Abstract
Canine Lyme disease is caused by the spirochete Borrelia burgdorferi after transmission by an Ixodes tick, typically resulting in joint pain, fever and lethargy. Lyme nephritis is a poorly characterized syndrome associated with severe glomerular and tubular renal injury and poor clinical outcome in young to middle-aged dogs positive for exposure to B. burgdorferi. The aims of this study were to identify associations between natural exposure to B. burgdorferi and the presence of microalbuminuria in nonclinical young Labrador and Golden Retrievers and to compare two commonly used serologic tests available to document B. burgdorferi exposure: the Western blot and the commercial point-of-care C6 peptide enzyme-linked immunosorbent assay (ELISA) tests. Microalbuminuria was assessed using a commercial point-of-care ELISA specific for canine albumin. Blood and urine samples from 268 asymptomatic Labrador and Golden Retrievers were included. Of these, 18.7% were positive for B. burgdorferi exposure according to the C6 ELISA; 21.2% were positive for natural exposure to B. burgdorferi and 11.5% for vaccinal antibodies according to the Western blot. The agreement rate was 93% between the two tests (kappa = 0.78, P < 0.0001) for natural exposure. Urine from 6.1% of the dogs was positive for microalbuminuria. There was no association between microalbuminuria and exposure to B. burgdorferi based on results of a Western blot (P = 0.57) or C6 ELISA (P = 0.53). Microalbuminuria is likely not a consequence of B. burgdorferi exposure in young nonclinical Labrador and Golden Retrievers.
Borrelia burgdorferi, the causative agent of Lyme disease, was first identified more than two decades ago 1 and has since been associated with a wide variety of clinical signs in approximately 5–10% of infected dogs. 8 , 13 , 17 , 18 Clinical signs of canine Lyme disease include transient fever, arthritis, monoarthropathy or polyarthropathy, local lymphadenomegaly, and intermittent lameness. 7 , 18 Although less common, the most severe manifestation of Lyme disease may be what is called “Lyme nephritis.” 7 , 18
Lyme nephritis is thought to be a rare and generally fatal manifestation of canine borreliosis that is rapidly progressive and characterized by an acute onset of clinical signs consistent with renal failure. 2 , 18 Dogs presenting with this condition are thought to rapidly develop azotemia, uremia, proteinuria, vasculitis and peripheral edema, thromboembolism, hypertension, and an active urine sediment in the absence of urinary tract infection. 2 , 18 Affected dogs are relatively young (average age, 5.6 years), there is often a recent history of joint disease, and patients are typically positive for B. burgdorferi antibodies. Labrador and Golden Retriever dogs have been reported to be overrepresented. 18 Affected kidneys have both glomerular and tubular lesions. Most commonly, a membranoproliferative glomerulonephritis with diffuse tubular necrosis and regeneration and lymphoplasmacytic interstitial inflammation has been reported. 2 , 5
In humans, microalbuminuria (MA) has been shown to be a strong predictor of progressive renal disease in individuals with and without diabetes mellitus. 20 , 22 , 24 Microalbuminuria has also been associated with neoplasia and other illnesses in humans. 19–21 Recent evidence in the veterinary literature suggests that the presence of microalbuminuria is associated with various disease states and may precede the development of overt proteinuria. 6 , 12 The prevalence of microalbuminuria in healthy dogs has ranged from 19–24.7%. 9 , 23 However, the prevalence of microalbuminuria in dogs with various illnesses that presented to referral hospitals ranged from 30–61%. In another study, more than half of the dogs that had microalbuminuria but did not have overt proteinuria had an underlying systemic disease. 25 More specifically, microalbuminuria in dogs has been associated with heartworm disease, lymphosarcoma, and osteosarcoma, and microalbuminuria frequently preceded overt proteinuria in these dogs and others with familial nephropathies. In light of these findings, it seems possible that dogs that have canine borreliosis and microalbuminuria are more likely to be those that develop overt proteinuria and Lyme nephritis. Microalbuminuria screening of Lyme disease-positive dogs to identify early cases of Lyme nephritis will only be useful if the test specifically identifies early cases, and it will not be useful if every Lyme disease positive dog is also microalbuminuria positive.
