Abstract
It is well known that tumor-infiltrating lymphocytes (TILs) and peripheral blood lymphocytes (PBLs) from patients with advanced-stage cancer have a poor immune response. Regulatory T cells (Tregs), characterized by the expression of a cluster of differentiation 4 and intracellular FoxP3 markers, can inhibit antitumor immunoresponse. In the present study, the prevalence of Tregs in peripheral blood and tumor tissue from dogs with oral malignant melanoma was evaluated by triple-color flow cytometry. The percentage of Tregs in the peripheral blood of the dogs with malignancy was significantly increased compared with healthy control dogs, and the percentage of Tregs within tumors was significantly increased compared with Tregs in peripheral blood of dogs with oral malignant melanoma. This finding suggests that the presence of tumor cells induced either local proliferation or selective migration of Tregs to tumor-infiltrated sites. A better understanding of the underlying mechanisms of Treg regulation in patients with cancer may lead to an effective anticancer immunotherapy against canine malignant melanoma and possibly other tumors.
The emergence of a tumor results from the disruption of cell growth regulation as well as from a failure of the host to provoke a sufficient immunologic, antitumor response. In human medicine, it is well known that tumor-infiltrating lymphocytes (TILs) and peripheral blood lymphocytes (PBLs) from patients with advanced-stage cancer have a poor immune response. 13
Regulatory T cells (Tregs), which are from a cluster of differentiation (CD)4+ T-cell lineage, are thought to be a functionally unique subset of T lymphocytes that play an important role in maintaining immune homeostasis. 18 Recently, intracellular detection of transcription factor FoxP3 has been shown to uniquely identify a highly enriched Treg population in rodents 5 and is considered the most specific Treg marker. In dogs, Tregs have been identified using an antibody specific for canine CD4 and murine FoxP3. 2 In addition, patients and experimental animal models of cancer showed that Tregs down-regulated the activity of the antitumor effector functions resulting in T-cell dysfunction in cancer-bearing hosts. 1,9,11,12 In human medicine, an increased population of Tregs in peripheral blood and tumors has been reported in patients with various cancers. 9,11 In veterinary medicine, a previous study reported that an increased population of Tregs was observed in the peripheral blood of dogs with various cancers 2 ; however, no previous reports have described Tregs in tumors from dogs with malignant cancer. In the present study, the prevalence of FoxP3+ CD4+ lymphocytes or Tregs was evaluated in the peripheral blood and tumors of dogs with oral malignant melanoma, using triple-color flow cytometry.
Seven dogs with oral malignant melanoma and 10 healthy control dogs were examined in the current study. Study animals had clinical signs associated with primary oral melanoma. Inclusion criteria consisted of owner consent for sample collection, procurement of a hepa-rinized blood sample before surgery, and surgical collection of a tumor specimen by incisional or excisional biopsy. One portion of the tumor specimen was used for detection of TILs, whereas the other portion was used for histologic diagnosis. Animals that had received immunosuppressive treatment (anticancer drug, radiotherapy, or corticosteroids) were excluded from the present study. The mean age of the dogs with oral malignant melanoma was 10 years (range: 8–15 years). The healthy control dogs also had a mean age of 10 years (range: 7–13 years). The present study was approved by the ethical committee at the Japan Animal Referral Medical Center.
Heparinized peripheral blood was obtained from dogs with malignant melanoma and from the healthy control dogs. Peripheral blood mononuclear cells (PBMCs) were isolated using a Ficoll density gradient according to a previous report. 7 For isolation of TILs, the tumor biopsy specimen was minced into 1-mm pieces, followed by enzyme digestion with 200 U/ml Dispase a for 2 hr. To determine immunophenotype, PBMCs and TILs were stained for cell surface molecules and intracellular FoxP3 using anti-CD4—fluorescein isothiocyanate, b anti-FoxP3—R-phycoerythrin (PE), c and anti-CD45—allophycocyanin (APC) d monoclonal antibodies. Intracellular FoxP3 staining was performed as previously described, 8 followed by triple-color flow cytometry. e Cell populations were then analyzed using flow cytometry software. e Lymphocytes were gated according to their CD45—APC intensity and side scatter characteristics (Fig. 1). The Treg percentage was derived from the percentage of FoxP3+ cells within the total CD4+ lymphocyte population (Fig. 2). The results were presented as mean values with associated standard deviations. Differences between dogs with malignant melanoma and healthy control dogs were assessed by the Mann—Whitney U test, and statistical analyses were performed using a spreadsheet program. f P values less than 0.05 were considered significant.
