Abstract
A case of fatal pulmonary hemorrhage in a 6–year-old American Paint mare with a 2–week history of intermittent coughing, fever, and epistaxis is described. Significant macroscopic abnormalities at postmortem examination were restricted to the respiratory system, and microscopically, severe pulmonary hemorrhage with suppurative bronchopneumonia was found. Actinobacillus equuli subsp. haemolyticus was cultured from a transtracheal wash performed antemortem as well as from the lungs at necropsy. The presence of airway-associated hemorrhage in conjunction with bacterial bronchopneumonia suggested endothelial damage caused by a locally elaborated bacterial toxin, possibly produced by the A. equuli strain isolated from the lungs. The objective of this report was to indirectly document the presence of hemolysin repeat in structural toxin (RTX) in the lungs of the reported mare. A real-time polymerase chain reaction (PCR) assay targeting the recently described aqx gene of A. equuli subsp. haemolyticus was established and validated. Transcriptional activity of the aqx gene was used as a surrogate method to document toxin production. Real-time PCR analysis of the transtracheal fluid and lung tissue of the affected mare confirmed the presence and the transcriptional activity of the aqx gene at the genomic (gDNA) and complementary DNA (cDNA) levels, respectively. The presence of pneumonia associated with hemorrhagic pulmonary fluid and the culture of large numbers of hemolytic A. equuli should prompt the clinician to consider endothelial damage caused by bacterial toxins.
A 6-year-old, 90-day-pregnant American Paint mare presented to the University of California Veterinary Medical Teaching Hospital (VMTH) for a 2-week history of intermittent coughing, fever, and bilateral epistaxis. Twenty-four hours before presentation, the mare developed increased respiratory effort, lethargy, and anorexia. No medical treatment was performed before admission to the VMTH. The mare was housed on pasture with other brood mares and was fed alfalfa and grass hay. Vaccinations (equine influenza, tetanus, equine eastern and western encephalitis, rhinopneumonitis, and West Nile) and de-worming were current. No prior history of illnesses or known exposure to toxins was reported by the owner. Upon arrival at the hospital, the mare was bright and alert, covered in sweat, with normal rectal temperature (37.6°C), tachycardie (60 beats per minute), and tachypneic (84 breaths per minute). The mare had a body condition score of 7/9. There was dried bloody nasal discharge from the right nostril. The mare displayed nostril flare and increased abdominal effort during expiration. The lungs auscultated harsh in all lung fields with rattling noise over the trachea. The mare had normal gastrointestinal borborygmi and was seen to urinate and defecate normally.
Initial blood work revealed low packed cell volume (25%, reference range: 30–46%), red blood cell count (4.97 × 106/μl, ref. range: 6.2–10.2 × 106/μl), and hemoglobin (10.5 g/dl, ref. range: 11.2–17.2 g/dl). Leukocyte count (6,370/μl ref. range: 5,000–11,600/μl), platelet count (127,000/μl, ref. range: 100,000–225,000/μl), plasma protein concentration (5.8 g/dl, ref. range: 5.8–8.7 g/dl), and plasma fibrinogen concentration (300 mg/dl, ref. range 100–400 mg/dl) were within normal limits. Mild lymphopenia (1,440/μl, ref. range: 1,600–5,800/μl) was attributed to stress. The clotting times activated partial thromboplastin time (42.2 seconds, ref. range: 31.5–48.5 seconds) and prothrombin time (14.9 seconds, ref. range: 14–17 seconds) were within normal limits. There was hyperglycemia (237 mg/dl, ref. range: 50–107 mg/dl) and hyperlactatemia (5.9 mmol/liter. ref. range: ≤ 2.0 mmol/liter), whereas electrolytes and venous blood gases were within normal limits. Thoracic ultrasound revealed severe and diffuse roughening of the pleura in all lung fields with focal areas of pulmonary consolidation bilaterally.
