Abstract
Bovine failure of passive transfer (FPT), defined as inadequate transfer of colostral immunoglobulins from the dam to the calf, has been associated with increased risk in neonatal mortality. Currently, radial immunodiffusion (RID) assay is considered to be the gold standard in determining FPT in serum samples from calves. There are 2 commercial RID assays routinely used for serodiagnosis of FPT in calves: VET-RID and SRID. Discrepancies between results of these RID assays were observed in the authors' laboratory. The objective of this study was to compare 2 commercial RID assays by testing a paired panel of 30 blood samples collected from newborn Holsteins at birth before, and 24 hr after, ingestion of colostrum, a commercial bovine reference serum, and a panel of different concentrations of 2 purified bovine immunoglobulin G (IgG) products. Overall, the results of this study showed a high level of discrepancy and poor agreement between the 2 RID kits. The interassay precision study revealed lower between-run coefficients of variation for the VET-RID kit compared with the SRID kit. The spiking and recovery study using purified bovine IgG products demonstrated that the VET-RID kit more closely approximates the expected concentrations of the purified bovine IgG products, whereas the SRID kit consistently overestimates the concentration of purified bovine IgG products. It was concluded that this may be due to inaccuracies in the internal standards of the SRID kit.
The transfer of maternal antibodies from the dam to the neonatal calf, referred to as passive transfer of immunity, is important in reducing mortality in calves. In ruminants, the syndesmochorial placenta prevents transmission of immunoglobulins to the fetus in utero so newborn calves need to absorb colostrum containing maternal immunoglobulin G (IgG) soon after birth. Failure of passive transfer (FPT) of adequate amounts of maternal antibody (≥1,000 mg/dl) is an important condition to identify soon after birth because newborn calves are predisposed to infections, which can lead to high morbidity and mortality. 1,2,4
Various methods have been used to measure the passive transfer status in neonatal calves. The radial immunodiffusion (RID) assay, 2 the turbidimetric immunoassay (TMI), 3 and the lateral-flow immunoassay 2,6 using species-specific antibody are methods that directly measure serum bovine IgG concentration. Other methods, such the use of refractometry to determine serum total solids concentration, 7,11 zinc sulfate and sodium sulfite turbidity tests, 5,9,11 glutaraldehyde coagulation tests, 10 and measurement of serum γ-glutamyltransferase activity 8,12 estimate serum IgG concentration based on concentration of total globulin or other proteins whose passive transfer is statistically associated with transfer of IgG. Currently, RID is considered the gold standard for determining FPT in calves. 2 Two commercial RID assays routinely used for serodiagnosis of FPT in calves include the VET-RID kit a and the SRID kit. b The VET-RID kit contains 3 internal standards with IgG concentration of 620, 2,500, and 5,000 mg/dl and 3 plates with 16 wells per plate requiring 5 μl of sample applied to each well. The SRID kit contains 4 standards with IgG concentration of 400, 800, 1,600, and 3,200 mg/dl and 5 plates with 12 wells per plate requiring 3 μl of sample applied to each well. Discrepancies in the results of these 2 RID assays were noted in the authors' laboratory (Clinical Immunology Laboratory, Kansas State University, Manhattan, KS). It was hypothesized that the variation in the results was due to inaccuracies in the internal standards of 1 of the kits. Therefore, the objective of the present study was to compare the bovine IgG concentration determined by these 2 commercially available RID kits. To the authors' knowledge, agreement between results of these 2 RID assays has never been reported.
Thirty neonatal Holstein calves from the Kansas State University Dairy were used in this study (Jan–Feb 2006). Blood samples collected before, and 24 hr after, ingestion of colostrum by jugular venipuncture were transferred into tubes without anticoagulant. Serum was obtained from clotted blood samples <18 hr after collection and frozen in aliquots. For the recovery study, a reference serum
c
with 2,800 mg/dl of IgG and 2 purified bovine IgG products from Sigma
d
(5,000 mg/dl of IgG concentration) and the National Veterinary Service Laboratories
e
(NVSL; 3,100 mg/dl of IgG concentration) were tested using both kits. A precolostral serum pool was prepared by mixing equal volumes of precolostral sera from 20 calves. Two-fold dilutions of the purified bovine IgG products designated as bovine IgG-SIGMA 1–4 and bovine IgG-NVSL 1–3 were prepared by spiking separate aliquots of the precolostral serum pool with the purified bovine IgG products to achieve the following concentrations: 5,000, 2,500, 1,250, and 625 mg/dl for bovine IgG-SIGMA, and 3,100, 1,550, and 775 for bovine IgG-NVSL. All samples were tested in duplicate and both kits were used simultaneously. The respective assays were performed according to the manufacturers' recommendations. After incubation at room temperature for 20 hr, the samples and the precipitin ring diameter of the internal standards were measured by using an immunoviewer.
