Abstract
The capacity of a commercially available gamma interferon (IFNγ) assay to detect infected sheep early in the pathogenesis of Johne's disease enables the removal of such animals from the flock before bacterial shedding and pasture contamination. However, nonspecific IFNγ responses in the assay have meant that to achieve high-test specificity, there has been a reduction in sensitivity. Although the optimal conditions for the use of the assay in cattle have been well documented, there have been few studies optimizing the assay for use in sheep. The current study details the effect of anticoagulant, duration of incubation, cell concentration, blood storage temperature, time of stimulation of cells with antigen relative to time of sample collection, and temperatures during transit on IFNγ synthesis. Maximal IFNγ synthesis occurred with incubation periods of 48 hr in blood collected into heparinized tubes. Decreasing the leukocyte population by diluting the total peripheral blood leukocyte concentration was associated with a decreasing IFNγ response. Conversely, concentrating the peripheral blood concentration 2-fold resulted in an increase in the IFNγ production. In field studies, immediate incubation of blood samples with antigen at 37°C resulted in larger IFNγ responses; however, significantly lower IFNγ values were obtained if the samples were transported at ambient temperature. The results of this study indicate that optimization of the IFNγ assay may enable increased synthesis of IFNγ during the stimulation phase of the assay and that future work may determine whether this translates to increased sensitivity of the assay in detecting early infections in sheep.
Introduction
The Bovigam sandwich assay a is a commercially available, biphasic, whole blood assay using an enzyme-linked immunosorbent assay (ELISA) to measure gamma interferon (IFNγ) production. The assay was originally developed for use in cattle in the diagnosis of tuberculosis, 1 and, with a modification to the antigen used in the whole blood stimulation phase of the assay, has subsequently been used in the diagnosis of bovine Johne's disease. 2 The assay detects the cell-mediated immune responses that, in diseases such as Johne's disease, typically occur earlier than the humoral responses that are often associated with onset of clinical disease. The optimal conditions for use of the Bovigam sandwich assay in cattle have been well documented. 3 It is recommended that bovine blood samples be collected in blood tubes in which heparin salts are used as the anticoagulant and that 16 hr is the optimum incubation time for the whole blood stimulation phase of the assay. 3 It is also recommended that the blood is stimulated within 8 hr of blood collection to obtain maximum IFNγ synthesis. 3,4 This is a situation that may be difficult to achieve, particularly with samples collected from farm animals in remote field settings.
Ovine IFNγ is also detected by the Bovigam sandwich assay 1,5 and, in recent years, the assay has been evaluated as a diagnostic test for use in ovine Johne's disease (OJD) control programs. 6,7 The assay's capacity to detect infected animals early in the disease process enables identification of potentially infected sheep before any bacterial fecal shedding. Irrespective of whether these animals progress to the clinical stage, removal of such animals from the flock has the potential to decrease contamination of pasture and, subsequently, to reduce infection of other sheep. However, because of nonspecific IFNγ responses, raised cutoff points have been required to achieve high specificity (≥98%), 7 which has resulted in a reduction of sensitivity to less than 50%, limiting its application for early detection of infection or certification of disease freedom. Optimization of the IFNγ production in ovine blood samples may improve the diagnostic sensitivity of the assay. The current study, therefore, aimed to improve the utility of the Bovigam IFNγ assay a in sheep by investigating the effect of anticoagulant, duration of incubation, cell concentration, blood storage temperature, time of stimulation of cells with antigen relative to time of sample collection, and temperature during transit on IFNγ production in whole ovine blood.
Materials and methods
Sheep
All experiments using housed or field animals in the present study were carried out with the approval of the University of Sydney Animal Ethics Committee. For the preliminary investigations, 2 Merino sheep approximately 7 months old were used. Only 2 animals were used in the study to maximize the number of incubation condition variables studied. The sheep had been vaccinated with an adjuvanted, killed
MAP antigen and blood collection
Mechanically disrupted MAP antigen was derived from strain 316V. c Blood was collected from the jugular vein into commercially available 10-ml blood collection tubes, d containing 144 United States Pharmacopeia (USP) units of spray-dried lithium–heparin for all experiments, except for those in which the effect of other anticoagulants was analyzed. In these experiments, blood was collected into commercially available, 10-ml, blood collection tubes d containing either potassium oxalate/sodium fluoride (100/sp, 1,000/ca) or potassium ethylenediamine tetra-acetic acid (EDTA; 15% solution, 0.117 ml, 17.55 mg [100/sp, 1,000/ca]).
Whole blood culture
Previous studies in cattle described the nonspecific release of IFNγ in some samples when whole blood was diluted in tissue culture media.
