Abstract
The blacklegged tick (Ixodes scapularis), which transmits Borrelia burgdorferi, the causative agent of Lyme disease, has undergone rapid range expansion in Ontario. In horses, Lyme disease remains an enigmatic disease, with limited understanding of the pathogenesis and many issues pertaining to selection and interpretation of laboratory tests. We evaluated B. burgdorferi seropositivity in naturally exposed horses over a 12-mo period and compared paired samples with 2 common serologic tests. Serum samples were collected in 2017, ~1 y after initial testing, from a cohort of 22 horses that were seropositive in a 2016 seroprevalence study. Samples were tested using a C6 ELISA and a multiplex ELISA targeting outer surface proteins A, C, and F. 1 y after initial testing, 14 of 22 (64%) horses remained seropositive; 7 (32%) were positive on the multiplex ELISA, 2 (9%) on C6 ELISA, and 5 (23%) on both tests. Repeatability was 100% for the C6 ELISA, and 95% for the multiplex ELISA, with no significant difference between paired sample multiplex titer values. Our results indicate strong intra-test reliability, although further investigation is required to determine the clinical significance of serologic testing.
There have been considerable increases in the abundance and diversity of ticks in many areas of Canada, with corresponding increases in tick-borne diseases.5,6,20 In Ontario, Lyme disease, which is caused by the spirochete Borrelia burgdorferi and is transmitted by the blacklegged tick (Ixodes scapularis), has been a growing concern.3,5,17,19,20 In horses, Lyme disease can manifest as complex infections, although the full clinical spectrum of equine Lyme disease is not fully understood. 8
Serologic testing is commonly used to determine if horses have been exposed to B. burgdorferi.10,22,23 Horses living in regions where B. burgdorferi is endemic may have antibodies as a result of previous exposure, but may never exhibit clinical disease.7,8,14 Using 2 ELISAs, a 2020 study in Ontario found 17% seroprevalence of B. burgdorferi in clinically healthy horses, although there were discrepancies in agreement between the 2 serologic tests used. 16 Additionally, a gold standard testing method does not exist for horses, which complicates clinical testing and assessment of available tests. 8
Two main Lyme disease serologic tests are used in North America. One is a multiplex ELISA developed and offered at the Cornell University Animal Health Diagnostic Center (CU-AHDC; Ithaca, NY) that targets 3 outer surface proteins (Osps) of B. burgdorferi: OspA, OspC, and OspF (Lyme disease, equine multiplex assay; CU-AHDC).21,22,25 It has been suggested that this test can help determine the infection stage or vaccination status of the horse given that the Osps are expressed transiently, and host antibodies develop at different stages of infection.12,22,23 The multiplex ELISA uses recombinant B. burgdorferi OspA, OspC, and OspF coupled to beads that are detected by a fluorescent conjugate, and values are expressed as median fluorescent intensities. 22 Antibodies to OspA are considered to be detectable in horses that either have been vaccinated with a canine B. burgdorferi vaccine or are in the early stage of infection, and are often undetectable by the time clinical signs appear.4,22,25 OspC seropositivity indicates acute infection, and may not be detected on follow-up testing.22,23 In contrast, OspF seropositivity has been reported to develop 5–8 wk after exposure, can persist for many years, and is interpreted as a marker of chronic infection.9,22,23
The other commonly used ELISA detects antibody response to the synthetic peptide C6, corresponding to a highly conserved region (IR6) of the B. burgdorferi surface lipoprotein VlsE. 11 This method has shown greater sensitivity and specificity as a single-tier option to the common 2-tier approach (ELISA and western blot) used in humans. 13 The C6 ELISA is a lateral-flow antibody immunoassay using a proprietary SNAP device that utilizes reversible chromatographic flow of sample and sequential flow of wash and enzyme substrate, in which color development on the sample spot indicates B. burgdorferi antibody presence (SNAP 4Dx Plus test; Idexx).10,18 The C6 ELISA offers point-of-care testing that is not influenced by vaccination and is often used for testing dogs, and although it has been validated for equine use, anecdotally it is not used commonly by equine practitioners.10,12,18
The agreement between these 2 serologic tests and their ability to detect B. burgdorferi infections can vary, and few studies have analyzed B. burgdorferi antibodies in horses at more than one time. 16 Our objectives were to evaluate B. burgdorferi seropositivity in naturally exposed horses over a 12-mo period, and to determine the repeatability of these 2 common serologic tests using paired serum samples.