In the past, the serodiagnosis of Lyme disease has been accomplished by using a kinetic enzyme-linked immunosorbent assay (ELISA) or indirect fluorescent antibody assay for B. burgdorferi and confirming positive results via Western blot. 14 The Western blot has traditionally been regarded as the diagnostic standard for detecting infection. 16 More recently, detection of antibodies against the invariable region of the surface antigen of B. burgdorferi VlsE, or the synthetic analogous peptide known as C6, has been found to be sensitive and specific for the serodiagnosis of infection and is not affected by vaccination. 15 A previous study was done in conjunction with the manufacturer to evaluate a new point-of-care version of the C6 ELISA. a , 14 In this study the point-of-care ELISA was found to be 100% sensitive and 100% specific (N = 18) and was negative for dogs exposed only to the vaccine. These results were 100% consistent with the Western blot results. 14 These findings are very promising, but the sample size was small and further investigation of this test was warranted.
The goals of the present study were 2-fold: to identify possible associations between natural exposure to B. burgdorferi and the presence of microalbuminuria in young, asymptomatic Labrador and Golden Retriever dogs and to evaluate the agreement between the results of the commonly used point-of-care ELISA a and the Western blot. The hypotheses were that microalbuminuria would not be associated with positive serology via the Western blot or the ELISA and that the point-of-care ELISA and Western blot tests would yield similar results.
Study subjects were 268 apparently healthy Labrador and Golden Retriever dogs (median age = 1.5 years, range = 1.5–12 years). A few dogs had missing values for some tests. All dogs were affiliated with a guide-dog facility in New York State. b More than 90% of the dogs had spent their first 18 months with foster families, largely in areas in which Lyme disease is endemic. Blood and urine samples were obtained from all dogs. All dogs received monthly flea and tick preventive medication. The dogs were vaccinated at the discretion of the foster families' veterinarians.
Blood and urine samples were collected from younger dogs when they returned to the guide-dog facility at the age of approximately 18 months for training or for an annual examination. Older dogs were active guide-dogs or members of the breeding colony located near the guide-dog facility, so blood samples were obtained at the time of study or during an annual examination. Blood was collected in non-anticoagulated and ethylenediaminetetraacetic acid-anticoagulated tubes and was then centrifuged for serum and plasma. Serum and urine were frozen at −20°C (−4°F) and sent to Cornell University in Ithaca, New York. Immediately after blood collection and separation, the point-of-care C6 peptide ELISA a was performed on the plasma samples. The test was performed according to the manufacturer's instructions by one of the authors (BAB). A kinetic ELISA and Western blot were performed on sera from all dogs in a large veterinary diagnostic laboratory. c The 2 tests were performed in a blinded fashion to the results of the point-of-care C6 peptide ELISA. Because of differences in antibody response and banding patterns, the Western blot has been shown to distinguish between dogs infected with and vaccinated against B. burgdorferi. 3 In this commercial laboratory, Western blot results are occasionally subjectively interpreted as equivocal. This means that either only 2 of the 6 bands associated with Lyme infection 11 are thought to be present, or that 3 or more are present but are too faint to be read with confidence. If this occurred in the interpretation of samples from this study they were considered to be low positive. Finally, the presence of microalbuminuria in urine was determined using the point-of-care ELISA d according to the manufacturer's instructions. This testing was performed according to the manufacturer's instructions at Cornell University by one of the authors (REG), who was blinded to all the serologic results for B. burgdorferi. Results of the serology and microalbuminuria tests were reported in 2 ways: positive or negative and, more subjectively, as negative, low, medium, or high positive. The manufacturers of the point-of-care C6 peptide ELISA a and the micro-albuminuria ELISA d include criteria for this more subjective, semiquantitative scoring, based on the intensity of the color change of the ELISAs. The commercial diagnostic laboratory c used its standard protocols 8 for determining a positive or negative result for natural infection or vaccination as well and a subjective scoring system of low moderate or high antibody concentrations for the Western blot. Briefly, this determination is made subjectively based on the number and intensity of the bands known to be associated with infection. 8 The presence of 3 of 6 bands, for example, is required for a low positive result; the presence of 6 bands or 5 very intense bands would be a strong positive.
