Abstract
An 11-year-old, neutered, male Golden Retriever cross dog was euthanized following a history of recurrent pericardial effusions. At necropsy, blood-tinged pericardial and intrathoracic effusions were seen along with numerous firm to hard plaque-like masses that studded the epicardial, pericardial, mediastinal, and costal pleural surfaces. Within the right thorax, the lesions coalesced into a large mass that occupied most of the cavity. Histologically, the masses were composed of solid sheets and papillary aggregates of medium-sized polygonal cells that contained abundant vacuolated to clear cytoplasm. Some of the cytoplasmic vacuoles stained positive with oil red O. The stroma contained metaplastic trabeculae of woven and lamellar bone. Immunohistochemically, the neoplastic cells expressed vimentin, pancytokeratin, and S-100 protein. Transmission electron microscopy corroborated the presence of intracytoplasmic vacuoles and demonstrated prominent intercellular junctional complexes and apically located microvilli. These findings are consistent with a lipid-rich variant of mesothelioma. To the authors' knowledge, this is the first report of a lipid-rich mesothelioma in a dog.
Mesothelioma is a malignant neoplasm arising from mesothelial cells that line the peritoneal and pleural surfaces. Epithelioid, predominant fibrous and biphasic variants of mesothelioma have been reported. 10,11 The subtypes that have been described in dogs include cardiac mesothelioma with granular cell morphology, 1 deciduoid peritoneal mesothelioma, 12 epitheliod, 11,13 cystic peritoneal mesothelioma, 5 and sclerosing peritoneal mesothelioma. 6,7 Most canine mesotheliomas arise within the pleura or pericardium, but mesothelioma is an uncommon neoplasm in dogs. 14
A case of unusual lipid-rich variant mesothelioma has been reported in an 80-year-old man with history of shortness of breath and pleural effusions. 2 The histomorphologic description given by the authors of that report includes branching glands and papillary structures lined by plump epithelioid cells with abundant intracytoplasmic lipid vacuoles. The neoplastic cells were confirmed to be mesothelial in origin based on morphologic, immunohistochemical, and ultrastructural analyses. 2 The present report describes a lipid-rich pleural mesothelioma in a dog with history of persistent pericardial effusions. The diagnosis was based on the tumor location, microscopic appearance, immunophenotype, and ultrastructural characteristics.
An 11-year-old, neutered, male Golden Retriever cross dog presented with weakness, tachypnea, and episodes of collapse. A pericardial effusion was clinically suspected, and a pericardiocentesis performed after echocardiography failed to demonstrate an intrathoracic mass. Approximately 700 ml of hemorrhagic pericardial effusions was removed. Treatment at the time consisted of 25 mg of metoprolol twice daily for arrhythmia. The dog presented 13 weeks later with renewed pericardial effusion. Therapeutic pericardiocentesis was performed a second time and the dog was discharged. A thoracoscopic pericardectomy window was scheduled for the following week. The night before the planned surgical procedure, the dog presented again with shortness of breath. A mild pericardial effusion was present. A thoracoscopic pericardectomy was attempted the following day but this could not be safely performed as a result of extensive intrathoracic adhesions. A limited-approach thoracotomy was performed to create a pericardial window and to biopsy the pericardium; a thoracostomy tube was placed postoperatively. The dog recovered well from the procedure. Prior to removal of the thoracostomy tube, 50 mg/m 2 of cisplatin was infused into the pleural space as local chemotherapy. Following surgery, the dog presented multiple times for recurrent pleural effusion. A thoracostomy tube was placed for extraction of fluid, and administration of cisplatin continued. Euthanasia was performed 23 weeks following the initial presentation as a result of repeated accumulation of pleural fluid and the poor prognosis.
At necropsy, the pericardium and thoracic cavity were filled with approximately 300 ml and 3 liters of hemorrhagic fluid, respectively. A nodular, firm, gritty to hard, tan to gray mass measuring 18.0 cm x 7.0 cm x 2.0 cm filled the right thoracic cavity and primarily involved the mediastinal aspect of the pleura (Fig. 1). Numerous smaller nodules of similar consistency, ranging in size from 0.1 cm x 0.1 cm x 0.1 cm to 0.5 cm x 0.5 cm x 0.5 cm, studded the epicardial, pericardial, and costal pleural surfaces.
