Abstract
Background:
Mesenchymal stromal/stem cells (MSCs) have potent immunomodulatory abilities, particularly in a milieu of hyperactive immune system, through secreting a number of cytokines, growth factors, bioactive compounds and peptides, and by cell-cell contact. During viral infection, failure of immuno-neutralization of the viral particles, recruits T-lymphocytes (T-cells) that clear the virally-infected cells. MSCs greatly potentiate T-cells anti-viral activity.
Objective:
The objective of this study is to assess the ability of the cytokine-primed MSCs to activated T-cells, towards an antiviral application.
Method:
Human umbilical cord MSCs (UC-MSCs) were isolated from Wharton Jelly of a consented donor. UC-MSCs were primed with interferon (INF)-γ and transforming growth factor (TGF)-β1. Peripheral blood T-cells were isolated using mini-max and CD3+ population was purified using anti-CD3 immuno-magnetic beads. Naïve or primed MSCs were co-cultured with naïve and phytohemagglutinin (PHA)-activated CD3+ T-cells. T-cell activation was evaluated by changes in their rate of proliferation by cell count, flowcytometric immuno-phenotyping for CD25 expression, and IL-6 secretion in the conditioned medium.
Results:
The findings revealed that CD3+ T-cells count nonsignificant differed comparing the five experimental groups; Naïve MSCs, Naïve T cells, coculture with naïve MSCs, coculture with primed MSCs, and upon phytohemagglutinin-activation, despite a nonsignificant reduction of proliferation in the last two groups’ coculture. Only the coculture with the primed MSCs showed significant activation of T-cells assessed as CD25 expression compared to the other groups (
Conclusion:
The cytokine-primed UC-MSCs significantly upregulated CD25+ expression on T-cells, while hindering IL-6, without affecting their proliferation rate. This may point to potentially stronger antiviral effects, while alleviating the viral infection-induced cytokine storm.
Keywords
Mechanism diagram

Introduction
The mesoderm-derived multipotent mesenchymal stromal stem cells (MSCs) locate to several body tissues and demonstrate specific characteristics and functions. 1 The tissue regenerative therapeutic potential of MSCs encompasses their tri-lineage osteogenic, chondrogenic, and adipogenic differentiation, angiogenesis, resident stem cells support through cytokine section and mitochondrial potentiation, immunomodulatory, and anti-inflammatory actions. Therefore, MSCs are widely investigated for such promising tissue-regenerative and immunosuppressive cell therapy potential. 2 The identification of MSCs relies on their in vitro plastic adherence ability with a fibroblast-like morphology, induced tri-lineage differentiation potential, and surface expression of cluster of differentiation (CD) proteins. They express CD73, CD90, and CD105, while HLA-DR, CD14, CD11b, CD19, CD34, CD45, and CD79α expression is repressed. 3 MSCs could be induced to differentiate not only into tissues of mesodermal origin, but also into other tissue lineage cells. 4 The best sources for MSCs’ isolation are bone marrow, adipose tissue, tonsils and umbilical cord, and even the menstrual blood. 5
The microenvironment of immune-mediated inflammation and tissue degeneration is harsh to MSCs. This is an unsolved challenge to the clinical application of MSCs-based therapies. During infections, exposure of MSCs to TOLL-like receptor-agonists (i.e. TLR priming) present critical changes in their phenotype, survival and proliferation, hematopoietic support and immunosuppressive/immunomodulatory capacity, multi-lineage potential, regenerative functions, and therapeutic efficacy. Such challenges require prior meticulous assessment and invention of ameliorative approaches, namely different priming approaches that include TLR ligand such as lipopolysaccharides.4,6,7 Priming/activating MSCs, by different small molecular weight effectors and cytokines, not only potentiate the magnitude of these effects, but it also affects their nature, and the homing ability of MSCs. IFN-γ is the most commonly used MSCs-priming agent.8,9
