Abstract
Influenza A virus (FLUAV) causes acute respiratory disease in humans and a variety of animal species. The virus tends to remain within the species of origin; nonetheless, naturally occurring cross-species transmission of FLUAV has been periodically documented. Multiple cross-species transmissions of FLUAV have been reported from companion animals and captive wild animals, neither of which is historically considered as natural hosts of FLUAV. In the fall of 2010, mink (Mustela vison) inhabiting a 15,000-head mink farm in the Midwest United States experienced persistent severe respiratory distress and nose and/or mouth bleeding. Mink losses averaged approximately 10 animals per day. Six dead mink at 6 months of age were submitted to the Iowa State University Veterinary Diagnostic Laboratory for diagnostic investigation. Gross and microscopic examinations revealed that all 6 mink had hemorrhagic bronchointerstitial pneumonia. Hemolytic Escherichia coli was isolated from lungs, probably accounting for hemorrhagic pneumonia. All animals tested negative for Canine distemper virus and Aleutian mink disease virus. Interestingly, FLUAV of H1N2 subtype, which contained the matrix gene of swine lineage, was detected in the lungs. Serological follow-up on mink that remained in the ranch until pelting also confirmed that the ranch had been exposed to FLUAV of H1 subtype (δ clade). The case study suggests that FLUAV should be included in the differential diagnosis when mink experience epidemics of respiratory disease. Since the source of FLUAV appeared to be uncooked turkey meat, feeding animals fully cooked ration should be considered as a preventive measure.
Influenza A virus (FLUAV; family Orthomyxoviridae, genus Influenzavirus A) causes acute respiratory disease with fever in humans and a variety of animal species. 23 In animals, FLUAV infection has been commonly identified in pigs, horses, marine mammals, and poultry. The virus and disease were typically named after its host of isolation (e.g., Swine influenza virus [SIV], Equine influenza virus, and Avian influenza virus [AIV]). Each of these viruses tends to remain within the species of origin and maintain distinct genetic profiles from each other. Nonetheless, naturally occurring cross-species transmission of FLUAV has been periodically documented.1,3,8 Because FLUAV can be transmitted between humans and animals,7,21 the virus is a zoonotic pathogen. Most of the cross-species transmission events are self-limiting without any further implication. In some cases, such an event introduces a new FLUAV strain to a new population through adaptation or reassortment process.5,11,14
Multiple cross-species transmissions of FLUAV have been reported from companion animals3,16-18and captive wild animals,4,9,13 neither of which is historically considered natural hosts of FLUAV. While exact mechanisms for successful cross-species transmission require further study, such an event brings a new perspective and challenge to the influenza ecology, which should be taken into consideration when dealing with respiratory problems in those animal species. The present report describes a natural outbreak of FLUAV infection in a mink (Mustela vison) farm.
In mid-October 2010, ranch-raised mink in the midwestern United States experienced persistent severe respiratory distress, with death loss of approximately 10 animals per day. The episode of widespread coughing and death persisted for a period of 6–8 weeks with a daily mortality rate up to 3% during the acute stage. The farm had 15,000 mink, of which 3,500 were breeding females and the rest kits and growers. The mink were housed in adjacent pens in 2 rows, 4 rows, or 6 rows in a shed configuration. At the time of outbreak, the mink were grouped in male–female pairs. No other domestic animals except farm dogs were housed on the ranch. The mink were fed poultry meat. Affected animals were lethargic, had mucoid and watery discharge from the eyes, as well as hemorrhage from the mouth and nose. The most notable clinical sign was severe cough, which could be heard throughout the ranch. The clinical problem affected both adults and kits, with affected mink spread randomly across the ranch. There was no warning as to which mink were more likely to be acutely affected and die. The death loss was also widespread across the ranch during the outbreak, although it was usually concentrated in a specific area of the farm on any particular day. Antibiotics administered during the outbreak were erythromycin, neomycin, and trimethoprim sulfa. The treatments slowed death losses, but after treatments ceased, the death losses escalated back to higher than expected rates.