3
This complication was not observed with ovine blood samples in the current study, so blood was diluted 1:2 in media to facilitate adequate mixing with antigen or mitogen. The culture medium used for all experiments consisted of Roswell Park Memorial Institute (RPMI)-1640 medium,
e
containing 5% fetal bovine serum (FBS),
f
Gamma interferon enzyme-linked immunosorbent assay
The whole blood culture method was followed by the Bovigam sandwich ELISA as previously described 1 and performed according to the manufacturer's instructions. All samples were assayed in duplicate or triplicate, and optical densities (ODs) were measured on an ELISA plate reader h at 450 nm. Samples were reassayed if duplicate or triplicate values differed from their means by more than 10%.
Effect of concentration of peripheral blood leukocytes and total and differential peripheral blood leukocyte counts on IFNγ production
Field trial investigations into the effect of varying transport temperature and time of adding antigen on IFNγ production
Group 1.—For each sheep, 3 polypropylene-capped tubes containing 1 ml of blood in 1 ml of media alone, 1 ml of media plus antigen, and 1 ml of media plus mitogen, respectively, were transported to the laboratory at ambient temperature, which was estimated to vary between 10°C and 20°C. Once at the laboratory, the blood was placed on a laboratory bench overnight at approximately 20°C, then incubated at 37°C for 48 hr before harvesting the supernatant.
Group 2.—For each sheep, 3 polypropylene-capped tubes containing 1 ml of blood in 1 ml of media alone, 1 ml of media plus antigen, and 1 ml of media plus mitogen were transported to the laboratory in a portable incubator j set at 37°C. Once at the laboratory, the blood was incubated at 37°C overnight and for a further 48 hr before harvesting the supernatant.
Group 3.—For each sheep, 3 polypropylene-capped tubes containing 1 ml of blood in 1 ml of media were transported to the laboratory at ambient temperature, which was estimated to vary between 10°C and 20°C. Once at the laboratory, the blood was placed on a laboratory bench overnight at approximately 20°C. The following morning, either MAP antigen or PWM was added to 2 of the 3 tubes, whereas the third tube remained as a control tube without additives. All tubes were then placed in the laboratory incubator at 37°C for 48 hr before harvesting the supernatant. Once harvested, all supernatants from all 3 groups were stored at −20°C before being assayed in the Bovigam sandwich ELISA according to manufacturer's instructions.
Statistical analyses
All statistical analyses were performed using GenStat version 10, k Minitab version 14, l or SAS release 9.1. m