After their recruitment to sites of tissue injury, the reparative anti-inflammatory effect of MSCs, particularly in the milieu of hyperactive immune response, employ several mechanisms. These mechanisms include cell-cell contact and by paracrine secretion of interleukins (e.g. IL-1, IL-10, and INF-γ), growth factors (e.g. platelet-derived growth factor (PDGF) and transforming growth factor (TGF)-β), and bioactive peptides. They also induce mitochondrial-supportive effects. MSCs also induce the expansion of IL-10-producing dendritic cells and B-cells, CD4+CD25+FOXP3+ T-cells, and CD8+CD28+ T-cells.10,11 This way, MSCs are able to modulate activity, differentiation/polarization and fate of macrophages, neutrophils, and B- and T-lymphocytes, towards adaptive and innate immunomodulation, immunosuppression, regeneration and regulatory T-cell expansion. MSCs enhance tissue homeostasis, remodeling, surveillance, and repair. They support hematopoiesis, resolve injury, improve regeneration and angiogenesis, and dampen inflammation and autoimmunity. These actions have corrective potential in degenerative, inflammatory, autoimmunity and infectious diseases afflicting heart, neurons, liver, skin, hematopoietic and respiratory systems, intestine, and kidney.6,12–16
MSCs’ antiviral immune effect is mediated by the cytotoxic T-cells. 17 T-cells come into play after failure of the neutralizing humoral immune response, and entry of viral particles into cells. The expressed neo-antigens in virus-infected cells are associated with the MHC class I complex. In the draining lymph nodes, both dendritic cells and naïve T-cells stimulate CD8+ T-lymphocytes by peptide-MHC class I complexes. The activated CD8+ T-cells release cytotoxic granules to induce apoptosis of the virus-infected cells. For the antiviral response, MSCs also induce the COX2-PGE2 pathway in monocytes and the stimulation of T-cell and the consequently secreted cytokines. 18 Severity of SARS-CoV-2 infection correlates impaired inflammatory innate and adaptive immune responses, mostly due to the exhaustion of T-lymphocytes and lymphopenia. COVID-19 cases exhibiting minimal or no lymphocyte activation response, with low activated CD4+ and CD8+ T-cells count, is possibly linked to decrease in frequency of Th1 cells. 19
The therapeutic potential of MSCs is modulated by changes in the in vitro and in vivo microenvironment. Cytokine-primed UC-MSCs cocultured with T-cells can enhance the anti-viral effects of the latter. Therefore, we planned the current in vitro study to assess the ability of interferon (INF)-γ and transforming growth factor (TGF)-β1-primed MSCs cells to modulate T-cells proliferation, CD25 expression and IL-6 secretion, as markers of antiviral T-cell activation.
Methods
Mesenchymal stem cell isolation and characterization
Umbilical cord Wharton Jelly MSCs (UC-MSCs) were isolated via explant method from the umbilical cord of a consented female delivered by Caesarian section at Women Health Hospital, Faculty of Medicine, Assiut University, Assiut, Egypt, where the study was ethically approved (Ethical approval# 04-2023-300276).
Inclusion and exclusion criteria used for this study
➢ For isolation of MSCs: Umbilical cord samples were taken from full-term healthy pregnant women free from chronic and autoimmune diseases and infection (bacteriological and virology screening tests were done), with written informed consent obtained from all donors.
➢ T-lymphocytes were isolated from healthy donors aging 15–60 years, after clinical and serological examination to confirm that they are free from infections, chronic and autoimmune diseases.