f
The IgG concentration in test samples was then determined by comparing the diameter of the zone of precipitation with a standard curve generated by assaying the internal standards of each kit. All statistics were performed using JMP software, version 6.
g
The interassay precision, expressed as a percentage of the coefficient of variation (% CV) was determined from the mean of duplicates of the diameter of the zone of precipitation of each kit's internal standards, the IgG concentrations of the reference serum, and different dilutions of the bovine IgG products from 8 separate runs; the data were expressed as mean (
In the present study, 60 serum samples from neonatal calves were collected. The samples included 30 precolostral serum samples and 30 postcolostral serum samples. For the 30 precolostral serum samples, the IgG concentration was below detectable limits of both assays (<400 mg/dl for the SRID kit and <620 mg/dl for the VET-RID kit). Eight of the 30 postcolostral sera were excluded because of having IgG concentrations higher than the detection limits of the 2 kits (>5,000 mg/dl for the VET-RID kit, 2 samples; >3,200 mg/dl for the SRID kit, 6 samples). Out of 22 postcolostral calf sera, 21 samples (95.5%) had different IgG concentrations using both RID kits (range of differences: 50–1,200 mg/dl). Twelve samples (54.5%) had higher IgG concentrations using the VET-RID kit (range of differences: 50–1,200 mg/dl), and 9 samples (40.9%) had higher IgG concentrations using the SRID kit (range of differences: 50–500 mg/dl). Using the VET-RID