In addition, logistic regression analyses were performed to evaluate the effect of blood cell counts or percentages on probability of classification of a sample as positive. Samples were designated as positive or negative using 2 stringency criteria. For the more stringent level, the sample was considered positive if the absorbance of the stimulated sample (either PWM or MAP antigen) was 0.100 absor-bance units greater than the absorbance achieved for the nonstimulated control well (media only) for that animal and if the absorbance of the stimulated sample was twice that of the unstimulated sample (i.e., OD MAP/OD media only ≥2 and OD MAP ≥ OD media only +0.1). This classification criterion was reported by researchers using the Bovigam sandwich assay for detection of IFNγ in cattle. 8 For the less-stringent criterion, the sample was considered positive if the absorbance of the stimulated sample (either PWM or MAP antigen) was at least 0.05 absorbance units greater than the absorbance achieved for the nonstimulated control well (media only) for that animal and if the absorbance of the stimulated sample was at least 1.3 times that of the unstimulated sample (i.e., OD MAP/OD media only ≥1.3 and OD MAP ≥ OD media only +0.05). This criterion has also been reported for the detection of IFNγ in sheep. 9,10
In initial linear and logistic regression analyses, all explanatory variables were tested individually to determine their unconditional association with the outcome. Subsequently, explanatory variables associated with the outcome at univariable
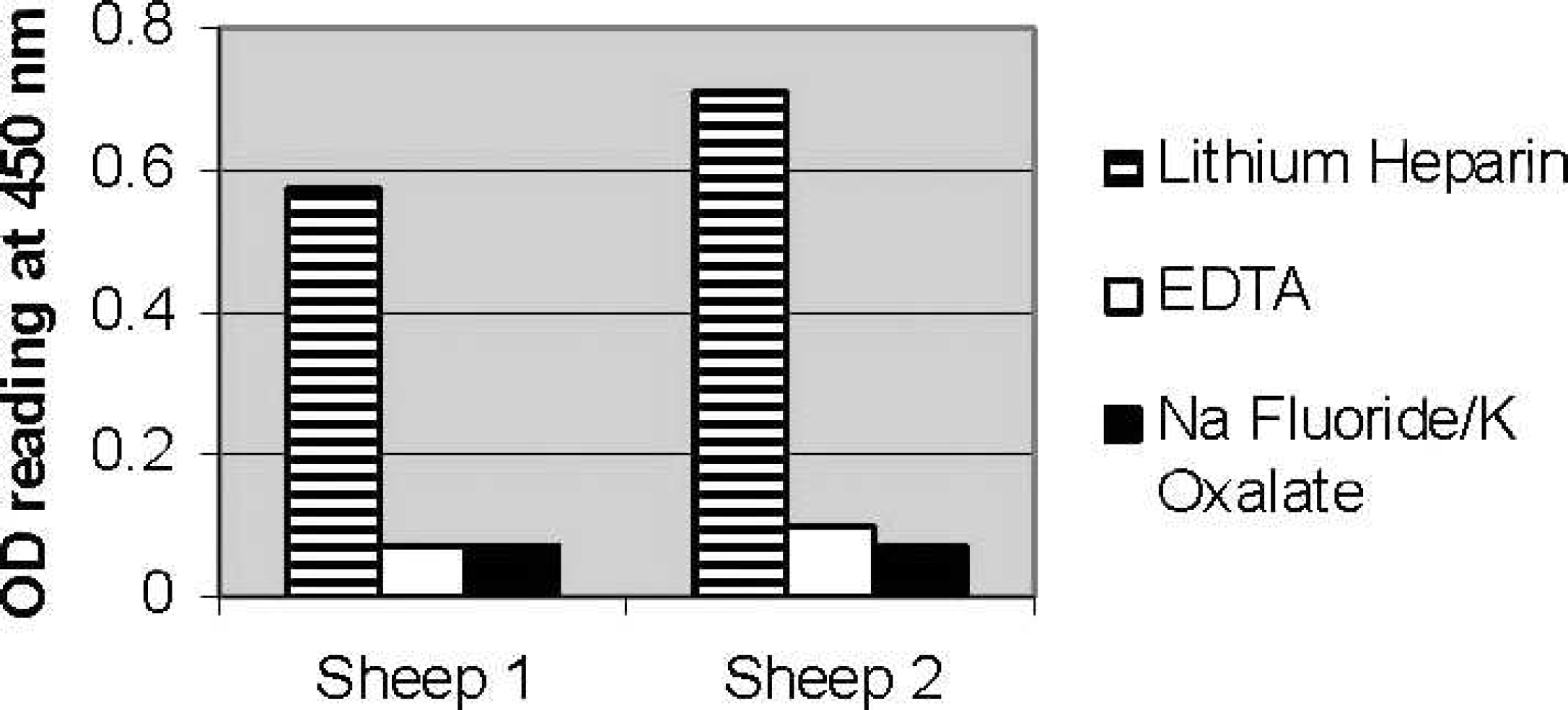
Effect of anticoagulant on gamma interferon production during incubation of whole blood with
Results
Effect of anticoagulant
The effect of anticoagulant on IFNγ production was evaluated in 2 vaccinated animals. Blood collected into tubes containing lithium heparin resulted in the optimal response with collection of blood into tubes containing potassium EDTA, and potassium oxalate/sodium fluoride, resulting in a complete abrogation of the IFNγ response (Fig. 1).
Effect of varying duration of incubation
The effect of varying duration of incubation on IFNγ production was evaluated in 2 vaccinated Merino sheep following stimulation in the whole blood assay with MAP antigen (final concentration of 5 μg/ml) and Con A mitogen (final concentration of 10 μg/ml). Blood was incubated for 24, 48, or 72 hr before harvesting the supernatant. In both sheep and with both MAP antigen and mitogen, optimum IFNγ production was obtained following 48 hr incubation (Fig. 2). The responses to MAP antigen were greater than those to ConA in both sheep.
Effect of blood storage temperature and duration
To account for field sampling and time to assay, the effect on IFNγ production of varying the temperature at which blood was stored for 4, 8, or 24 hr before incubation in the whole blood assay was evaluated in 2 vaccinated animals. Adding the MAP antigen at the time of blood collection consistently resulted in lower IFNγ production than the levels obtained when the antigen was added just before incubation irrespective of the time or temperature at which the blood was held before incubation (Table 1). For both sheep, regardless of when antigen was added, there were clear indications that the longer the delay to incubation, the less IFNγ was produced.
