Abstract
False-positive results on serologic assays for Mycobacterium avium subsp. paratuberculosis (MAP) are believed to occur due to cross-reacting antibody produced by Corynebacterium pseudotuberculosis (C. pstb) infection in goats. This issue of compromised specificity was evaluated by testing 771 adult goats from 10 Midwestern goat herds in 2004. Assays for MAP infection included radiometric fecal culture and 2 enzyme-linked immunosorbent assays (ELISAs); ELISA-positive samples were tested by agar gel immunodiffusion (AGID). A synergistic hemolysin inhibition assay (SHI) was used to detect C. pstb antibody. Four infection status categories were evaluated. Category 1 goats (free of both MAP and C. pstb infection) tested negative on all MAP fecal cultures and SHI tests. Five of 181 goats were positive in both ELISAs, and 2 more were positive in ELISA-1 only. For Category 2 (MAP infected; no C. pstb infection), all animals were SHI negative. Six goats were fecal culture positive and strongly positive in both ELISAs; 2 more goats were positive only in ELISA-1. For Category 3 (C. pstb infected or vaccinated; no history of MAP infection), all fecal cultures were negative and 91% were SHI test-positive. In this population, only 2 goats were positive in both MAP ELISAs, while 84 additional goats were test-positive only on ELISA-1. In the absence of C. pstb infection, both ELISAs performed comparably, but when C. pstb infection was present the performance of ELISA-1 was significantly perturbed. Use of the ELISA-2 for goats is an effective and efficient method for Johne's disease surveillance in any goat herd.
Mycobacterium avium subsp. paratuberculosis (MAP) infection in goats causes chronic and contagious granulomatous enteritis called paratuberculosis or Johne's disease (JD). Most goats are believed to acquire the infection through fecal-oral exposure as kids 15 but months to years may pass before clinical signs of the infection occur. Fecal shedding of the organism occurs intermittently throughout the infection, increasing in frequency and organism number as the goat becomes clinically affected. 10 Rapid weight loss is the primary sign of JD in goats as diarrhea does not occur in all cases. Antibody production in response to the infection usually commences with the onset of clinical disease.
Test and cull programs are the usual approach for controlling the infection in goat herds since neither effective treatment nor vaccine is available for small ruminants in the United States. Relatively little research has been done on JD assays for small ruminants. A factor believed to complicate serologic surveillance for JD in goats is caseous lymphadenitis (CLA). 1,8 This prevalent small ruminant disease, caused by Corynebacterium pseudotuberculosis (C. pstb) infection, is characterized by abscesses in either external or internal lymph nodes, or both. Antibody produced in response to C. pstb infection is believed to cause false-positive JD serology results in small ruminants by cross-reacting with JD antigens used in JD ELISAs, although evidence for this is primarily anecdotal.
This study evaluated whether the presence of C. pstb infection, C. pstb vaccination or both disturbed the diagnostic performance of 2 commercially available JD enzyme-linked immunosorbent assays (ELISAs) and 1 commercially available agar gel immuno-diffusion (AGID) JD assay that were routinely used as formulated in 2004 to diagnose JD in goats.
Ten Midwestern goat herds were selected for the study. Each herd was categorized according to its MAP and C. pstb infection status based on prior diagnostic testing and clinical data. A herd was considered infected with MAP if at least 1 tissue-culture confirmed case of MAP with clinical disease was documented within 1 year of the study. A herd was considered free of MAP if it had been closed for more than 2 years with no prior history of clinical JD; most herds in this category had been tested previously and were ELISA negative. A C. pstb-infected herd was defined as one with confirmed cases of CLA in goats with documented superficial lymph node abscesses. Herds free of C. pstb infection were defined as those that were closed with no clinical history of or vaccination for CLA; some herds had been tested previously and were C. pstb synergistic hemolysin inhibition assay (SHI) test-negative.