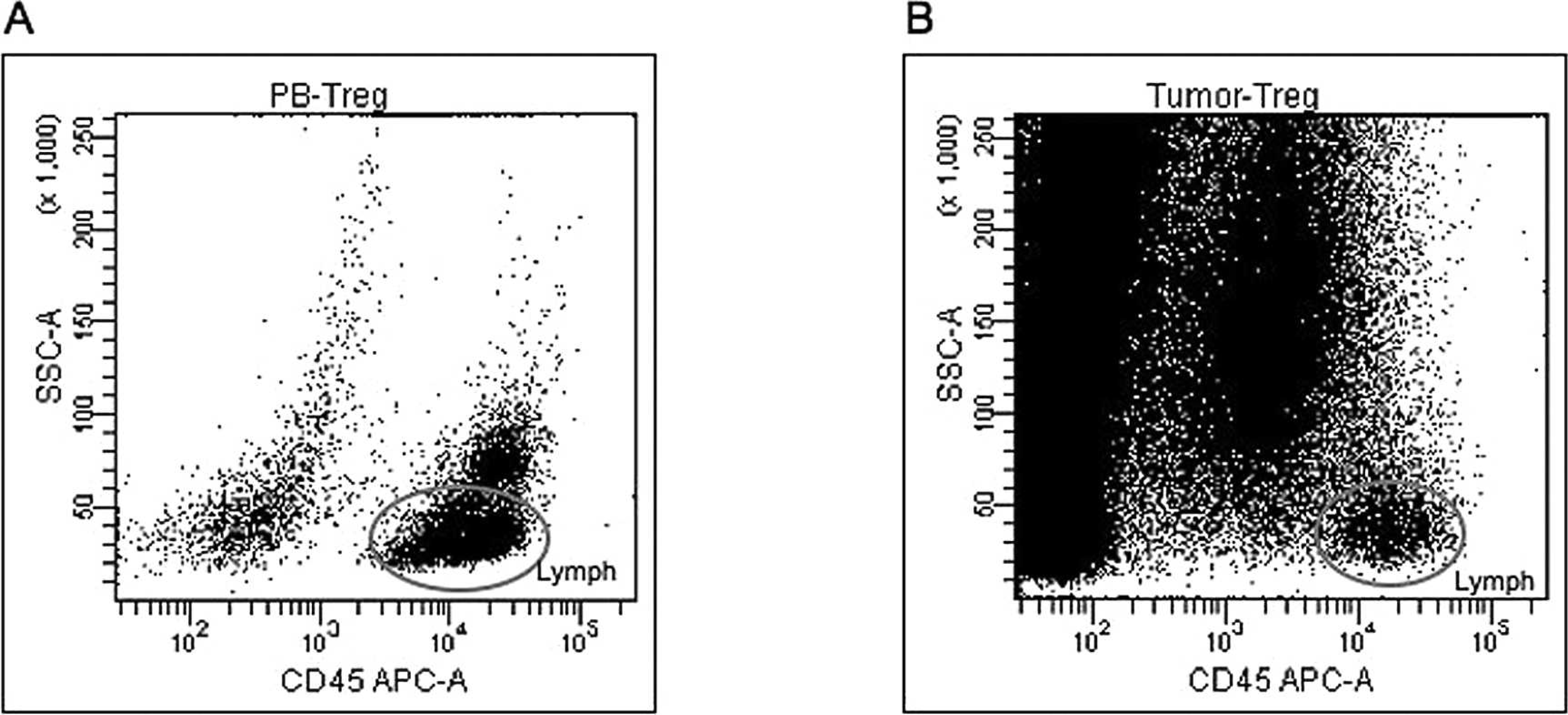
Identification of canine lymphocyte using a cluster of differentiation (CD)45 antibody. Lymphocytes were gated according to their CD45—allophycocyanin (APC) intensity and side-scatter characteristics (SSC).
