Abstract
Systemic infection by Nocardia asteroides was diagnosed in a reindeer (Rangifer tarandus tarandus). At necropsy, the animal had enlarged tracheobronchial lymph nodes, pleural and peritoneal effusions, and numerous, multifocal to coalescing, yellow, firm nodules with inspissated pus in the lung, pleura, omentum, liver, heart, adrenal glands, and left kidney. Microscopically, the nodules consisted of foci of pyogranulomatous inflammation. Microscopic lesions were present in the grossly affected organs as well as spleen and brain. Sections stained with Gram and modified Fite-Faraco histochemical stains had numerous gram-positive, variably acid fast, beaded and branching filamentous organisms in the necrotic centers. N. asteroides was isolated from the lung, bronchial lymph nodes, liver, and left kidney. To the authors' knowledge, this is the first report of systemic N. asteroides infection in a reindeer.
Nocardiosis is a suppurative or, less frequently, pyogranulomatous disease caused by aerobic bacteria of the genus Nocardia. 3 These gram-positive, branching and partially acid fast bacteria cause localized or systemic disease. The latter is commonly seen in immunocompromised and massively exposed hosts. 3 Nocardial infections are reported in several species including humans, dogs, cats, cattle, llama, goats, horses, antelopes, pigs, birds, whales, and dolphins. 3 , 4 In cattle and small ruminants, N. astreoides is an uncommon cause of chronic mastitis. 5 This bacterium is additionally a rare cause of abortion in horses, cattle, sheep, and swine. 3 The present report describes a systemic nocardial infection in a reindeer.
A 1.5-year-old male, farmed reindeer was submitted to Purdue Animal Disease Diagnostic Laboratory (ADDL), West Lafayette, IN, for necropsy. The animal died following intermittent fever of approximately 10-day duration. Significant clinical findings included anemia, hypoproteinemia, hypoglycemia, and hypocalcemia. Fecal examination revealed heavy parasitic burden. On presentation to ADDL, the animal was thin and lacked body fat and had reduced muscle mass. At necropsy, the pleural and peritoneal cavities respectively contained approximately 1 and 5 liters of turbid brown effusion. The right caudal lung lobe had an approximately 25 × 20 × 13 cm firm tan area of consolidation, with multifocal to coalescing, yellow to tan, firm, 2–4 mm nodules on the