The 10 herds were assigned to 1 of 4 categories based on prior MAP and C. pstb infection status (i.e., MAP/C. pstb: negative/negative; positive/negative; negative/positive; positive/positive). Category 1 was believed free of both MAP and C. pstb infection and comprised 4 goat farms (181 samples). Category 2 contained 2 herds with a history of MAP infection but no evidence of C. pstb infection (58 samples). Category 3 consisted of 4 goat herds with a history of C. pstb infection but no prior evidence of MAP (344 samples; a majority of these goats had been vaccinated against C. pstb). The fourth category, included to illustrate a commonly found situation where both CLA and JD are present, included 1 herd (188 samples) with a history of both infections.
Blood and fecal samples were collected from 771 adult (>18 months old) goats. Feces was collected directly from the rectum and processed within 24 hours for the BACTEC radiometric liquid culture methods. a , 2 Blood samples were collected from the jugular vein into tubes without anticoagulant and centrifuged within 24 hours for serum collection. Each serum sample was tested with 2 commercially available JD ELISAs: one licensed for use in small ruminants (Parachek) b and the other, in 2004, containing a protein G conjugate (HerdChek) c known to be capable of binding antibodies from multiple species, 9,12,13,16 18 including goats. Both assays are used globally for JD surveillance in small ruminants (unpublished survey, 8th Int Colloq Paratuberculosis). 4 Commercially available AGID d and SHI e assays were also used. The latter assay detects C. pstb antibody and was performed by the California Animal Health and Food Safety Laboratory (Davis, CA). All assays were performed and interpreted according to manufacturers′ instructions. Acid-fast isolates were identified using an in-house multiplex polymerase chain reaction (PCR) assay; isolates that were 16S and IS900 positive were identified as MAP. A fecal culture-positive goat was considered MAP infected. Results were analyzed by McNemar's chi-square and and McNemar exact significance probabilities to assess the marginal differences between each ELISA per infection category. f
The data are summarized in Figure 1 and Table 1 below. Results for all 4 herd categories confirmed the predetermined classifications for each herd's MAP and C. pstb infection status. In category 1 (181 goats from 4 herds believed to be free of both MAP and C. pstb infection and test-negative by fecal culture and SHI), 7 (3.9%) were test positive in ELISA-1. c Five of these samples (2.8%) were also test positive in ELISA-2. b The 7 ELISA-positive sera were AGID negative. Statistically, the 2 ELISAs performed comparably in this MAP and C. pstb infection-free population (chi square = 2; exact McNemar significance probability = 0.50).
Nine of the 58 goats in category 2 (from 2 MAP-infected herds free of C. pstb infection and SHI negative) were positive in both ELISAs and the AGID; 7 of these were also fecal culture positive for MAP. Two additional goats were positive in the 2 ELISAs, but AGID negative; 1 of these was fecal culture positive. Two further goats were test-positive on ELISA-1 only. In this MAP-infected, C. pstb-free population, the 2 ELISAs again performed comparably (chi square = 2; exact McNemar significance probability = 0.50).
All 344 goats from the 3 herds in category 3 (C. pstb infected and/or vaccinated but free of MAP infection) were MAP fecal culture negative. For the SHI test, 91% (313/ 344) were positive with SHI titers ranging from >1:8 to 1:256. Three of these goats (0.8%) were MAP test-positive in both ELISA-1 and ELISA-2. An additional 84 goats (or 26% of goats in this category) were positive in the ELISA-1 assay only with optical density values ranging from 0.25 to 1.31. All AGIDs on ELISA-positive samples were negative. There was a statistically highly significant difference between the results provided by the 2 ELISAs in this C. pstb-infected, MAP-free population (chi square = 84; exact McNemar significance probability = 0.00).
