Abstract
The i-STAT portable clinical analyzer (PCA) was evaluated for performance in avian species. With the EG7+ cartridge, which provided results for hydrogen ion concentration, oxygen tension, carbon dioxide tension, sodium, potassium, ionized calcium, hematocrit, and various calculated parameters, analytical accuracy and precision were tested by comparing obtained values to those of established traditional blood gas and chemistry analyzers. Deming's regression and bias plots were used to compare i-STAT results with those obtained by laboratory professionals using benchtop analyzers. The reliability of the i-STAT PCA with EG7+ cartridges was good, with 0–5.7% system failures in measured values. Regression statistics were good for all blood gas analytes and acceptable for electrolytes and calculated parameters, except for potassium and base excess, for which the regression data or the discrepancy between the methods was too large. The system was reliable and easy to use and had an overall acceptable accuracy in avian species. These features, together with portability and small required blood volumes, make the i-STAT suitable for point-of-care use in critical avian patients, although single values require careful interpretation.
Introduction
Growing knowledge in avian medicine has resulted in better critical care and in more sophisticated and challenging surgeries. Simultaneously, the need for better diagnostic evaluation and anesthetic monitoring has increased. Blood gases, electrolytes, and hematologic parameters are used to assess the efficiency of breathing, tissue oxygenation, acid-base balance, and the cardiovascular system. 3 Immediate identification of disorders by rapid determination of representative parameters could be life-saving. Their measurement, however, is complex and requires high- quality instrumentation, 14 which is limited to clinical laboratories or large veterinary clinics with skilled personnel. Small body size and blood volume in most avian species are further limiting factors in diagnostic analysis. In addition, most available equipment is designed for use in mammals. Physiological (e.g., excretion of nitrogenous waste as uric acid) and anatomical (e.g., air sacs, nucleated and larger erythrocytes) differences of avian species might require the use of different analytic techniques or even different analytes in comparison to mammals. Systems might not work accurately, and therefore the evaluation of analytic techniques is of major importance.
The development of so-called “point-of-care” devices for blood analysis has resulted in many systems that have become fairly commonplace in emergency and critical care settings in both human and veterinary clinics. 2,7,10,15,17–19 Results of studies in humans and various mammalian species documented the accuracy and reliability of portable clinical analyzer (PCA) results. These devices were developed to provide necessary improvement in convenience, patient care, and results availability and have facilitated the stabilization of critical patients and assisted clinicians in determining more specific diagnostic procedures and therapy. 10,15,19 The i-STAT PCA (i-STAT, Axonlab, 5405 Baden-Dättwil, Switzerland) a is a hand-held analyzer of acceptable size and weight that requires a small sample volume of approximately 65 μl and performs simultaneous assays of whole blood in less than 2 minutes. 2
In the current study, the i-STAT PCA was evaluated the first time in avian species, and analytical accuracy and precision were tested by comparing obtained values to those of established traditional blood gas and chemistry analyzers. The purpose of this study was to explore the practical application of a PCA in the evaluation of selected blood constituents that may reflect metabolic or cardiovascular stress and that may be used to assess the critical avian patient.
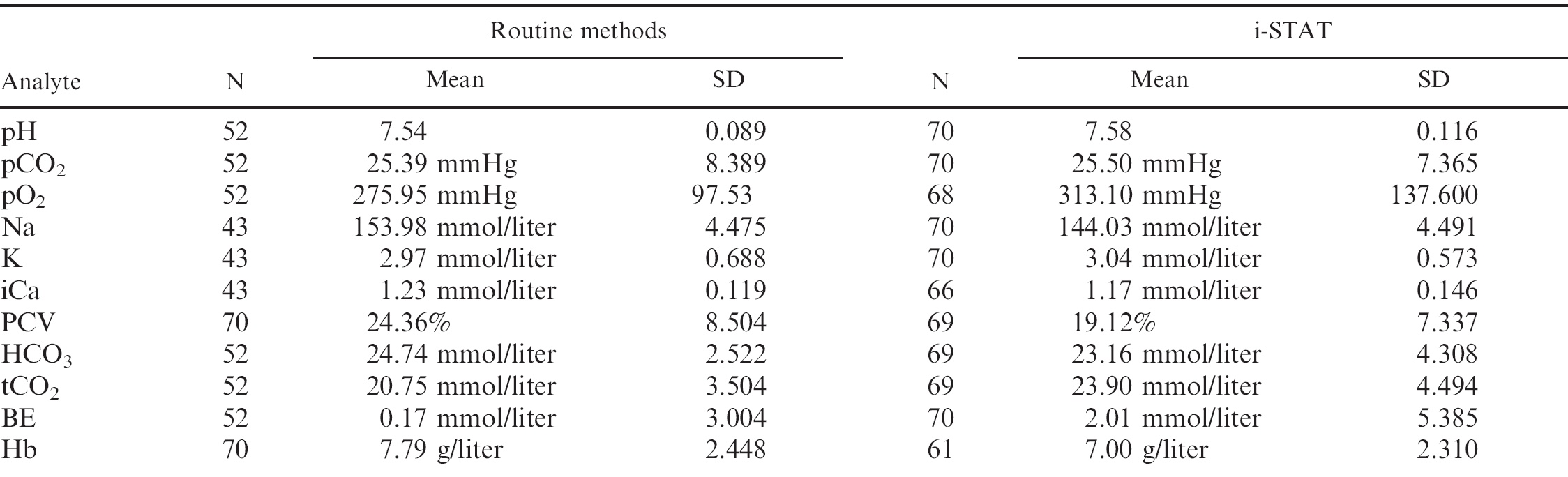
Blood gases, electrolytes, and hematologic parameters for chickens (Gallus gallus) obtained from whole samples using the i-STAT portable clinical analyzer and from whole blood or serum using standard laboratory benchtop analyzers.
