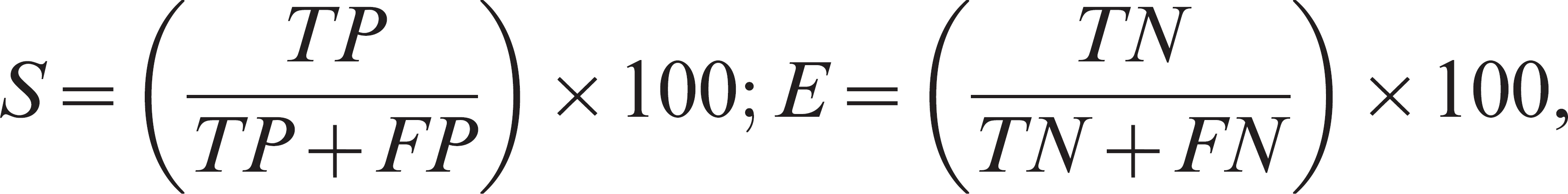Abstract
In the current study, the applicability of the quantification of gamma interferon (IFN-γ) levels for the detection of animals infected with Corynebacterium pseudotuberculosis and for determining caseous lymphadenitis (CLA) clinical status was evaluated. Peripheral blood leukocytes were collected from CLA nonendemic areas animals, from CLA seropositive animals without clinical signs of the disease, and from seropositive animals presenting CLA clinical signs. The leukocytes were stimulated with C. pseudotuberculosis—secreted antigens that were concentrated by the three-phase partitioning technique. An ovine IFN-γ enzyme-linked immunosorbent assay was used to quantify IFN-γ production. Goats and sheep with CLA had higher IFN-γ levels than uninfected seronegative animals. Leukocytes from sheep with CLA chronic abscesses produced higher IFN-γ levels when compared with seropositive sheep without CLA clinical signs, but this difference was not significant in goats. The sensitivity of the assay was 55.8% and 56%, whereas the specificity was 100% and 93%, for goats and sheep, respectively. In conclusion, IFN-γ is a potential marker for the determination of CLA infection status in small ruminants; however, further research is needed to improve assay sensitivity.
Keywords
Introduction
Corynebacterium pseudotuberculosis is the etiologic agent of caseous lymphadenitis (CLA), 3 a disease that is characterized by the formation of chronic abscesses in several organs in small ruminants. 28 Nonapparent infections are common, and failure to diagnose infection in a commercial herd results in high levels of animal-to-animal transmission and significant economic losses. 3 The disease is found worldwide and has a severe economic impact on the goat and sheep industry, which leads to decreased milk production, wasting, and condemnation of carcasses because of internal abscesses. 10
Currently, CLA diagnosis is based on the identification of characteristic clinical signs represented by the development of caseous lesions in the lymph nodes and the detection of C. pseudotuberculosis in these abscesses. 6 Serologic diagnosis is of major importance because subclinically infected animals are a potential source of infection. Many immunologic assays have been developed to detect infection, the majority of which detect the humoral antibody response to infection, 2,4,5,8,22,23,25,26 with varying degrees of success.
Sensitivity and specificity are important factors to be considered when making the choice of diagnostic assay(s) for a screening program. A test with a lower specificity can lead to false positives, whereas a reduced sensitivity can lead to false-negative results. Problems with sensitivity and specificity in immuno-diagnostic assays can be caused by many factors, especially in animals with a low or undetectable humoral response against C. pseudotuberculosis 2 as well as with cross-reactivity to other related microorganisms. 6,28
Because C. pseudotuberculosis is a facultative intracellular pathogen, cell-mediated immunity is an important component of the protective immune response. 9 Previously, a whole-blood gamma interferon (IFN-γ) assay was found to be a promising detection tool for the eradication of CLA from small ruminant flocks, 11,12,24 An alternative approach to the immunodiagnosis of CLA is the assessment of the cellular immune response to C. pseudotuberculosis antigens, 11,12,20,24 carried out concurrently with serologic assays.
In the current study, quantification of IFN-γ induced in leukocytes stimulated with secreted antigens derived from C. pseudotuberculosis T1 strain was evaluated. The objective was to determine its use in the detection of C. pseudotuberculosis infected goats and sheep, and for differentiating between subclinical and clinical CLA.
Material and methods
Serum and whole blood samples
Five milliliters of serum and heparinized whole blood were collected from goats and sheep divided into 3 groups; each group was composed of 30 goats and 30 sheep: 1) the noninfected group: healthy animals from a CLA nonendemic area, presenting negative results in an enzyme-linked immunosorbent assay (ELISA); 2) the infected-subclinical CLA group: seropositive animals, without clinical symptoms of the disease; and 3) the infected-clinical CLA group: seropositive animals presenting clinical symptoms of CLA and from which C. pseudotuberculosis was isolated from the lesions.
To perform a validation study, 57 blood and serum samples (5 ml each) from seropositive goats with caseous lesions and with bacterial infection confirmed by bacteriologic assays (described later) and multiplex polymerase chain reaction (PCR) 15 (named truly positive) were screened. Also, 30 samples from seronegative animals from a nonendemic area (named truly negative) were used. By using the same criteria, 42 truly negative and 42 truly positive samples from sheep were also obtained.
