Abstract
A 2-month-old female llama with a history of tetraparesis was presented for necropsy. This cria was apparently normal until it became ataxic during its second week of life. It had diminished flexor reflexes of the left forelimb and both hind limbs. Hematology and serum biochemistry revealed neutrophilia, elevated alkaline phosphatase, elevated phosphorus, and modest hyperglycemia. Radiography of the cervical spine demonstrated an abnormal C3–C4 intervertebral disc and bony proliferation of the vertebral end plates. The llama was treated with antibiotics and corticosteroids but failed to respond. Postmortem examination revealed cervical intervertebral disc disease and vertebral exostosis. Microscopically, there was necrosis of intervertebral connective tissue and focal malacia of the spinal cord. Pseudomonas aeruginosa was isolated from the intervertebral disc space and from cerebrospinal fluid. To the authors' knowledge, this is the first description of Pseudomonas-associated discospondylitis in a llama.
A 2-month-old female llama weighing 12.7 kg was examined. It had developed tetraparesis after an apparently normal first week of life. The animal was ataxic and unable to nurse. On physical examination, the llama was thin, bright, and alert, with normal heart rate, respiratory rate, and pink mucous membranes. All joints were normal; flexor reflexes of the hind limbs and left forelimb were weak. The animal was able to hold its head upright with no head tilt but was unable to stand without support. Cervical radiography suggested an abscess between C3 and C4. The animal was treated with florfenicol (300 mg) and flunixin meglumine (20 mg) once daily. In addition, single doses of dexamethasone (2 mg) and B-complex (1 ml) were given. Flunixin was discontinued after 2 days and ceftiofur sodium was begun on day 13 after discontinuing florfenicol. There was a neutrophilia (25,700 cells/μl; reference range for