Abstract
A protein chip based on surface plasmon resonance imaging (SPRI) was developed for detecting fish iridovirus antibody using a recombinant 50-kDa fragment of major capsid protein (MCP) as an antigen. The diagnostic potential of SPRI for measuring antibodies to the iridovirus MCP was compared with that of a conventional enzyme-linked immunosorbent assay (ELISA) using 40 juvenile rock bream (Oplegnathus fasciatus) serum samples in a nursery. There was a strong positive correlation between the SPRI and ELISA (n = 40, r = 0.939, P < 0.01). Therefore, this recombinant 50-kDa MCP can be used as an antigen for serological studies, and the SPRI, which is a label-free and high-throughput method, is potentially a valuable tool in the serodiagnosis of an iridoviral infection.
Iridoviruses are icosahedral cytoplasmic double-stranded DNA viruses with a genome size of between 170 and 200 kb. 21 Outbreaks of fish iridovirus infections are characterized by their wide geographic distribution and broad host spectrum, particularly at water temperatures of 25°C to 27°C. The cumulative mortality attributed to this disease is 50%-90% over a 2-month period. Histological lesions in affected fish include enlarged basophilic cells in the gill, kidney, heart, liver, and spleen. 19 Iridoviruses are associated with systemic disease in an increasing assortment of wild and cultured fish populations. 1,5,20 Iridoviruses cause significant mortality in important food fish species, 1 and there is the potential for the spread of iridoviral diseases between wild and cultured fish as well as between imported and indigenous species. 16
Several diagnostic methods for detecting iridovirus have been developed, which include cell culture, 14 immunofluorescence assay to detect viral antigen, 13 polymerase chain reaction (PCR), 11 and enzyme-linked immunosorbent assay (ELISA) to detect the antibodies (Abs) to the iridovirus. 10 Although these methods are quite reliable, they are time- and labor-intensive protocols. Most protein arrays currently developed rely on detection technologies that employ enzymatic or fluorescent tags. In contrast, surface plasmon resonance imaging (SPRI) is a label-free, surface-sensitive spectroscopic technique used to examine the bioaffinity interactions on thin gold films. 17 SPRI detects the changes in the refractive index within a short distance from the surface of a thin metal film as variations in the light intensity reflected from the back of the film and therefore does not require labeling. 2,3,9 SPRI has been successfully used to screen various bioaffinity interactions using proteins. 8 In this study, the 50-kDa fragment of the major capsid protein (MCP) gene 10 was used to develop a protein chip based on SPRI for detecting antibodies to iridovirus in rock bream sera. The diagnostic efficacy of SPRI in determining the presence of an iridoviral infection was compared with that of conventional ELISA.
Recombinant MCP (rMCP) was prepared using a prokaryotic expression system as described previously. 9 Briefly, the MCP gene (GenBank no. AY310918) was amplified by PCR using previously described primers (Table 1). 19 The PCR products were cloned into the prokaryotic expression vector pRSET. a , 18 The recombinant DNA was transformed into the BL21(DE3)pLysS host cells. The transformants were grown in 25 ml of Luria-Bertani medium b containing ampicillin c and chloramphenicol c to an optical density (OD) of 0.5. Protein expression was induced by adding isopropyl β-D-thiogalactopyranoside (final concentration of 1 mM). c The proteins were purified under denaturing conditions using an affinity purification system. d Polyclonal antibody was prepared by immunizing five 8-week-old ICR mice e subcutaneously with the purified rMCP (100 μg per mouse in 100 μl of phosphate-buffered saline [PBS] mixed with 100 μl of complete Freund adjuvant) followed by 2 booster injections at 2-week intervals (with 100 μl of incomplete Freund adjuvant). The mice were bled 7 days after the final booster injection for serum collection, and the serum was heat inactivated at 56°C for 30 minutes and stored at −80°C until needed.