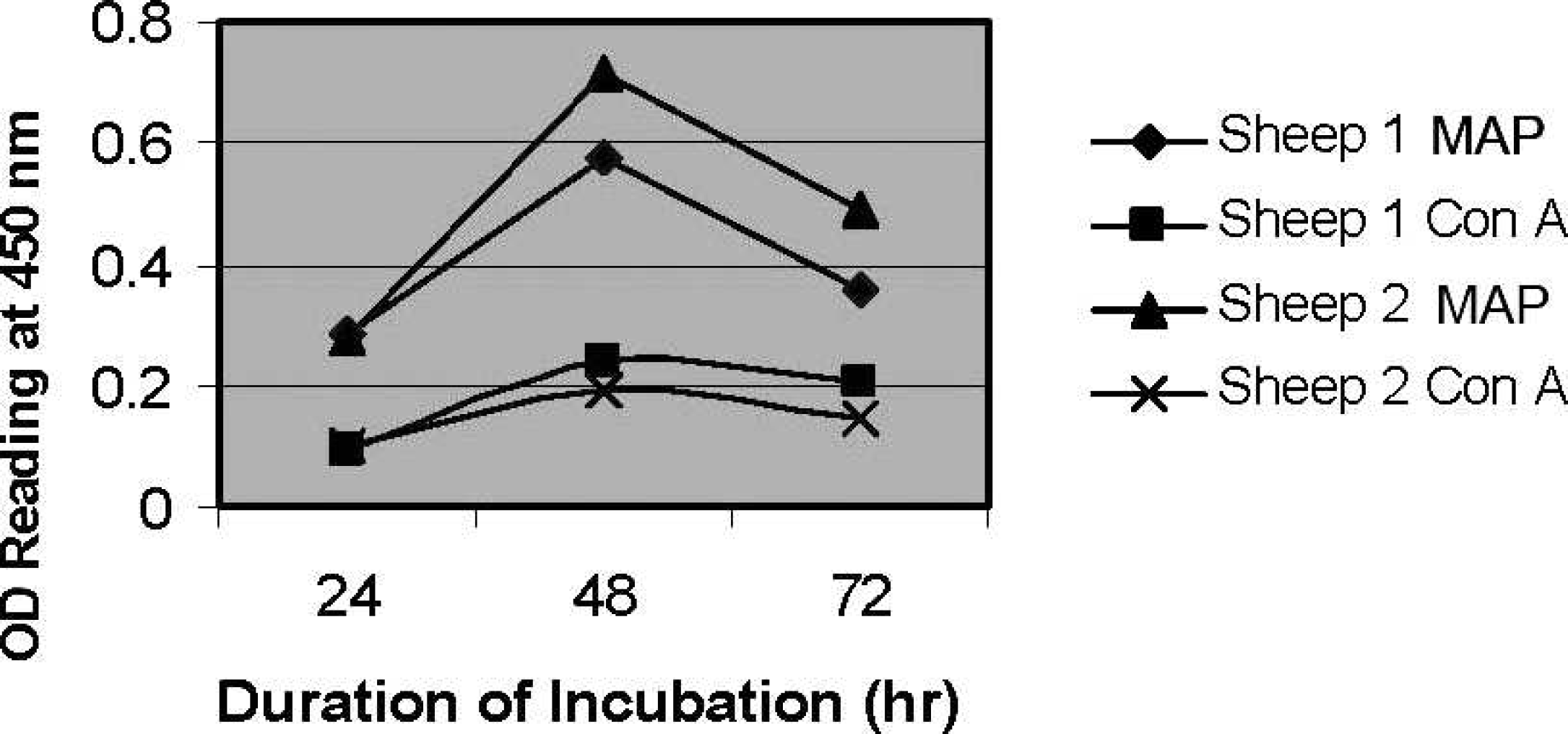
Effect of varying whole blood incubation time at 37°C on gamma interferon production in peripheral blood obtained from 2 vaccinated Merino sheep in response to stimulation with
Effect of total white blood cell concentration and differential cell count
Increasing and decreasing cell concentration in naturally infected sheep.—The effect of both concentrating and titration of the peripheral blood leukocyte concentration with media was examined in 4 naturally infected Merino sheep. In all 4 sheep, concentrating the peripheral blood concentration 2-fold resulted in a significant increase in IFNγ production (
Effect of delay in peripheral blood sample processing, and the temperature at which the sample was stored throughout that delay, on gamma interferon production in response to stimulation with
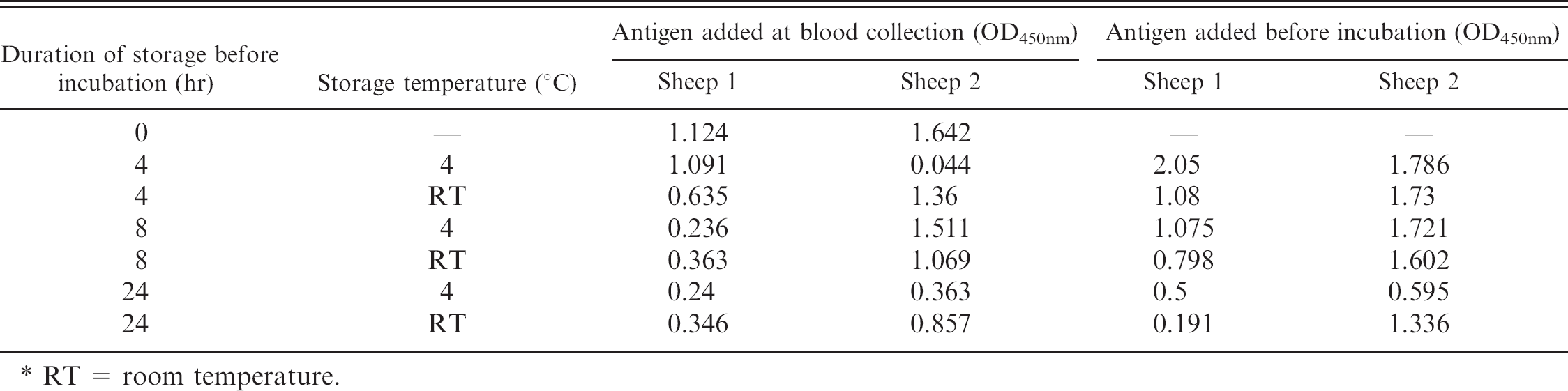
RT = room temperature.