For the cytology performed on the pericardiocentesis sample, direct smears were made and stained with Wright-Giemsa stain. Representative samples of the masses were taken, fixed in 10% neutral buffered formalin, embedded in paraffin, and routinely processed according to accepted histologic technique. For light microscopy, 5-μm-thick sections were stained with hematoxylin and eosin and immunohistochemically stained for vimentin, a pancytokeratin, a and S-100 a protein. In addition, 5-μm-thick frozen sections of formalin-fixed tissue were stained with oil red O. For electron microscopy, the tissue was fixed in Trump fixative, postfixed in 1 % osmium tetroxide in a sodium cacodylate buffer, stained en block with 5% aqueous uranyl acetate, and embedded in Epon-812. One-micrometer-thick sections were cut and stained with toluidine blue and examined by light microscopy. Appropriate areas were cut into 50-nm to 70-nm thin sections, stained with uranyl acetate and lead citrate, and examined with a Philips EM 201 transmission electron microscope.
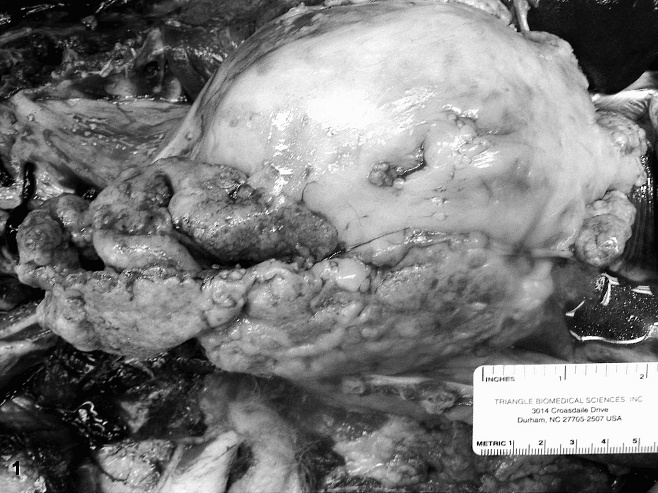
Thoracic mesothelioma; dog. A partially folded thoracic mass within the mediastinal pleura of the right side of the thoracic cavity.
Cytologic examination of the pericardial fluid revealed numerous erythrocytes along with large, round to polygonal, atypical cells that exhibited foamy cytoplasm. Possible cell types for the atypical cells included large epithelioid macrophages, reactive mesothelial cells, or neoplastic cells. The pericardial biopsy sample consisted of 3 sections of pericardium. These were thickened by mature hyalinized bundles of collagen with few scattered lymphocytes, plasma cells, and hemosiderin-laden macrophages. The mesothelium varied from one to several cell layers thick and focally formed papillary structures. Occasional cystic structures lined by the neoplastic cells extended into the collagenous stroma. The mesothelial cells were intermediate in size and contained either scant eosinophilic or abundant vacuolated cytoplasm. The nuclei were round to oval and hyperchromatic. Anisocytosis and anisokaryosis were moderate to marked. Mitotic figures (MFs) varied in number but rarely exceeded 1 MF per high-power field. The pleural and mediastinal masses were unencapsulated and composed of well-delineated aggregates of neoplastic cells. Centrally, the neoplastic cells were arranged in solid sheets and in papillary formations along the surfaces; the latter were associated with a stroma that contained numerous trabecular arrays of woven and lamellar bone (Fig. 2, inset). The neoplastic cells were polygonal in shape, measured approximately 15 μm in diameter, had distinct borders, and contained abundant rarefied to vacuolated clear cytoplasm (Fig. 2). Many of the vacuoles contained oil red O-positive droplets. The nuclei were round to oval, hyperchromatic, and often eccentrically located. Anisocytosis and anisokaryosis were limited. On average, 2 MFs per 10 high-power fields were identified.