Using rMCP as an antigen, evidence of an iridoviral infection was determined using 40 rock bream serum samples to detect the viral MCP-specific antibodies using an ELISA, as described elsewhere. 4 Briefly, a 96-well plate f was coated with rMCP (1 μg per well in 90 μl). The plate was blocked with PBS containing 1% skim milk and washed with PBS + 0.05% Tween-20 (PBS-T; pH 7.4). The sera for antibody analysis were obtained from fish (weight = 5–6 g, length = 5–7 cm, from more than 6 months old from 5 regions in the southern Korean sea, selected randomly) and tested at 1: 100 dilution in PBS-T. A mouse antirock bream-IgM monoclonal antibody (MAb) 6 diluted 1: 10 was used as the secondary antibody. Horse radish peroxidase-conjugate goat antimouse IgG (whole molecule) g diluted 1: 100 was used as the tertiary antibody. After incubating the diluted serum samples (90 μl/well) for 1 hour at 37°C, the plate was emptied and washed 3 times with PBS-T. Positive and negative rock bream control sera (5 samples each), h which were previously screened by rock bream iridovirus-specific PCR were used. In addition, 5 sea bream sera were used as the species-specific controls. For this, mouse MAb antisea bream IgM i was used as the secondary antibody. The presence of any bound tertiary antibody was detected using a substrate solution (0.1 M citric acid buffer, pH 4.0, 10 ml; 0.05 M 2,2′-azinobis[3-ethylbenzothiazoline-6-sulfonic acid] 250 μl; 35% H2O2 50 μl). The plate was stored in the dark at room temperature for 15 minutes to allow the color reaction to occur. After incubation, 50 μl of a 50 g/l sodium dodecyl sulfate solution was added to stop the reaction. The absorbance at a wavelength of 405 nm was read using an ELISA reader. j The results were expressed as the average ± standard deviation of the OD values.
Primers used in this study (reference: GenBank no. AY310918).

Underlined = restriction enzyme sites.
The surface modification of a gold chip for the specific binding of antigen was carried out as described elsewhere. 12 Briefly, a patterned glass slide chip with a gold film k was used to prepare the gold chip. Prior to coating with ProLinker B, l the gold-coated glass slides were cleaned using a freshly prepared piranha solution (3: 1 mixture of concentrated H2SO4 and 30% H2O2). After washing with deionized water, the glass slides were dried under a N2 stream. The gold chip was prepared by soaking a clean gold-coated chip in 3 mM ProLinker B solution for 1 hour followed by rinsing sequentially with chlorofom, acetone, ethanol, deionized water, and drying. For quantitative assays of antibodies with a minute amount of serum samples (1 μl per well), the antigens in PBS were spotted onto the ProLinker B-coated gold chip using the ProteoChip l with miniaturized micro wells. After the spotting, the chip was incubated for 20 minutes in 80% humidity at 37°C. The spotted chip was rinsed 3 times with PBS-T, followed by rinsing with deionized water. After adding 1 % bovine serum albumin (BSA) in PBS for 10 minutes to block the chip surface, the test samples (antibody or serum samples in a PBS buffer containing 0.1 mg/ml BSA) was applied to the chip for 15 minutes. The chip was then dried and analyzed using the SPRI system. m A Student's t-test was used to examine the differences in the ELISA OD values. A P value less than 0.01 was considered significant. The Pearson correlation coefficient n was used to determine the strength of the association between the SPRI and ELISA.
In the ELISA for detection of iridovirus-specific antibodies in the rock bream sera, an OD value of ≥0.451 (corresponding to 3 standard deviations above the negative controls;  = 0.307, SD = 0.048) was considered positive. Using this cutoff value, 37 samples (92.5%) tested positive (
= 0.307, SD = 0.048) was considered positive. Using this cutoff value, 37 samples (92.5%) tested positive ( = 0.876, SD = 0.358), indicating that an iridoviral infection is quite common in Korea, as reported previously.