Bacterial identification
Tissue samples from enlarged peripheral lymph nodes (1 from each animal) with lesions characteristic of caseous lymphadenitis were aseptically collected from clinically ill animals and plated onto brain heart infusion (BHI) agar a supplemented with 5% defibrinated sheep blood. The plates were incubated aerobically for approximately 48 hr at 37°C. Colonies that morphologically resembled C. pseudotuberculosis were Gram stained. Gram-positive colonies were further tested for urease activity, synergistic hemolytic activity with CAMP (Christie, Atkins, Munch-Peterson) factor from Rhodococcus equi and carbohydrate fermentation (glucose, lactose, sucrose). 5 Strains that were positive for urease and glucose fermentation, negative for lactose and sucrose fermentation, and that enhanced the hemolysis of R. equi were identified as C. pseudotuberculosis. 6
To characterize the tissue samples retrieved, a multiplex PCR assay was performed as previously described. 15 The assay was performed in a final reaction volume of 10 μl that contained 1.5 UI AccuPrime Taq DNA polymerase, b PCR Buffer II b (200 mM Tris-HCl, 500 mM KCl, 15 mM MgCl 2 , 2 mM each deoxyribonucleotide triphosphate, AccuPrime protein, b 10% glycerol) and 2 mM of each of the primers 16S-F/16S-R, C2700F/C3130R, and PLD-F/PLDR2. The template concentration was approximately 30 ng of DNA extracted from the samples. Reactions were carried out in a thermal cycler c under the following conditions: initial denaturation at 95°C for 3 min; 40 cycles of 95°C for 1 min, 58°C for 40 sec, and 68°C for 1 min and 30 sec; final extension at 68°C for 7 min. The amplified products were resolved by electrophoresis on 1.0% agarose gels and visualized by ethidium bromide staining. This procedure had the objective to provide a more sensitive determination of the presence of C. pseudotuberculosis in infected animals.
Corynebacterium pseudotuberculosis secreted antigen production for ELISA
The preparation of the C. pseudotuberculosis secreted antigen was carried out as previously described. 14 Briefly, the C. pseudotuberculosis T1 strain was cultivated in BHI broth a at 37°C for 72 hr. The culture was centrifuged for 30 min at 10,000 X g. The supernatant was filtered through a 0.22-μm membrane d and then kept at —20°C until use. The quantification of protein was determined by the Lowry modified method 19 by using a commercial detergent compatible protein assay kit. e
Corynebacterium pseudotuberculosis secreted antigen concentration
The C. pseudotuberculosis antigen preparation was concentrated by using the three-phase partitioning (TPP) technique as previously descibed. 17 Briefly, 15% ammonium sulfate was added to the culture supernatants at room temperature, and the mixture was stirred gently. The pH was adjusted by the addition of HCl, and the same volume of n-butanol was added. The mixture was vortexed for 1 min and allowed to rest for 1 hr. The mixture then was centrifuged at 1,350 X g for 10 min, and the interface precipitate was collected and dissolved in Tris (20 mM, pH 7.4) buffer (500 μl buffer per 5 ml culture supernatant). Extracts were dialyzed by ultrafiltration with a 10-kDa membrane d cutoff against Tris 20 mM, pH 7.4. The protein content of the extracts was determined by the Lowry modified method 19 by using a commercial detergent compatible protein assay kit. e
ELISA for the detection of specific immunoglobulins against C. pseudotuberculosis
The ELISA was carried out as previously described, 4,22 with the following modifications: ELISA microplates f were coated with BHI-culture antigen, diluted 1:100 in carbonate–bicarbonate buffer (pH 9.6), and incubated at 4°C for 12 hr. The plates then were washed twice with phosphate buffered saline solution (PBS) and 0.05% Tween 20 (PBST), blocked with 5% skimmed milk in PBST, and incubated for 2 hr at 37°C. After 2 additional washes with PBST, 100 μl of sheep or goat serum samples, diluted 1:100 or 1:200, respectively, in PBST with 1% skimmed milk were added, and the mixture was incubated for 1 hr at 37°C. Each serum sample was tested in duplicate. The plates were washed 4 times with PBST. Rabbit immunoglobulin (Ig)G anti-sheep IgG g or donkey anti-goat IgG b , both conjugated with horseradish peroxidase, were diluted at 1:20,000 and 1:10,000 dilutions, respectively, in PBST, and were added to the plates and incubated for 45 min at 37°C. After incubation, the plates were washed, and the reaction was developed with tetramethylbenzidine i chromogen and hydrogen peroxide substrate (100 μl/well) for 15 min, and the reaction was stopped with 4 N sulfuric acid. Results were read in an ELISA plate reader e at 450 nm.
The plates were adjusted by defining the assay with the higher optical density (OD) value for the positive control (pool of 10 sera samples from animals presenting CLA confirmed by bacterial isolation), the lowest OD for the negative control (pool of 10 sera samples from animals from a CLA nonendemic area), and the lowest OD result for the blank (with no sera addition) as the standard plate. A correction factor was developed by dividing the positive control OD value of the standard plate by the positive control OD value found in the assayed plate, and all serum samples OD values of this same plate were corrected by this factor. The same negative and positive controls were used in all plates. The cutoff values for the ELISA were applied as described before. 4,22
Gamma interferon quantification
Quantification of IFN-γ in blood cultures was performed in samples collected from the experimental animal groups, as previously described, 12,16 with some modifications. Briefly, 2 hr after collection, 1 ml of heparinized blood was incubated at 37°C for 48 hr in a humidified 5% carbon dioxide atmosphere in a 24-well culture tray f with 20 μg of TPP antigen, 17 5 μl of pokeweed mitogen (1 mg/ml) as a positive control, or PBS as a negative control. After this period, the blood was centrifuged and IFN-γ was quantified in the supernatant by using a commercial ovine IFN-γ quantification kit. j
Statistical analysis
The comparisons between the IFN-γ quantification results from noninfected and seropositive animals, and results from noninfected, CLA subclinical and clinically infected animals, were carried out by using the Mann—Whitney nonparametric test with the SPSS v13.0 software, k and a P value < 0.05 was considered significant. The cutoff point for IFN-γ quantification was calculated as previously described, 7 with a confidence interval of 99.8%, which corresponded to the mean of the negative sera ODs, plus 3 standard deviations. Fifty-seven blood samples from seropositive goats with caseous lesions and confirmed bacterial infection (truly positive group) and 30 samples from seronegative goats from a nonendemic area (truly negative group) were evaluated. By using the same criteria, 42 truly negative and 42 truly positive sheep samples were evaluated.