Nonparametric statistical tests were used to analyze the study data. Cross-tabulations and Wilcoxon's rank-sum tests e were performed to investigate whether or not the microalbuminuria score (negative, low, medium, or high positive) was related to results of the Western blot or the point-of-care B. burgdorferi C6 ELISA test. Results of the serology tests were grouped in 2 ways: positive or negative and high/medium positive or low positive/negative. The Wilcoxon's rank-sum test e was also used to determine if microalbuminuria score was related to age (age < 5 years or age ≥ 5 years). Fisher's exact tests e were used to assess whether or not microalbuminuria score was related to serology findings; all results were grouped as dichotomous variables: positive or negative. Additionally, kappa f was used to quantify any agreement (beyond that expected by chance) between the point-of-care B. burgdorferi C6 ELISA and Western blot test results. A P value of ≤ 0.05 (2-sided) was considered to be significant.
Of the 268 dogs, 50 (18.7%) were positive for B. burgdorferi exposure according to the point-of-care B burgdorferi C6 ELISA test. Western-blot analysis showed that 55 of 259 dogs (21.2%) were positive for natural exposure to B. burgdorferi and 30 of 260 dogs (11.5%) were positive for vaccinal antibodies. Agreement rate between the 2 tests of natural exposure to B. burgdorferi was 93% (kappa = 0.78, P < 0.0001) (Table 1). All dogs with Western blot tests that were positive exclusively for vaccinal antibodies, based on Western blot analysis, had negative point-of-care ELISA C6 tests. There were 14 dogs that had Western blot tests positive for B. burgdorferi exposure (10 low, 3 medium, 1 high positive) and negative point-of-care B. burgdorferi C6 tests. Four dogs that were positive according to the point-of-care B. burgdorferi C6 ELISA (all low antibody level) were negative according to Western blot (Table 1).
Cross-tabulation of Lyme results: Western blot (exposure) vs. in-house Borrelia burgdorferi (Bb) ELISA.*

Agreement = 93%, K = 0.78, P < 0.0001.
Sixteen of 262 dogs (6.1%) were positive for microalbuminuria (9 low, 3 medium, 4 high positive). There was no association between semiquantitative microalbuminuria score and exposure to B. burgdorferi based on results of the Western blot (P = 0.57) or the point-of-care C6 B. burgdorferi ELISA (P = 0.53). Serology data were then grouped as follows: medium and high positive scores together and negative and low positive scores together. With this grouping, there was still no association of Lyme disease positive results with microalbuminuria via Western blot (P = 0.37) or the point-of-care B. burgdorferi C6 ELISA (P = 0.61). Of the 16 dogs with microalbuminuria, 3 were positive for B. burgdorferi according to Western blot and 4 according to the point-of-care B. burgdorferi C6 ELISA. Microalbuminuria data were grouped into dichotomous variables (positive or negative), and still no association was found between microalbuminuria and the Western blot (P = 0.74) or the point-of-care B. burgdorferi C6 ELISA (P = 0.74) (Tables 2 and 3). Furthermore, there was no relationship between the presence of microalbuminuria in older dogs (age ≥ 5 years) compared to younger dogs (age < 5 years; P = 0.14) or between microalbuminuria and B. burgdorferi vaccination based on Western blot analysis (P = 0.16).
The results from the present study demonstrate very good agreement between the point-of-care B. burgdorferi C6 peptide ELISA and the referral laboratory standard Western blot for antibodies associated with natural exposure to B. burgdorferi in asymptomatic young Labrador and Golden Retrievers. This finding is consistent with the prior study of these two diagnostic methods. 14 The percent agreement in the present study (93%) is likely more accurate than the previously reported 100% because of the much larger sample size. In most cases in which results of the 2 tests were discordant, the Western blot was generally positive and the point-of-care B. burgdorferi C6 peptide ELISA was negative. When used semiquantitatively, the readings on the Western blot were occasionally higher than on the point-of-care B. burgdorferi C6 peptide ELISA. It is impossible to say whether these findings are a result of the subjectivity of the semiquantitative assessment of the 2 tests or a tendency for overreading the Western blot or under-reading the point-of-care B. burgdorferi C6 peptide ELISA, as there is no other gold standard test for comparison. It may be that the present findings arise from slightly different thresholds for identifying antibodies at very low concentrations. Obviously, a slightly higher threshold would decrease sensitivity but would increase specificity and vice versa.
Cross-tabulation of Lyme results: Western blot (exposure) vs. microalbuminuria.*

Fisher's exact test (2-sided), P = 0.74.