At the Stem Cells Center, Assiut University, a piece of the umbilical cord (~5 cm) was resected and washed with PBS solution containing 10% penicillin-streptomycin-amphotericin B (Cat# PB180121, Elabscience Biotech Inc., Houston, Texas, USA). The cord was longitudinally opened, the vessels were removed and the cord was incubated in 1:1 (v/v) mixture of trypsin (TrypLE™ Express Enzyme, Cat# 12604013) and collagenase (Collagenase Type I, Cat# 17100017) at 37ºC for 45 min (Thermo-Fisher Scientific, USA). After discarding the solution and washing the cord with PBS, the Wharton Jelly was scraped off with a sharp scalpel. The Wharton Jelly particles were plated in T25 flasks without medium for 4 h to allow for adherence. DMEM containing 20% fetal bovine serum and 1% penicillin-streptomycin-amphotericin B was added and was refreshed every other day till the growth reached 85%–90% confluence. MSCs were trypsinized, counted and tested for viability tests using trypan blue exclusion test (Cat# 15250061, Thermo-Fisher Scientific, USA). Cells were flowcytometrically immuno-phenotyped for CD105, CD271, CD90, CD45, and CD34 (Anti-CD105 (105C02), Anti-CD45 (ab10559), and Anti-CD90 (ab23894), and Anti-CD34 (ab762), Abcam, Cambridge, UK, and, Anti-CD271 (ME20.4), and, Alexa Fluor™ 488, Cat# 53-9400-42, eBioscience™, USA). Our lab is experienced in isolation and characterization of stem cells, and their in vitro and in vivo therapeutic applications.12,20,21
Cytokine enhancement of UC-MSCs
After passage 3, the harvested MSCs were divided into two groups. Group A was subcultured in presence of INF-γ (500 U/mL) and TGF-β1 (10 ng/mL) for 48 h, aka, cytokine-primed MSCs (Cat# 285-IF and 240-B, Bio-Techne, UK). Group B cells were subcultured without the priming cytokines to stay as the naïve MSCs.
Isolation of peripheral blood mononuclear T-lymphocytes
We collected 5-mL peripheral blood samples on heparin from ten consented healthy donors, for the isolation of peripheral blood mononuclear cells (PBMCs). Cells were separated from by using Ficol Hypaque density gradient centrifugation. The mononuclear T-cells were recovered, washed and counted (Cat# M8997, Thermo-Fisher Scientific, USA). Separation of T-lymphocytes was flowcytometrically achieved using mini-max separation kit and anti-CD3 immuno-magnetic beads into pure CD3+ T-cells (MagniSort™ Human CD3 Positive Selection Kit, Cat# 8802-6830-74, Thermo-Fisher Scientific, USA). Then, they were washed and counted. 22
Co-culture of T-lymphocytes and UC-MSCs
In triplet wells of 12-well plates, an equal number of naïve or cytokine-primed UC-MSCs and T-cells (1000 cells/well) were incubated in the complete DMEM medium for 48 h. After 48 h, the plates were gently hand shaken, and the conditioned medium was recovered and centrifuged to isolate floating T-cells. The clear supernatant was collected and aliquot stored at −20°C. T-cells were activated with the T-cell mitogen phytohemagglutinin (5 mg/mL; Cat# R30852801, Thermo-Fisher Scientific, USA). 22 The study was repeated three times.
Evaluation of T-lymphocyte activation
T-lymphocyte activation was assessed by T-cell counting, flowcytometric CD25 versus CD3 immuno-phenotyping and secretion of IL-6. Results were compared for the five experimental groups; (1) coculture 5 mg/mL phytohemagglutinin-activated T-cells with the naïve UC-MSCs, (2) coculture of 5 mg/mL phytohemagglutinin-activated T-cells with the cytokine-primed UC-MSCs and, (3) cytokine-primed MSCs, (4) Naïve MSCs, and (5) Naïve T cells. 22
IL-6 was quantitatively assayed using specific immunoassay kit that utilizes two human IL-6-specific antibodies, for binding and detection. The latter is biotinylated to fix the avidin-conjugated horseradish peroxidase that proportionally converts the substrate into chromogen (lower detection limit is 0.94 pg/mL, Cat# E-EL-H6156; Elabscience Biotech. Inc., Houston, Texas, USA). Three serial dilutions of each sample, 10 replicates for each group, were tested to insure IL-6 concentration that overlap with the standard curve.
Statistical analysis of the data
Data were analyzed using IBM SPSS software (version 20.0; IBM Corp., Armonk, NY). Quantitative normally distributed data were expressed as range (minimum—maximum), mean ± standard deviation, and median. The continuous data were tested for normality by using the Shapiro-Wilk test. One-way ANOVA tests with Tukey Post Hoc tests (for pairwise comparison between each two groups) were used for comparing the five studied groups.