10
This highlights the need for a proper prevention program to protect juvenile rock bream in a nursery against natural infections. The mean OD value for positive controls was 1.081 with a standard deviation of 0.222, while the mean OD value for the species control (sea bream) serum samples was 0.226 with a standard deviation of 0.038. Figure 1 shows the relationship between the signal intensity in the SPRI as a function of the control antibody dilution. The figure shows that the signal intensity in the SPRI increased linearly with increasing Ab concentration. The detection limit of the SPR protein chip corresponded to a 1: 100 antibody dilution, which corresponded to a signal intensity of 60 (
= 0.876, SD = 0.358), indicating that an iridoviral infection is quite common in Korea, as reported previously.
10
This highlights the need for a proper prevention program to protect juvenile rock bream in a nursery against natural infections. The mean OD value for positive controls was 1.081 with a standard deviation of 0.222, while the mean OD value for the species control (sea bream) serum samples was 0.226 with a standard deviation of 0.038. Figure 1 shows the relationship between the signal intensity in the SPRI as a function of the control antibody dilution. The figure shows that the signal intensity in the SPRI increased linearly with increasing Ab concentration. The detection limit of the SPR protein chip corresponded to a 1: 100 antibody dilution, which corresponded to a signal intensity of 60 ( = 60.1, SD = 6.3). Thirty-seven samples (92.5%) also tested positive by SPRI, suggesting that a chip based on SPRI was as sensitive as ELISA (the lowest detection limit corresponded to a 1: 100 dilution). Mouse anti-50-kDa antibody (positive control) and no primary antibody (negative control) were used in the SPRI, which required only 1 hour. There was a good correlation between SPRI signal intensity and ELISA OD values for the 40 serum samples (r = 0.939, P < 0.01; Fig. 2).
= 60.1, SD = 6.3). Thirty-seven samples (92.5%) also tested positive by SPRI, suggesting that a chip based on SPRI was as sensitive as ELISA (the lowest detection limit corresponded to a 1: 100 dilution). Mouse anti-50-kDa antibody (positive control) and no primary antibody (negative control) were used in the SPRI, which required only 1 hour. There was a good correlation between SPRI signal intensity and ELISA OD values for the 40 serum samples (r = 0.939, P < 0.01; Fig. 2).
The newly developed SPRI was used successfully to detect anti-50-kDa antibodies in rock bream sera. Surface plasmon resonance and SPRI assays are not competitive with ELISA in terms of cost. However, this comparative study showed that although ELISA is quite reliable, it is time-consuming. Recently, many SPR assays have been described for detection of antibodies against pathogens such as porcine circovirus type 2, 2 classical swine fever virus, 3 and Mycoplasma hyopneumoniae. 9 However, the SPR assay can be applied only to 2 to 4 samples simultaneously. Compared with the SPR assay, the newly developed SPRI protein chip with miniaturized micro wells proved to be a rapid (with an assay time of 1 hour), high-throughput, and valuable tool for the serodiagnosis of rock bream iridoviral infection.
