Abstract
To investigate the prevalence and features of postweaning multisystemic wasting syndrome (PMWS) in Japan, an epidemiological study was conducted in 692 weaned pigs with various clinical signs, commonly including wasting or weight loss, collected from 129 swine farms between 2000 and 2003. The presence of PMWS was diagnosed by the detection of characteristic histological lesions and moderate to large amounts of porcine circovirus type 2 (PCV2) antigen within the lesions in multiple lymphoid tissues. Postweaning multisystemic wasting syndrome was positive in 23.4% of pigs (162/692) over the course of the study, and occurred in 50.4% of the farms (65/129). Mortality in 30–120-day-old pigs in the farms positive for PMWS varied from 0.1 to 32.0%. No significant difference in mortality was seen between PMWS-positive and -negative farms (P = 0.1). However, mortality was significantly higher in the PMWS-positive farms where PMWS was diagnosed in more than 50% of the pigs examined compared to farms negative for PMWS (P = 0.02). These findings indicate that PMWS has spread widely in Japan. Moreover it may exist in variable forms in swine farms, including an epidemic form or a subtle endemic or sporadic form. A case-control study suggested that risk factors for the occurrence of PMWS include porcine reproductive and respiratory syndrome (PRRS) pneumonias and Mycoplasma hyorhinis infection.
Introduction
Postweaning multisystemic wasting syndrome (PMWS) is an emerging disease of pigs. The syndrome was first observed in Canada in 1991, 14 and outbreaks have since been recorded globally. 2 Pigs affected with PMWS exhibit wasting after weaning with or without dyspnea, diarrhea, pallor, and jaundice, 15 and develop multiple characteristic histopathological changes, including histiocytic cellular infiltrates with or without basophilic intracytoplasmic inclusion bodies, depletion of lymphocytes, syncytial multinucleated giant cells in lymphoid tissues, and granulomatous inflammation in any tissue. 2,38 The newly recognized porcine circovirus (PCV) type 2, which is antigenically and genomically different to PCV type 1 (PCV1), which establishes persistent infections in porcine cell lines, was initially recognized as the causal agent of PMWS. 10,30 However, subsequent investigations revealed that PCV2 infection alone usually does not result in PMWS, because most pigs experimentally inoculated with PCV2 isolates develop only subclinical infections; 4,11,26,35 moreover, field surveys have found high proportions of pigs with antibodies to PCV2 in pig farms in which PMWS had not been observed. 24,27
The detection of PCV2 is thus not sufficient to establish a diagnosis of PMWS, and PMWS case definition remains debatable. An accurate diagnosis for individual pigs is currently based on the presence of all of the following three conditions: 1) clinical signs including progressive wasting after weaning; 2) characteristic microscopic lesions; and 3) detection of PCV2 within the lesions. 43 In commercial swine farms, PMWS is typically manifested by a persistent increase in the number of uneconomical, slow-growing pigs. 14 An excessive increase in mortality compared with a historical level in a herd is thus proposed as a key criteria in a current case definition on herd level. 42 The pathogenesis of PMWS also remains unclear. Infectious co-factors, either singly or in combination, are thought to be risk factors for the development or exacerbation of PMWS. 13 Experiments with dual infection have identified porcine parvovirus (PPV), 1,22 porcine reproductive and respiratory syndrome virus (PRRSV) 3,16,41 and Mycoplasma hyopnemoniae 33 as candidate co-factors. There is also evidence that the administration of certain vaccines 32 or immunostimulation 23 may predispose to the development of PMWS.
Differences in the prevalence of PMWS among countries may assist in understanding the pathogenesis of this condition. To date, a number of epidemiological studies of the disease have been reported. 21,25,34,36,37,46,47 In Japan, PMWS was first described in 1997, 31 and a subsequent serological survey in 1999 detected PCV2 antibodies in 94.6% of clinically healthy pigs and 96.6% of randomly selected farms. 19 These findings indicated that PCV2 infection is common among the pig population in Japan. However, the prevalence and other epidemiologic features of PMWS in Japan remain poorly understood.
Here, to better understand the diagnosis and pathogenesis of PWMS, we conducted an epidemiological study to determine the prevalence of this condition in Japan. We also describe the mortality, microscopic lesions, and infectious agents associated with the disease.