Based on the presenting signs, blood work, and thoracic ultrasound, an acute, diffuse, inflammatory pulmonary disease was suspected. Thoracic radiographs revealed diffuse, irregularly margined, nodular, interstitial opacities in all lung fields. A transtracheal wash for cytology, culture, and serum for Coccidioides immitis serology were collected. The transtracheal wash was grossly hemorrhagic, interpreted as marked purulent, septic inflammation, and revealed large numbers of red blood cells and moderately to markedly degenerate neutrophils, with fewer macrophages and rare lymphocytes. With Wright-Giemsa staining, many rod-shaped bacteria were seen extracellularly, often in clumps, and intracellulary in neutrophils and macrophages. Culture grew large numbers of Actinobacillus equuli subsp. haemolyticus in pure culture. The mare was seronegative for C. immitis by agar gel immunodiffusion.
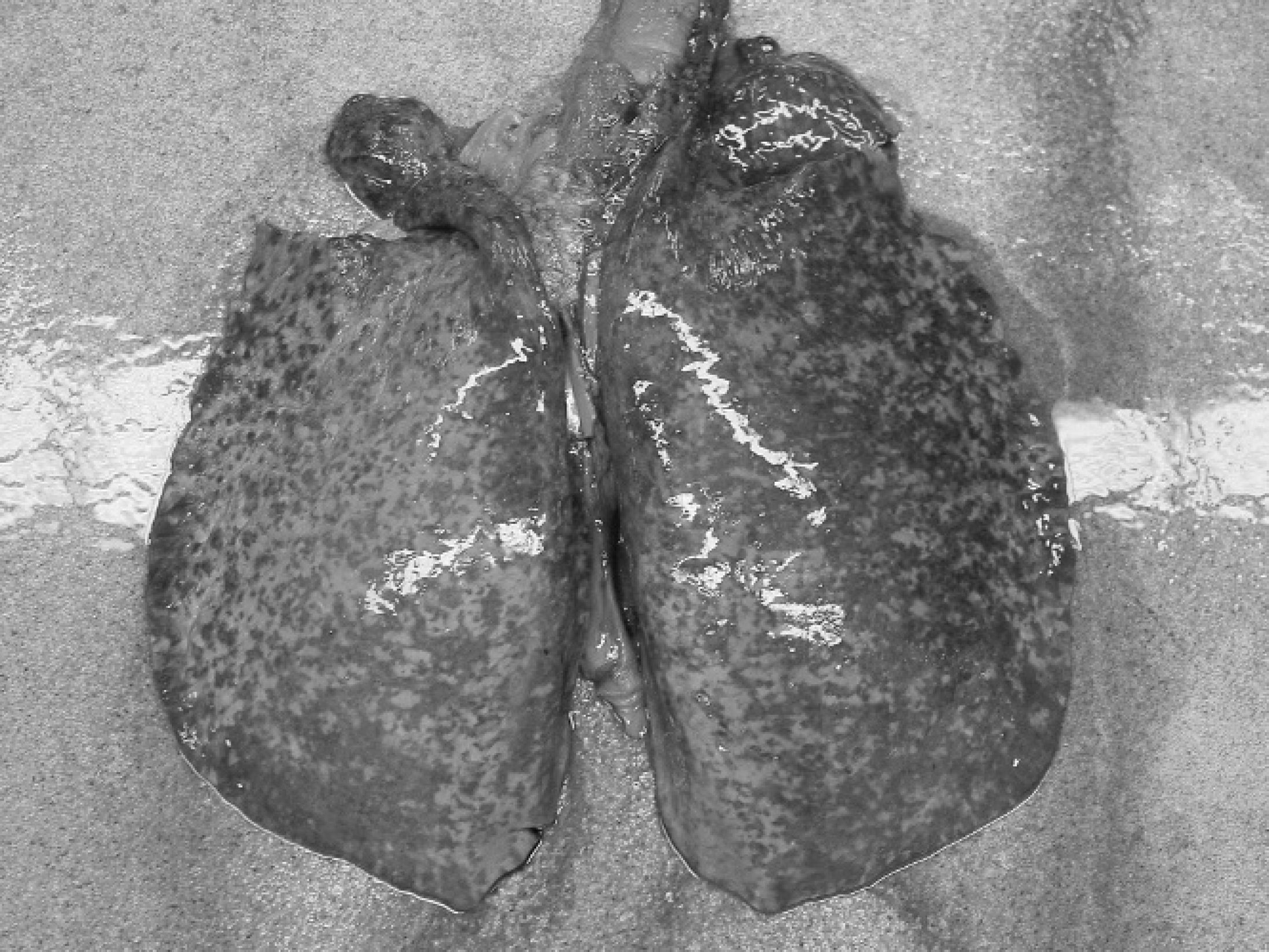
Gross specimen of lungs showing generalized lobular hemorrhage throughout the pulmonary parenchyma.