young llama: 6,400–19,200 cells/μl), and the serum biochemical profile revealed modest hyperglycemia (170 mg/dl; ref. range for young llama: 80–145 mg/dl), elevated alkaline phosphatase (188 U/liter; ref. range for young llama: 41–92 U/liter), and mildly increased phosphorus (7.4 mg/dl; ref. range for young llama: 4.6–7.1 mg/dl). Additional radiographs revealed focal mineralization of disc material and increased density of vertebral end plates at C3–C4 (Fig. 1). On day 16, the animal was unable to stand, and breathing was agonal. The cria became unresponsive and died.
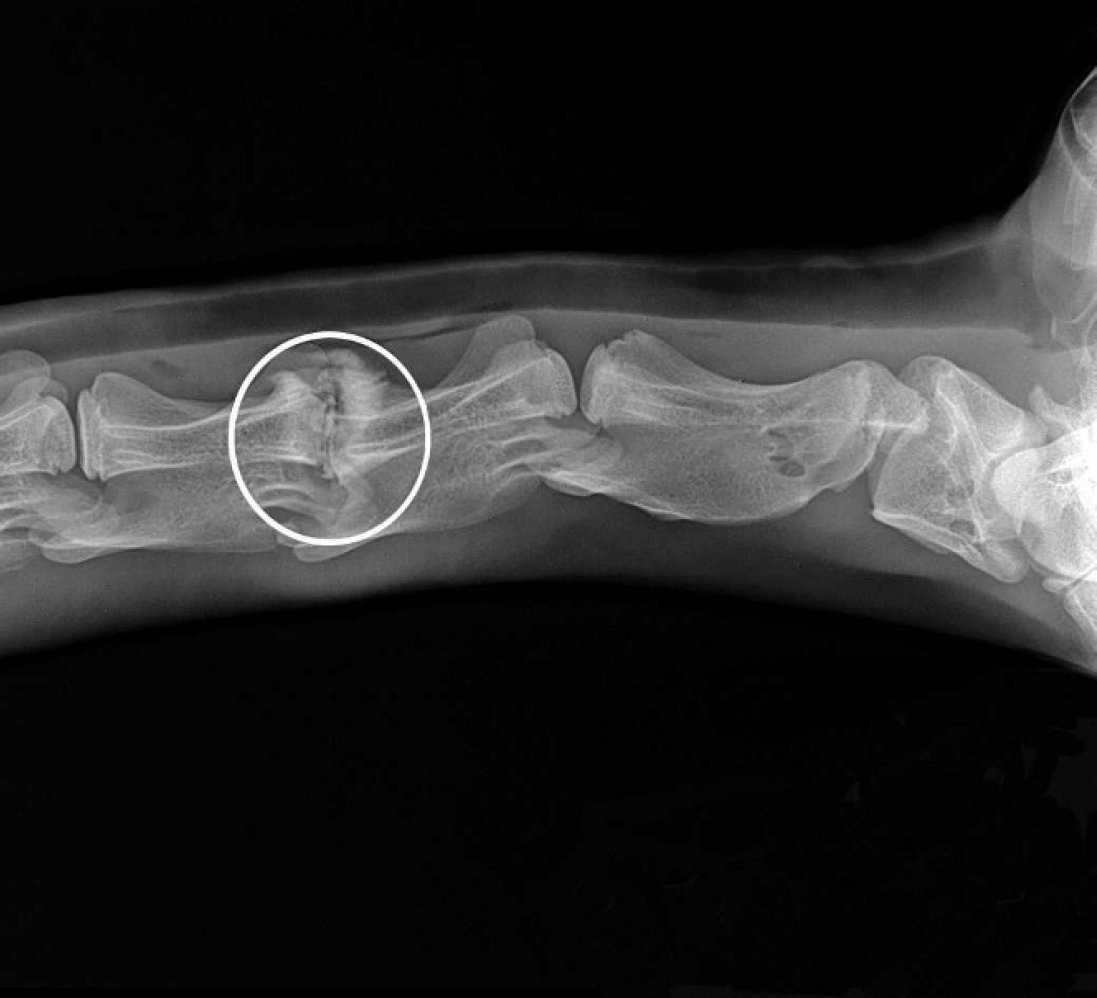
There is mineralized disc material in the intervertebral space of C3–C4 (circled), increased density of vertebral end plates and exostoses.
On external examination at necropsy, there was a prominent kyphosis of the cervical spine at C2–C3 vertebrae. After removal of the skin and surrounding musculature, cerebrospinal fluid was collected aseptically with syringe and needle by dorsal puncture at the intervertebral articulation of C3–C4. As part of the dissection, the vertebral column was sagittally split with a band saw. After searing the cut surface, the intervertebral disc space at C3–C4 was swabbed with a sterile cotton-tipped applicator stick. This was streaked onto 5% blood agar, incubated at 37°C under microaerophilic conditions, and examined after 24 and 48 hr.
The cervical spinal canal at C3–C4 was narrowed due to marked thickening of the vertebral end plates (Fig. 2). The spinal cord at this site was compressed and soft. The intervertebral space contained sparse red-brown granular gelatinous material, and there appeared to be dissolution of nucleus pulposus and annulus fibrosus. This was accompanied by spondylosis of the ventral surfaces of C3 and C4 vertebrae, with thickening of the dorsal and ventral margins of the caudal edge of the C3 vertebral body and anterior edges of the C4 vertebral body.