Materials and methods
Experimental design
The investigation was conducted by local livestock hygiene service centers in 26 of 47 prefectures in Japan (Aomori, Iwate, Akita, Miyagi, Yamagata, Fukushima, Niigata, Ishikawa, Ibaraki, Gunma, Kanagawa, Shizuoka, Shiga, Kyoto, Osaka, Tottori, Shimane, Yamaguchi, Kagawa, Ehime, Tokushima, Kochi, Fukuoka, Saga, Nagasaki, and Okinawa) and in the authors' laboratory at the National Institute of Animal Health, Shichinohe, Japan between 2000 and 2003. The areas examined had a widely dispersed geographic distribution that broadly covered most of Japan. Each center examined 1–6 farrow-to-finish swine farms in their prefecture, and 2–9 piglets with poor growth after weaning at 25–180 days of age (average 76.8 ± 30.6 days of age) in each herd. Clinical signs varied, including respiratory disorders and diarrhea, but all had poor growth in common. A total of 692 diseased pigs were examined in 129 swine farms over 4 yrs, ranging from 113 to 200 pigs in each year. The average number of piglets examined per farm was 4.2 ± 1.6 piglets (2 piglets in 26 farms, 3 in 24, 4 in 27, 5 in 12, 6 in 34, 7 in 4, 8 in 1, and 9 in 1). By herd size, 38 farms (29.5%) had <100 sows, 62 (48.1%) had 101–500 sows, 17 (13.2%) had 501–1000 sows and 12 (9.3%) had > 1,001 sows. The number of piglets examined corresponded well with herd size; for example, 2 piglets were examined in farms with <100 or 101–500 sows and more than 6 piglets were examined in farms with 501–1,000 or >1,001 sows. Various tissues (tonsil, spleen, lymph nodes, lung, liver, kidney, and ileum) were collected from each pig and portions were stored at −80°C until use or fixed with 10% neutral buffered formalin for histopathological examination. For bacterial isolation, clinical specimens were cultured on MacConkey agar plates, sheep blood agar plates, or chocolate agar plates for 18 hr at 37°C. Bacterial isolates were identified by standard proto-cols. 17 Also, selected tissues were assayed by polymerase chain reaction (PCR) for PCV1, PCV2, PRRSV, PPV, Mycoplasma hyopneumoniae, and Mycoplasma hyorhinis. In addition, immunohistochemical (IHC) staining for PCV2 and PRRSV was performed on the formalin fixed tissues.
Because some local livestock hygiene service centers did not examine some pathogens in some pigs, the number of pigs examined differed by pathogen in PCR detection and in IHC for PRRSV. Mycoplasmal detection started from 2001 and thus fewer pigs were examined for Mycoplasma than for the other pathogens.
Diagnosis for individual pigs was based on characteristic histological lymphoid lesions 2,38 and positive IHC staining of PCV2 in the lesions. Pigs having these lesions with a moderate to large amount of PCV2 antigens in multiple lymphoid tissues were considered as positive, because slight lymphoid lesions in a single lymphoid tissue with a small amount of PCV2 antigen were observed even in pigs with subclinical infections in experimental infections. 4,11,20,26,35 In addition to the lymphoid lesions noted above, granulomatous lesions or lymphohistiocytic infiltrations with positive IHC staining of PCV2 were examined in lung, liver, and kidney.
The prevalence of PCV2 and PMWS in farms was examined on the condition that farms that had at least 1 pig with each factor were counted as positive. Each center also investigated current mortality data in the swine farms using a written questionnaire and mortality data was compared between PMWS-positive and negative farms. Moreover, the proportion of farms with a high frequency of cases, defined as a diagnosis of PMWS in more than 50% of animals examined, and the proportion of PMWS-positive or -negative farms with the coexistence of PRRS were also examined. PRRS was diagnosed in individual pigs by the presence of interstitial pneumonia with intralesional PRRSV antigen. Farms that had at least one pig with PRRS were counted as positive. For comparison of mortality data, farms were subgrouped as all PMWS-positive farms, farms positive for both PMWS and PRRS, farms positive for PMWS and negative for PRRS, farms positive for PMWS (50%,), farms positive for PMWS (50%,) and positive for PRRS, farms positive for PMWS (50%,) and negative for PRRS, all PMWS-negative farms, farms negative for PMWS and positive for PRRS, and farms negative for both PMWS and PRRS.