Results
Characterization of the isolated UC-MSCs and T-Cells
Figure 1 presents the flowcytometric assessment of the harvested MSCs cells. They lacked CD45 expression and looked positive for CD90, CD271, and CD73 (Figure 1). MSCs were harvested in large numbers and their viability assessed by trypan blue exclusion test was 98% as one pool. We harvested a total of 3410 ± 919.5 lymphocytes/mL peripheral blood (with a range of 1400–4500 T-cells/mL) from our ten blood donors. Out of which, 910.0 ± 204.5 cells/mL (with a range of 550–1200 cells/mL) were CD3 positive. They showed very high viability (98% ± 1.247% and a range of 95%–99%).

The flowcytometric assessment of phychoerythrin-stained MSCs isolated from human umbilical cord Wharton Jelly. (a) Cells are negative for CD45 (APC). (b) Cells are positive for both of CD271 (in the left part) and CD73 (in the right part) versus CD90 (FITC for both).
T-cells activation by co-culture with UC-MSCs
CD3+ mature T-cells Sorted flowcytomerically, five groups were used Naïve MSCs and Naïve T cell and primed MSCs groups. Activated T by phytohemagglutinin, then incubated with naïve MSCs, cytokine-primed MSCs. Results showed nonsignificant differences in their proliferation rate, as assessed by cell counting (910 ± 204.4 (550–1200), 901 ± 194.1 (580–1200), 1003 ± 227.7 (600–1300) cells/well;
Comparison between the five studied groups according to total T-cells (CD3+ve).

SD: standard deviation;
Means with any common letter (a–c) are not significant (OR means with totally different letters (a–c) are significant).
Statistically significant at

Comparison between the five studied groups according to total T-cells (CD3+ve).
The rate of CD25 expression as a marker for T-cell activation was assessed. Activated T-cells incubated with naïve MSCs showed a rate of CD25 expression of 39.9% ± 12.5% (with a range of 20%–58%), while cytokine-primed MSCs cells had a rate of CD25 expression of 54% ± 11.5% (with a range of 35%–73%), and primed MSCs incubated with T-cell activated with phytohemagglutinin showed a rate of CD25 expression of 43.2% ± 11.8% (with a range of 26%–60%). While Naïve MSCs group had a rate of CD25 expression of 0.46b ± 0.07 ( with a range of 0.10–0.50) and group of Naïve T cells had a rate of CD25 expression of 2.35b ± 0.75 with a range of (1.10–3.50) The cytokine-primed MSCs showed significantly higher CD25 expression compared to each of the coculture with the naïve MSCs (
Comparison between the five studied groups according to activated T-cells (CD25%).

SD: standard deviation;
Means with any common letter (a–c) are not significant (OR means with totally different letters (a-c) are significant).
Statistically significant at
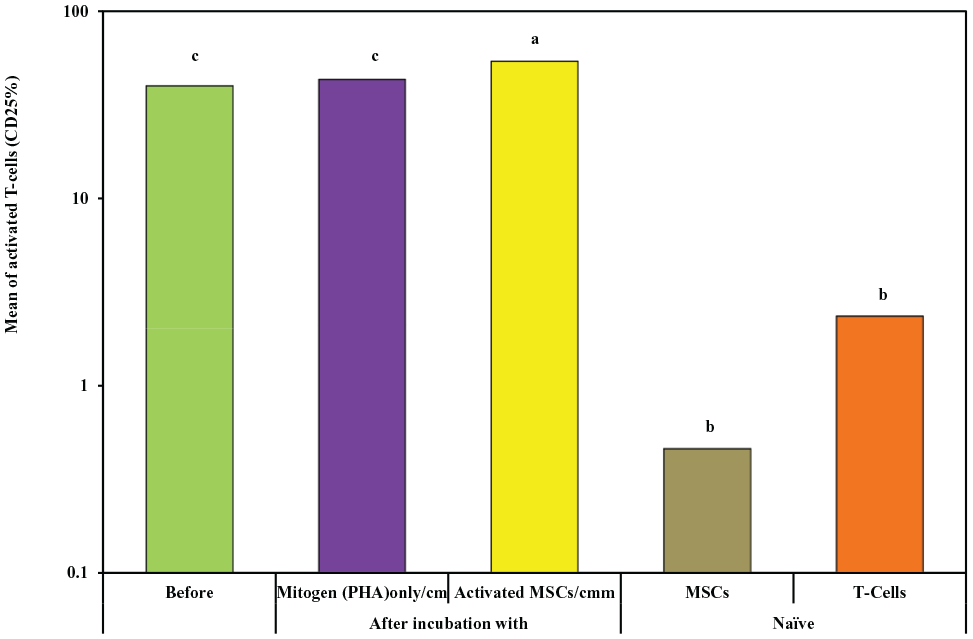
Comparison between the five studied groups according to activated T-cells (CD25%).