Cross-tabulation of Lyme results: In-house Borrelia burgdorferi (Bb) ELISA vs. microalbuminuria.*
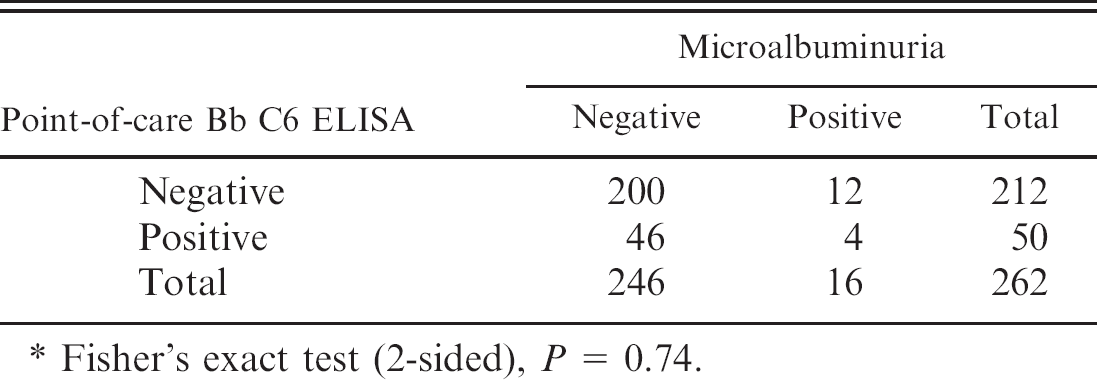
Fisher's exact test (2-sided), P = 0.74.
Additionally, the results confirm that the point-of-care B. burgdorferi C6 peptide ELISA does not detect vaccinal antibodies; there were no dogs with positive point-of-care B. burgdorferi C6 peptide ELISA results that had only been exposed to vaccinal antigens as detected by Western blot. This finding is consistent with previous research showing that the ELISA based on the C6 peptide is specific for detecting infection versus vaccination. 24 Overall, the point-of-care B. burgdorferi C6 peptide ELISA appears to be a reliable test with a sensitivity and specificity similar to that of the Western blot for detecting exposure to but not vaccination against B. burgdorferi.
The prevalence of B. burgdorferi exposure in this study (18.7–21.2%) is less than that typically seen in many areas in which Lyme disease is endemic. In some areas, more than 50% of healthy dogs have been shown to have antibodies against B. burgdorferi. 13 The low prevalence in this study is most likely attributable to the fact that all of the dogs received monthly tick preventive treatment. Tick control has been recommended as an effective way to reduce the risk of Lyme disease and transmission of B. burgdorferi. 18 Additionally, the population of dogs in the present study was relatively young, and prevalence of exposure to the etiologic agent of Lyme disease might increase with age.
The prevalence of microalbuminuria in this study (6.1%) is consistent with what has been reported in the past for dogs that are 1.5 years of age. 9 Although no relationship between microalbuminuria and age was found in this study, there were few dogs older than 10 years, and geriatric dogs appear to have the highest incidence of microalbuminuria. The results showed no relationship between the presence of microalbuminuria and exposure to B. burgdorferi via either the Western blot or the point-of-care B. burgdorferi C6 peptide ELISA test in clinically healthy dogs. Furthermore, vaccination against Lyme disease, based on Western blot findings, was not associated with the development of microalbuminuria. The current findings are similar to those of a previous study of Bernese Mountain Dogs, where microalbuminuria was also unrelated to the presence of B. burgdorferi antibodies. 4
The results of this study indicate that microalbuminuria is not a common consequence of Lyme disease exposure in healthy young Labrador and Golden Retrievers. Additional studies are warranted to evaluate the possible occurrence of microalbuminuria before the development of overt proteinuria in dogs that do eventually develop Lyme nephritis.
Acknowledgement. Source of Funding for this study: HESKA Corporation, Fribourg, Switzerland.
Footnotes
a.
SNAP-3Dx heartworm, Ehrlichia canis, and Borrelia burgdorferi test, IDEXX Laboratories, Westbrook, ME.
b.
The Guiding Eyes for the Blind, Yorktown Heights, NY.
c.
New York State Animal Health Diagnostic Center, Cornell University, Ithaca, NY.
d.
Heska E.R.D.—Healthscreen, HESKA Corporation, Fribourg, Switzerland.
e.
Statistix 8 Analytical Software, Tallahassee, FL.
f.
Epi Info 6 v. 6.04b, CDC, Atlanta, GA.