The sensitivity (S) and specificity (E) of the IFN-γ assay were calculated as follows:
where TP = the number of truly positive samples, TN = the number of truly negative samples, FP = the number of false-positive samples, and FN = the number of false-negative samples. 4
Results
Pokeweed mitogen (used in the experiment as a stimulation positive control) induced IFN-γ production by peripheral blood cells in both seronegative and seropositive goats and sheep, without significant statistical difference among all groups. There were significant differences in the IFN-γ levels between seronegative and seropositive goats (Mann–Whitney test; Fig. 1a) as well as for sheep that were negative or positive for the presence of C. pseudotuberculosis—specific antibodies (Fig. 1b), which suggests that IFN-γ quantification was able to differentiate between seronegative and seropositive groups.
When the IFN-γ quantification in samples from goats with different clinical status of CLA was examined, there was a significant difference in induced IFN-γ levels between uninfected animals and CLA subclinically and clinically infected animals (Fig. 2a); however, the levels in goats with a subclinical infection were not significantly different to animals with C. pseudotuberculosis clinical infection. In contrast, sheep with CLA clinical signs produced significantly more IFN-γ than subclinically infected animals and noninfected animals. However, there was no statistically significant difference in IFN-γ levels between the subclinical group and the noninfected group (Fig. 2b).
The IFN-γ production in sheep and goats was compared to determine if the cellular response to C. pseudotuberculosis antigens differed between these 2 species (Fig. 3). However, no significant differences in IFN-γ levels were observed between these species, regardless of CLA clinical status.
