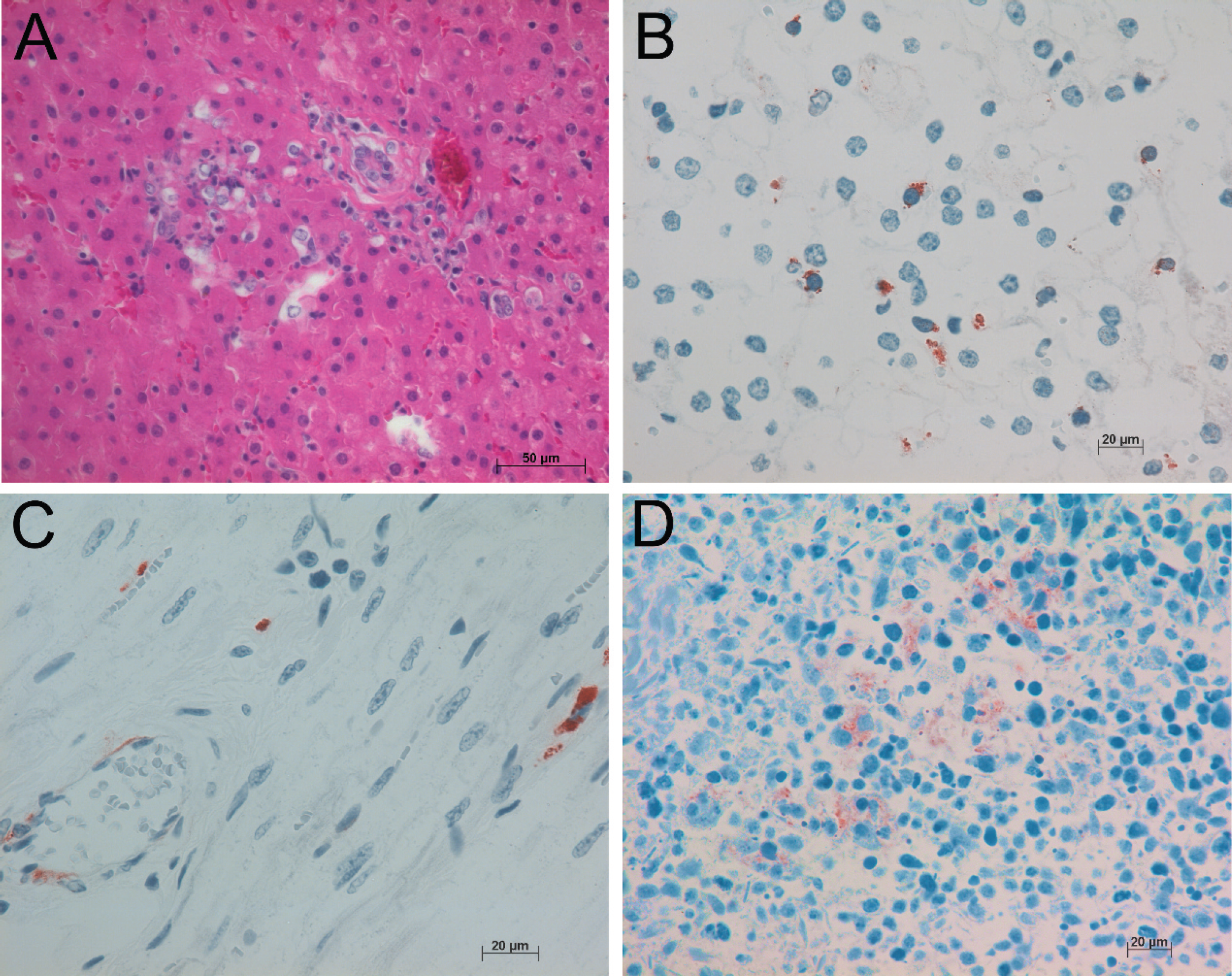
A, necrotizing hepatitis in goat 1. Hematoxylin and eosin.