pleural surface. On cut section, these nodules extended into the parenchyma and contained thick, pale tan, caseous exudate (Fig. 1). Similar but fewer nodules were present in the parenchyma and pleura of the remaining lung lobes. The tracheobronchial lymph nodes were almost 4 times the normal size with similar lesions effacing the nodal architecture. The left kidney, which was almost twice the normal size, had multifocal to confluent, pale tan nodules (1-10 mm in diameter) that effaced approximately two-thirds of the renal cortex and variably extended into the underlying medulla at 1 pole. The cortex and medulla at the opposite pole were spared (Fig. 2). Similar lesions were also present in the liver, heart, adrenal glands, and, omentum. Other macroscopic findings included white purulent material in the anterior chamber of the right eye (hypopyon). Samples of multiple tissues including the affected organs were collected and fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 μm and stained with hematoxylin and eosin (HE). Selected tissue sections were stained with Brown and Brenn Gram stain, periodic acid-Schiff (PAS), Gomori methenamine silver (GMS), and modified Fite-Faraco acid-fast histochemical stains. The homogenates of lung, bronchial lymph nodes, liver, and left kidney were submitted for bacteriologic examination and subjected to grow on blood agar and Sabouraud agar at 25, 37, and 45°C, respectively. 2
Histologically, the pulmonary parenchyma of the right caudal lung lobe was effaced by variably sized, multifocal to coalescing foci of necrosis. The necrotic foci were characterized by abundant eosinophilic and karyorrhectic debris, surrounded by large numbers of degenerate and viable neutrophils, surrounded by moderate numbers of lymphocytes and histiocytes, and by scant to moderate immature fibrous connective tissue (Fig. 3). Rare foci contained few, foreign body-type giant cells. The remaining pulmonary parenchyma was diffusely congested. In some lobules, less affected alveolar spaces contained fibrin, hemorrhage and lymphocytes, plasma cells, neutrophils and histiocytes. The left kidney, spleen, liver, adrenal glands, heart, brain, and bronchial lymph nodes had similar necrotizing and inflammatory foci of variable severity. No intralesional causative agent was observed in HE, PAS, and GMS stained tissue sections. Staining of affected lung sections by the Gram method