The ELISA data did not have a significant correlation with the amount of IFN-γ produced by peripheral blood cells after stimulation with the C. pseudotuberculosis antigens in the seropositive and clinically infected goats and sheep (r = 0.027 and 0.0204; Fig. 4a, Fig. 4b, respectively). The distributions of the individual results obtained for IFN-γ levels in goats and sheep are shown in Figure 5a and 5b, respectively. There was no positive result for seronegative goats above the cutoff (204 pg/ml). However, the cellular response of seropositive animals was more variable, with a broad range between the maximum and the minimum results. A similar response pattern was observed in seropositive sheep. Furthermore, some sheep exhibited significant IFN-γ production even though they were seronegative.
A specificity of 100% and a sensitivity of 55.8% were observed for goats when using a cutoff of 204 pg/ml. Out of a total of 87 samples, no false positives were found, but 45 false negatives were identified (Table 1). The same procedure was used to evaluate sheep (Table 1). Of 84 samples, 3 presented false-positive results, and 33 were false negatives. The sensitivity and specificity of the assay were 56% and 93%, respectively, and the cutoff point was calculated to be 508 pg/ml.
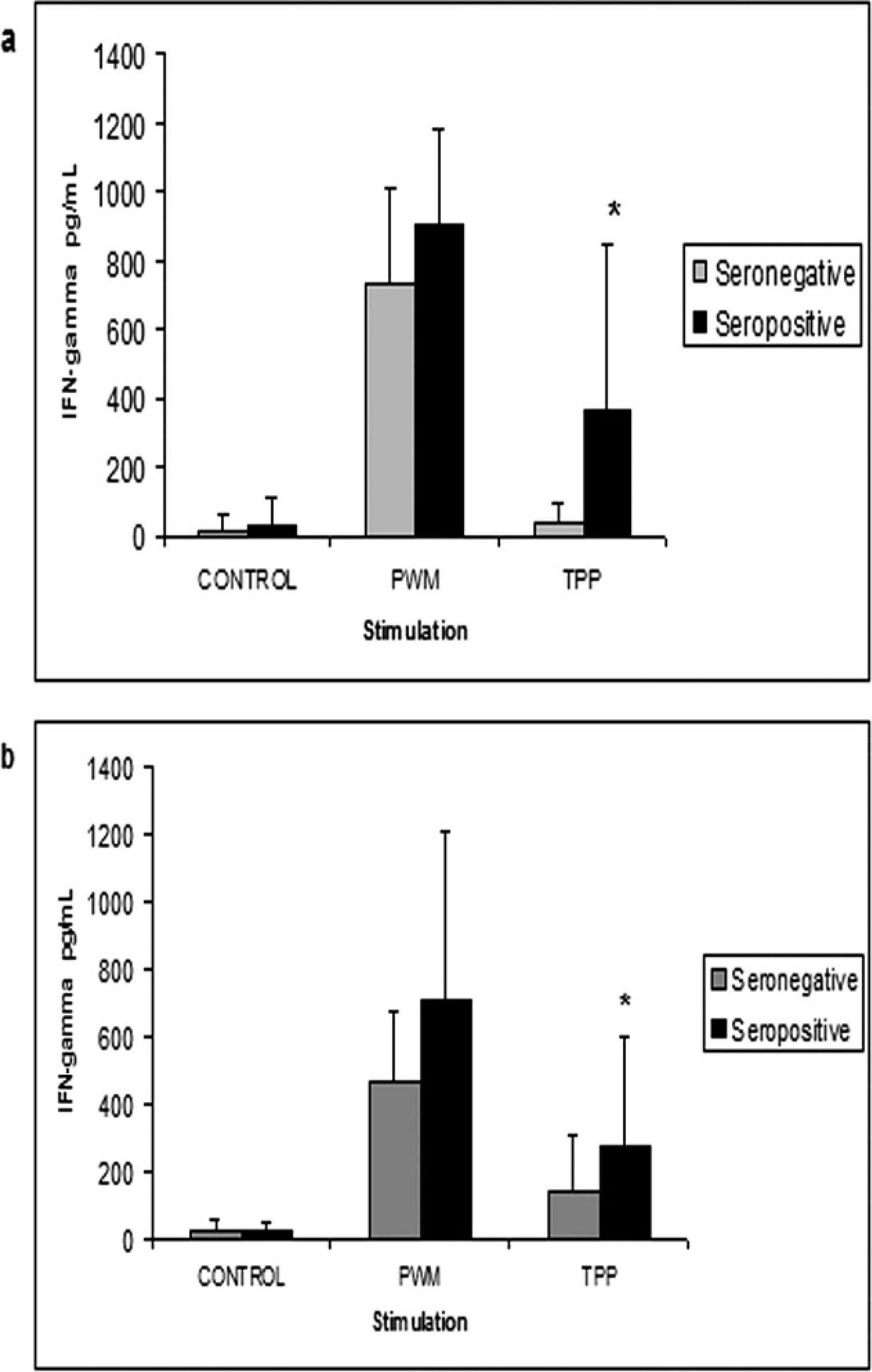
Peripheral blood leukocyte production of gamma interferon in goats (
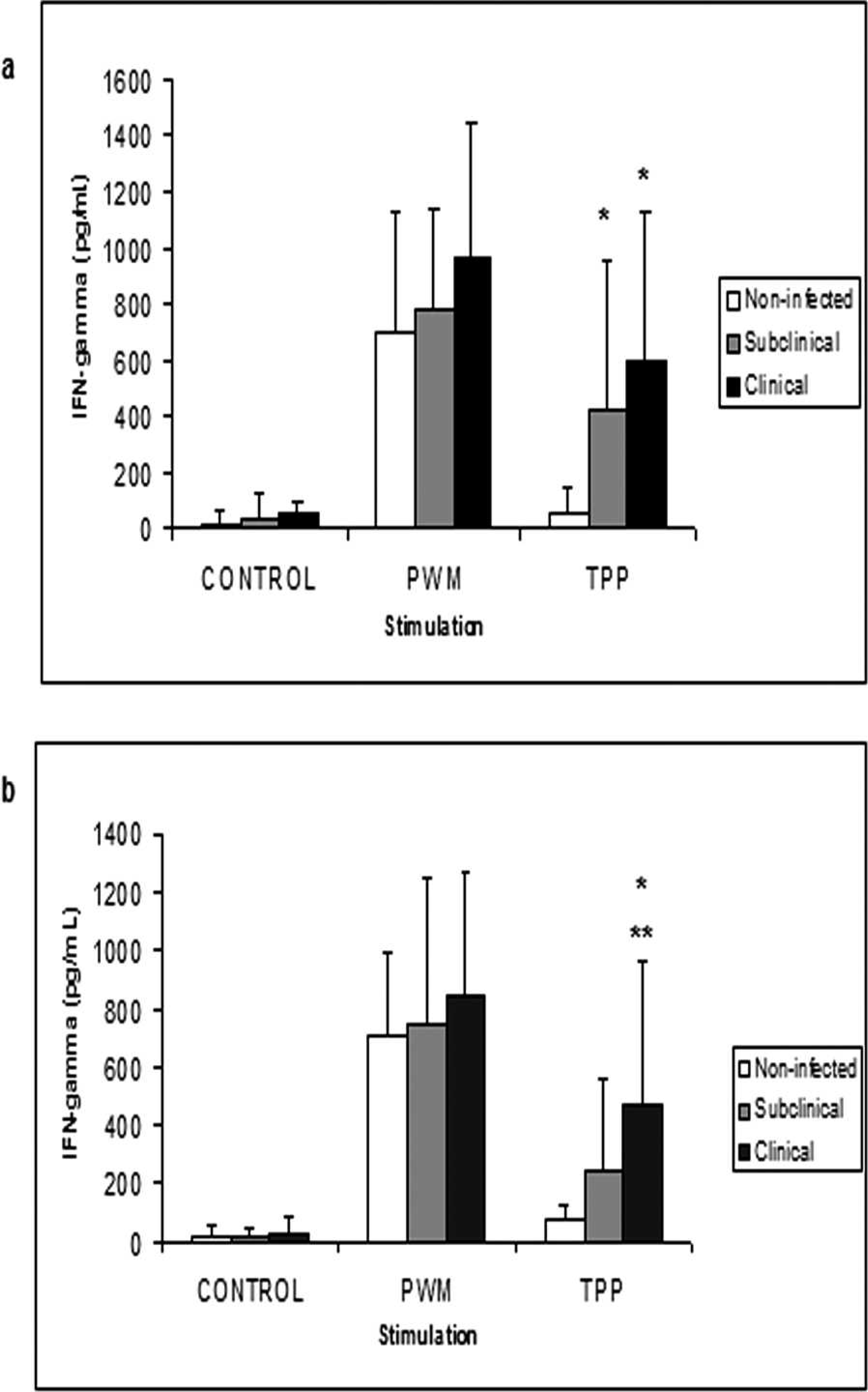
Quantification of gamma interferon (IFN-γ) produced by peripheral leukocytes stimulated with pokeweed mitogen (PWM), three-phase partitioning (TPP) concentrated secreted and/or excreted antigens, and in the absence of stimulation (control). Results for blood samples retrieved from goats (
Discussion
Several aspects of C. pseudotuberculosis infection in sheep and goats have been widely studied because of the importance of CLA in commercial herds. However, although some information concerning cellular and humoral responses to this bacterium is now available, few studies have focused on the development of reliable diagnostic tools and immunoprophylaxis. 2,4,11,21 The applicability of IFN-γ quantification after stimulus of peripheral blood leukocytes with TPP-concentrated secreted C. pseudotuberculosis antigens was examined, and the efficacy of this method was evaluated for the determination of infection and clinical CLA status in sheep and goats.