Histopathology and immunohistochemistry
Tissues were fixed in 10% neutral buffered formalin, then dehydrated, embedded in paraffin wax, sectioned at 5 μm, and stained with hematoxylin and eosin (HE). Immunohistochemical staining for PCV2 was performed using porcine anti-PCV2 serum raised against a PCV2 isolate as described previously. 20 In brief, tissue sections were deparaffinized, rehydrated, and treated with pronase. Each section was incubated with optimally diluted biotinylated PCV2 antiserum and visualized using a biotin-streptavidin diaminobenzidine system. a Sections were counterstained with Methyl green. PRRSV antigen in lung tissue was detected by IHC using rabbit anti-PRRSV serum as previously described. 18
Polymerase chain reaction
Polymerase chain reaction assays were used to detect specific nucleic acids of PCV1 12 and PCV2 20 in a homogenate of tonsil or lymph nodes; PPV in kidney 28 ; and PRRSV, 7 M. hyopnemoniae and M. hyorhinis in lung 5 from each animal. DNA or RNA was extracted from each sample using a commercial kit according to the manufacturer's instructions. 13 Polymerase chain reaction primers and details of thermocycling conditions are described in the respective references above and summarized in Table 1. In each assay, virus or virus-infected cells and mock-infected cells were included as positive and negative controls, respectively. The presence of target sequences in samples was visualized by 1.5% agarose gel eletrophoresis of the PCR amplicon with molecular size determination.
The primers and cycling conditions in PCR for detecting the pathogens.

Statistical analysis
Distribution of positive results for infectious agents between cases and controls was evaluated using the chisquare test. Strength of association between PMWS and an agent was evaluated using the odds ratio. Average mortality among farm groups was compared using the Mann-Whitney test. 8 Statistical significance was set at P <0.05 unless noted otherwise.
Results
Characteristics of PMWS in affected pigs
Table 2 shows the proportion of pigs positive for PCV2 and PMWS between 2000 and 2003. In total, PCV2 was positive by PCR in 556 of 672 pigs (82.7%). Twenty pigs were not examined for PCV2 by PCR. PMWS was diagnosed in 162 of 692 pigs (23.4%), from 25–150 days of age, with an average age of 74.6 ± 21.7 days. The majority (98.1%) were from 30 to 120 days old, with 2 peaks on 10-day stratification at 50–60 (22.8%) and 80–90 days (24.1%). The average age of unaffected pigs was 76.8 ± 33.5 days old and no average age difference was observed between affected and unaffected pig groups.
The characteristic microscopic lesions of PMWS in the lymphoid tissues of cases (n = 162) were, in descending order, cytoplasmic inclusion bodies (72.2%), lymphocyte depletion (69.8%), macrophage infiltration (43.2%), infiltration of multinucleate giant cells (31.5%), and eosinophil infiltration (5.6%). Figure 1 shows the characteristic lesions and IHC staining in a significant case. However, similar lesions were observed in the lymphoid tissues of pigs not diagnosed with PMWS (n = 469), except for cytoplasmic inclusion bodies, with lymphocyte depletion in 15.8%, macrophage infiltration in 3.6%, infiltration of multinucleate giant cells in 6.0% and eosinophil infiltration in 1.5%.
The proportion of pigs with PMWS-related lesions (n = 162) and PCV2 antigen (the number examined differed among tissues) was determined in lung, liver, and kidney of case animals. Interstitial pneumonia including alveolar thickening and lymphohistiocytic or multinucleate giant cell infiltration was the most frequent lesion (50.6%), followed by lymphohistiocytic or histiocytic infiltration in interstitial areas in the kidney (46.3%), and lymphohistiocytic infiltration or hepatocyte necrosis in the liver (38.9%). PCV2 antigen was detected in 50.3% (75 of 149) of lung samples, 39.3% (42 of 107) of kidney and 30.3% (36 of 119) of liver in PMWS cases.