Table 3 presents IL-6 secretion results. While IL-6 was almost undetectable in the conditioned medium of naïve MSCs, naïve T-cells secreted considerable amounts of the cytokine (
Comparison of the interleukin-6 (pg/mL) into the conditioned media as an indicator for as a proxy biological function of T-cells activation among the treatment conditions.

Secretion of interleukin-6 (pg/mL) into the conditioned media from naïve (Naïve MSCs) and INF-γ/TGF-β1-primed umbilical cord mesenchymal stem cells (primed MSCs) compared to naïve and phytohemagglutinin-activated T-lymphocytes (activated T-Cells), and their cocultures. Data shown are mean ± SD (range), and
Discussion
We planned to study the effect of INF-γ/TGF-β1-primed UC-MSCs on cocultured healthy peripheral blood CD3+ T-cells with a hope to potentiate the expected immunomodulatory/antiviral activities assessed as rate of cell proliferation, CD25 expression and IL-6 secretion. CD3 is involved in activating both of T helper cells (CD4+ naive T-cells) and the cytotoxic T-cell (CD8+ naive T-cells). Both of UC-MSCs and lymphocytes were properly characterized and purified to a viability of 98%. Reportedly, naïve MSCs are hypo-immunogenic and have several immune evasion abilities. However, in vitro or in vivo priming of MSCs with pro-inflammatory cytokines (e.g. IL-1β, IL-17, IFN-γ, and TNF-α), as is the case within the inflammatory, degenerative, infected tissue milieu, upregulates the class I/II MHC and co-stimulatory molecules expression on MSCs. This imparts to them improved survival and proliferation with enhanced immunomodulatory and immunosuppressive functions, albeit on the expense of their hastened aging and clearance, and induced immuno- and tumori-genecities. Primed MSCs, dose- and time-dependently, secrete kynurenine upon modulating indoleamine-2,3-dioxygenase (IDO-1) expression, the ligands for receptor programmed cell death 1 (PD-1) system (PD-L1 and 2), interleukins (IL-6 and -10), nitric oxide, prostaglandin-E2, hepatocyte growth factor, TGF-β1, and, leukemia inhibitory factor and human leukocyte antigen-G molecules. These factors empower the MSCs immuno-suppressive and/or modulating activity, to reduce rate of proliferation and functional activity of T-cells, NK cells, and dendritic cells, along with Th17 differentiation.6,9,22–24
T-lymphocytes have a potential antiviral immunity. Viral antigens are presented on activated antigen-presenting cells associated with MHC class II molecules to naïve CD4+ T-cells in draining lymph nodes. This results in the differentiation of naïve CD4+ T cells to mature antiviral cells. Specific antiviral T-cells have unique surface markers with upregulated CD25 receptor expression and its expansion augments the anti-viral potential. Moreover, they have a Th1 phenotype with increased IFN-γ production, which along with IL-12, enhanced the proliferation of this subset of cells, while inhibiting other T-cell subsets’ development.11,25,26 MSCs are implicated in the antiviral process through multifactorial immune-modulatory effects on T-cell-mediated antiviral activity, despite an antiproliferative action on T-cells. 27 COVID-19 virus-induced immune disturbances are proposed as the initiator of tissue injury, and the disturbed INF response exacerbates the production of inflammatory cytokines that hyperactivate macrophages. As a result, active B- and T-cell lymphocytes, and NK cells could be depleted. Moreover, neutrophil extracellular traps are deployed, resulting in thrombo-inflammation. 28 Therapeutic and preventive measures of COVID-19 infection must, therefore, target the disturbed/stimulated immune system, through, for example, the therapeutic potential of MSCs. 29 The variable effects of MSCs on T-lymphocyte activity in antiviral immune response needs to be explored.