Materials and methods
System description. The i-STAT system is composed of 2 main parts: the PCA and the disposable cartridges with microfabricated biosensors. For clinical analyses, the blood specimen is introduced into the cartridge by using a syringe; the cartridge is then inserted into the analyzer, and operator and patient identification are entered into the system. A calibration is automatically performed just before sample analysis. Results are displayed on the screen and stored in the analyzer's memory; in addition, results can be sent by infrared port to a printer. A detailed technical description of the PCA and its use is described elsewhere. 2
Experimental study protocol. Forty healthy Lohmanns selected leghorn chickens (Gallus gallus) were anesthetized with 4% isoflurane b in oxygen delivered by face mask and subsequently intubated by endotracheal tubus. c Anesthesia was maintained by administration of isoflurane (expired isoflurane fraction: 1.9-2.0%).
Blood for analyses was collected with 2-ml preheparinized syringes d from arterial puncture of the Arteria ulnaris or Arteria tibialis cranialis during a shock trial. Forty blood samples were collected in the healthy anesthetized chicken, 15 samples were collected 10 min after 50% loss of blood volume, and 15 samples after resuscitation. The whole-blood sample was tested without delay on the i-STAT PCA. Before analysis, syringes were gently mixed and the blood specimen was directly introduced into the cartridge. The cartridge was then inserted into the analyzer, patient and operator information were entered, and analysis was started immediately. Disposable EG7+ cartridges e were used for hydrogen ion concentration (pH), carbon dioxide tension (pCO2), oxygen tension (pO2), sodium (Na), potassium (K), ionized calcium (iCa), and hematocrit (PCV) measurement; in addition, the i-STAT provided bicarbonate (HCO3), total carbon dioxide (tCO2), base excess (BE), oxygen saturation (sO2), and hemoglobin (Hb) as calculated values based on the measurements.
The remaining blood sample was sealed airtight without air bubbles, kept on ice, and immediately transported by hand to the in-house hematology laboratory (Clinical Laboratory, Vetsuisse Faculty, University of Zurich), where the sample was analyzed within less than 20 minutes after collection time. Blood gas analyses (pH, pCO2,pO2, HCO3, tCO2, BE, sO2) were performed directly from remaining blood of preheparinized syringes on an automatic benchtop analyzer. f Hematocrit was manually obtained by centrifugation g of an aliquot of the whole blood specimen for 3 min at 16,000 × g in a microcentrifuge. Electrolytes, Na, and K were analyzed from serum h and iCa from serum. i All testing was done according to manufacturers' recommended procedures.
Statistical analysis. The data was tested by a Kolmogorov-Smirnov test for normal distribution. Comparisons of traditional laboratory analyzers with i-STAT results were performed according to standard recommendations for comparing analytical techniques based on Deming's regression and Bland-Altmann bias representation. 8,12 Calculations were performed using a personal computer-based statistic software. j Results are given as mean values, with 95% confidence intervals in parenthesis. Significance level was set at P = 0.05.