Shortly after admission and initial work up, the mare collapsed and died despite resuscitation attempts. Significant macroscopic abnormalities at postmortem examination were restricted to the respiratory system. The lungs had multifocal to coalescing hemorrhage throughout the parenchyma (Fig. 1). Small and large bronchi, trachea, nasopharynx, and nasal cavity, all contained clotted blood. Aerobic and anaerobic cultures of lung tissue grew large numbers of A. equuli subsp. haemolyticus in pure culture. Tissue samples were fixed in 10% buffered formalin, embedded in paraffin, and serially sectioned for routine hematoxylin and eosin staining. Lung tissue was also frozen at −80°C.
Microscopically, there was severe pulmonary hemorrhage and moderate, subacute, suppurative bronchopneumonia. The hemorrhage had a multifocal to coalescing distribution within the parenchyma and was more closely associated with airways than small blood vessels. Numerous bronchi and bronchioles contained free erythrocytes and clotted blood; in some areas, alveoli were distended with erythrocytes and fibrin, but alveolar architecture was preserved and there was minimal inflammation (Fig. 2). Although the hemorrhage was primarily acute, the presence of small numbers of hemosiderin-laden macrophages indicated that it had likely been occurring at a low level for at least a few days. Other microscopic lesions included mild, multifocal peribronchial and peribronchiolar fibrosis, which was consistent with chronic bronchitis. The presence of airway-associated hemorrhage in conjunction with bacterial bronchopneumonia suggested endothelial damage caused by a locally elaborated bacterial toxin, possibly produced by the A. equuli strain isolated from the lungs.
To test the hypothesis that the isolated strain of A. equuli subsp. haemolyticus produced hemolysin RTX toxins, the authors established a real-time polymerase chain reaction (PCR) assay to document transcriptional activity of the A gene of the aqxCABD operon gene, as a surrogate method for toxin production. The assay is based on the detection of a specific 128 base-pair-long product of the aqx A gene of A. equuli subsp. haemolyticus (GenBank accession number AF381185; oligonucleotides sequences 5′–3′: forward primer AGTGCCGGTTTTGCATTAGC, reverse primer CGTTGCGCAAGAATATAGCCT, probe 6-FAMCTGCTGGA). Amplification efficiency was calculated from the slope of a standard curve generated on 10–fold diluted A. equuli subsp. haemolyticus-positive DNA sample. High amplification efficiency for the target gene of 93% indicated a high analytical sensitivity. Analytical specificity was verified by sequencing PCR products for the target gene, in addition to testing non-aqx A gene harboring A. equuli subsp. equuli isolates from 5 septicemic foals and a variety of different microorganisms, including Klebsiella pneumoniae, Enterobacter spp., Bordetella bronchiseptica, Salmonella spp., Pasteurella spp., Pseudomonas spp., Enterococcus spp., Streptococcus equi subsp. zooepidemicus, and Staphylococcus aureus. Extraction of nucleic acids (genomic DNA [gDNA] and RNA) from transtracheal fluid samples and frozen lung tissue of the study case was performed according to the manufacturer using a commercial available extraction kit. a Complementary DNA (cDNA) was generated using previously reported protocols. 12 PCR reactions contained 400 nM of each primer, 80 nM of the TaqMan probe and master mix, b and 1 μl of gDNA or 5 μl of cDNA sample in a final volume of 12 μl. The samples were amplified in a combined thermocycler/fluorometer c for 2 minutes at 50°C, 10 min at 95°C, and then 40 cycles of 15 seconds at 95°C and 60 seconds at 60°C. Real-time PCR analysis of the transtracheal fluid and lung tissue of the affected mare confirmed the presence and the transcriptional activity of the aqx A gene at the gDNA and cDNA level, respectively. None of the 5 A. equuli subsp. equuli isolates from septicemic foals and none of the selected non-Actinobacillus microorganisms tested PCR-positive for the aqx A gene, confirming the specificity of the PCR assay.