Field trial investigations into the effect of varying transport temperature and time of adding antigen on IFNγ production
Effect of titration of white blood cell concentration on gamma interferon production in response to stimulation with
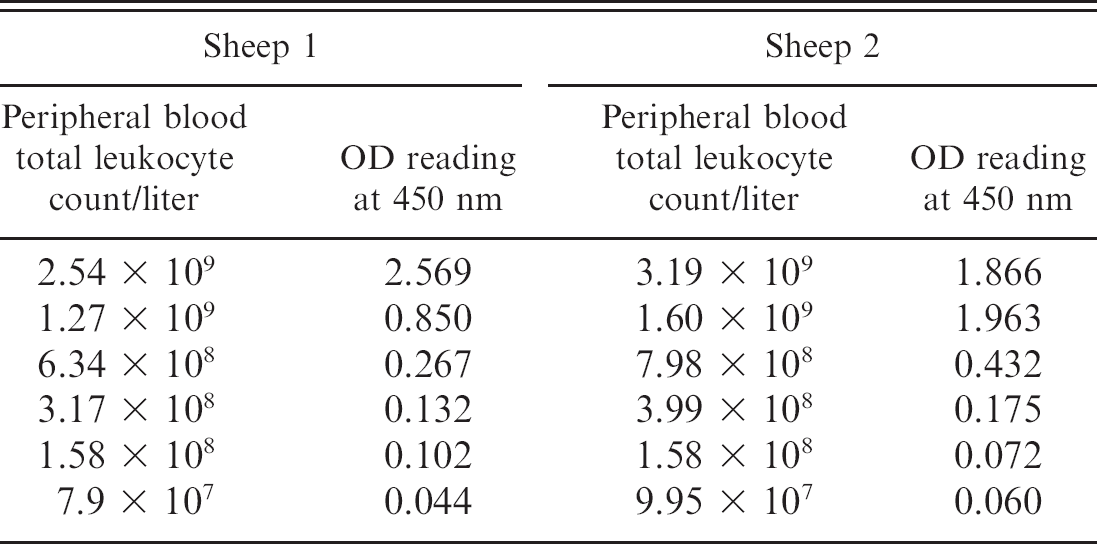
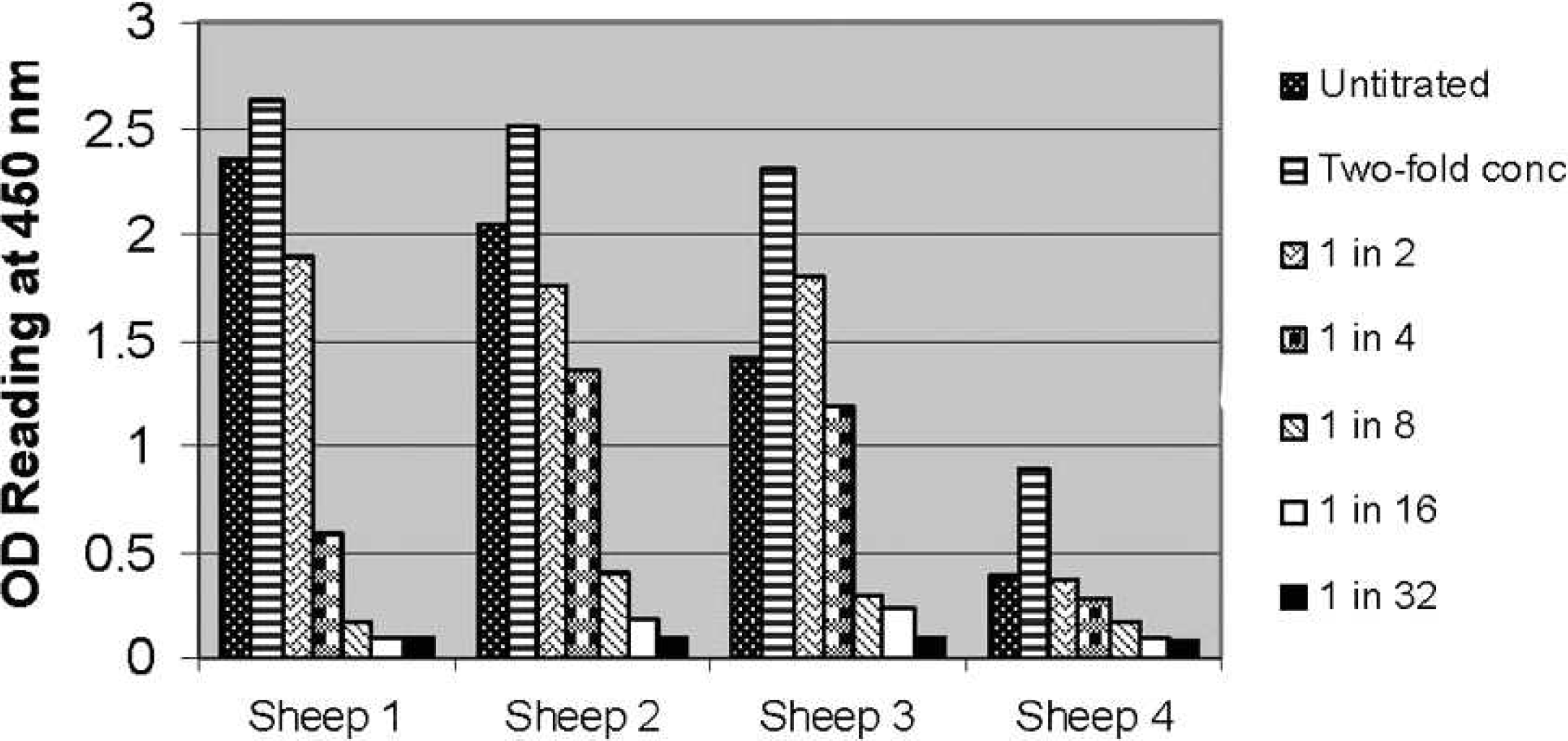
Effect of concentrating (2-fold) and diluting (down to 1 in 32) the leukocyte concentration on gamma interferon response measured as optical density (OD) at 450 nm in the Bovigam sandwich assay in peripheral blood samples obtained from 4 naturally infected Merino sheep.
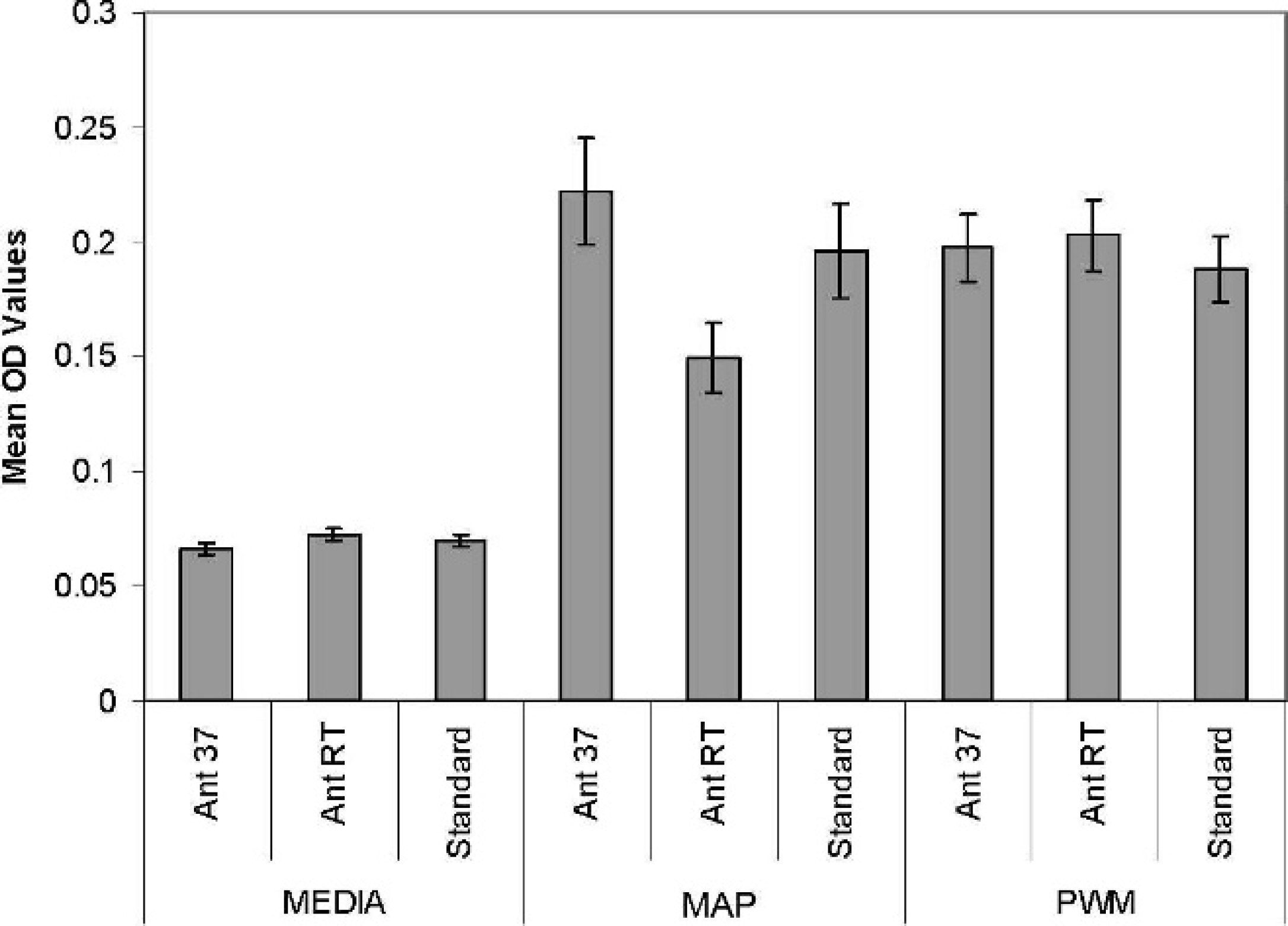
Effect of timing of addition of antigen and transport temperature on IFN-γ response measured as optical density (OD) at 450 nm in the Bovigam sandwich assay
a
in peripheral blood samples obtained from 50 vaccinated Merino sheep. Ant RT =
Discussion
The current study analyzes a suite of variables impinging on the reliable and reproducible assay of IFNγ from whole blood cultures in sheep. The finding that lithium heparin was the only anticoagulant enabling optimal IFNγ production with almost complete abrogation of the IFNγ response occurring with the use of sodium fluoride/potassium oxalate and potassium EDTA was consistent with results observed when the Bovigam sandwich assay was evaluated in cattle. 3,9 Similar results have been obtained in whole blood culture techniques for evaluating cytokine production 11 and lymphocyte reactivity 12 in humans.