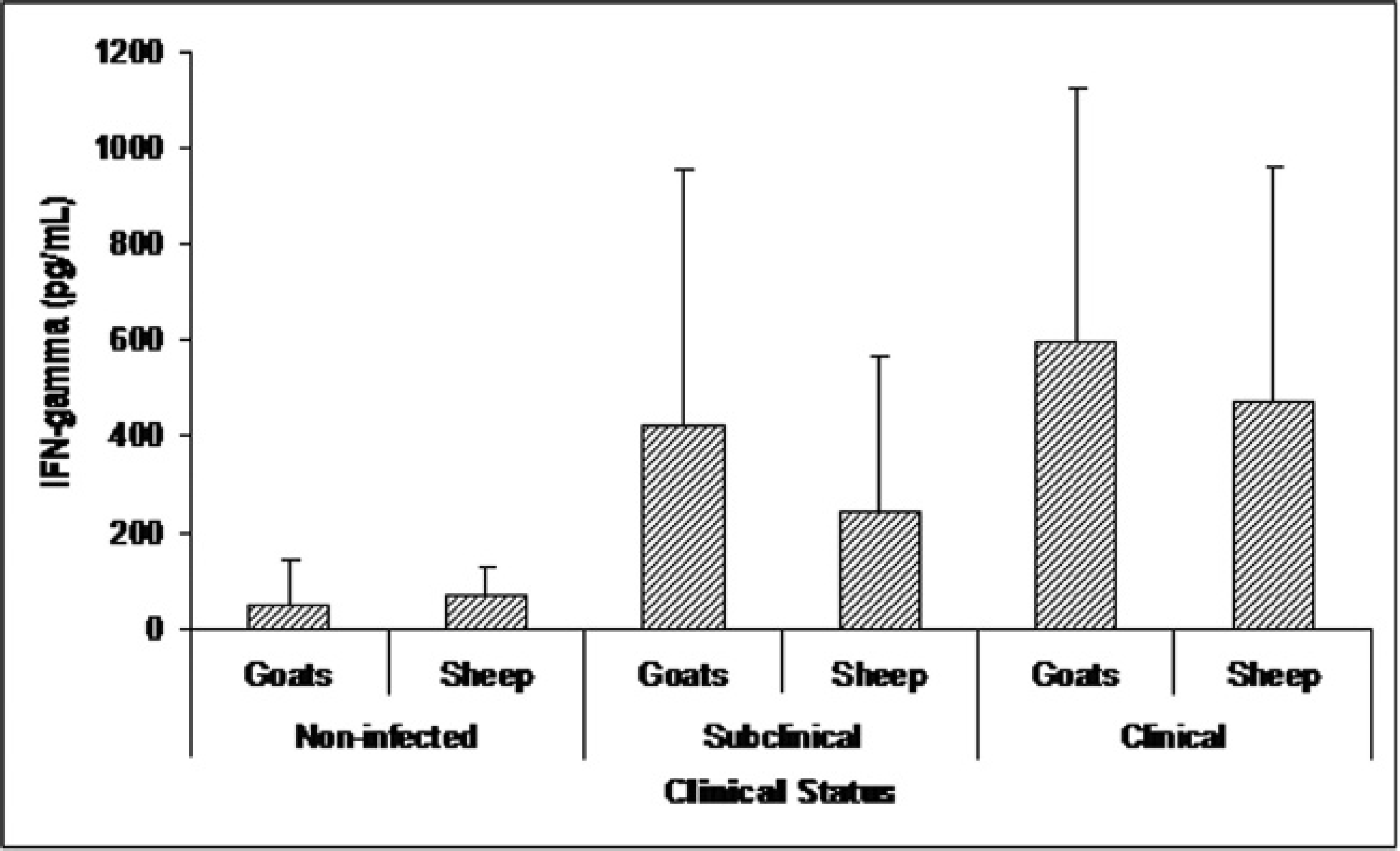
Comparison of the gamma interferon production between noninfected (from a nonendemic area, seronegative in enzyme-linked immunosorbent assay (ELISA) and without caseous lymphadenitis clinical lesions), subclinically (seropositive in ELISA and without clinical signs of caseous lymphadenitis) and caseous lymphadenitis clinically infected (seropositive in ELISA and presenting characteristically caseous lymphadenitis lesions, with Corynebacterium pseudotuberculosis presence confirmed by bacteriologic assays) sheep and goats, after stimulation with secreted and/or excreted C. pseudotuberculosis antigens. Thirty goats and sheep were tested in each group. The results represent the means of each group, and the bars indicate the standard deviations. No statistical difference was observed between the goats and sheep in any of the groups studied.