Results
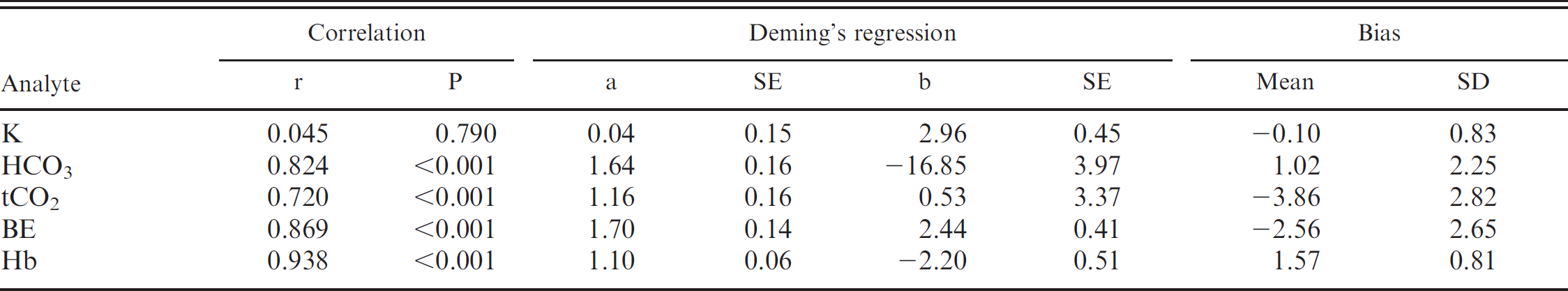
A total of 75 EG7+ i-STAT cartridges were used; cartridge filling errors (air/too small or too large sample volume) occurred in 5 (6.67%) occasions. Analysis of pH, pCO2, Na, K, and BE provided results in all 70 cartridges (100%), whereas system failures of single or multiple values were noted in 1–9 (1.5-12.9%) cartridges (HCO3, tCO2, PCV: 1 cartridge [1.4%], pO2: 2 cartridges [2.9%], iCa: 4 cartridges [5.7%], Hb:9 cartridges [12.9%]). Data were normally distributed (P > 0.05). Mean and standard deviation (SD) of analyzed parameters divided by routine methods and i-STAT analysis are displayed in Table 1. For the parameters pH, pCO2, pO2, iCa, and PCV, the i-STAT gave good accuracy (Figs. 1–6), with results correlating very well (P < 0.001) with control values (correlating