In the present study, optimum IFNγ production was obtained after incubation of the ovine blood with the antigen or mitogen for at least 48 hr compared with overnight (16 hr) in cattle.
3
Interestingly, the finding that longer incubation times are required for optimum IFNγ production in sheep is consistent with that obtained in some studies with human blood.
11
In one study using a commercially available IFNγ assay
n
in humans, prolonged incubation for 6 days enhanced the sensitivity of the assay to detect latent
A dramatic decline in IFNγ production was observed associated with relatively minor reductions in the total white blood cell count. Cell density has been demonstrated to play a role in the reactivity of lymphocytes to stimulants in human studies 12 with marked decreases in lymphocyte responses demonstrated when cells are dispersed over larger surface areas, such as occurs if larger welled plates or flat bottom vessels are used for culture. This is presumably due to intercellular interactions that are hindered if the cells are not in close contact, which might occur with reductions in the total white cell count. Although the cell population predominantly responsible for production of IFNγ in sheep has not yet been clarified, the population in cattle has been identified as cluster of differentiation (CD)4 T lymphocytes. 14 In sheep, preliminary studies at the authors' laboratory suggest that this population comprises approximately 30% of the peripheral white blood cell count (D. Begg, unpublished data, 2009), and therefore, serial dilutions may result in a relatively low number of cells capable of producing IFNγ. In locations or circumstances where endemic parasitism could engender whole blood loss via the gastrointestinal tract, reductions in leukocyte number could potentially affect the sensitivity of the test. Similarly, sensitivity may also be affected by variations in leukocyte number because of physiologic and stress responses, such as the endogenous glucocorticoid-mediated lymphopenia that occurs in response to periods of chronic stress.
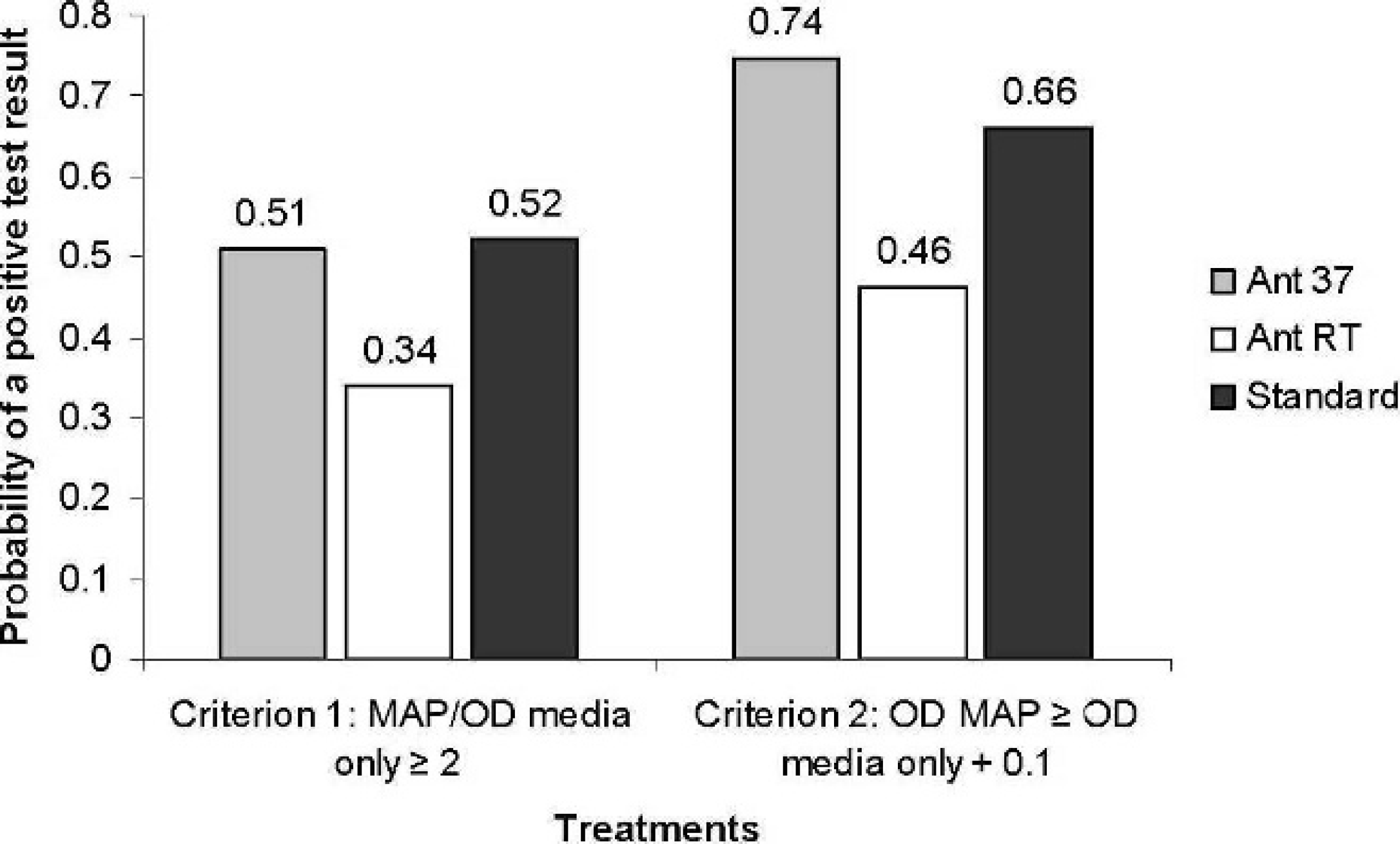
Logistic regression analysis to determine the effect of timing of addition of antigen and temperature during transport on the classification of the level of gamma interferon as positive or negative based on 2 classification criteria. Ant RT =
Field studies with blood samples from children in remote areas of South Africa have demonstrated that immediate incubation with antigen at 37°C and throughout transportation to the laboratory resulted in optimal IFNγ production as measured by flow cytometry (Stewart DJ: 2005, MLA Harvest Year Conference Programme.
There have been other novel ways of potentiating IFNγ responses in production animals where the delay from sample collection on farm to assay in the laboratory is prolonged. One such study (Jungersen G, Grell SN, Clemensen A, et al.: 2005, Interleukin-12 potentiation of the interferon-gamma test rescues day-old blood samples for diagnosis of paratubercu-losis PPD specific cellular mediated immune responses.