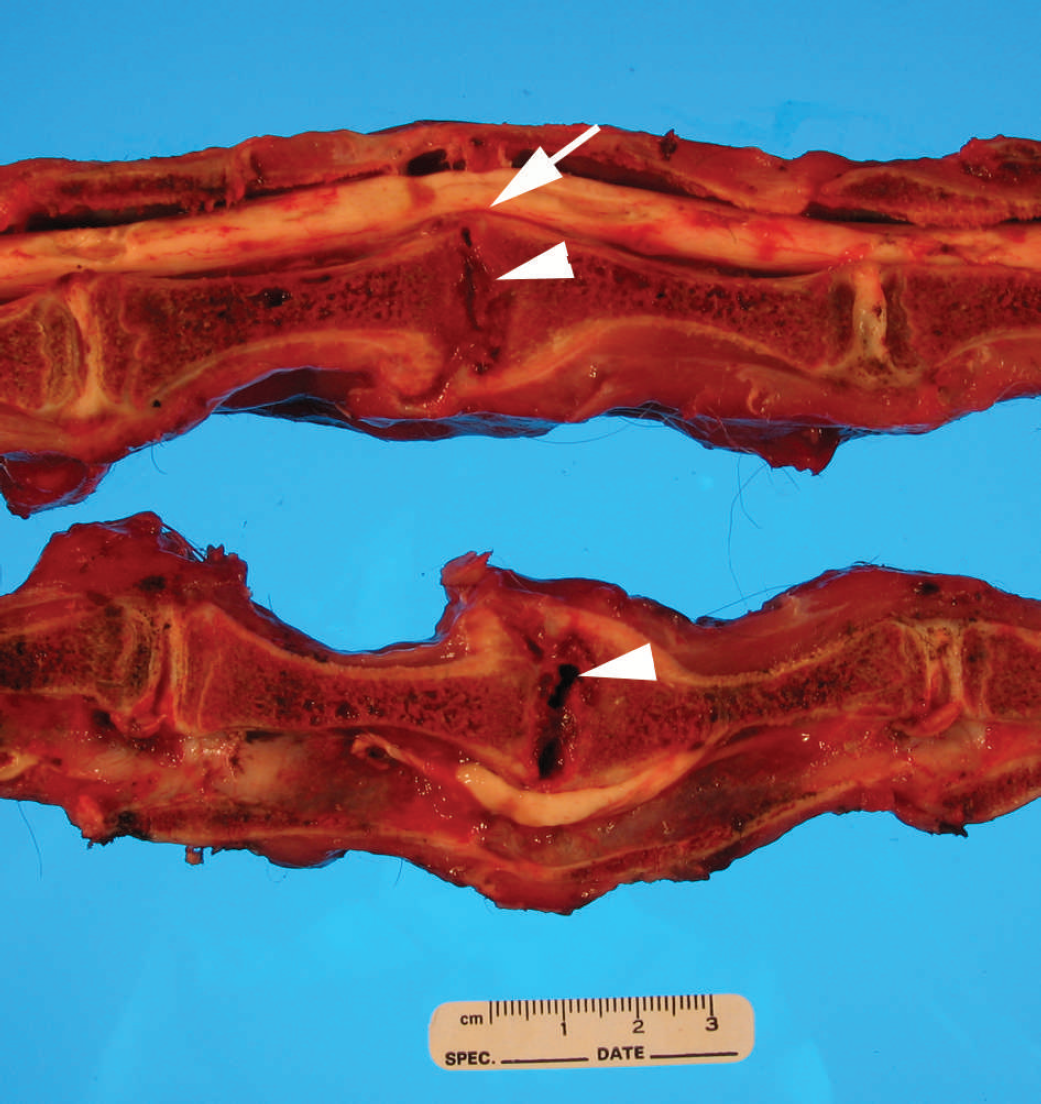
Photograph of both halves of a parasaggital section of the cervical vertebral column. Note the focal spinal cord compression (arrow) produced by thickening of the C3–C4 vertebral end plates. The intervertebral space contains a lytic zone (arrowhead) encompassing the C3–C4 disc, the posterior end plate of C3, and anterior end plate of C4. There is spondylosis of the ventral surfaces of C3 and C4 vertebrae.
Multiple organs, including the cervical vertebrae, spinal cord, and brain, were fixed in 10% neutral buffered formalin. All the bony tissues were decalcified in 1.35 N hydrochloric acid for 56 hr. Following the decalcification, sections of the vertebrae were processed routinely for histologic examination and stained with hematoxylin and eosin, Masson's trichrome, Prussian blue, Brown and Brenn's modified Gram stain, and Grocott's methenamine silver stain.