Six dead 6-month-old mink nearing maturity were received by the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL; Ames, IA) for diagnostic investigation. Gross examination revealed that all 6 mink had locally extensive or diffuse hemorrhagic pneumonia. No gross lesions were apparent in other body systems. Microscopically, lungs had severe diffuse congestion with locally extensive mixed bronchointerstitial pneumonia. Terminal airways had necrotizing bronchiolitis, plugs of mixed inflammatory and necrotic debris, and had moderate nonsuppurative peribronchiolar and perivascular cuffs. There was mild acute mixed periportal hepatitis in sections of liver. Some of the spleens had reactive follicles, and 1 mink had multifocal necrosis in the spleen. Mild nonsuppurative encephalitis was noted in the brain from 1 of the 6 mink.
Hemolytic Escherichia coli was isolated from lung tissue from each of the 6 animals submitted. No nucleic acid of Aleutian mink disease virus was detected in spleens by a real-time polymerase chain reaction (PCR), which was performed at the Wisconsin Veterinary Diagnostic Laboratory (Madison, WI). Lungs, brains, and bladder swabs tested negative for Canine distemper virus by a fluorescent antibody test of frozen tissue sections and/or reverse transcription (RT)-PCR assays. 15 Because lung lesions were suggestive of epitheliotropic viral insult, the samples were tested for FLUAV by immunohistochemical staining 22 and RT-PCR assay. 6 Virus antigen (nucleoprotein) was detected within lesions. Viral nucleic acid corresponding to matrix (M) and nucleoprotein (NP) genes was detected in fresh lung tissue. Hemagglutinin (H) and neuraminidase (N) genes of the virus were subtyped as H1N2 using the RT-PCR assay. By sequence analysis, 12 the virus appeared to be of swine origin as its M gene (GenBank accession no. JN409444) showed close genetic relatedness to the M gene of U.S. endemic SIV (Fig. 1). Partial sequence (241 bases) of NP gene also showed genetic relatedness to the NP gene