Acknowledgements
Research was funded by Meat and Livestock Australia (MLA). The authors would like to thank Ms. Natalie Schiller, Mrs. Anna Waldron, and Mr. Craig Kristo (Farm Animal Health Group, The University of Sydney); Mr. Jeff Eppleston (Rural Lands and Parks Board, Bathurst); and Mr. Hilton Brown (“Grovesdale,” Trunkey via Bathurst, New South Wales) for their excellent technical and animal assistance. Bovigam test kits were supplied by Pfizer Animal Health Australia with technical assistance provided by Ms. Eurife Abadin. Dr Stephen Jones (Cellestis International Pty Ltd.) kindly provided the portable incubator for the field trial.
Footnotes
a.
Bovigam®, CSL Ltd., Parkville, Victoria, Australia.
b.
Gudair®, Pfizer Animal Health, West Ryde, New South Wales, Australia.
c.
Provided by NSW Department of Primary Industries, Menangle, New South Wales, Australia.
d.
Vacutainer® tubes, BD, Franklin Lakes, NJ.
e.
RPMI medium 1640, Gibco-Invitrogen, Grand Island, NY.
f.
Sigma Chemicals Pty Ltd., Castle Hill, New South Wales, Australia.
g.
BD, Franklin Lakes, NJ.
h.
Multiscan Biochromatic 348, Helsinki, Finland.
i.
Diff-Quik, Lab Aids, New South Wales, Australia.
j.
Cellestis International Pty Ltd., Carnegie, Victoria, Australia.
k.
VSNI, Hemel Hempstead, Herts, United Kingdom.
l.
Minitab Inc., State College, PA.
m.
SAS Institute Inc., Cary, NC.
n.
QuantiFERON-TB®, Cellestis International Pty Ltd., Carnegie, Victoria, Australia.