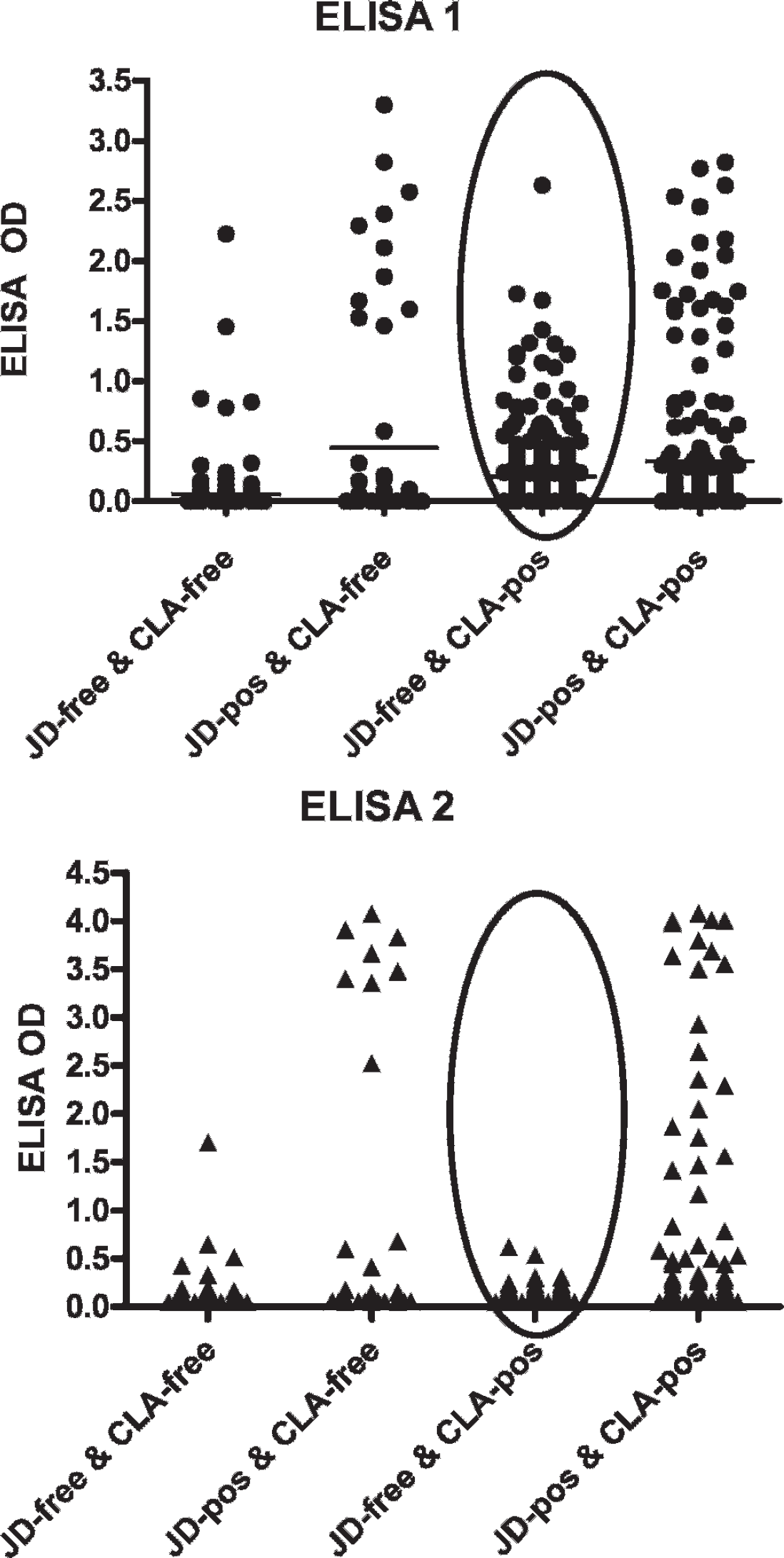
Performance of ELISA-1 and ELISA-2 in goat herds infected with neither, one, or both organisms (Mycobacterium avium subsp. paratuberculosis, Corynebacterium pseudotuberculosis).
