Abstract
The detection of myocardial injury in cattle caused by the ingestion of cardiotoxic compounds or cardiac diseases would be facilitated by the availability of a rapid and specific quantitative serum assay for cardiac troponins. Therefore, the accuracy of the only cardiac troponin T (cTnT) immunoassay to receive approval by the US Food and Drug Administration for the measurement of cTnT in human serum was evaluated to quantify the protein in bovine serum. Recovery experiments were performed by the addition of purified bovine cTnT to normal bovine serum. Cardiac troponin T was quantified using an immunoassay commonly used for the measurement of cTnT in human serum. The immunoassay demonstrated a well correlated (
Cardiac troponin T (cTnT) is a cardiac specific marker used in the diagnosis of myocardial cell injury in humans and is elevated in myocardial infarction,
7
congestive heart failure,
20
cardiomyopathy,
19
sepsis,
21
and myocarditis.
9
Moderate increases in cTnT may be observed in patients with clinically stable renal disease but without acute coronary syndromes compromising the specificity of the biomarker.
11
There is a need to accurately and quickly determine myocardial injury in nonhuman animal models of cardiac injury and in routine veterinary medicine. Several cardiotoxic compounds cause morbidity and mortality in cattle, including ionophore feed additives, gossypol, and cardiac glycosides found in plants such as
The immunoassay used for this report is a third-generation electrochemiluminescent immunometric assay designed for the in vitro quantification of human cTnT in serum. This assay uses 2 monoclonal antibodies, M7 and M11.7, recognizing independent adjacent epitopes composed of amino acid positions 125–131 and 136–147, respectively, located in the central part of the 288 amino acid protein. 3,12,18 The assay coefficient of variation (CV) is 10% at a concentration of 0.04 ng/ml (human cTnT). 16
The accuracy of the immunoassay to detect bovine cTnT was determined using recovery experiments. Two different lots of a commercial preparation of bovine cTnT b that is >98% pure by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and designed for use as an immunoassay standard were used. Normal bovine serum was obtained from a healthy Holstein cow maintained by the University of California-Davis School of Veterinary Medicine. Bovine cTnT (100 μg) was reconstituted with either normal bovine serum or a Tris buffer recommended by the manufacturer of the protein (20 mM Tris, pH 7.5, 5 mM EDTA, 7 M urea, 15 mM mercaptoethanol) to a final concentration of 20 μg/ml and added to normal bovine serum to yield expected cTnT concentrations of 10, 20, 50, 100, and 1,000 ng/ml. Cardiac TnT concentrations were determined in quadruplicate in 2 separate recovery experiments.
The immunoassay demonstrated a well correlated (
One possible explanation for the low recovery was incomplete reconstitution of the commercial cTnT. The initial concentration of 20 μg/ml was not verifiable, as this material had been reconstituted in normal bovine serum. Therefore, the recovery experiments were repeated using a different lot of commercial bovine cTnT reconstituted in Tris buffer to achieve a concentration of 20 μg/ml. A Bradford protein assay
1
used in accordance to the manufacturer's instructions
c
confirmed the final protein concentration was 20.5 μg/ml. The concentration of buffer in the prepared specimens did not interfere with the analytical performance of the immunoassay (data not shown). Consistent with the initial experiments, the immunoassay demonstrated a well correlated (
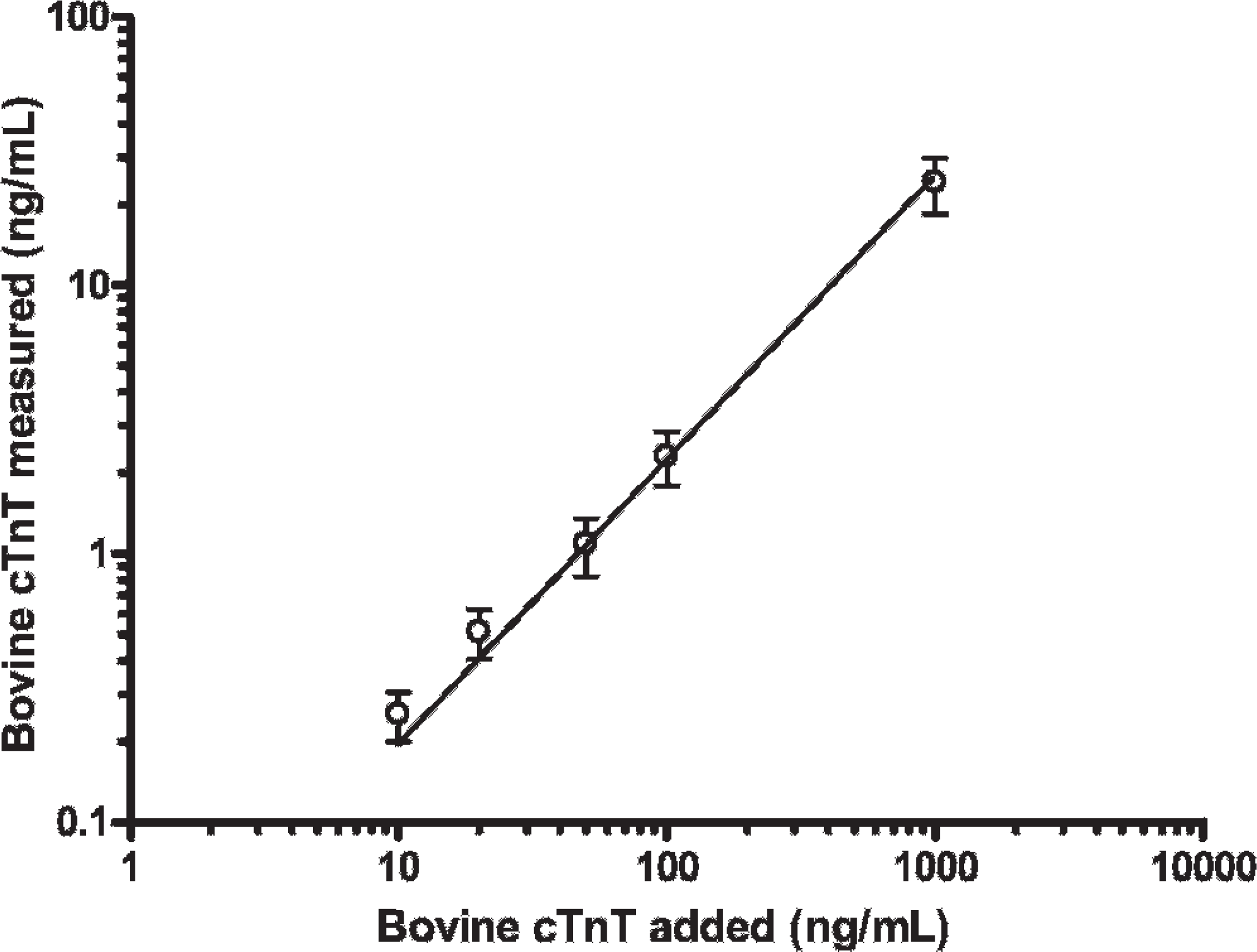