coefficient r = 0.944 for pH, r = 0.953 for pCO2, r = 0.901 for pO2, r = 0.776 for iCa, r = 0.944 for PCV). The correlation coefficient for Na was low (r = 0.327), but still significant (P = 0.045). Deming's regression slopes were close to 1.00 for all measured values and intercepts acceptably close to 0 (see legends of Figs. 1–6). Potassium values obtained using the i-STAT PCA did not correlate with those obtained using the control systems (r = 0.045, P = 0.789); K failed Deming's regression analysis as well (F = 0.073, P = 0.789) (Table 2).
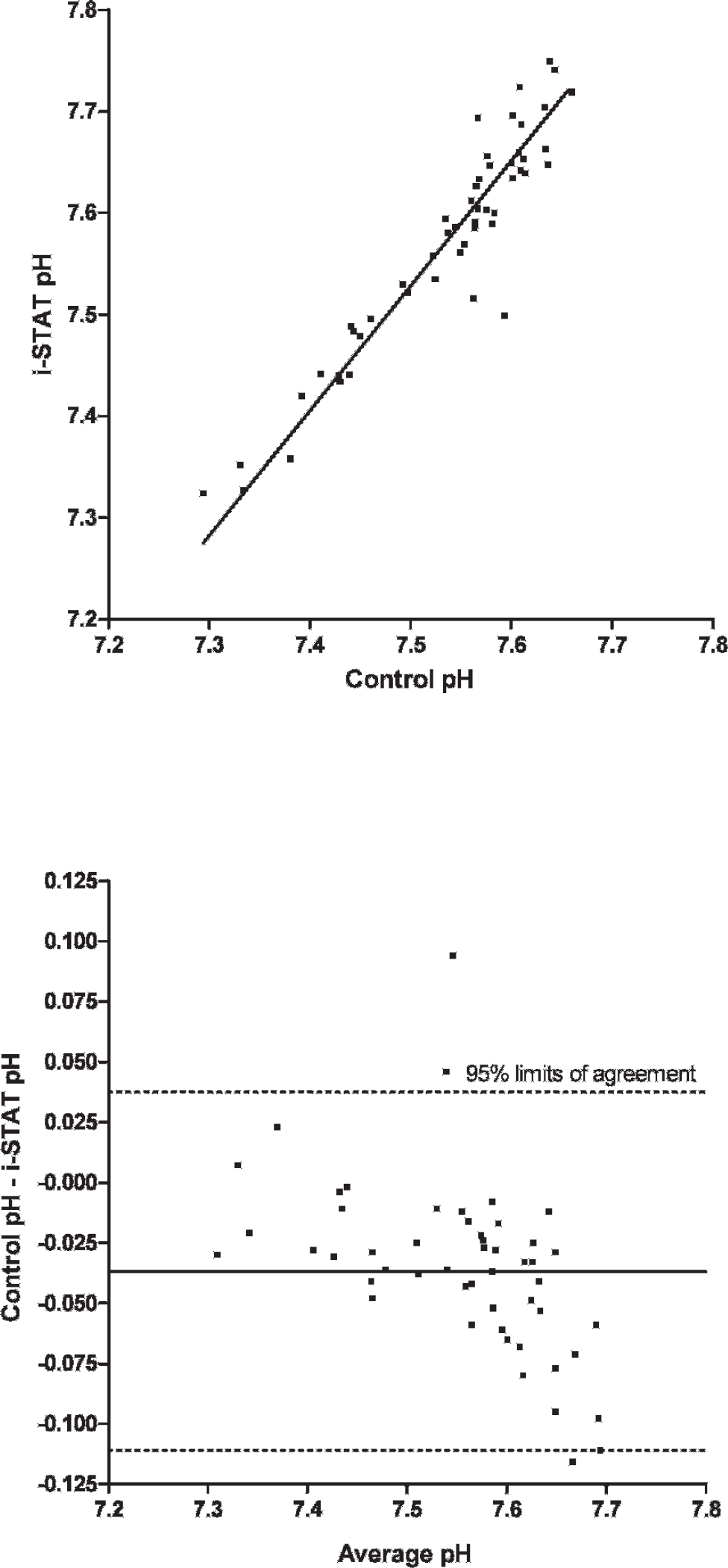
Scatterplot of pH measurements in chickens (Gallus gallus) with the i-STAT system versus the control blood gas analyzers
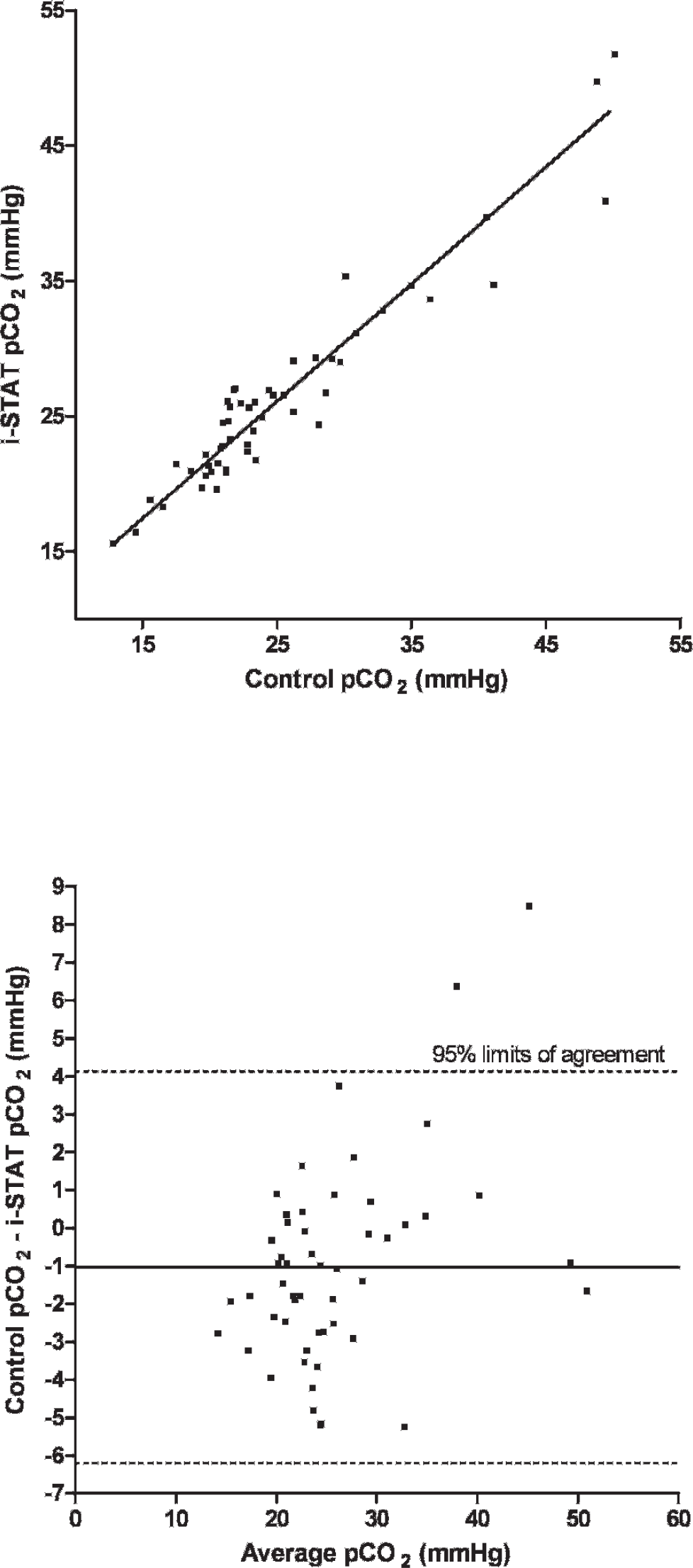
Scatterplot of carbon dioxide tension (pCO2) measurements in chickens (Gallus gallus) with the i-STAT system versus the control blood gas analyzers