We observed nonsignificant reduction in proliferation of T-cells incubated with primed MSCs as compared to those incubated with naïve cells and hemagglutinin. An antiproliferative effect for primed MSCs was frequently reported in the literature, although the effect was differential depending on the T-cell subset.30–32 Naïve and IFN-γ-primed UC-MSCs secrete a very low level of IL-6 and TGF-β. While coculture with naïve UC-MSCs inhibited the proliferative response of Th1 and Th17, it augmented Th2 and Treg proliferation. Primed UC-MSCs reduced level of IFN-γ, TNF-α, and IL-2, while increasing IL-6 and IL-10 secretion from phytohemagglutinin-CD3Ab-IL-2-activated PBMCs. Priming of UC-MSCs with IFN-γ before coculture significantly furthered the reduction in IFN-γ and TNF-α, the increase in IL-6 and IL-10 secretion. IFN-γ-primed UC-MSCs increased the frequency of CD4 + CD25 + CD127dim/- T-cells and decreased the frequency of Th17. 22 Priming bone marrow-derived MSCs with the inflammatory cytokines, IFN-γ and TNF-α, increased immunosuppressive MSC functions, but decreased their proliferative ability.13,23 IFN-γ-preconditioned MSCs, though IDO expression, inhibit cytotoxic T-cell degranulation and T-cell proliferation. Compared to untreated MSCs, the primed cells resist host T-cell cytolysis, without affecting their homing characteristics. 33 While IFN-γ priming induces the expression of surface class-II HLA (HLA-DR) molecules on early passage young MSCs, this does not happen with senescent MSCs. 34
Expression of CD25 (IL-2Rα) is induced upon T-cell activation towards Treg and Teff pathways and autoimmune immunosuppression. CD25 locus and the serum levels of soluble CD25 were associated with susceptibility to a number of autoimmune disorders.35,36 Our results showed that only the primed UC-MSCs were able to significantly induce CD25 expression on T-cells and reduced their ability to secrete IL-6, which can be associated with stronger antiviral effects and dampen the cytokine storm. In HIV-1-infected immune non-responders, UC-MSCs therapy had anti-inflammatory and suppressive effects on the overactivated immune system, as well as an immunomodulatory effect by reducing number of circulating activated CD8+ T-cells, while increasing CD4+ T-cells. 37 IFN-stimulated genes (ISGs) in primed MSCs enable them to target viruses at different stages during the viral cycle; cell membrane penetration, endocytosis, nuclear mRNA entry, transcription, and translation.38–40 These viral infections include COVID-19, SARS, dengue, influenza A viruses and ebola. Moreover, susceptibility to Chikungunya virus infection is increased by repressing the expression of ISGs in MSCs. 39 MSCs cell therapy, for patients with hepatitis B virus-related liver cirrhosis, corrects the Treg/Th17 cell imbalance by upregulating Treg cells and reducing Th17 cells, with a significant increase in serum TGF-β levels and a reduction in TNF-α, IL-6, and IL-17A levels. 41 Treatment with MSCs, in patients with acute severe refractory A/H1N1 influenza-induced respiratory distress syndrome, exerted beneficial anti-inflammatory actions. This was evidenced by decreased pulmonary infiltrates via maintaining the integrity of alveolar-capillary barrier, and reducing permeability. This was coupled with lower IFN-γ and IL-8 levels in plasma, and IL-6 levels in broncho-alveolar lavage fluid.42–45 Moreover, non-coding miRNAs contained in released extracellular vesicles from MSCs, such as Let-7f, miR-145, miR-199a, and miR-221, exerted potent antiviral activity by inhibiting hepatitis C virus (HCV) replication. 40 Experimental skin wound healing was accelerated by IFN-γ/TNF-α-primed UC-MSCs secreted IL-6 that induced M2 polarization of macrophages to significantly enhance vascularization and collagen deposition. 46 This effect is similar to our observation that primed UC-MSCs secreted huge amount of IL-6 as compared to the naïve ones, very close to the level observed for naïve CD3+ T-cells. Although our naïve MSCs were able to augment IL-6 production in the coculture by activated CD3+ T-cells, the primed MSCs significantly reduced such production. It is possible that the presence to TGF-β1 in the priming cocktail complicated the interactions and molecular mechanisms employed, as it is known to enhance the chondrogenic differentiation of UC-MSCs. 47 Increases in the secretion of IL-6 and IL-10, and decreases in the secretion of IFN-γ and TNF-α were noticed upon coculturing the activated lymphocytes with IFN-γ-primed UC-MSCs. Moreover, the frequency of Treg and Th17 cells decreases, the count of CD4+CD25+CD127dim/− T-cells increases. 