of SIV. Unfortunately, sequencing for other genes and virus isolation attempts were unsuccessful due to the suboptimal condition of the samples. Approximately 2 months after the initial outbreak, serum samples were randomly collected from 20 mink during pelting and tested for FLUAV antibody. Nineteen out of 20 mink were strongly seropositive for FLUAV using a commercial enzyme-linked immunosorbent assay, 2 further supporting that the mink from this ranch were exposed to FLUAV. Hemagglutination inhibition (HI) tests 25 against 4 H1 subtype FLUAVs and 1 H3 subtype FLUAV demonstrated that the animals had significant levels of HI antibody (≥1:40) against H1 of δ clade 20 and no HI activity against H1 viruses of α, β, or γ clades or H3 virus of cluster 4 (Fig. 2), suggesting that the animals were exposed to a H1N2 FLUAV (δ clade).
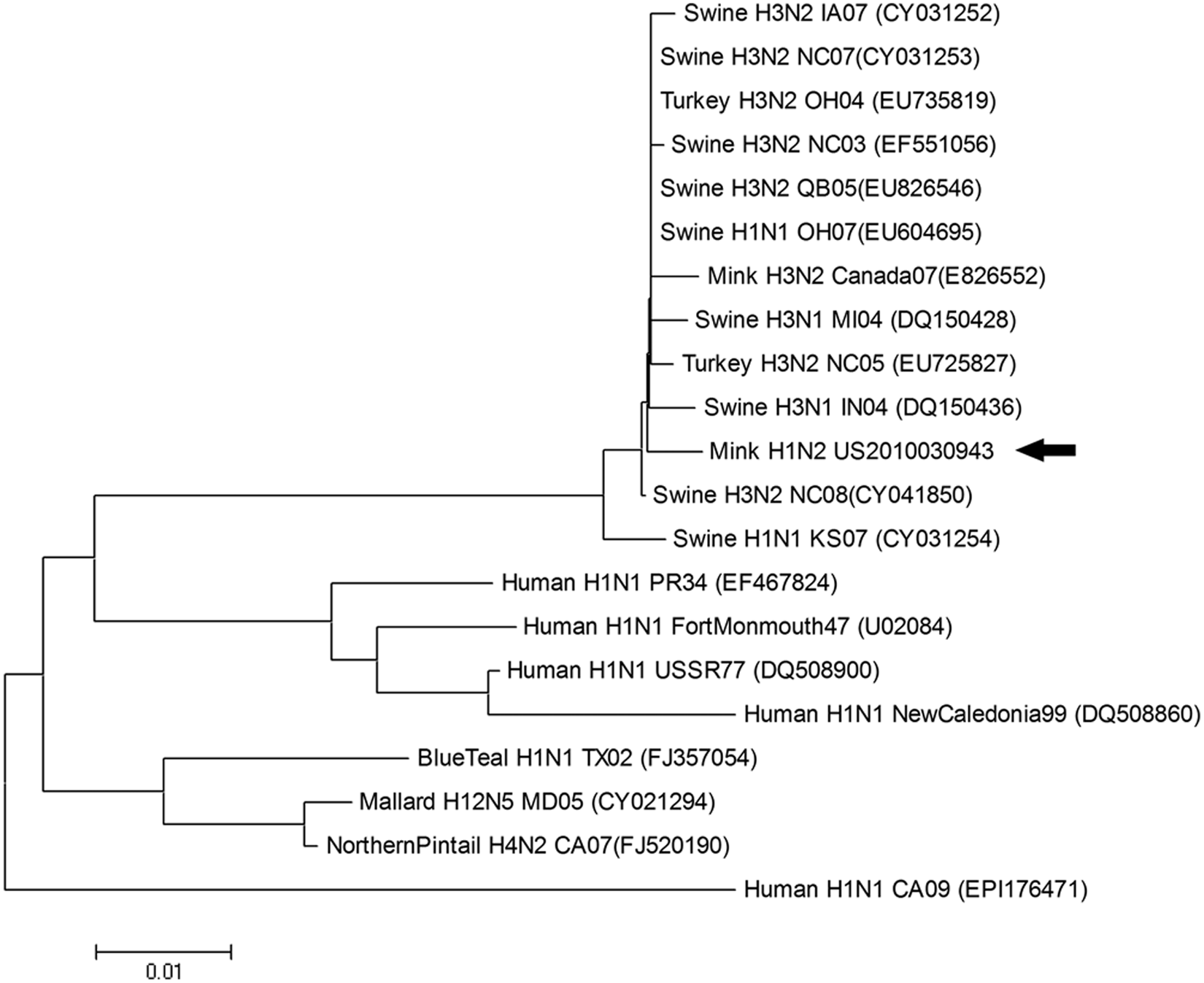
Phylogenetic relationship between H1N2 Influenza A virus from a Midwest U.S. mink ranch (US2010030943; arrow) and other influenza A viruses of human, swine, and avian origin based on nucleotide sequence of the matrix gene. GenBank accession numbers of reference viruses are in the parentheses. Phylogenetic analyses were conducted using MEGA (version 4) using the neighbor-joining tree building method, with 1,000 bootstrap replicates.
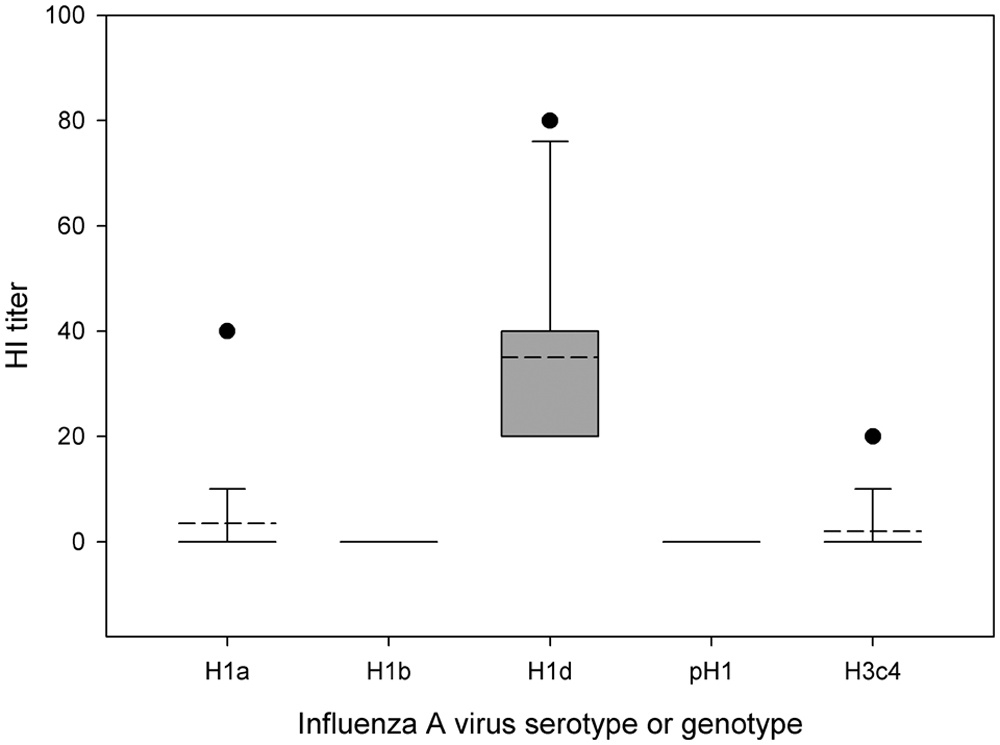
Hemagglutination inhibition (HI) antibody titers of mink sera (n = 20) against various H1 Influenza A virus clusters (alpha [H1a], beta [H1b], delta [H1d]), pandemic 2009 H1 strain (pH1), and H3 cluster 4 influenza virus (H3c4). The sera were randomly collected from surviving mink 2 months after initial confirmation of H1N2 influenza infection in the index farm. Dashed line represents the mean. When not present, dashed line and solid line are the same. Box (or solid line) represents 10–90 percentiles of data points or 5 and 95 percentile in case no error bar is present. Error bar represents 5 and 95 percentiles of data points. Solid closed circles display outliers.