showed gram-positive, beaded to filamentous and occasionally branching organisms in the centers of the necrotic lesions (Fig. 4). Fite-Faraco staining of the kidney lesions revealed variably acid-fast, filamentous and beaded organisms. These organisms were morphologically consistent with Nocardia spp. or Actinomyces spp. 3 N. asteroides in pure culture was isolated from the lung, bronchial lymph nodes, liver, and left kidney. The organisms grew on blood agar and Sabouraud agar at 25 and 37°C, but not at 45°C. Growth was evident in 4–5 days of incubation and colonies were irregularly folded, raised, smooth or granular, and yellow orange. The colonies were gram-positive, partially acid fast, and branching filamentous elements, distinguishing them from saprophytic and atypical Mycobacterium spp. 2 In addition, the organisms were positive by urease test and unable to decompose tyrosine. The laboratory findings from the isolation protocols in this case also ruled out the presence of systemic fungi, Actimyces spp., Actiobacillus spp., and other Nocardia species except N. asteroides. 2 Isolation of N. asteroides from the tissues mentioned above were confirmed by the bacteriology laboratory in the National Veterinary Services Laboratories, Ames, IA.
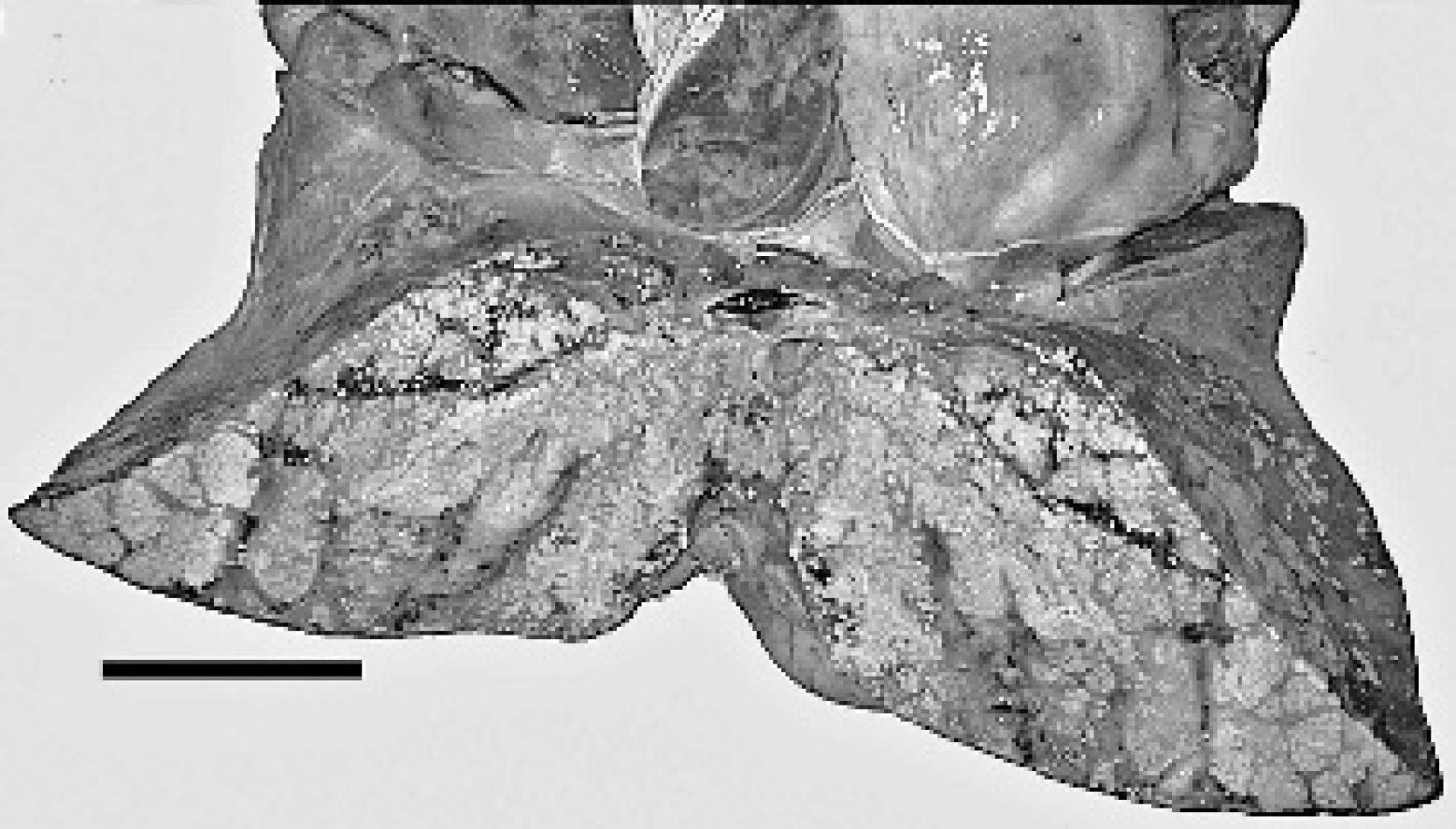
Lung, reindeer. Cross section of lung showed numerous, multifocal to coalescing, yellow to tan, firm, 2–4 mm foci with caseous exudate. Bar = 10 cm.
Systemic nocardiosis was confirmed based on the morphology and staining characteristics of the bacteria associated with the lesions, and culturing N. astreroides from several affected tissues. Nocardial infections in animals are either localized or multisystemic as presented here. In ruminants, reports of localized nocardial infections include mastitis, pneumonia, dermatitis, and nasal granulomas. 8 , 10 , 11 Disseminated infection by Nocardia spp. was reported in several species of ruminants including a group of slender-horned gazelles and a North African antelope. 1 , 7 The infection was confirmed by culturing Nocardia sp. from the affected tissues in the gazelles and antelope; however, a specific species of Nocardia was not identified in these cases.
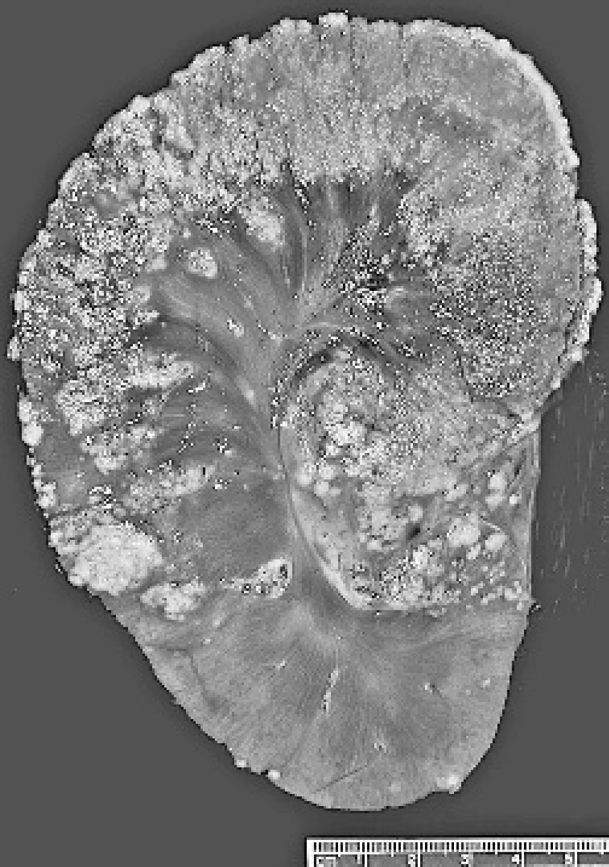
Kidney, reindeer: Left kidney had multifocal to confluent, 1–10 mm, pale tan nodules that effaced the majority of the cortex and about half of the medulla.