Prevalence of PCV2 genome or PMWS in pigs with poor growth after weaning.
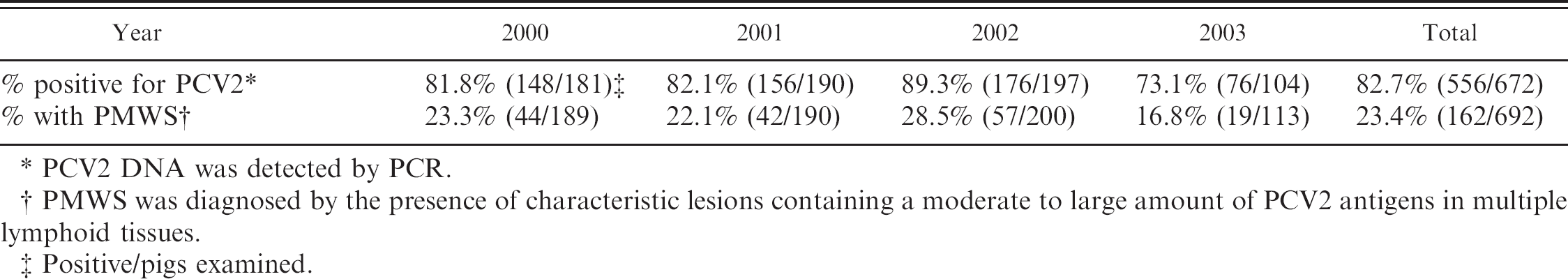
PCV2 DNA was detected by PCR.
PMWS was diagnosed by the presence of characteristic lesions containing a moderate to large amount of PCV2 antigens in multiple lymphoid tissues.
Positive/pigs examined.
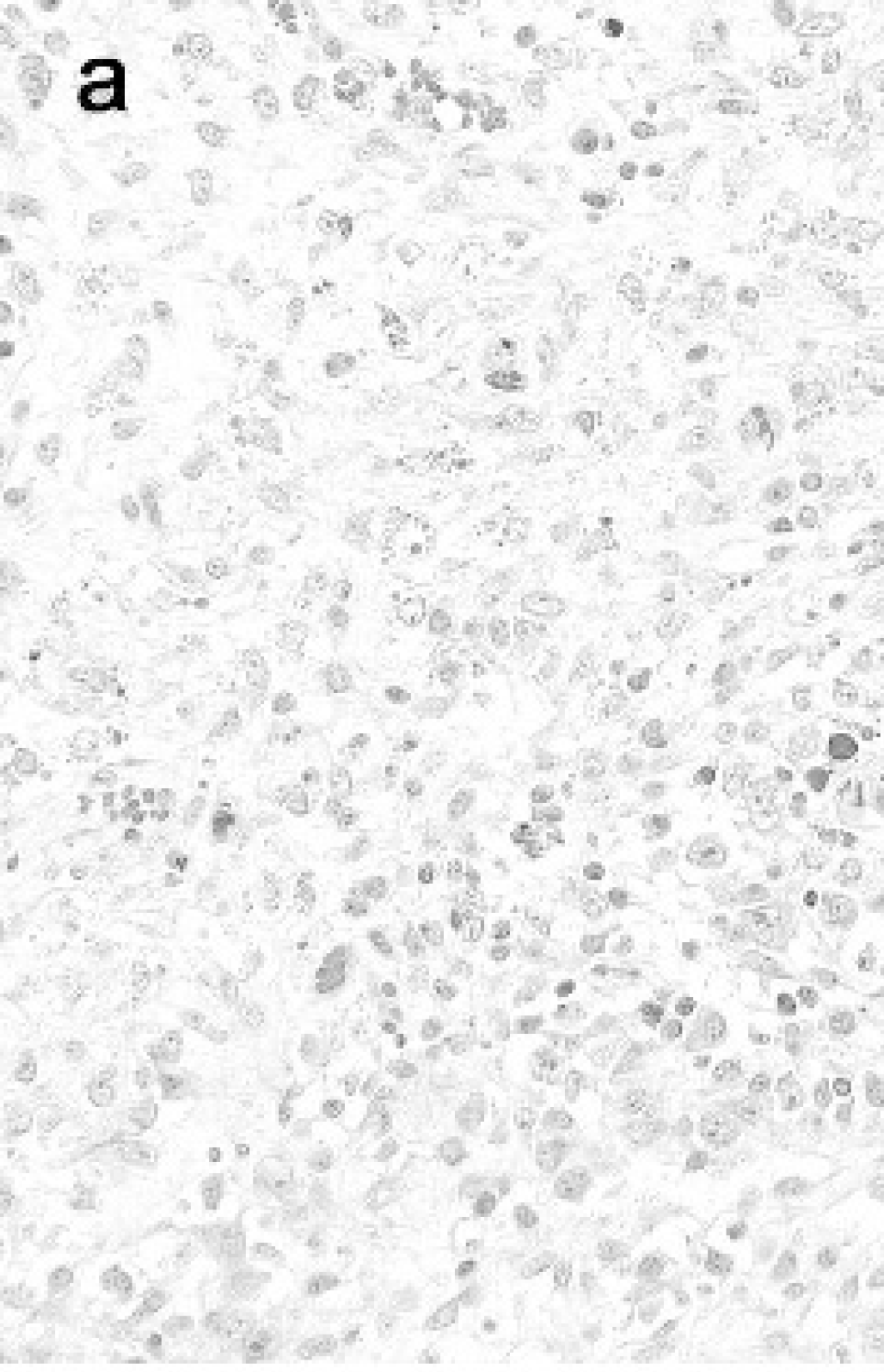
a, Lymphocyte depletion with intracytoplasmic inclusion bodies (arrows) in lymphoid follicles of lymph node in a significant case of PMWS. HE stain, b, Large amount of PCV2 in lymph node in the same pig as
Forty-one pigs with characteristic PMWS lesions only in 1 lymphoid tissue with a small amount of PCV2 antigen within the lesions, 15 pigs without antigen in lymphoid tissues but with antigen in lesions in lung or kidney, and 5 pigs with the immunohistochemical presence of PCV2 but no lymphoid tissue lesions failed to meet the inclusion criteria and were excluded from the count of PMWS-positive pigs. These pigs were also excluded from PMWS-negative cases in a case-control study, because pigs with mild PMWS lesions or extralymphatic lesions may be in a convalescence stage of the disease.
The prevalence of various pathogens in PMWS-positive and -negative cases is summarized in Table 3. By PCR, 100% of PWMS cases were positive for PCV2. Although there was a significant difference in the proportion of positive pigs among groups (P < 0.001), 75.1% of negative pigs were positive for PCV2. PCV1 occurred in varying proportions of pigs examined, but