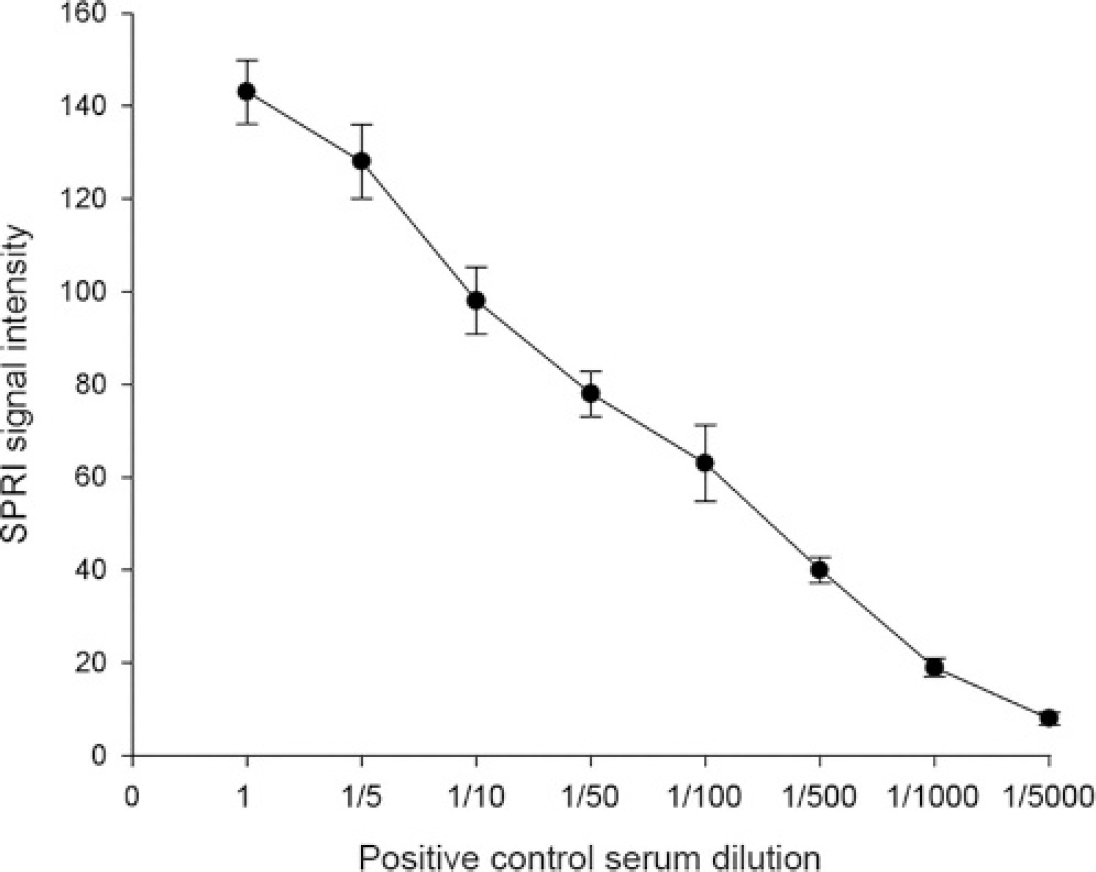
The changes in the SPRI signal intensity due to binding between the Ag (rMCP) immobilized ProLinker B and various dilutions of positive control antibody (anti-50-kDa mouse polyclonal).
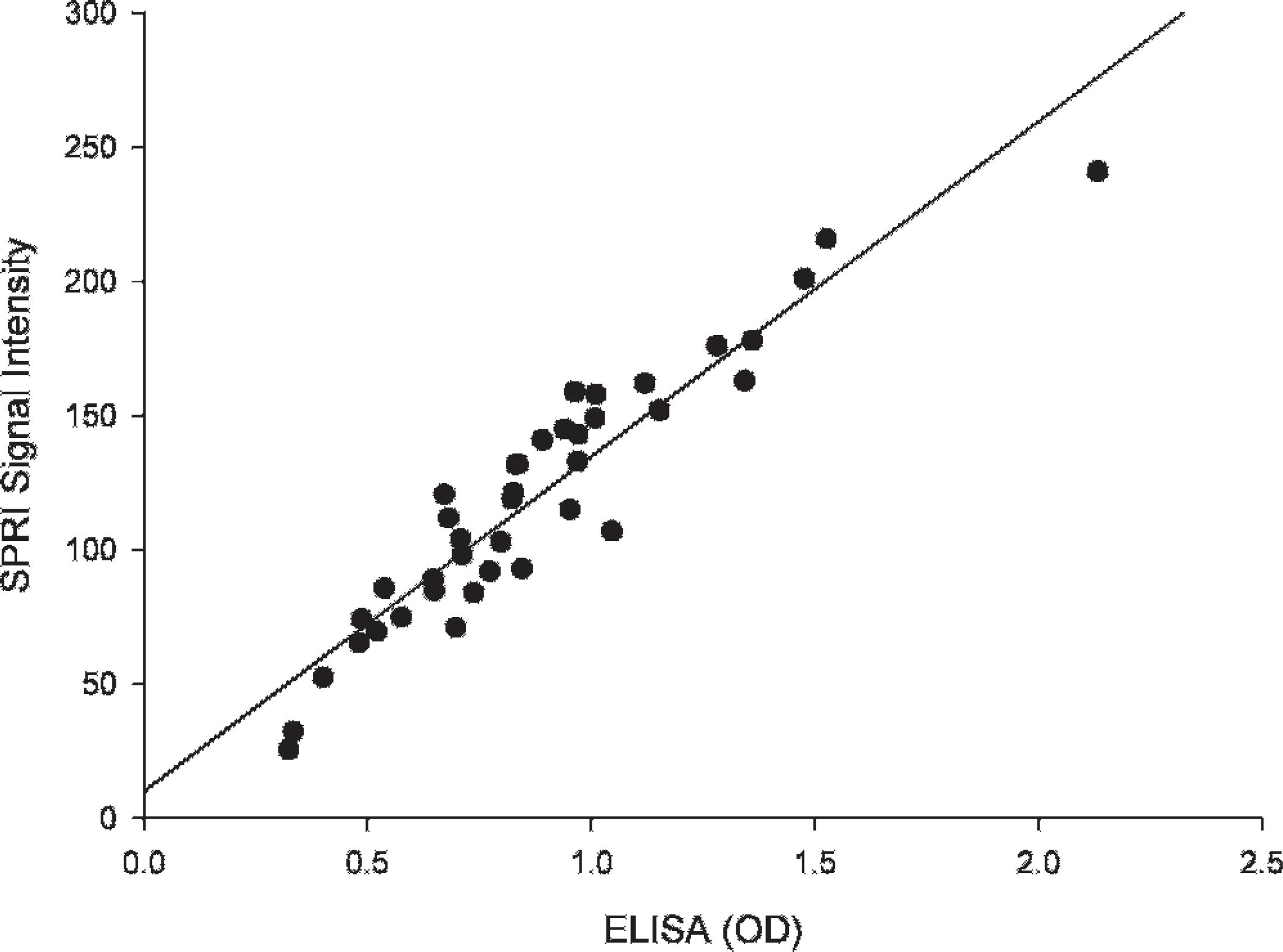
Correlation between the antibody OD values of the iridovirus in the rock bream sera determined by ELISA and signal intensity determined by SPRI (Pearson correlation coefficient, n = 40, r = 0.939, P = 0.01).
Acknowledgements. T. J. Kim acknowledges a graduate fellowship provided by the Korean Ministry of Education and Human Resources Development through the Brain Korea 21 Project.
Footnotes
a.
pRSET, Invitrogen, Carlsbad, CA.
b.
BBL™, Becton Dickinson and Company, Sparks, MD.
c.
Sigma, St. Louis, MO.
d.
Probond™, Invitrogen, Carlsbad, CA.
e.
Charles River, Yokohama, Japan.
f.
Maxisorp™, Nunc, Roskilde, Denmark.
g.
Pierce, Rockford, IL.
h.
Fish Disease Control Center, National Fisheries Research and Development Institute, Busan, Korea.
i.
Aquatic Diagnostic, Stirling, UK.
j.
Multiskan EX, Thermo LabSystems, Beverly, MA.
k.
K-Mac, Daejeon, Korea.
l.
Proteogen, Seoul, Korea.
m.
m. SPRi LAB, K-MAC, Daejeon, Korea.
n.
n. SPSS Base 12.0, SPSS, Chicago, IL.