Horses that had been identified as positive on a multiplex ELISA or C6 ELISA during a 2016 prevalence study in Ontario, Canada, were eligible for enrollment. 16 Twenty-two of 91 (24%) seropositive horses were available for retesting 1 y after the initial test. Our study was approved by the University of Guelph Animal Care Committee (Animal Use Protocol 3257).
Serum samples were submitted between August and September of 2017 and saved as 4 aliquots per horse. The sera were stored at −80°C and thawed in a water bath at 37°C at the time of testing. Two aliquots from each horse were tested simultaneously for the presence of B. burgdorferi antibodies using the C6 ELISA (Idexx), with results reported as positive or negative, according to the manufacturer’s instructions. The other 2 aliquots were tested for antibodies against B. burgdorferi Osps A, C, and F via the multiplex ELISA (CU-AHDC). The multiplex result is reported with titers for antibodies to each Osp antigen, with interpretation of positive, negative, or “equivocal” (indicates very early infection or can be induced by nonspecific serum reactions).
The Cohen kappa test with 95% confidence intervals was employed to determine the agreement between the 2 tests. Paired t-tests were used to compare initial and follow-up testing titers. A significance level of α = 0.05 was used for all statistical analyses. Statistical analysis was conducted using STATA v.14 (StataCorp).
The 22 initially positive horses that were available for resampling ~1 y later comprised 14 geldings and 8 mares with a median age of 13 y (range: 6–23 y). No horses had clinical signs consistent with Lyme disease at the time of initial sampling, and none had developed signs attributed to Lyme disease during the intervening 1-y period. The initial sample collection was completed in July–August 2016, and the follow-up sampling was in August–September 2017.
1 y after initial testing, 14 of the 22 (64%) initially positive horses were positive on at least one of the B. burgdorferi tests. Five of the 14 (36%) horses were positive on both tests, 7 of 14 (50%) horses were positive to 1 or more of Osps A, C, or F, and 2 of 14 (14%) were positive on the C6 ELISA only (Table 1). There were no significant differences between OspF and OspA titers from initial testing in 2016 and follow-up testing in 2017 (p > 0.05). Nine of 14 (64%) horses had the same test results as the previous year. Five of the 10 (50%) horses that had a positive OspF titer in 2016 changed to an equivocal OspF titer value (Fig. 1), and 1 horse that was OspA positive initially had an equivocal result 1 y later (Fig. 2); another previously negative horse became positive. No horses tested positive for OspC during follow-up testing.
Change in serum antibody titers against Borrelia burgdorferi in 22 horses tested in July–August 2016 and in August–September 2017.
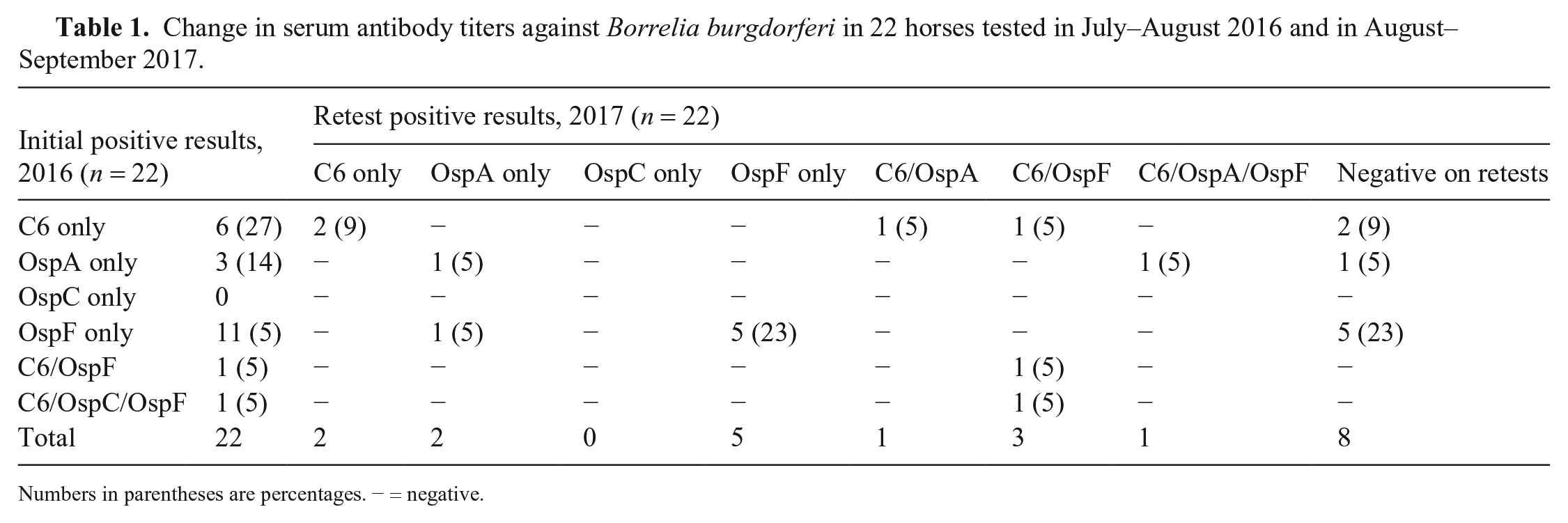
Numbers in parentheses are percentages. − = negative.