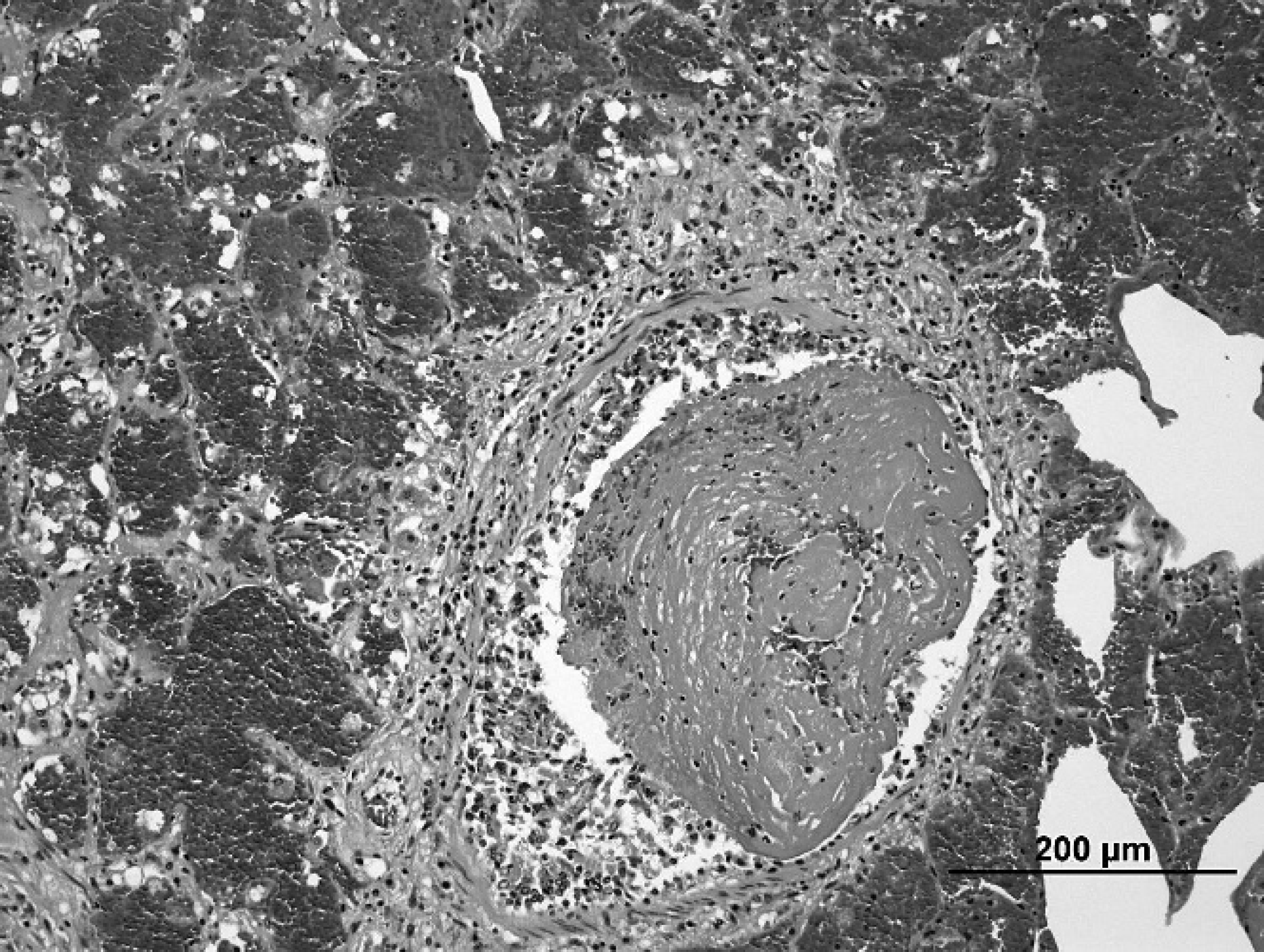
Section of lung showing intrabronchiolar clotted blood and fibrin, and hemorrhage within alveoli in surrounding parenchyma. Hematoxylin and eosin stain. Bar = 200 μm.