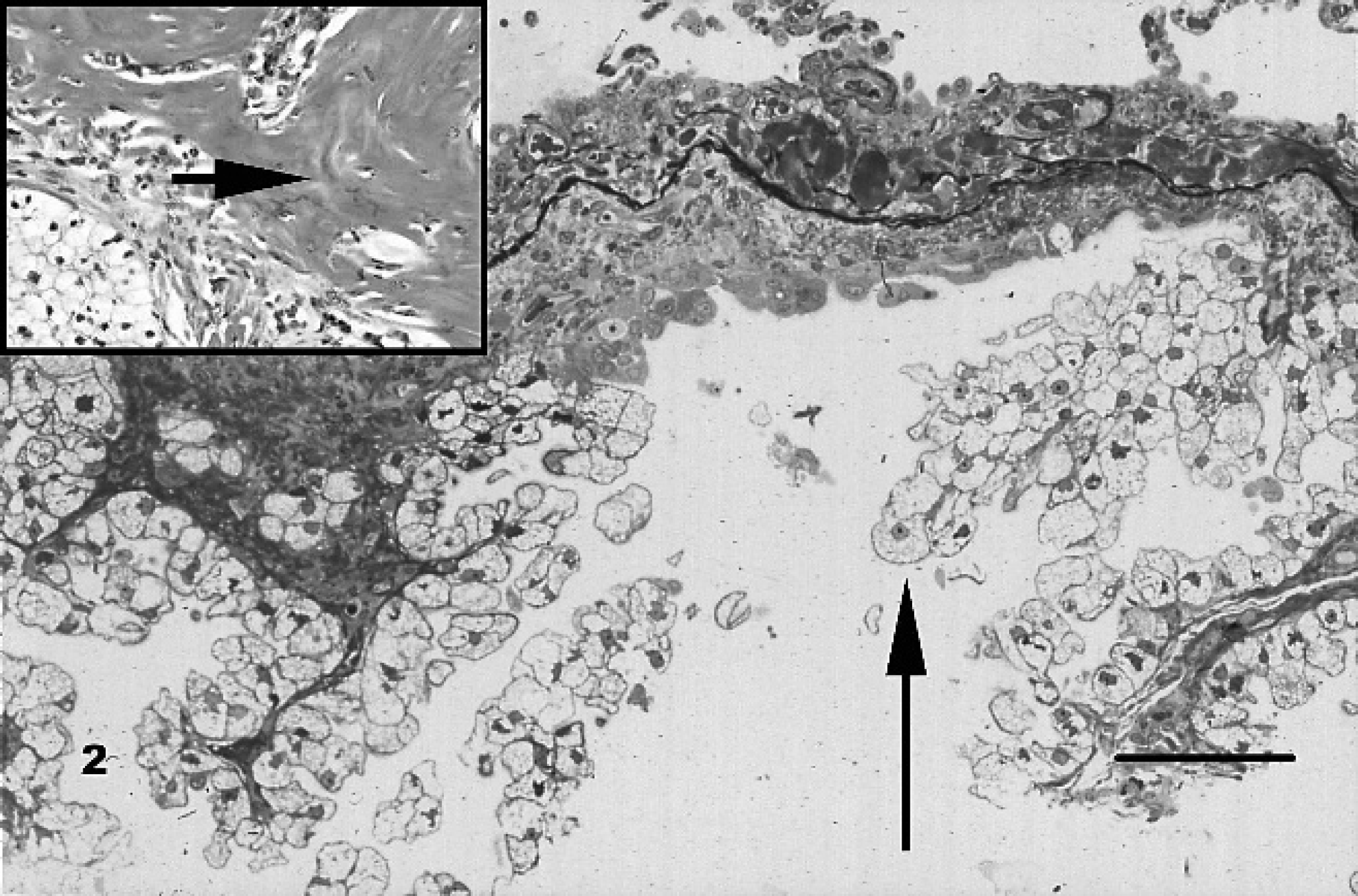
Thoracic mesothelioma; dog. Papillary projections of lipid-laden neoplastic mesothelial cells (arrow) are seen in association with a thick fibrous stroma. Inset: Woven and lamellar bone (arrow) adjacent to sheets of neoplastic cells. Hematoxylin and eosin. Bar = 125 μm.
Immunohistochemically, the tumor cells expressed vimentin (mild and multifocal), pancytokeratin (moderate to marked and diffuse), and S-100 (moderate and diffuse). Ultrastructural analysis revealed numerous clear cytoplasmic vacuoles consistent with lipid droplets (Fig. 3a). Prominent microvilli were present along the apical borders of neoplastic cells. Cells had visible intercellular junctions and were attached to a basement membrane (Fig. 3b). Based on the location of the tumor within the thoracic cavity, its morphological features, ultrastructural characteristics, and immunophenotypic profile, the diagnosis of mesothelioma was rendered. The numerous cytoplasmic vacuoles that were focally oil red O-positive in frozen sections and evident on ultrastructural evaluation allowed classification of this neoplasm as a lipid-rich pleural mesothelioma.