kit, the serum IgG concentration ranged from 700–4,100 mg/dl. Using the SRID kit, the serum IgG concentration ranged from 1,200–3,100 mg/dl. Using the VET-RID kit, 1 calf sample had IgG concentration of 700 mg/dl, which is below the cutoff value of 1,000 mg/dl used as a criterion for FPT, whereas all serum samples had IgG concentrations higher than 1,000 mg/dl using the SRID kit (Fig. 1). Results of interassay precision evaluations for the diameter of the zone of precipitation of each kit's internal standards are provided in Table 1. Using reference serum and different concentrations of bovine IgG products, the range of interassay precision for the VET-RID kit was 1–4.2% and for the SRID kit was 3–4.5%.
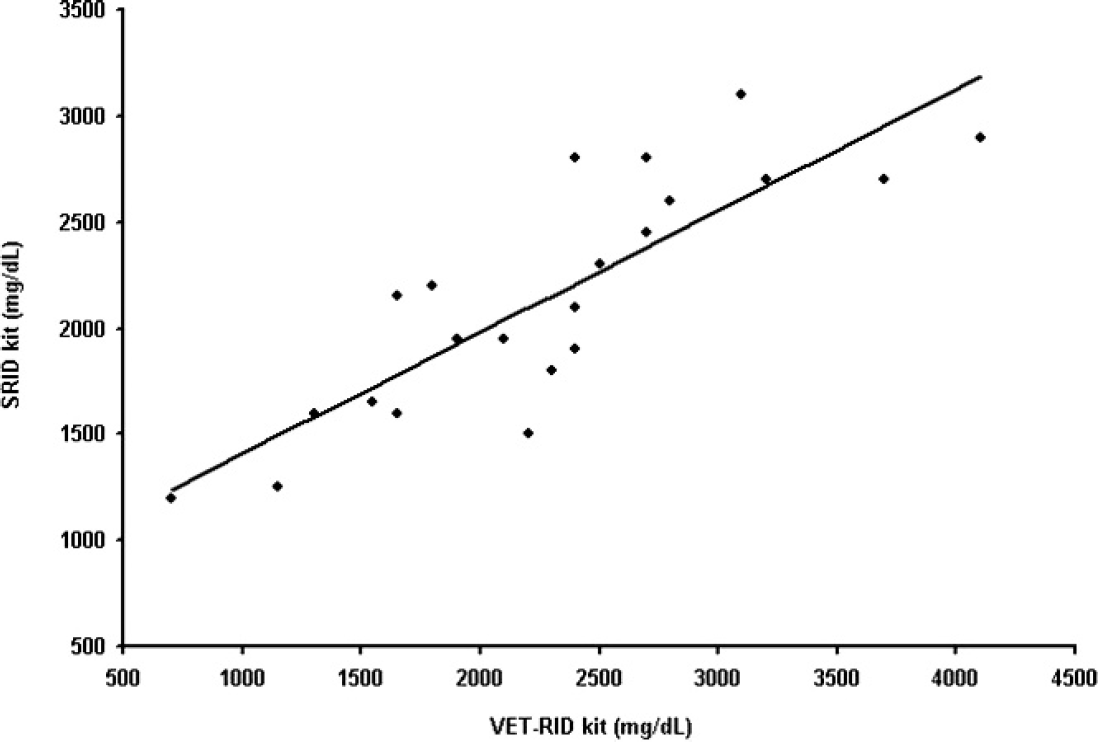
Scatter diagrams illustrating the associations between bovine immunoglobulin G concentrations (mg/dl) in 22 postcolostral calves sera measured by the VET-RID and SRID kits. Solid black line indicates the regression line (
The coefficient of determination (
Interassay precision of the diameter of the zone of precipitation (mm) of the internal standards of the VET-RID and SRID kits from 8 separate runs.*
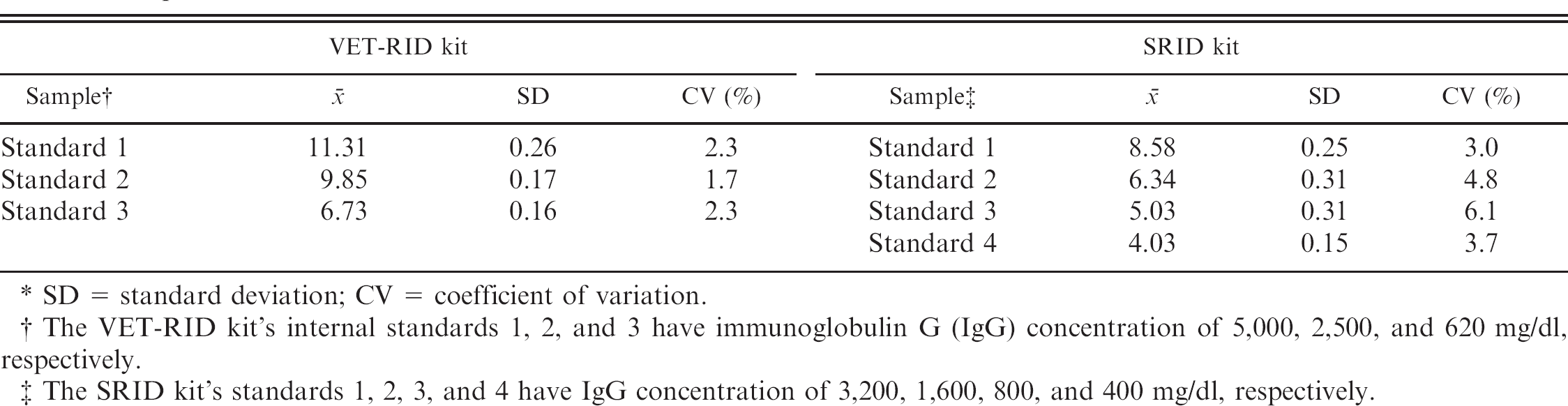
SD = standard deviation; CV = coefficient of variation.
The VET-RID kit's internal standards 1, 2, and 3 have immunoglobulin G (IgG) concentration of 5,000, 2,500, and 620 mg/dl, respectively.
The SRID kit's standards 1, 2, 3, and 4 have IgG concentration of 3,200, 1,600, 800, and 400 mg/dl, respectively.
Recovery study using commercial standards and the VET-RID kit.