RTX toxins are pore-forming proteins of various Gram-negative bacteria that act as virulence determinants for several human and animal pathogens. 8 Aqx protein, a new member of the RTX family, has recently been shown by genetic and phenotypic assays to be present only in hemolytic A. equuli isolates from horses. 2,10 These hemolytic strains have been reclassified as A. equuli subsp. haemolyticus, whereas nonhemolytic isolates are classified as A. equuli subsp. equuli. 6
Both hemolytic and nonhemolytic A. equuli strains are part of the normal flora of the oral cavity, the upper respiratory tract, and the gastrointestinal tract of horses. 14 These opportunistic pathogens have been associated in adult horses with pneumonia, pleuropneumonia, arthritis, mastitis, metritis, peritonitis, endocarditis, meningitis, and stillbirth. 1,5,7,9,11,15,16 Interestingly, only A. equuli subsp. equuli is associated with septicemia in foals. 6 Pulmonary hemorrhage can have many etiologies with exercise-induced pulmonary hemorrhage, lung abscesses, and pleuropneumonia most commonly recognized in horses. A. equuli subsp. haemolyticus has, to our knowledge, not been directly associated with pulmonary hemorrhage. The generalized distribution of the hemorrhage and inflammation throughout the lung fields is highly suggestive of endothelial damage caused by a bacterial toxin. It is very likely that the A. equuli subsp. haemolyticus isolate in this case elaborated toxins that contributed to pulmonary alveolar capillary damage and hemorrhage in the study case. These findings are supported by the detection of the aqx hemolysin gene of A. equuli subsp. haemolyticus and its transcriptional activity from the transtracheal wash and the lungs. However, the detection of gene transcripts is not equal to the detection of the toxin, but instead, represents a surrogate method for protein synthesis. The study case shares some common clinical and pathological features with porcine pleuropneumonia caused by A. pleuropneumoniae, an obligate pathogen of the porcine respiratory tract. Clinical signs of affected pigs are strikingly similar to the study horse and include depression, fever, anorexia, severe respiratory distress, and hemorrhagic nasal discharge. 13 Further, pulmonary lesions associated with peracute and acute A. pleuropneumoniae infection are characterized by edema, inflammation, hemorrhage, and necrosis. 3 The pathophysiologic mechanisms associated with Actinobacillus pleuropneumoniae infection in pigs are well characterized and can mostly be attributed to RTX toxins, which exert direct cytotoxic effects on various cell types, such as endothelial cells, as well as indirect effects by stimulating release of inflammatory mediators from activated phagocytes. 4 It is suspected that a similar mechanism may be responsible for the pathological findings observed in the mare, because A. equuli subsp. haemolyticus produces similar virulence determinants (i.e., Aqx toxin).
This case represents an unusual presentation of A. equuli subsp. haemolyticus infection. The presence of pneumonia associated with hemorrhagic pulmonary fluid and the culture of large number of hemolytic A. equuli should prompt the clinician to consider endothelial damage caused by bacterial toxins. Although no treatment was able to be performed in the study case, the A. equuli subsp. haemolyticus isolate was susceptible to a routine panel of tested antimicrobials, and early treatment would most likely have lead to clinical improvement.
Footnotes
a.
DNeasy Tissue Kit, Qiagen, Valencia, CA.
b.
TaqMan Universal PCR Master mix, Applied Biosystems, Foster City, CA.
c.
ABI PRISM 7700 Sequence Detection System, Applied Biosystems, Foster City, CA.