The TPP-concentrated secreted antigen from the T1 strain of C. pseudotuberculosis has been reported to cause significant stimulation of leukocytes from peripheral blood collected from small ruminants. 17 Use of this assay in goats after stimulation with secreted and somatic antigens from C. pseudotuberculosis has also been described. 12 Secreted antigens in a chemically defined medium 15 presented an increased stimulation, with higher production of IFN-γ, when compared with the stimulation induced by a somatic antigen. However, the weak induction of IFN-γ by a somatic antigen has not been observed by other researchers 11,18,20 ; this discrepancy can be explained by the utilization of different pathogenic, attenuated, or genetically modified strains, 17 or because of different antigen purification methods. Some researchers 24 were not able to obtain significant results when a somatic antigenic lysate was used to stimulate leukocytes collected from re-infected sheep, whereas better results were observed when peripheral blood cells were stimulated with formalin-inactivated bacteria.
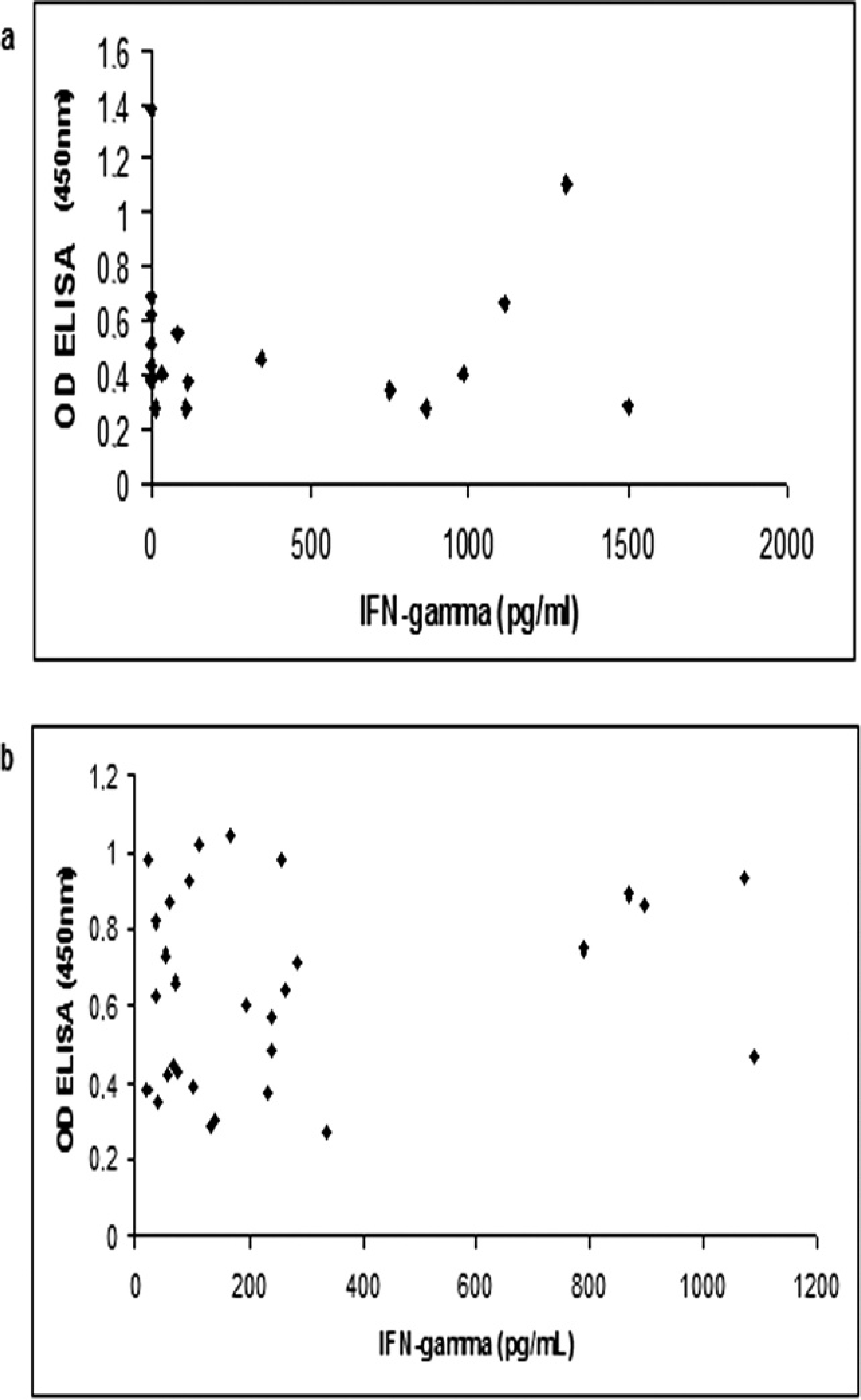
The correlation between the enzyme-linked immunosorbent assay (ELISA) optical density (OD) results for the detection of specific antibodies against Corynebacterium pseudotuberculosis in ELISA and gamma interferon levels after stimulation with C. pseudotuberculosis three-phase partitioning concentrated antigen in goats (
In the present study, when seronegative and seropositive animals were tested to look for differences in IFN-γ levels, significant differences were found in both sheep and goats (Fig. 1a, Fig. 1b). However, this response was extremely variable, especially when individual results were analyzed (Fig. 5a, Fig. 5b). Similar diverse cellular responses to secreted antigens from C. pseudotuberculosis, in both naturally and experimentally infected animals, have been reported previously. 14
The IFN-γ levels were also investigated in animals presenting different CLA clinical status. Goats with subclinical or clinical CLA could be distinguished from negative cases in the current study, as could sheep with clinical signs from healthy animals. Subclinically infected sheep presented a significantly lower IFN-γ production compared with animals with clinical disease (Fig. 2a, Fig. 2b). The importance of IFN-γ for protection against progression of this disease and as a major intermediate cytokine in the cellular response against this microorganism has been previously reported. 9,18 However, results of the current study contradict the hypothesis that some animals with clinical lesions can have decreased IFN-γ levels in response to bacterial antigens and that seropositive animals with no apparent symptoms of disease can produce high levels of IFN-γ. 12 This situation may be caused by a strong stimulation of the immunologic system by C. pseudotuberculosis presence, or by secreted molecules produced by this bacteria.