Pathology (goat 2)
All necropsy tissues from goat 2 were moderately autolyzed. In the lungs, several bronchioles were severely distended and filled with large numbers of neutrophils admixed with fibrin. One blood vessel in the heart was surrounded by lymphocytes and plasma cells. Occasional aggregates of neutrophils were scattered throughout the liver. The renal interstitium was infiltrated by small numbers of lymphocytes and plasma cells. Scattered macrophages throughout the lung (Fig. 1) and spleen had intracytoplasmic immunoreactivity for BVDV antigen. Samples of lung and mesenteric lymph nodes were positive for BVDV via RT-qPCR and virus isolation. Bacterial cultures were positive for Corynebacterium pseudotuberculosis and mixed bacteria (consistent with postmortem overgrowth) from the lung. Cultures of intestinal contents for Salmonella spp. were negative.
Summary of serological and virological findings in exposed mountain goats, white-tailed deer, and mule deer.*
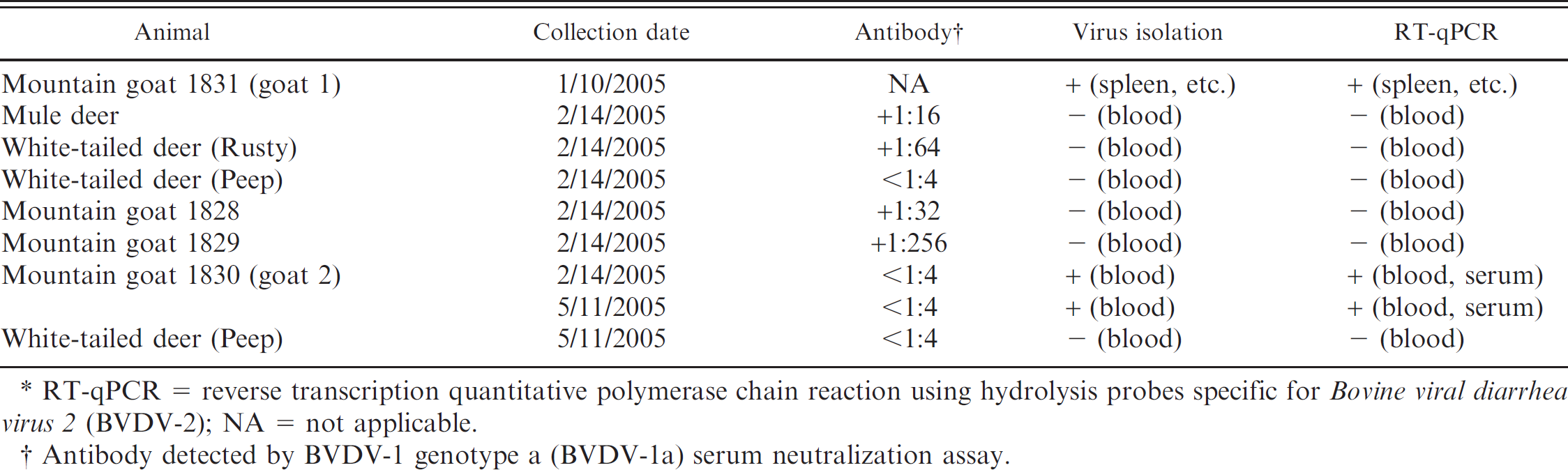
RT-qPCR = reverse transcription quantitative polymerase chain reaction using hydrolysis probes specific for Bovine viral diarrhea virus 2 (BVDV-2); NA = not applicable.
Antibody detected by BVDV-1 genotype a (BVDV-la) serum neutralization assay.