Histologic lesions were confined to the C3–C4 articulation, where there was widened intervertebral space and vertebral exostoses. The intervertebral connective tissue and vertebral growth plates were extensively replaced by fibrin, hemorrhage, and mineralized disc substance. The primary and secondary bone trabeculae were frequently discontinuous, and few microfractures were observed. Exostoses extended both dorsally and ventrally around the joint space. Serial sections of the cervical spinal cord at the site of compression showed asymmetrical areas of rarefaction and necrosis of the dorsal horns and funiculi (Fig. 3). Marked Wallerian degeneration occurred throughout the affected regions, and there were numerous clear vacuoles that contained small numbers of macrophages (myelin digestion chambers). Spongiosis and focal gliosis of the white matter, with variable dilation of myelin sheaths, occurred immediately cranial and caudal to the site of most significant damage. Eosinophilic to pale-gray swollen axons (spheroids) were present in both gray and white matter. Pseudomonas aeruginosa was cultured from both the spinal fluid and intervertebral disc space.
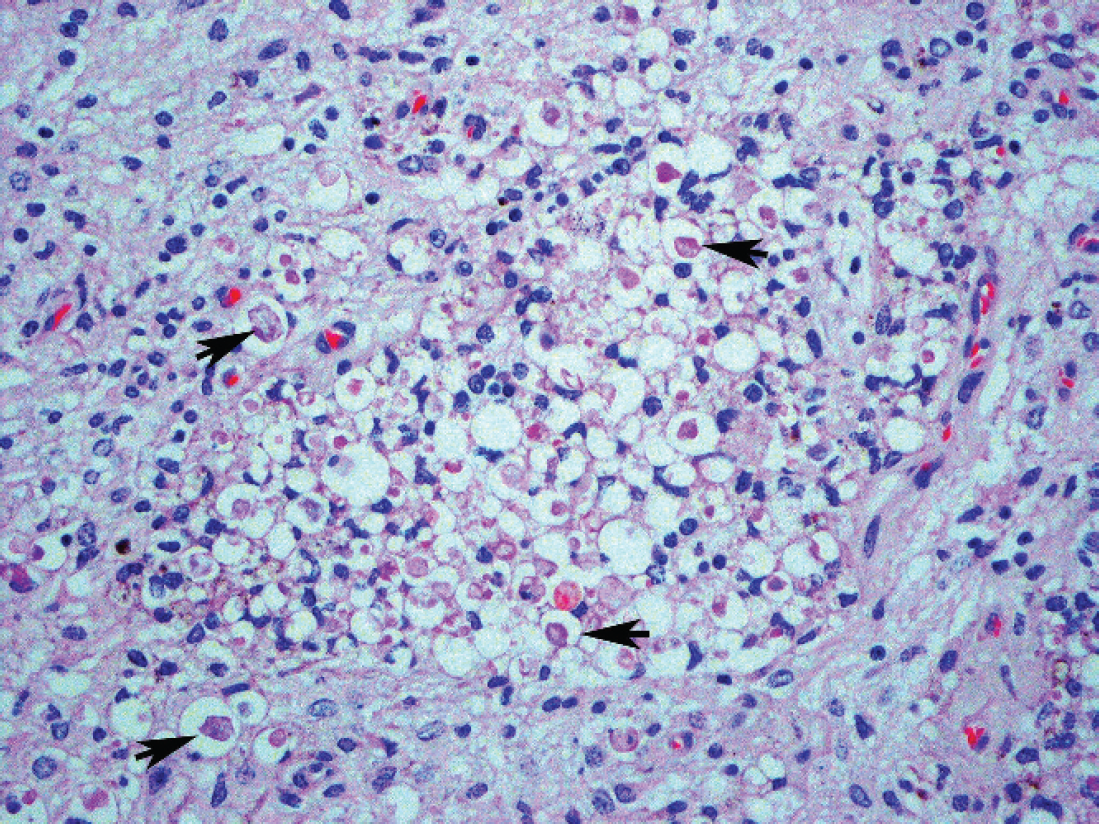
Photomicrograph of the cervical spinal cord at the site of compression showing the affected region. There is status spongiosis as a consequence of rarefaction and necrosis of the dorsal horn. Also note the spheroids in dilated axonal sheaths (arrows). Hematoxylin and eosin. Original magnification X400.