22 Increases in IL-6 secretion in coculture of primed UC-MSCs and activated PBMCs in this study, is contrary to our finding of a reduction after priming, while an increase with naïve MSCs. This could be reasoned to different nature of our priming cytokines, incubation time and dosing. 48
Mechanistically, IFN-γ-preconditioned MSCs suppressed NK activation and inhibited their IFN-γ secretion more efficiently than naive MSCs. This effect is partially mediated by increases in prostaglandin-E2 (PGE-2) and IDO expression. Moreover, reduction in the expression of the activating ligand NKG2D, and increases in expression of class I HLA molecules provide protection for IFN-γ-primed MSCs against NK cytotoxicity. 49 IFN-γ-treated human bone marrow-derived MSCs (BM-MSCs) significantly differentially expressed 210 proteins. Among the majority of proteins that were overexpressed were ICAM-1, VCAM-1, IDO, BST-2, and PDL-1). Proteins that were repressed include (41 proteins) include NPR3, ANTXR1, FADS2, and APCDD1L. 50 Human and murine IFN-γ-preconditioned MSCs induce immunosuppression through inhibition of mTOR activity and the activating phosphorylation of signal transducer and activator of transcription (STAT1/STAT3). Consequently, genes related to proliferation, stemness, and differentiation were downregulation, while those functional in immune-regulation were upregulated. 51 MSC-induced suppression of proliferation of PBMCs is a complex mechanism and may function differently, depending on T-cell mitogenic stimuli. While inhibiting prostaglandin-synthesis, using adding indomethacin, restored the inhibitory ability of MSCs on PBMCs activated by phytohemagglutinin. Same was not achieved using allo-antigens. 52
Limitations of the study include our shortage of analyzing the complete secretome and the intracellular mechanisms underlying UC-MSCs priming, particularly with the infrequently used combination of INF-γ/TGF-β1, and for the activation of and coculture with CD3+ T-cells. One priming cytokines concentration and incubation time, and MSCs: T-cells cell number ratio were used; and not a dose-/time-depended manner.
Conclusions
The INF-γ/TGF-β1 primed MSCs’ potential antiviral mechanism might be exerted by the upregulation of CD25+ expression on T-lymphocytes, while hindering their IL-6 secretion towards subsiding the viral infection-induced cytokine storm.
Footnotes
Acknowledgements
The authors like to express their gratitude to Prof. Dr. Tarek H. El-Metwally, Biochemistry Division, department of Pathology, College of medicine, Jouf University, Sakaka, Saudi Arabia, for his supervision of IL-6 assay and proof-reading the manuscript.
Authors contributions
The authors have contributed in all aspect of this work, including conceptualization, design, execution, data collection and analysis, and, manuscript preparation and revision, and approval for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to the Deanship of Scientific Research at Jouf University for funding this work through the research grant #CV-44-41.
Institutional review board statement
The study was ethically approved by IRB of the Faculty of Medicine, Assiut University, Assiut, Egypt—Approval# 04-2023-300276.
Ethics approval
Ethical approval for this study was obtained from Women Health Hospital, Faculty of Medicine, Assiut University, Assiut, Egypt, where the study was ethically approved (Ethical approval# 04-2023-300276).
Informed consent statement
All volunteering blood and the umbilical cord donors signed an informed consent after being brief for their rights, the study aims and its publication. Written informed consent was obtained from legally authorized representatives before the study.
Trial registration
In vitro study.
Data availability statement
All data are presented in the manuscript.