To the authors' knowledge, this is the first report that documents lipid-rich pleural mesothelioma in a dog. Pleural mesothelioma is best known in humans, in whom it is associated with exposure to asbestos; asbestos exposure may also be an etiologic agent in some of the mesothelioma cases in dogs. 8,9 Most canine mesotheliomas present clinically with pleural or pericardial effusions. Grossly they manifest as granular to velvety plaques or nodules that coat the mesothelial surface. 14 Histologically, conventional mesothelioma consists of arborizing papillary structures lined by cuboidal to polygonal cells with abundant eosinophilic cytoplasm. 14 Ultrastructurally, the neoplastic cells have numerous characteristic long microvilli that may cover the cells circumferentially. The cells are joined by numerous desmosomes, and the cytoplasm contains bundles of tonofilaments arranged around the nucleus. 14 Immunohistochemically, mesothelial cells express both epithelial cytokeratin and mesenchymal vimentin markers. 3,14 Furthermore, in humans, sarcomatous mesothelioma may express S-100 protein, 3 and S-100 expression is consistently found in liposarcoma. 4 Based on the lipid-rich nature of the tumor cells in this case, liposarcoma was considered; however, coexpression of cytokeratin and vimentin and ultrastructural features are characteristic of mesothelioma. 3,14
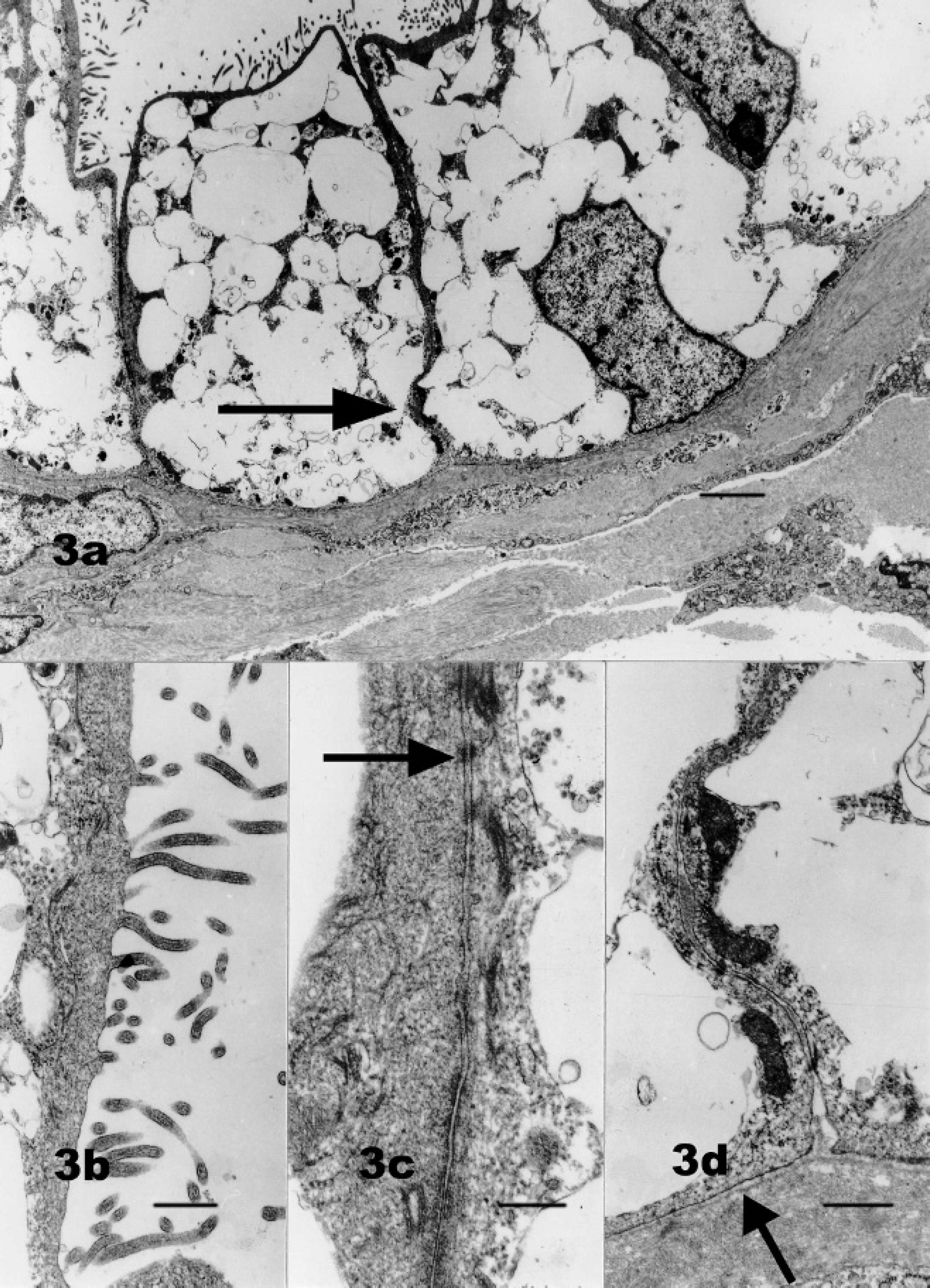
Thoracic mesothelioma; dog.
In conclusion, the authors of this report document for the first time a lipid-rich pleural mesothelioma in a dog. The mesothelial origin of this neoplasm is based on histomorphologic, immunohistochemical, and ultrastructural findings. It is hoped that the current report will aid veterinary laboratory diagnosticians in identifying this rare variant of mesothelioma in dogs.
Acknowledgements. The authors wish to thank Annet Shepard-Barry and Inna Lomakin for their technical help.
Footnotes
a.
Pancytokeratin, S–100, and vimentin, Ventana Medical Systems Inc., Tucson, AZ.