Summary of laboratory findings in mountain goats 1 and 2.*

IHC = immunohistochemistry for Bovine viral diarrhea virus (BVDV); EDTA = ethylenediamine tetra-acetic acid; NA = not applicable; RT-qPCR = reverse transcription quantitative polymerase chain reaction using hydrolysis probes specific for BVDV-1 and BVDV-2.
Molecular identification of BVDV and phylogenetics of the isolate
Table 1 compares the serologic and antemortem RT-qPCR whole-blood results over time to identify a PI animal (goat 2). The 2 mountain goats that succumbed to disease (goats 1 and 2) were positive for BVDV-2 by RT-qPCR (Table 2). Phylogenetic analysis based on the 5′UTR indicated that these isolates belonged to the BVDV-2 species within the genus Pestivirus (Fig. 2). In addition, the isolates obtained from goats 1 and 2 are genetically distinct with approximately 95.7% nucleotide identity.
Discussion
In the original mountain goat (goat 1), there is evidence of systemic infection with BVDV as indicated by viral isolation, RT-qPCR, sequencing, and immunohistochemistry. However, none of the BVDV antigen-positive cells are degenerate or necrotic. Since the bacteria isolated are most consistent with postmortem contamination or overgrowth, the underlying cause of enteritis is not determined. In addition, the pattern and distribution of hepatitis in goat 1 is suggestive of septicemia. Therefore, death may be due to enteritis and systemic bacterial infection, and it cannot be determined with certainty whether BVDV infection is fatal in goat 1. In goat 2, death is most likely due to corynebacterial pneumonia, and the role of BVDV infection is similarly unclear. Although BVDV infection is likely not the proximate cause of death in either goat, the presence of BVDV-infected cells without directly associated necrosis in multiple organs is suggestive of systemic infection possibly leading to immunosuppression, runting, increased susceptibility to secondary bacterial infection, and death.

Dendrogram based on the sequenced 5′UTRs from multiple isolates of Border disease virus (BDV) and Bovine viral diarrhea virus 1 (BVDV-1) and 2 (BVDV-2), including virus from goat 1 (postmortem tissue samples, 05–320) and goat 2 (antemortem serum samples, 05–1761 [02/2005] and 05–5265 [05/2005]).
Bovine viral diarrhea virus was isolated from goat 2 twice over the course of 3 months while the animal was consistently seronegative. The current standard for definitive determination of PI status in cattle requires 1 of 2 criteria: identification of virus in 2 separate serum or buffy coat samples with a minimum of 21 days between sample collections 16 , 21 and identification of BVDV antigen by immunohistochemistry on skin biopsy specimens (preferably repeated after 30 days). 17 , 20 , 25 , 28 Although the additional evidence of seronegativity over time could provide further evidence of persistent infection in goat 2, the sensitivity of the virus neutralization assay is compromised since BVDV-1a was used as the challenge virus. Nevertheless, goat 2 fully meets the criteria for persistent infection with BVDV since the virus was directly detected with both virus isolation and RT-qPCR from multiple samples collected over a 3-month period.
Since goat 1 lacked longitudinal evidence of prolonged BVDV-2 infection, it is not possible to definitively determine whether goat 1 was acutely or persistently infected. Previous studies in cattle have shown that immunoreactivity to BVDV in acutely infected animals is generally limited to lymphoid tissues and is mostly eliminated by 13 days post-infection; in contrast, immunoreactivity in PI animals is found in a variety of tissues and is not necessarily associated with lesions. 24 Although the history of poor weight gain and distribution of virus throughout the viscera suggest that goat 1 was also persistently infected, acutely infected animals occasionally may have wide distribution of the virus throughout tissues, including the skin. 9 , 20 Consequently, it is not possible to definitively determine whether goat 1 was persistently or acutely infected.