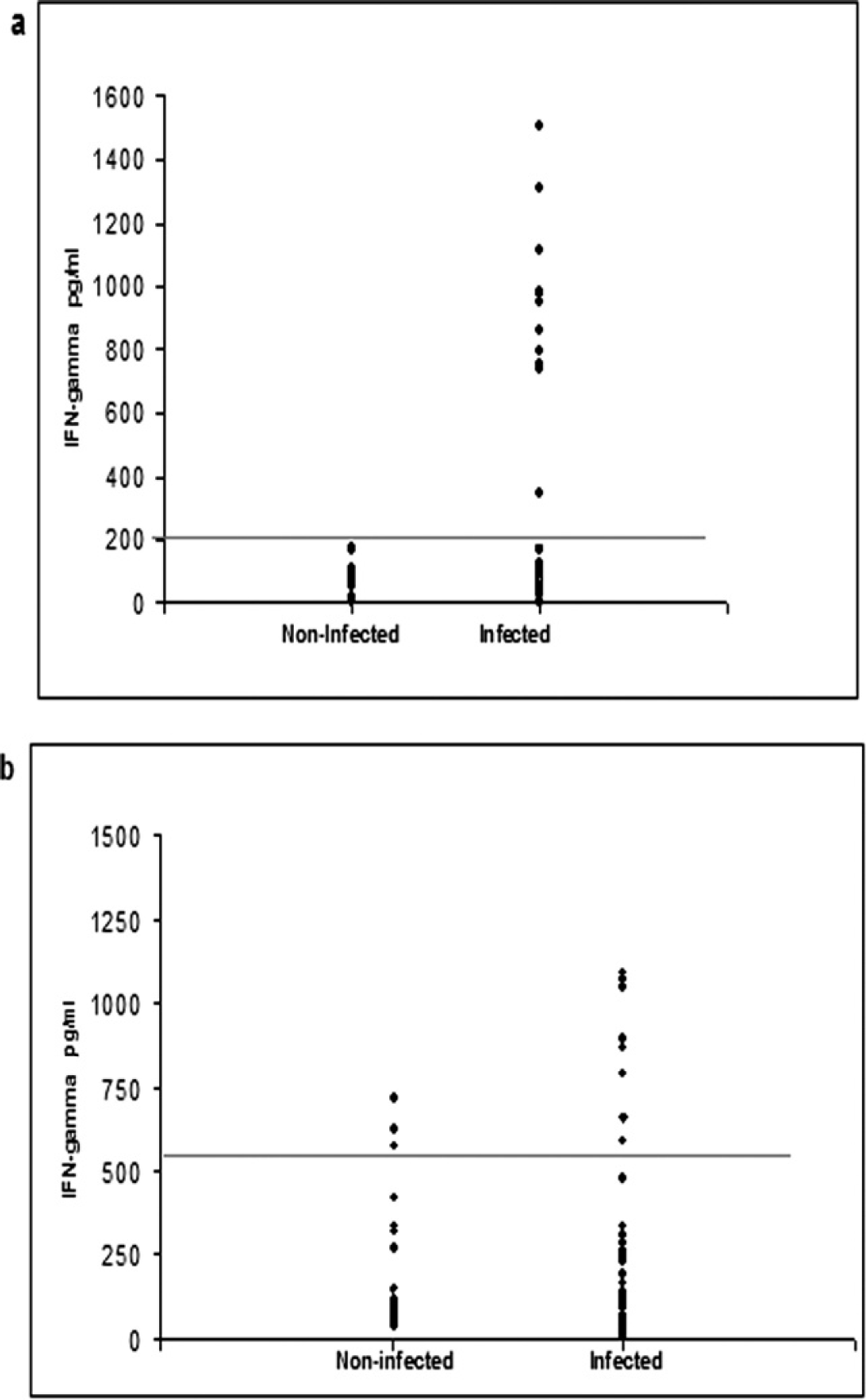
Distribution of the individual results obtained for gamma interferon levels in the supernatant of peripheral blood culture after stimulus with three-phase partitioning (TPP) concentrated antigen in goats (
Validation parameters for the gamma interferon quantification assay, after stimulation of peripheral blood leukocytes with three-phase partitioning concentrated secreted and/or excreted Corynebacterium pseudotuberculosis antigens, in goats and sheep. Eighty-seven and 84 samples obtained from goats and sheep, respectively, were analyzed. The criterion standard used was the C. pseudotuberculosis isolation and identification. The cutoff corresponds to the mean of the negative samples results plus 3 standard deviations.