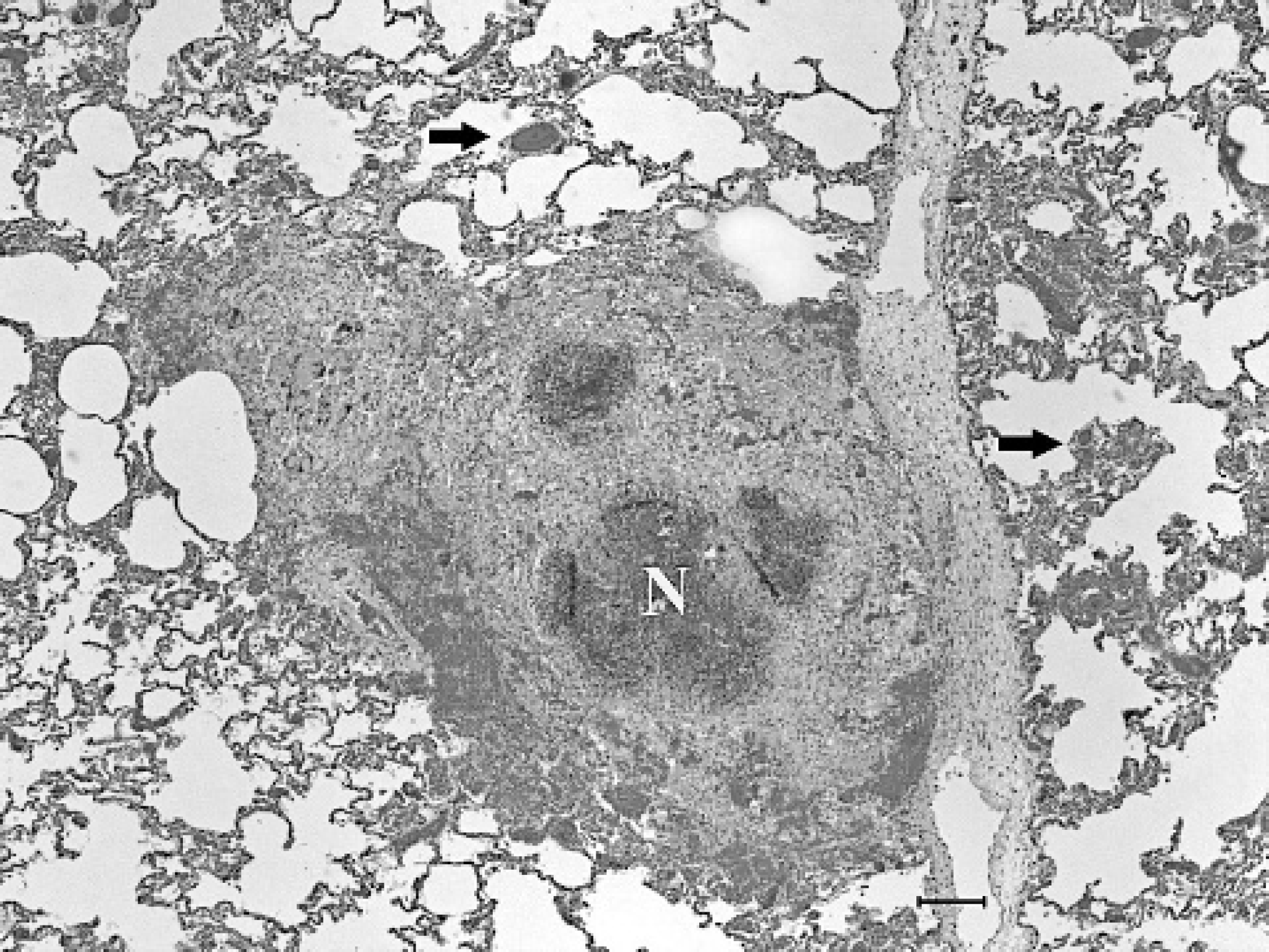
Lung, reindeer. The pulmonary parenchyma contains variably sized, central foci of necrosis (N) with degenerate neutrophils and cell debris, surrounded by macrophages, multi-nucleate giant cells and immature fibrous connective tissue. The alveolar capillaries adjacent to the necrotic foci were congested (arrows). HE. Bar = 150 μm.