The percentage of Tregs in peripheral blood of dogs with malignant melanoma was significantly increased compared with that of healthy control dogs (healthy control dogs, 4.1% ± 0.98; dogs with malignant melanoma, 10.1% ± 3.39; P < 0.001; Fig. 3). Similarly, the percentage of Tregs within tumor tissue was significantly increased compared with Tregs in peripheral blood (25.0% ± 7.32; P < 0.001; Fig. 3). In human medicine, several published reports are similar to the findings in the present study. A previous human study reported that the percentage of Tregs in patients with melanoma was significantly increased compared with that in healthy donors, and the percentage of Tregs in tumor tissue was significantly increased compared with Tregs in peripheral blood. 10 A higher prevalence of FoxP3+ CD4+ T cells was observed in tumor-infiltrated tissues, suggesting that the presence of tumor cells induced either local proliferation or selective migration of FoxP3+ CD4+ T cells into sites of tumor infiltration. One possible scenario could be that some cytokines, such as C-C motif chemokine ligand 22 (CCL22), transforming growth factor-β (TGF-β), or prostaglandin E2 (PGE2) secreted by the tumor microenvironment, possibly facilitated the attraction and local proliferation of FoxP3+ CD4+ T cells, as well as the in situ conversion of CD4+ T cells into Tregs. 4,6,19 In this way, a tumor cells could promote their growth through selective expansion of Tregs. According to recent studies, canine malignant melanoma expressed TGF-β and cyclo-oxygenase-2—PGE2. 3,14,15 These growth factors could provoke the expansion of Tregs, as observed in the current study. To the authors' knowledge, no published studies have assessed the possible role of canine CCL22 in intratumor lymphocyte migration. Further studies are necessary to substantiate these hypotheses.
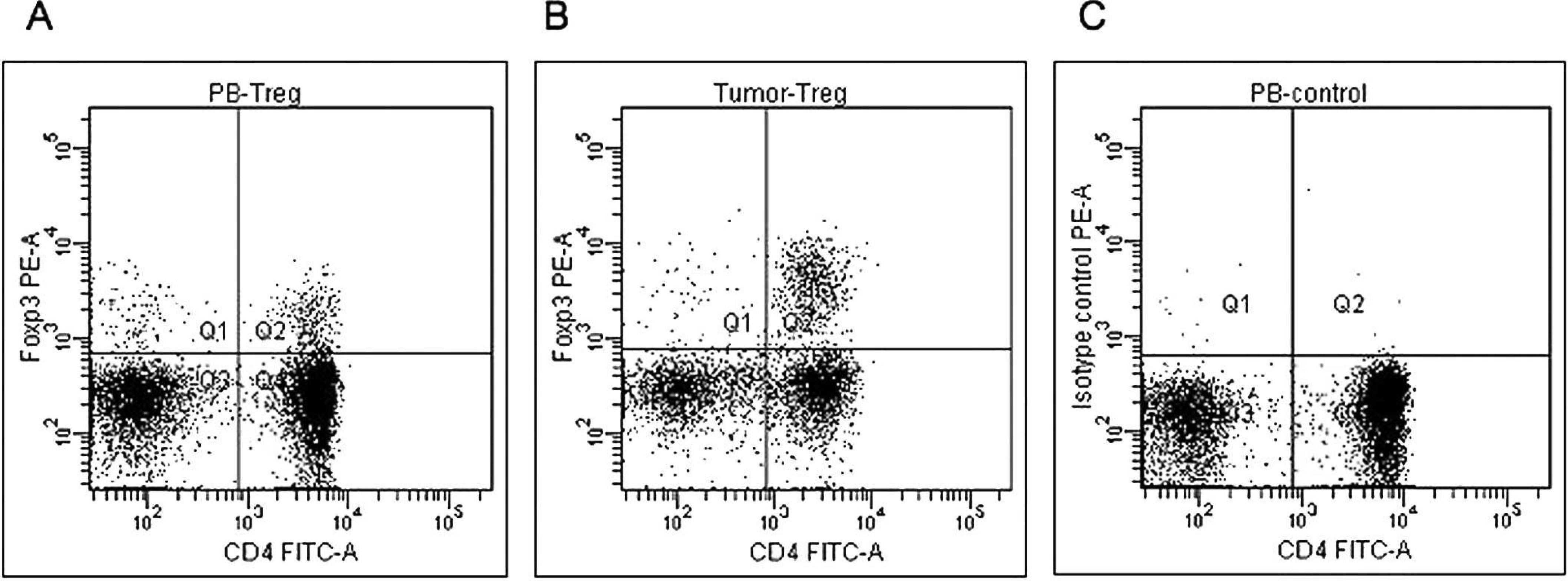
Identification of canine lymphocytes using FoxP3 antibody. Specimens of
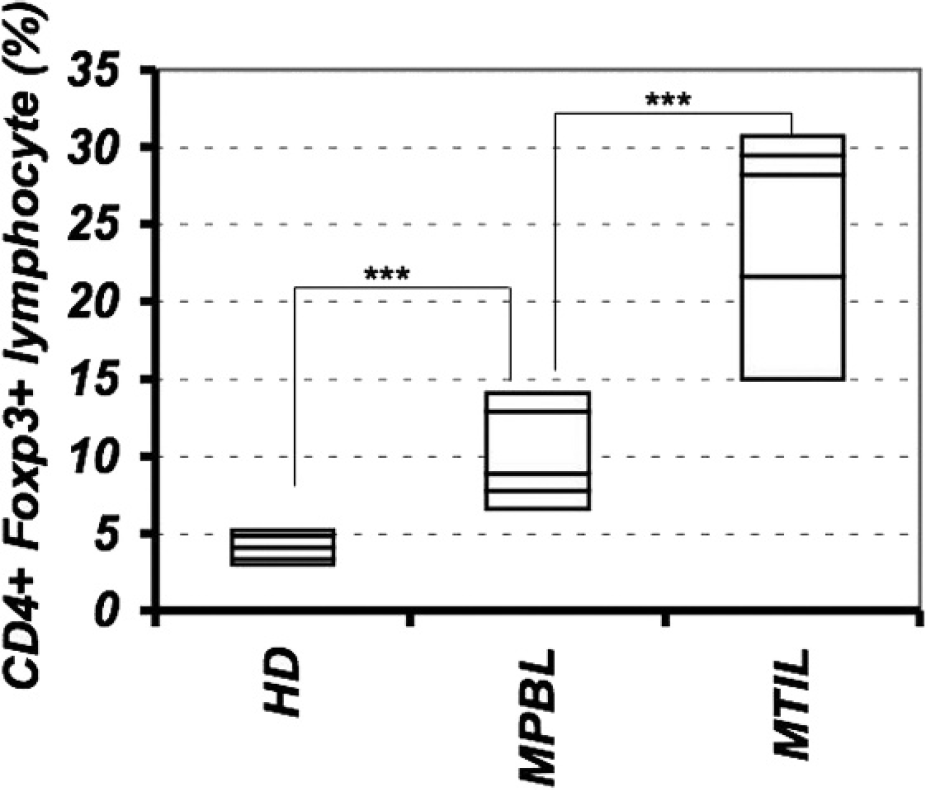