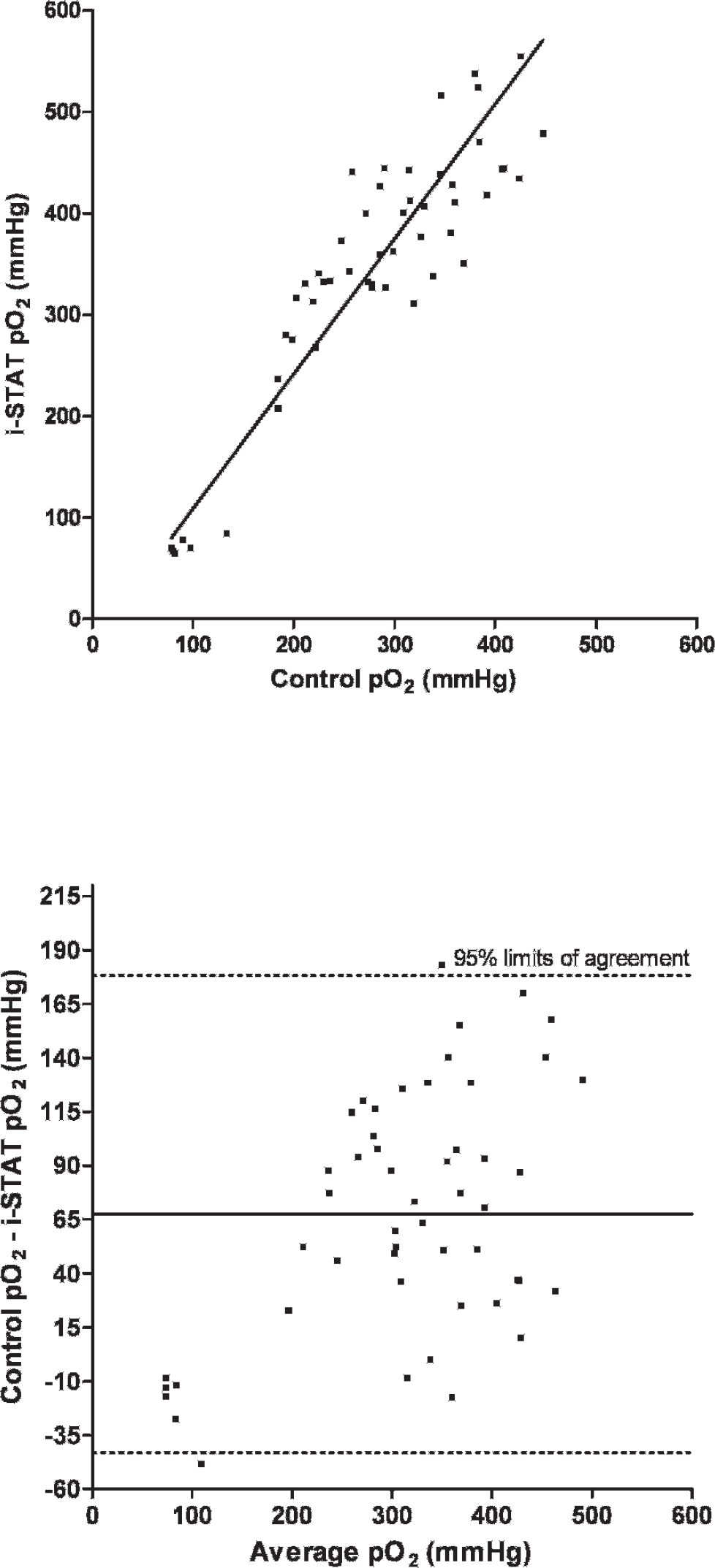
Scatterplot of oxygen tension (pO2) measurements in chickens (Gallus gallus) with the i-STAT system versus the control blood gas analyzers
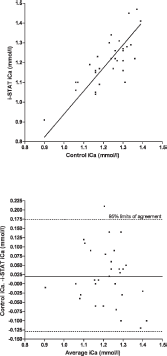
Scatterplot of ionized calcium (iCa) measurements in chickens (Gallus gallus) with the i-STAT system versus the control analyzers
Bland Altman plots for pH, pCO2, Na, and iCa showed an acceptable bias with a fairly consistent variability, whereas the average discrepancies between methods were wider in pO2 and PCV (Figs. 1–6). Table 2 presents the results of calculated parameters (HCO3, tCO2, BE, Hb, sO2). Again values obtained using the i-STAT PCA correlated well with those obtained using the control systems, but mean biases were moderate high.