In the category 4 herd (1 herd with both infections; 188 goats), 38 goats were MAP fecal culture positive. Fifty-one goats were ELISA-1 positive (25 shedding MAP); 43 of these were also ELISA-2 positive (23 of which were shedding). Twenty-one AGIDs were positive (13 of which were shedding). Just over 80% were SHI test-positive for C. pstb antibody (137 of 171 tested; an unknown number of these had been vaccinated). The performance of the 2 ELISAs differed significantly (chi square = 8; exact McNemar significance probability = 0.001).
Summary of herd results by infection category. *
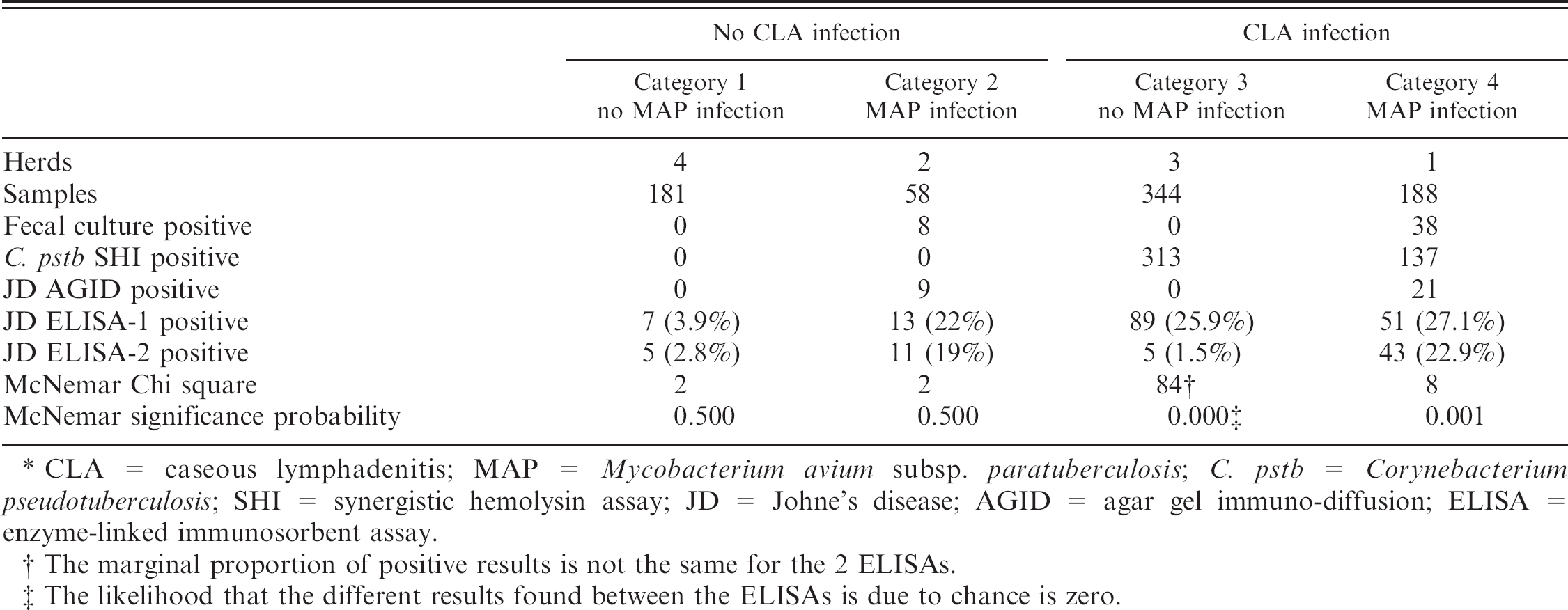
CLA = caseous lymphadenitis; MAP = Mycobacterium avium subsp. paratuberculosis; C. pstb = Corynebacterium pseudotuberculosis; SHI = synergistic hemolysin assay; JD = Johne's disease; AGID = agar gel immuno-diffusion; ELISA = enzyme-linked immunosorbent assay.