Increased populations of regulatory T cells (Tregs) in the peripheral blood and tumor tissue of dogs with oral malignant melanoma. Treg percentage was the percentage of FoxP3+ cells within the total cluster of differentiation (CD)4+ lymphocyte population in the peripheral blood of healthy control dogs (HD) or dogs with oral malignant melanoma (MPBL) or in tumor tissue (MTIL). Regulatory T cells in the peripheral blood were significantly higher in dogs with oral malignant melanoma than in healthy control dogs, and Tregs in tumor tissues were significantly higher than those in the peripheral blood were. *** P < 0.001.
The observation in the present study that dogs with malignant melanoma had increased numbers of Tregs in the peripheral blood and in tumor tissues compared to healthy control dogs is remarkably similar to the findings in human patients with melanoma. 10 Regulatory T cells are known to block antitumor activity. 16 Indeed, a previous study 17 in human patients with metastatic melanoma demonstrated that Treg depletion with diphtheria toxin conjugate and recombinant interleukin 2 (DAB/IL2; denileukin diftitox, Ontak) increased CD8+ T cells that recognized melanocyte differentiation antigens (such as Mart1, gp100, and tyrosinase) and caused regression of melanoma metastases. A better understanding of the underlying mechanism of Treg regulation or of the strategy for controlling Tregs in dogs may lead to more effective immunotherapy for canine malignant melanoma.
Acknowledgements The authors thank Mr. Ryudo Gouhara, Mr. Sho Kimijima, Miss Yoko Okumura, Miss Madoka Nakamura, Miss Aya Nishiyama, and Miss Youko Kurihara for their veterinary technical support.
Footnotes
a.
Godo-Shusei Co., Tokyo, Japan.
b.
Clone YKIX302.9, AbD Serotec Ltd, Oxford, United Kingdom.
c.
Clone FJK-16s, eBioscience Inc., San Diego, CA.
d.
Clone YKIX716.13, AbD Serotec Ltd, Oxford, United Kingdom.
e.
FACSCanto™ II, FACSDiva™; BD, Franklin Lakes, NJ.
f.
Excel®, Microsoft Corp., Redmond, WA.