An initial blood count in the llama showed neutrophilia, a finding that could be attributed to either acute or chronic bacterial inflammation, stress, or both. 26 Serum biochemistry revealed modest hyperglycemia. Neonatal camelids are known to develop hyperglycemia, as well as hypernatremia and hyperosmolarity, in response to a combination of stress and inadequate water intake. 7 Lysis and remodeling of vertebral end plates in the present case may have been the cause of the elevated alkaline phosphatase; however, in young animals, bone growth may also cause elevation. Alternatively, the lipopolysaccharide of P. aeruginosa can induce osteoclastogenesis and bone resorption. 27
Discospondylitis has not been previously reported in llamas. 1,22 In camelids, neurologic disease has been reported to occur as a consequence of aberrant migration of Parelaphostrongylus tenuis, 3,6,13 congenital vertebral malformations, 16 or idiopathic spinal cord degeneration. 18,19 In 2006, intervertebral disc protrusion and compressive myelopathy were documented in a 12-year-old llama. 24 A 2-cm mass of extruded nucleus pulposus originated from the C6–C7 intervertebral disc. In another case, discospondylitis and paraparesis were reported in an adult alpaca; failure to culture bacteria from the disc space was attributed to prior treatment with antibiotics. 26
Discospondylitis is inflammation of the intervertebral disc and adjacent vertebral bodies, characterized by necrosis of the disc and erosion of the end plates. Instability of adjacent vertebral bodies occurs, leading to collapse of the intervertebral space, and microfractures. 1 This condition is best understood in dogs, in which the lumbosacral spine is most often affected, commonly resulting in localized pain, progressive paresis, and ataxia. 23 Other clinical signs include inappetence, weight loss, depression, fever, and reluctance to move. In early stages of the disease, many animals show little or no neurologic deficit or only mild paresis. 26 In dogs, discospondylitis is frequently caused by bacterial infections, with Staphylococcus intermedius the most frequently isolated bacterium. Other organisms identified as causes include Staphylococcus aureus, Streptococcus, Brucella canis, Escherichia coli, Pasteurella, Corynebacterium, Mycobacterium, Nocardia, Actinomyces, and Bacteroides. 5,11,12,14 In rare cases, Fusarium, Aspergillus, Mucor, Paecilomyces variotii, 5,11,12,14 P. aeruginosa, 1 Enterococcus faecalis, 1 and Staphylococcus epidermidis 1 have been recovered from the infected discs of dogs. Penetration and migration of barbed grass awns is also known to cause discospondylitis. 14
In pigs, the most frequently isolated bacteria are Erysipelothrix rhusiopathiae, Brucella suis, Arcanobacterium pyogenes, and Staphylococcus aureus. 23 In horses, Corynebacterium equi, Streptococcus equi subsp. zooepidemicus, Brucella abortus, E. coli, and Salmonella are commonly isolated. 23 In other farm species, Arcanobacterium pyogenes and Staphylococcus have been recovered. 15
It is believed that trauma to the cartilaginous end plates, vertebral bodies, and intervertebral discs may contribute to the development of discospondylitis. 17 Trauma produces microfractures and compromises the end plate vasculature. The capillary loops and anastomosing vessels make the vertebral end plates vulnerable to bacterial infections. 4 This is more common in dogs and horses. 2,8,9,17 The cervical vertebral column of the llama is comparable to that of the horse, where there is a relatively high degree of flexibility. 20 The vertebral bodies are supplied by a rich vascular network allowing microbes easy access via nutrient vessels. 21,25 Colonization of the disc and surrounding end plates by bacteria are also reported to occur via venous blood flow from abdominal and pelvic viscera to the ventral vertebral venous sinus and vertebrae. 4,10,21,25 In this llama, it is unclear if the finding of P. aeruginosa in cerebrospinal fluid was real or the result of sampling contamination from insertion of the needle through C3–C4.
The issue in the present case was the cause of discospondylitis and the origin and role of P. aeruginosa. Given the age of the animal, there is the possibility that umbilical infection occurred and that the organism localized in the disc of C3–C4. Alternatively, some traumatic event may have damaged the neck of this young animal, enhancing the potential for bacterial localization, be it from umbilicus or some other portal of entry.