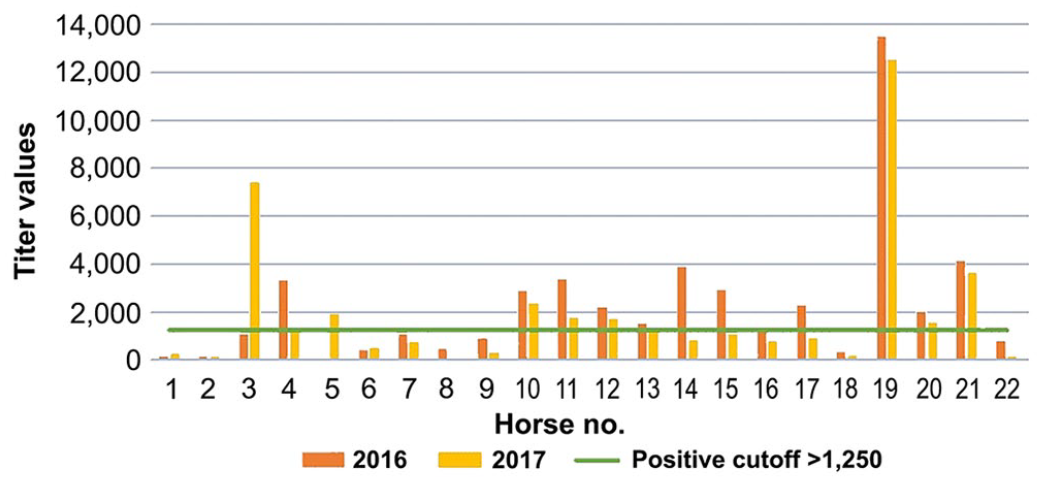
Borrelia burgdorferi outer surface protein F (OspF) titers for each of 22 horses in the multiplex ELISA for 2016 (orange bars) and 2017 (yellow bars). Cutoff >1,250 (green line), equivocal 750–1,250, and negative titers <750.
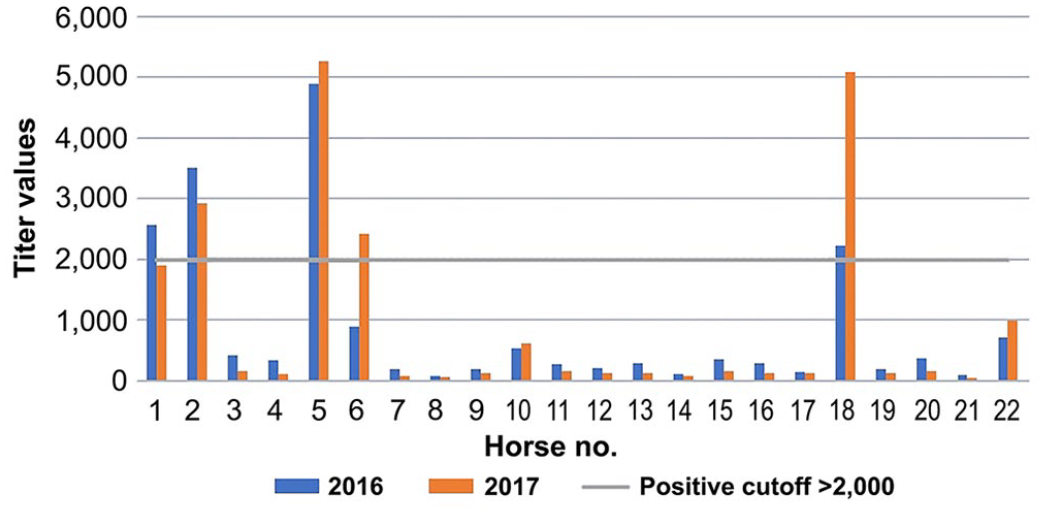
Borrelia burgdorferi outer surface protein A (OspA) titers for each of 22 horses in the multiplex ELISA for 2016 (blue bars) and 2017 (orange bars). Cutoff (gray line), equivocal 1,000–2,000, and negative titers <1,000.