Discussion
Measurements of blood gas, electrolytes, and hematologic parameters are used to monitor and control respiratory efficiency, tissue oxygenation, acid-base balance, and the cardiovascular system. 3 The required blood sample volumes made blood gas analysis a rarely used tool in avian medicine. The present study was designed to examine the analytical accuracy and precision of the i-STAT analyzer when used in avian patients.
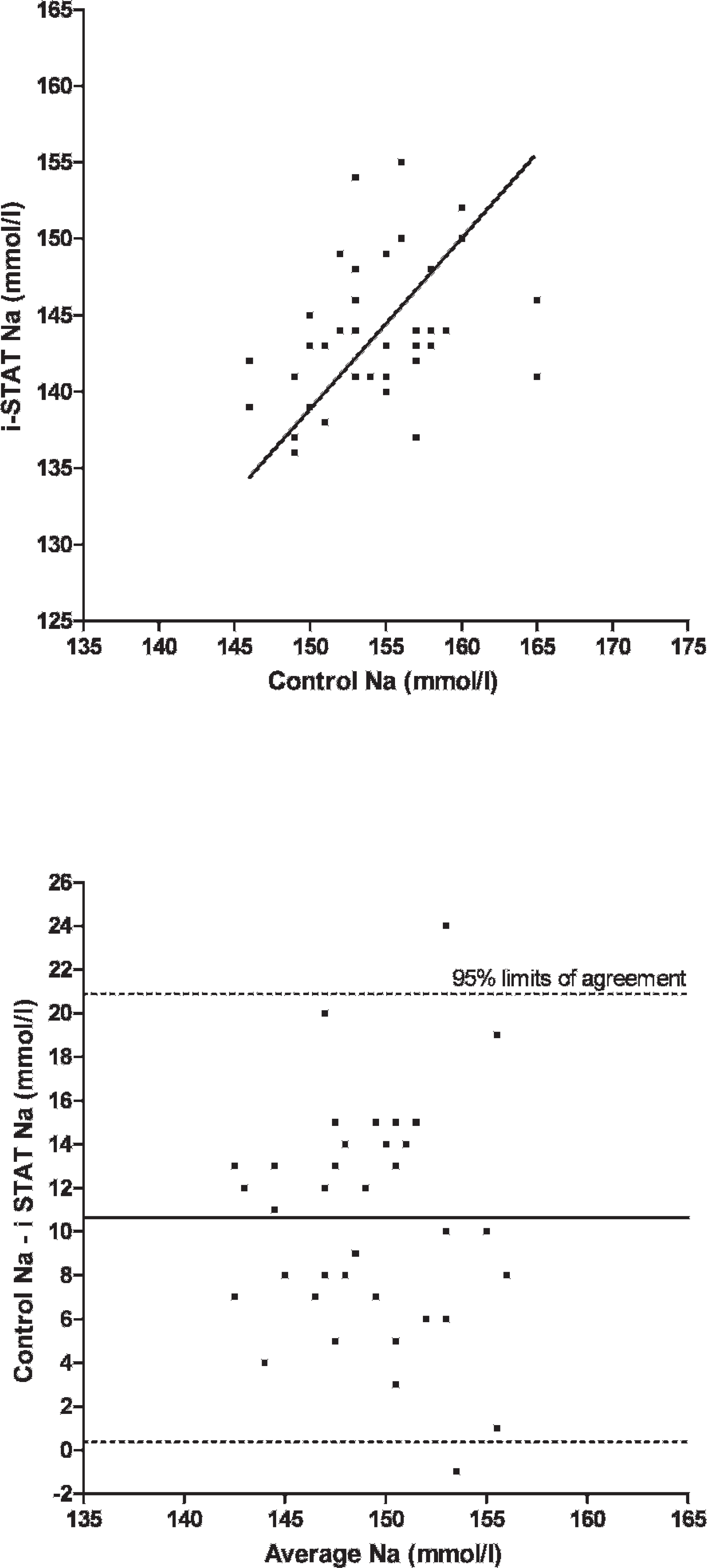
Scatterplot of sodium (Na) measurements in chickens (Gallus gallus) with the i-STAT system versus the control analyzers
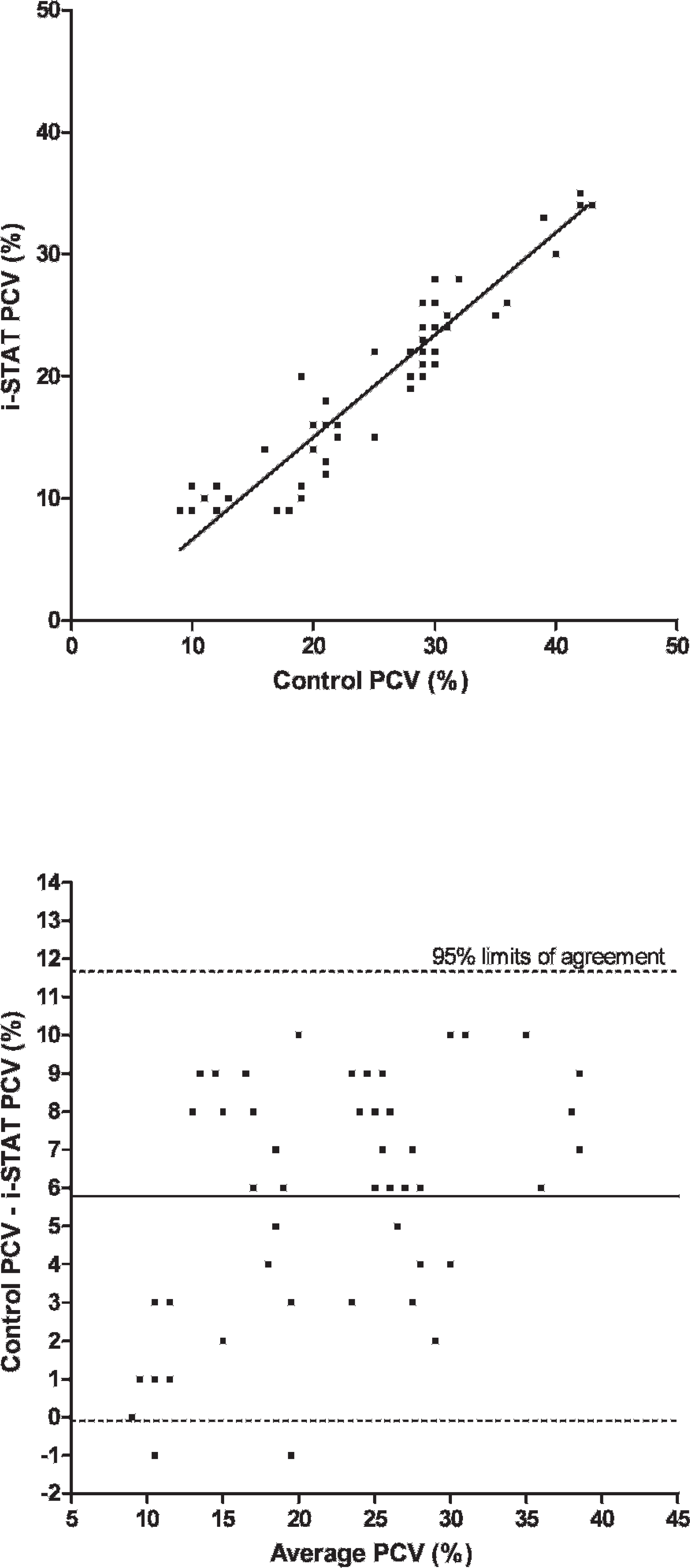
Scatterplot of packed cell volume (PCV) measurements in chickens (Gallus gallus) with the i-STAT system versus the control analyzers
The reliability and performance of the i-STAT PCA measuring pH, pO2, pCO2, Na, iCa, PCV and calculating HCO3, tCO2, Hb, sO2 were judged to be very acceptable, confirming earlier studies in other species. 15,19 In the current study, the i-STAT PCA with the EG7+ cartridge showed good accuracy for measurements in comparison to traditional laboratory analyzers across the entire range of values measured, with the exception of K and BE.
The small number of rejected results or system failures in the current study were similar as in a similar study of human blood analysis 2 (chi-square 0.69, P = 0.41) and indicates that the i-STAT PCA could become a more frequently used monitoring device in avian medicine. The system failures in the PCA require repeated sampling and analyses, but still facilitate improved turnaround time for testing because of the short analysis time and the availability of the system in veterinary clinics and in the field.
Comparison of results obtained with the i-STAT (y) and results obtained with routine laboratory benchtop analyzers (x) for hemoglobin, potassium, and calculated parameters of acid-base status in chickens (Gallus gallus).