The marginal proportion of positive results is not the same for the 2 ELISAs.
The likelihood that the different results found between the ELISAs is due to chance is zero.
This study shows that in goat herds free of C. pstb infection, the 2 JD ELISAs yield similar results with an accuracy comparable to their performance in cattle herds. 3 However, when C. pstb infection is present in a goat herd (or goats have been vaccinated for C. pstb) the results from the 2 ELISAs differ significantly. False-positives occurred with the JD ELISA-1 in 25% of the MAP-free population from C. pstb-infected herds, whereas the performance of JD ELISA-2 and the AGID was unaffected by the presence of C. pstb infection. These false-positive results support the hypothesis that antibody produced by C. pstb infection or vaccination may cross-react in some JD ELISAs, presumably due to the presence of surface antigens used in the JD assays that are shared between the genetically related Corynebacteria and Mycobacteria.
The JD ELISAs performed comparably when CLA was not present in the herds, as indicated by the comparable number of false-negatives in infected populations and the low, statistically nonsignificant, number of false-positives (using fecal culture as the reference test) in uninfected herds (category 1). It is possible that these few ELISA-positive goats were truly MAP infected despite the herds' lack of JD case history, their good health, and lack of shedding at the time they were sampled. It is also possible that the few false-positive results were due to antibody produced by another mycobacterial infection, as has been seen in other species. 6,17
Fecal culture is the reference test for JD surveillance. The presence of CLA does not affect fecal culture results, and recent advances in liquid culture methods with the use of sample pooling have made this approach a cost-effective diagnostic tool for small ruminant producers. 7 The AGID is another useful test for identifying goats with JD, especially animals in the later stages of the infection. In this study, CLA infection did not cause false-positive AGID reactions. The AGID assay was, however, less sensitive than the ELISAs in herds with JD, identifying only one third of the fecal culture-positive goats (additional herd data not shown). 11
The ELISA is a sensitive, fast, and inexpensive method of determining the prevalence of MAP infection in cattle herds and has also been used effectively in sheep, elk, and deer populations. 5,6,17,18 In the United States, the ELISA serves as the basis for the national Voluntary Bovine Johne's Disease Control Program. While this program focuses on MAP infection in cattle, some states (e.g., Wisconsin, Alaska, Colorado) are now incorporating goats as an eligible species for assistance in controlling the infection. The data from this study should be considered when deciding which assays are best for determining the MAP infection status of goat herds. The ELISA platform provides producers with a cost-effective and rapid way to establish a herd's MAP seroprevalence, and the United States Department of Agriculture (USDA)-validated ELISA for goats is minimally affected by C. pstb cross-reacting antibody. Studies evaluating the ELISA platform with milk instead of serum as a method to determine herd infection status are also promising. 14 As with JD herd certification in cattle, however, when serologic evidence of the infection is first found in a goat herd, the diagnosis should be confirmed by fecal culture.
Acknowledgements. Funding for this study was provided in part by the Johne's Testing Center (School of Veterinary Medicine, University of Wisconsin) and by a USDA-APHIS-VS National Center for Animal Health Programs (NCAHP) grant. Our thanks to the goat farm owners who permitted the sampling of their animals for this study; to Dr. Roy Berghaus, Population Health and Reproduction, University of California, for his comments on data analysis; and to the Johne's Testing Center Lab staff for invaluable assistance in processing the samples.
Footnotes
a.
Becton Dickinson, Franklin Lakes, NJ.
b.
Prionics USA Inc., Lincoln, NE.
c.
IDEXX Laboratories, Westbrook, ME.
d.
Immucell Corp., Portland, ME.
e.
CA Animal Health and Food Safety Laboratory, Davis, CA.
f.
GraphPad Software, San Diego, CA.