Testing of the aliquots from 2017 with the C6 ELISA identified complete agreement, with 7 paired aliquots testing positive for B. burgdorferi. Eleven of 12 paired aliquots tested using the multiplex ELISA yielded consistent qualitative (positive, negative, equivocal) results (kappa = 0.91). The paired aliquot with discordant findings was positive for OspF (titer = 1,548) on one aliquot but equivocal (titer = 1,095) on the other (equivocal range: 750–1,250) 22 ; this difference in antibody titer was not statistically significant (p = 0.78).
Our results show that naturally exposed horses may remain seropositive for at least 12 mo after initial testing by either serologic test. Although duration of seropositivity has been assessed minimally in horses, it is important for understanding the pathogenesis and immune response of Lyme disease and for assessment of test results.
Differentiation of persistent antibodies from antibodies generated by re-exposure is challenging in horses that remain in endemic regions. 8 Although this is a major limitation of our study, there was limited evidence for widespread re-exposure using the current interpretation of antibody expression.6,19 No horses had positive OspC titers at the time of follow-up testing, suggesting that there was no exposure in the past 3–5 mo.23,24 In our study, this timeframe equates to spring or early summer, when the likelihood of exposure is much higher given the higher activity of adult and nymphal ticks, respectively.6,19 Risk of re-exposure would also have been high during the fall after the initial sampling as a result of peak adult tick activity. 19
Previously, in dogs, it was observed that OspC developed prior to C6 and OspF, and C6 prior to OspF. 21 Following that study, antibody expression was studied in horses, and it appeared that OspC developed early as well and that horses became seropositive to OspF prior to C6.21,23
When examining titers over the 2 sampling periods, OspF was the most commonly detected Osp during both years of testing. A positive OspF antibody titer is thought to indicate historical exposure and may remain above the positive threshold for years.2,15,22,25 On the C6 ELISA, 5 of 7 horses remained seropositive, which is consistent with a study in B. burgdorferi experimentally infected dogs in which serum antibodies were detectable up to 69 wk post-infection. 11 Comparable data are not available for horses, but our study provides evidence that there may be similar persistence of C6 antibodies in horses. C6 and OspF antibodies might both be under continuous expression, given that both appear to be markers of chronic or historical exposure. 23 Further analysis is necessary to examine why some of the horses in our study only expressed C6 or OspF antibodies at both sampling times; whether persistence of antibody expression has any clinical relevance requires more research.
It has been suggested that a positive OspA titer can be from vaccination or may be a very early indicator of infection; however, data from our study are inconsistent with that suggestion.1,18,21,22 None of the horses in our study that had positive OspA titers had been vaccinated against Lyme disease. Furthermore, 3 horses that had positive OspA titers initially remained OspA positive, with only one of those subsequently developing antibodies against OspF and C6. Another horse, which was previously positive for OspA and OspF, was only seropositive for OspA on retesting. These findings raise questions about the recommended interpretation.
Although it is reasonable to consider the potential impact of antimicrobial treatment on development of Osp titers following early infection, none of our horses was reported to have been treated with antimicrobials during the period shortly after initial sampling, and results from the initial sample were not reported to owners and veterinarians until months later. Therefore, antimicrobial-associated impacts on Osp titers are unlikely.
The precision within repeated test results is important for any laboratory test. Although agreement between the 2 tests was limited, the use of paired samples confirmed that intra-sample repeatability was high with both tests. Although it is difficult to determine test accuracy at this time, the tests appear to perform with strong precision.
The small sample size is a limitation of our study, given that only a subset of positive horses was available for retesting. Studies of larger populations will provide more precise information, as will studies with increased frequency of resampling. The potential for re-exposure is an ever-present risk and can only be eliminated with a controlled population in laboratory-based studies.