Both sequencing and phylogenetic analysis identified the isolates as BVDV-2, and, to date, BVDV-1 or BVDV-2 has not been reported in mountain goats. Nucleotide sequence relatedness is the most reliable criterion for differentiating pestivirus species. The variation between the isolates from goats 1 and 2 is greater than that typically seen between isolates from animals exposed to the same source. 36 While all 4 mountain goats were obtained from the same source in Montana, the identification of distinct isolates suggests multiple transmission sources, supporting the hypothesis that BVDV infection in these goats is not an isolated event.
Pestiviruses are not host specific, and all 4 recognized pestivirus species will replicate in domestic sheep and pigs. While BDV has been reported in cattle, 11 these infections are apparently rare, with the predominance of pestivirus isolates from cattle being BVDV-1 and BVDV-2 strains. There is currently no definitive evidence that BVDV spreads from free-living animals to domestic ruminants. 47 However, experimentally infected PI mouse deer can transmit BVDV to domestic cattle, 42 , 43 and PI cattle can transmit BVDV to domestic goats. 6 The original source and transmission path of BVDV was not determined in the present mountain goat study. However, the BVDV isolates from the mountain goats replicated in bovine cells in vitro, and this suggests that this BVDV strain would probably be able to circulate between mountain goats and domestic cattle. Furthermore, serology of collection cohorts also suggests that there may have been transmission between mountain goats and other species. Because mountain goats and cattle may share range, there is potential for transmission of BVDV between wild and domestic populations, and further studies examining transmission between these 2 species are warranted.
Both PI and acutely infected cattle contribute to the introduction and continued circulation of BVDV within a herd, and the results of the current study provide more evidence of similar persistence mechanisms in captive wild ruminants. The existence of PI mountain goats suggests that they could serve as a reservoir of BVDV in captive populations. In dense populations such as animal parks or zoos, PI animals may compromise breeding and conservation programs for endangered species. There is little evidence to suggest that clinical disease in wild herds is currently prevalent. However, as wild ruminant populations increasingly lose critical habitat, creating increased herd densities and increased contact with domestic ruminants, BVDV may pose a more significant threat. Consequently, biosecurity practices including vaccination protocols, herd screening, quarantine procedures, and testing of new introductions may be necessary to avoid transmission and persistence of BVDV within captive wild ruminants.
Acknowledgements
This work was supported by the Washington Animal Disease Diagnostic Laboratory and the Veterinary Microbiology and Pathology Department of the College of Veterinary Medicine at Washington State University in Pullman, Washington. The authors thank John Vanderschalie and Pam Robertson for laboratory assistance.
Footnotes
a.
Protease type 14 (P5147-5G), Roche Diagnostics GmBH, Mannheim, Germany.
b.
Testing Laboratories for Comparative Medicine, Diagnostic Laboratory, College of Veterinary Medicine, Cornell University, Ithaca, NY.
c.
Dr. Tim Baszler, Veterinary Microbiology and Pathology Department, Washington State University, Pullman, WA.
d.
Covance-Signet 32246 ultrastreptavidin detection kit, Covance Research Products Inc., Dedham, MA.
e.
Dako K3464 AEC Substrate Chromogen, Ready-to-use, Dako North America Inc., Carpinteria, CA.
f.
Mediatech Inc., Manassas, VA.
g.
TaqMan probes, Biosearch Technologies Inc., Novato, CA.
h.
QIAamp Viral RNA Mini Kit (#52904), Qiagen Inc., Valencia, CA.
i.
Applied Biosystems, Foster City, CA.
j.
Invitrogen Corp., Carlsbad, CA.
k.
Fischer Scientific Co., Fair Lawn, NJ.
l.
Bio-Rad Freeze N Squeeze kit, Bio-Rad Laboratories, Hercules, CA.
m.
Amplicon Express, Pullman, WA.
n.
Perkin Elmer Applied Biosystems Prism Dye Terminator Kit, Applied Biosystems, Foster City, CA.
o.
Applied Biosystems, Foster City, CA.
p.
Scientific & Educational Software, Cary, NC.
q.
Hitachi Software Engineering America Ltd., San Francisco, CA.