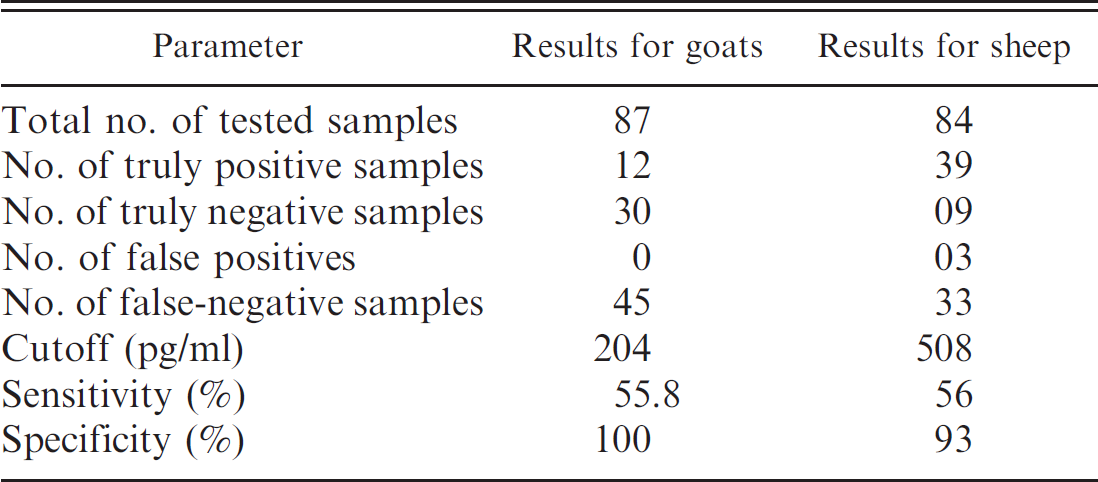
The sensitivity and specificity values obtained in the current study were 55.8% and 100% for goats, respectively, and were 56% and 93% for sheep, respectively (Table 1). The IFN-γ quantification assay used in the current study has characteristics similar to several serologic assays that detect specific C. pseudotuberculosis antibodies. 11,23,26 There were large differences in the intensity of the cellular response elicited by the bacteria, and it is already described that, in the case of Mycobacterium tuberculosis, a microorganism that is phylogenetically related to C. pseudotuberculosis, experimentally infected bovines were reported to develop a highly variable cellular immune response against culture filtrate antigens derived from the bacteria, and the response was reported to vary, depending on the stage of infection. 21
Sensitivity is a major validation parameter in a diagnostic assay that is being considered for use in eradication programs, principally to ensure that no infected animals are misdiagnosed, because such animals can be important sources of infection. 8 High values of sensitivity (91%) for IFN-γ quantification were reported in an assay when using formalin-inactivated somatic antigen stimulated sheep-derived cells 24 ; however, only 3 infected animals were evaluated, and the calculation of sensitivity and specificity was based on 33 observations on these animals. The study also reported that the IFN-γ assay failed to give consistent results over time.
The standardization of an IFN-γ assay, when using secreted C. pseudotuberculosis antigens, has been described previously. 12 A somatic antigenic lysate and secreted antigens were used, and it was concluded that secreted antigens were better for stimulating goat blood cells. In the current study, a larger number of animals were used, with the objective to test this assay as a diagnostic tool. The main pitfall in the present study was the poor sensitivity of the assay; but, the specificity obtained for sheep and goats was high (Table 1).
In mice and humans, IFN-γ is part of the cellular immune response, directing the response pattern toward a Th1 profile. 13,27 Interferon-γ also plays an important role in the humoral response, particularly in isotype switching, inhibiting changes that are IL-4 dependent, and inducing production of certain IgG subclasses. 1 However, in sheep and goats, the conditions that induce a Th1 or Th2 profile are not well understood, and little is known about isotype switching in B lymphocytes. When taking this into consideration, the correlation between specific IFN-γ levels and the presence of serum antibodies specific for the antigen used for blood-cell stimulation were examined in the present study. A significant correlation between IFN-γ levels and antibody production in animals presenting clinical symptoms of the disease was not found (Fig. 4a, Fig. 4b), but this conclusion applies only to this disease, because it cannot be extended to the small ruminant immune response in general.
In the current study, a TPP-concentrated secreted antigen was used. The preparation of this antigen is very simple, relatively fast, and has low production costs. This antigenic formulation represents an inexpensive and accessible method for use in the IFN-γ quantification assay. The sensitivity of this method requires further development, including improvements in blood-cell culture conditions, antigen extraction, and stimulation protocols, as well as the sandwich ELISA for IFN-γ quantification to enhance its sensitivity.
Acknowledgements
The authors are grateful to Bahia State Agency for Animal Defense for helping to obtain ovine samples, and Francisca Soares for technical support. The current work was supported by FAPEX, FUNDECE, and RENORBIO. R. W. Portela and R. Meyer are research fellows of FAPEX, and V. Azevedo of CNPQ. M. F. Rebouças and D. D. Lima are postgraduate students with grants from FAPEX.
Footnotes
a.
Himedia Laboratories, Mumbai, India.
b.
Invitrogen Corp., Carlsbad, CA.
c.
MJ Research Inc., Waltham, MA.
d.
Millipore Corp., Billerica, MA.
e.
Bio-Rad Laboratories, Hercules, CA.
f.
Nunc™, Thermo Fisher Scientific, Rochester, NY.
g.
Bethyl Laboratories Inc., Montgomery, TX.
h.
Dako North America Inc., Carpinteria, CA.
i.
Moss Inc., Pasadena, MD.
j.
MabTech AB, Nacka Strand, Sweden.
k.
IBM SPSS Statistics, Chicago, IL.