In the current study, the PCA showed good accuracy for blood-blood-gas and pH measurement across the range of values measured in chickens (Figs. 1–3). Bias was very low and fairly constant, although minor proportionality of bias was observed for pO2 similar to a previous study in dogs. 19 But even here, discrepancy would not result in normal/abnormal misclassification. Remarkably, correlation coefficients for electrolytes were not as good as in humans, 2 and no correlation was detected between measured potassium in the i-STAT PCA and benchtop analyzers. Nevertheless, the performance of the i-STAT for measuring iCa and Na were judged to be acceptable (Figs. 4 and 5) with Deming's regression slopes of 1.17 for iCa and 1.12 for Na. A significant bias was not found between the i-STAT and the in-house analyzer for iCa and Na. The failure of K might be explained by the differences in sample type (whole blood vs. serum) and/or by the relatively high artifactual change in plasma potassium values as has been shown in various avian species where potassium levels were decreased from 30% to 60% within a 2-hour period. 6,11 The results obtained for hematocrit determination by the PCA showed smaller values (by ∼7%) than those reported by the comparison system, but nevertheless the precision of the PCA appeared acceptable for clinical purposes. Smaller values in the PCA might be caused by mechanical interferences in the cartridge due to erythrocyte size. Avian erythrocytes are nucleated and are generally larger in size than human erythro-cytes. 5
The accuracy for calculated values (HCO3, tCO2, Hb, sO2, BE) was not as good, since correlation coefficients ranged from 0.72 to 0.94 and Deming's regression slopes ranged from 1.10 to 1.70. However, all biases with the exception of BE were acceptably low and constant enough to preclude clinical misinterpretation. The bias of BE was high, and further analyses are needed. To counter bias, it would be necessary to determine specific ranges of references values, which could be obtained from large samples of healthy animals.
The EC7+ cartridge was chosen because of its potential application in avian critical care. Oxygenation and ventilation are important factors in the treatment of emergency patients, mammalian as well as avian. The ability to obtain reliable measurements with current available methods, such as pulse oximetry and capnometry, for the assessment of oxygenation and ventilation can be limited by abnormal physiologic states commonly seen in emergency patients. In emergency situations (e.g., shock, bleeding), an abnormal ventilation/perfusion relationship affects end tidal CO2 measurements, and the absence of an adequate pulse signal can result in the failure of pulse oximetry to measure arterial hemoglobin saturation. Furthermore, acute carbon monoxide poisoning constitutes a particular problem as, in this situation, many pulse oximeters report overestimated oxygenation results, which wrongly indicate adequate oxygen saturation. 9 Additionally, severe peripheral hypoxia also leads to lactate production and reduces pH, a parameter that can be determined by blood gas analysis. Therefore blood gas analyses would be desirable for assessing oxygenation and ventilation in avian critical care.
Electrolyte status, specifically Na and iCa, seem to play an important role in water homeostasis and cardiac pathophysiology. 4,16 Whereas calcium plays an important physiological role in avian species and disorders of calcium metabolism in captive birds are well known, only limited reports of sodium and potassium abnormalities exists. 13,16 The measurement of serum iCa in consideration of potential acid base imbalances provides a more precise estimate of calcium status than does total serum calcium, especially in the diseased patient. 16 Although iCa is essential for myocardial contraction, its blind application during cardiac failure is not recommended because of the inherent risk of hypercalcemia, which could result in an irreversible myocardial contraction as it is known from human critical care. 1 Therefore, measurements if iCa and Na might be of increasing interest in avian critical care.
From a practical point of view, the i-STAT PCA is easy to use and can be used as a blood analyzer in critical avian patients. Most measured and calculated blood-gases values correlated well with gold standard analyzers. Thus the i-STAT appears a valid substitute for expensive benchtop analyzers when a limited number of tests is performed or when mobility is required, although single values (K, BE) have to be interpreted carefully.
Acknowledgements
The authors thank the technicians at the Clinical Laboratory and the keepers at the Exotic Pet Clinic, Vetsuisse Faculty, University of Zurich, for their help during the study. The corrections and input of Dr. Marcus Clauss are gratefully acknowledged.
Footnotes
a.
i-STAT, Axonlab, 5405 Baden-Dättwil, Switzerland.
b.
Attane, Provet AG, 3421 Lyssach, Switzerland.
c.
Cole endotracheal tubus, Cook Veterinary Products, Medical Solutions, 6312 Steinhausen, Switzerland.
d.
Preheparinized syringes, Labor-Systeme Flückiger, 5737 Menziken, Switzerland.
e.
EG7+ cartridges, Axonlab, 5405 Baden-Dättwil, Switzerland.
f.
Rapidlap 800, Bayer, 8045 Zürich, Switzerland.
g.
Hematocrit 20, Hettich AG, 8806 Bäch, Switzerland.
h.
Cobas Integra, Roche Diagnostics, 6343 Rotkreuz, Switzerland.
i.
Nova CRT 8 electrolyte analyzer, Nova Biomedical, Waltham, MA.
j.
GraphPad Prism, version 4.00, GraphPad Software, San Diego, CA.