The outbreak of severe hemorrhagic pneumonia in the index mink ranch was due to concurrent infection with FLUAV and E. coli based on microscopic lesions and identification of the corresponding agents. While the severity and persistence of clinical disease were due to FLUAV infection, the fatal hemorrhagic pneumonia and high mortality rate were likely attributed to secondary bacterial infection; a high mortality is not a common feature of FLUAV infection in mammals although FLUAV was reported to be the sole etiology of hemorrhagic pneumonia in some incidences. 3 A similar clinical presentation has been reported with FLUAV infection in racing dogs when the animals were concurrently or subsequently infected with bacteria. 24 The clinical outcome, particularly mortality, perhaps could be mitigated with timely use of appropriate antibiotics in acute outbreaks of influenza.
The mink were not fed pork byproducts, and there were no swineherds nearby; therefore, it was unexpected to identify FLUAV of swine origin. Interestingly, the owner of the mink ranch mentioned feeding of animals with turkey meat when chicken meat could not be supplied. Although a formal epidemiological study was not conducted, it is reasonable to speculate that the source of FLUAV was turkey because cross-species transmission of SIV, particularly H1N2 and H3N2, to turkeys commonly occurs, particularly after triple reassortant H3N2 FLUAV emerged in U.S. swine.1,19 Since the source of meat may have a role in bacterial and/or viral contamination in ration, the clinical episode in the mink ranch emphasizes the importance of carefully screening the source of meat as a preventive measure when mink are fed an uncooked ration. As a better intervention, feeding animals with fully cooked ration should be considered.
Mink, as well as some small mammals such as ferrets, guinea pigs, and mice, are known to be susceptible to FLUAV and have been frequently used in laboratories to study FLUAV derived from humans and other animal species. 23 Hence, it is not totally unexpected that mink or ferret fur farms undergo influenza outbreak when animals are fed FLUAV-contaminated meats. Indeed, there is a previous report of naturally occurring H3N2 FLUAV infection in a mink farm in Canada where animals were fed uncooked pork byproducts. 4 Therefore, veterinary professionals and mink producers should include FLUAV in the differential diagnosis when animals experience epidemics of respiratory disease.
With respect to influenza ecology, it has been reported that farmed mink can escape to the wild and establish a wild mink population or occasionally mingle with wild mink. 10 It would be of interest to assess whether free-ranging mink or other small mammals play a role in maintaining or transmitting FLUAV to humans or livestock. Furthermore, a careful investigation should be done to determine if these susceptible but historically unnatural host animals can be a “mixing vessel” of influenza viruses under captive status when exposed to FLUAV, either by feeding uncooked virus- contaminated rations or from farm workers with influenza-like illness. To this end, these species should be closely monitored for FLUAV infection.
Footnotes
Acknowledgements
The authors thank the Veterinary Diagnostic Laboratory staff (necropsy, histopathology, bacteriology, virology) for technical assistance in diagnostic investigation. Suzy Block is acknowledged for her excellent assistance in serological evaluation of mink sera.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The in-depth investigation was made possible, in part, by funding through cooperative agreement with the U.S. Department of Agriculture APHIS Wildlife Service for influenza virus in wild birds (no. 10-7100-0284-CA) and Veterinary Diagnostic Laboratory Research and Development Fund.