N. asteroides isolated from multiple organs of this reindeer is the most commonly isolated species from animals affected with nocardiosis. Other less frequently isolated Nocardia spp. that can cause infections in animals include N. brasilienses, N. caviae, and N. africana. 3 , 6 Infection by Nocardia spp. can occur through inhalation, ingestion, or mechanical introduction via trauma. 3 Because of the extensive involvement of the lung, inhalation was considered to be the likely route of entry for the bacteria in this reindeer; however, possible hematogenous spread to the lungs cannot be entirely ruled out. In the present case, the inflammation was primarily pyogranulomatous owing to the presence of epitheloid macrophages and giant cells that surrounded the foci of liquifactive necrosis. Actinomyces spp. cause similar gross and histlologic lesions and resemble Nocardia spp. morphologically in tissue sections. In addition, lesions produced by Actinomyces spp. usually contain sulphur granules, which are colonial masses surrounded by Splendore-Hoppli material. 3 Sulfur granules are rarely associated with nocardial infections and were not observed either grossly or histologically in the affected tissues of this reindeer. Furthermore, Actinomyces spp. are non-acid fast organism and do not stain with the modified Fite-Faraco technique. Other reported causes of granulomatous inflammation in reindeer include non tuberculous mycobacterium, fungi (Zygomycetes spp., and Aspergillus spp.), and parasites (Onchocerca tarsicola). 7 , 9 , 12 Some of these agents involve multiple systems and usually affect animals with compromised or underdeveloped immunity. 7 , 9
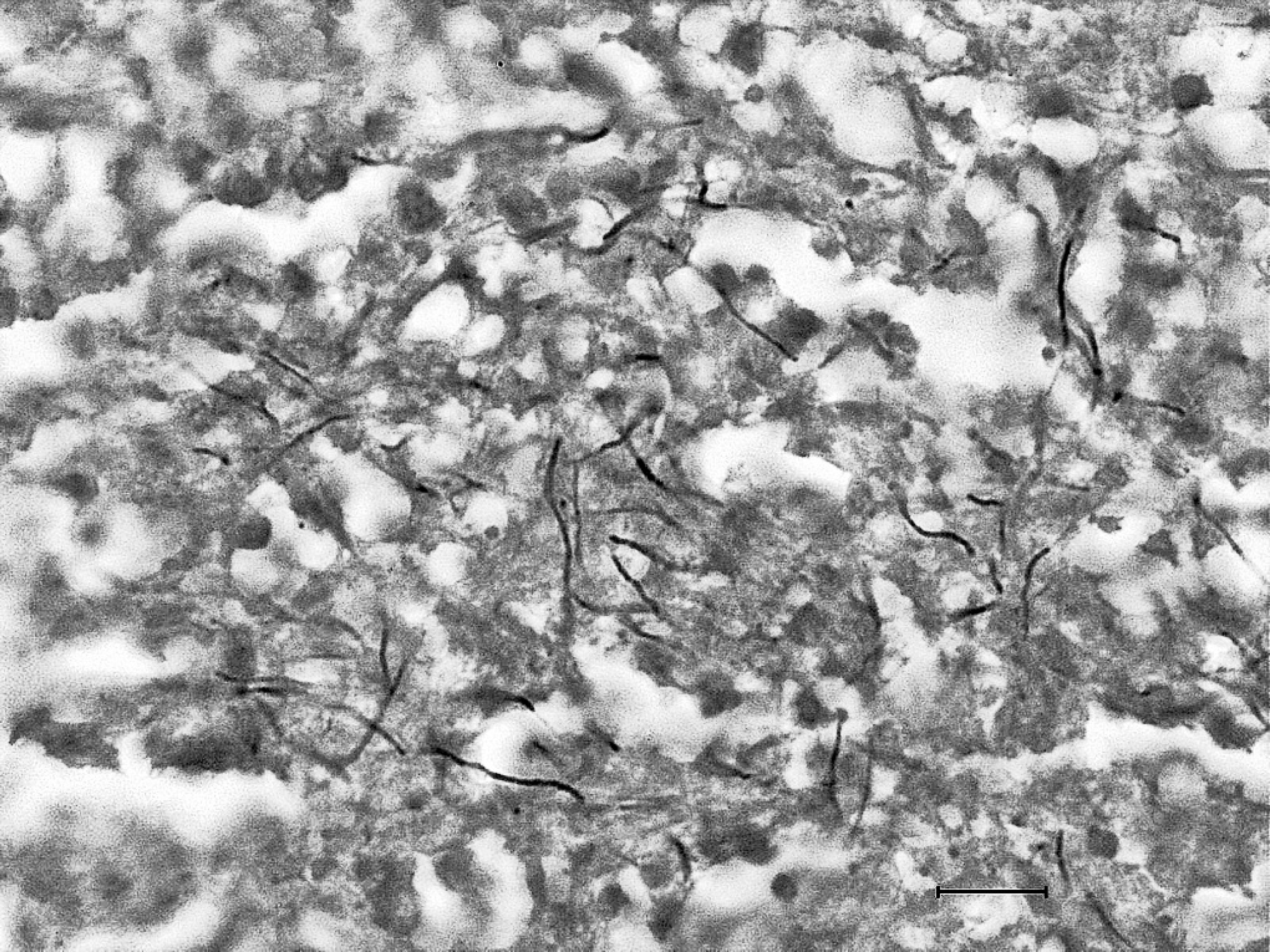
Lung, reindeer. Numerous gram-positive, 0.8–0.1 μm in diameter, beaded to filamentous bacteria morphologically consistent with Nocardia sp. were present in the centers of lesions. Brown and Brenn. Bar = 10 μm.