The immunoassay demonstrates a linear, dose-dependent response to bovine cTnT but recovers only 2.4% of added protein (Y = 0.024(X) −0.05). The points show the average of quadruplicate measurements from 2 separate experiments. The solid line is the best fit line, and the error bars represent the standard error of the mean.
To determine if the low recovery was because of interferences that might have been present in the normal bovine serum, immunoassay standards were added across a broad concentration range to the bovine serum, and cTnT was quantified in singlicate. Recovery over a cTnT range of 0.16-20.0 ng/ml averaged 131% indicating that there was nothing in the bovine serum that would account for the low recovery of bovine cTnT.
Bovine cTnT is homologous to the cTnT of other animals, such as chicken, rabbit, quail, and rat.
10
Bovine and human cTnT amino acid sequences were compared by querying the Entrez Protein Database (NCBI) and found human (NP_001001430, NP_777196, P13789) and bovine (Bos
These findings demonstrate that this immunoassay is not sufficiently analytically sensitive to accurately quantify bovine cTnT despite the similarity of amino acid sequences in the recognized epitopes between the human and bovine proteins. The 2 antibodies used in this third-generation immunoassay were previously used by the manufacturer in its second-generation assay. 12 Interestingly, the standard for the second-generation assay used a bovine cTnT standard, and an earlier report indicated that the M11.7 antibody had a low affinity for bovine cTnT, largely because it was produced using a human cTnT immunogen. 6 The third-generation assay uses recombinant human cTnT as standards.
The successful use by the manufacturer of bovine standards in the second-generation assay and the good linearity identified in this study from the third-generation assay both indicate that the third-generation assay may actually be able to accurately quantify bovine cTnT if appropriately calibrated with bovine standards. Alternatively, the assay might still be clinically useful if used to simply detect increased concentrations of cTnT in bovine serum. Based on the linear regression equation obtained from the combined recovery data and the 0.04 ng/ml functional sensitivity of the assay, a bovine cTnT concentration >3.75 ng/ml is quite likely to be clinically significant. Indeed, a rapid qualitative assay d with an analytical sensitivity of 0.08 ng/ml developed for the detection of cTnT in human serum produced a positive result with serum collected from a calf with histologically confirmed myocardial necrosis. 5 Regrettably, this case report provided no quantitative cTnT data, making it difficult to evaluate the analytical performance of the device. However, to be most clinically effective, any assay for cTnT (qualitative or quantitative) requires meaningful information regarding the upper reference limit of cTnT in cattle, and this remains unknown. Although other markers of myocardial injury were not investigated, such as cardiac troponin I (cTnl), a report by O'Brien et al. suggests that some cTnI immunoassays may produce accurate quantification of cTnI in bovine serum. 15
In summary, the only immunoassay to receive approval by the US Food and Drug Administration for the measurement of cTnT in human serum has poor accuracy for quantifying cTnT in bovine serum. This could limit its usefulness in detecting myocardial necrosis in cattle.
Footnotes
a.
Roche Elecsys Troponin T STAT immunoassay, Roche Diagnostics, Mannheim, Germany.
b.
Bovine cTnT, HyTest Ltd., Turku, Finland.
c.
Quick Start Bradford Protein Assay, BioRad Laboratories, Inc., Hercules, CA.
d.
TropT, Roche Diagnostics, Mannheim, Germany.