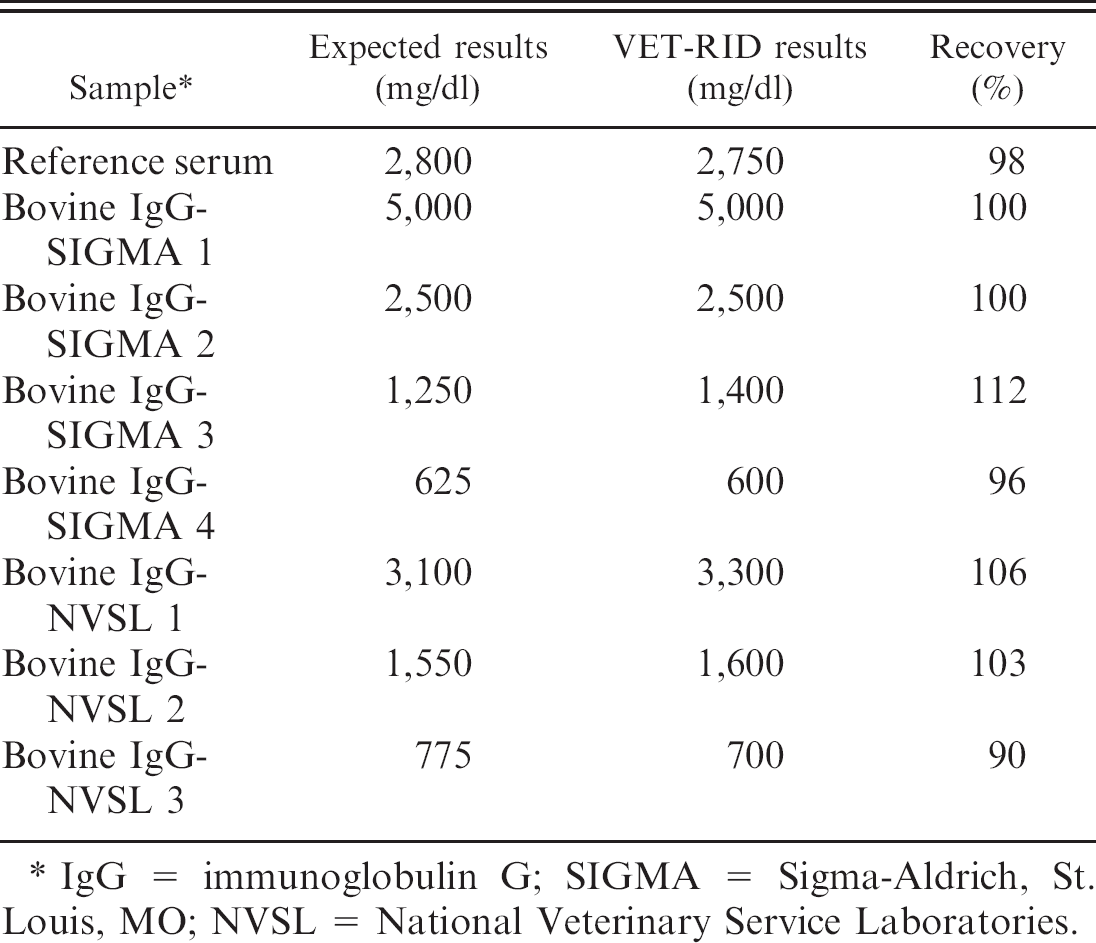
IgG = immunoglobulin G; SIGMA = Sigma-Aldrich, St. Louis, MO; NVSL = National Veterinary Service Laboratories.
Failure of passive transfer of humoral immunity in calves is defined as inadequate transfer of colostral immunoglobulin from dam to calf on the day of birth. Calves with FPT are at risk for life-threatening infections because of inadequate concentrations of protective maternal antibodies. Therefore, an accurate test is essential for targeted intervention and treatment strategies. The RID assay has long been recognized as the gold standard for determining IgG concentrations in bovine serum. Using both commercially available RID kits for measuring IgG concentration in bovine sera, bovine IgG was reported as not detectable in precolostral bovine sera. Failure of the RID kits to measure IgG concentrations in sera from 8 calves (VET-RID kit: 2 samples; SRID kit: 6 samples) in the present study was because IgG concentrations were higher than the high-concentration internal standards provided in both RID kits. Overall, the large bias and wide limits of agreement showed a poor agreement between the 2 RID assays. The interassay precision study revealed lower between-run CVs for the VET-RID kit compared with the SRID kit. The spiking and recovery study using purified bovine IgG products demonstrated that the VET-RID kit more closely approximates the expected concentrations of the purified bovine IgG products, whereas the SRID kit consistently overestimates the concentrations of purified bovine IgG products. It was concluded that this may be due to inaccuracies in the internal standards of the SRID kit.
Recovery study using commercial standards and the SRID kit.