Nocardial infections are on the rise in recent years because of increased and prolonged use of antibiotics and corticosteroids in both humans and animals. 3 Increased susceptibility to opportunistic infections associated with long-term use of antibiotics and corticosteroids has been implicated as the factor that predisposed a llama to concurrent infection by N. asteroides and Aspergillus sp. 4 Drug-induced immunosuppression might have contributed to systemic spread of the disease, but was unlikely the predisposing factor to the infection in this case because the reindeer was treated with nonstreoidal anti-inflammatory drugs for only 1 week prior to death. The animal's poor body condition suggests that the illness was of a longer duration than reported. Further, repeated inbreeding among a herd of slender horned gazelles has been indicated to increase susceptibility to infection by Nocardia spp. 7 The current animal was a farmed animal, but information regarding breeding practices on this farm was not available. In some localized forms of nocardiosis, antibiotic therapy combined with excision of granulomas can provide good therapeutic results. 3 However, in systemic infections prognosis is grave owing to poor response to treatment.
In summary, diagnosis of systemic nocardiosis was made based on the presence of consistent microscopic lesions and isolation of N. asteroides from the affected tissues. Though uncommon, Nocardia spp. should be considered as a differential diagnosis if pyogranulomatous lesions are present in tissues of domestic and wild ruminants. To the authors' knowledge, this is the first reported case of systemic nocardiosis in a reindeer.
Acknowledgements. The authors would like to thank Dr. L. Gallatin for submitting the case for necropsy and Dr. I. Langohr for critical evaluation of the manuscript.