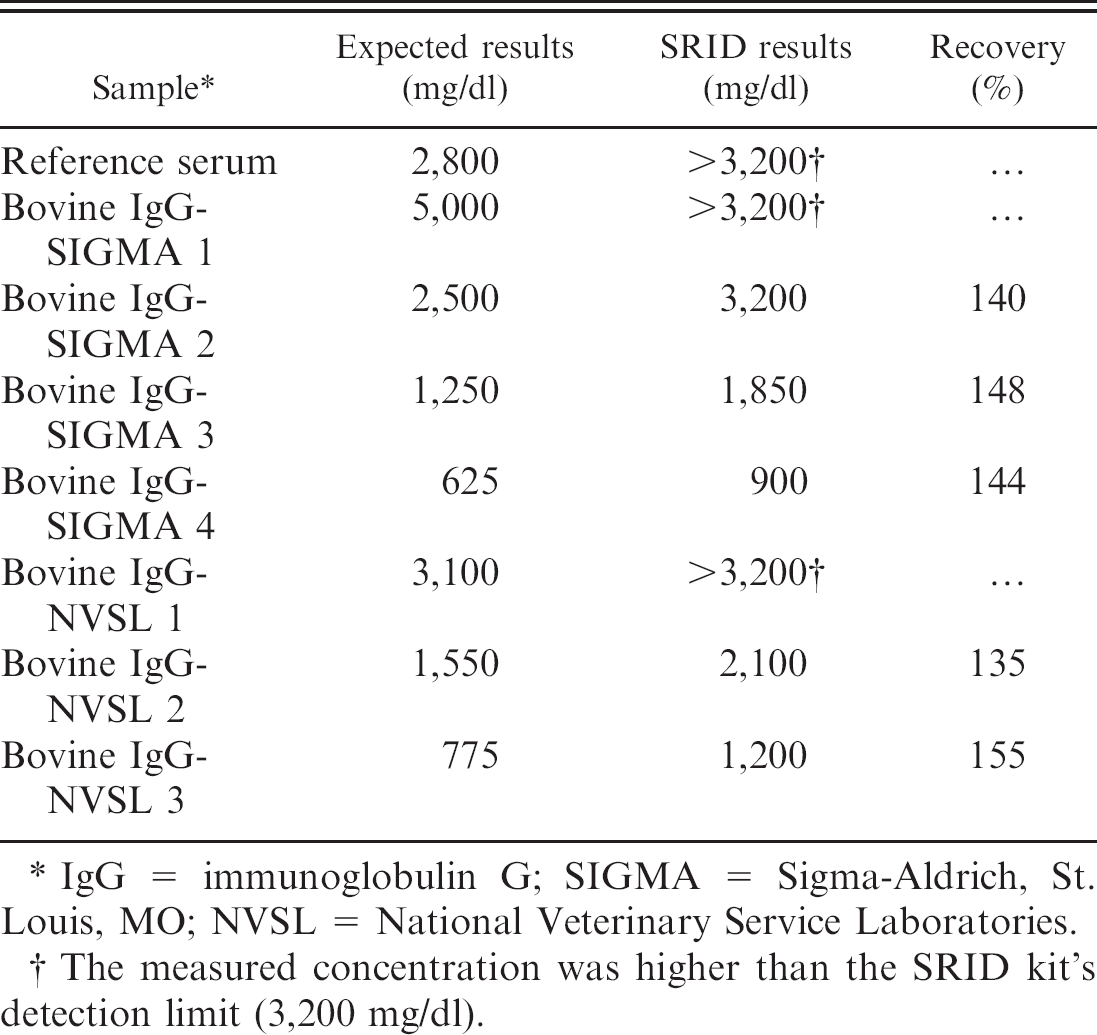
IgG = immunoglobulin G; SIGMA = Sigma-Aldrich, St. Louis, MO; NVSL = National Veterinary Service Laboratories.
The measured concentration was higher than the SRID kit's detection limit (3,200 mg/dl).
Footnotes
a.
Bethyl Laboratories, Montgomery, TX.
b.
VMRD Inc., Pullman, WA.
c.
Bethyl Laboratories, Montgomery, TX.
d.
Sigma-Aldrich, St. Louis, MO.
e.
National Veterinary Service Laboratories, U.S. Department of Agriculture, Ames, IA.
f.
Grafar Corp., Detroit, MI.
g.
SAS Institute, Cary, NC.