positive pigs were almost equally distributed between case and negative pigs (P = 0.24). PPV and M. hyopneumoniae were detected by PCR in 5.0% (7/139) and 14.0% (15/107) of case pigs, respectively, but these ratios were less than those in negative pigs (P = 0.05, P = 0.04). M. hyorhinis was detected by PCR in 56.5% of case pigs (61/108), significantly higher than in negative pigs (P = 0.002). The odds ratio for the strength of the association of M. hyorhinis with cases was 2.0 (95% confidence interval = 1.3, 3.2). Pasteurella multocida (19.8%, 32/162) and Streptococcus suis (7.4%, 12/162) were common bacterial isolates, but were again distributed almost equally among case and negative pigs (P = 0.69, P = 0.33). Among bacteria pathogenic for pigs, Haemophilus parasuis (5.6%, 9/162), Actinobacillus pleuropneumoniae (4.9%, 8/162), Salmonella choleraesuis (4.9%, 8/162), and Arcanobacterium pyogenes (2.5%, 4/162) were isolated from pigs with PMWS. Porcine reproductive and respiratory syndrome virus was detected by PCR in 59.0% of cases (85/144), and this frequency was significantly different from that in negative pigs (P = 0.03). In addition to PCR detection, IHC revealed PRRSV antigen in lung tissue of 39.9% of cases (63/158) and 19.7% of PMWS-negative pigs (90/456), with this difference being significant by the chi-square test (P < 0.001). Odds ratio for the IHC association of PRRSV with cases was 2.7 (95% confidence interval = 1.8, 4.0).
Prevalence of various agents in PMWS-positive and negative pigs.
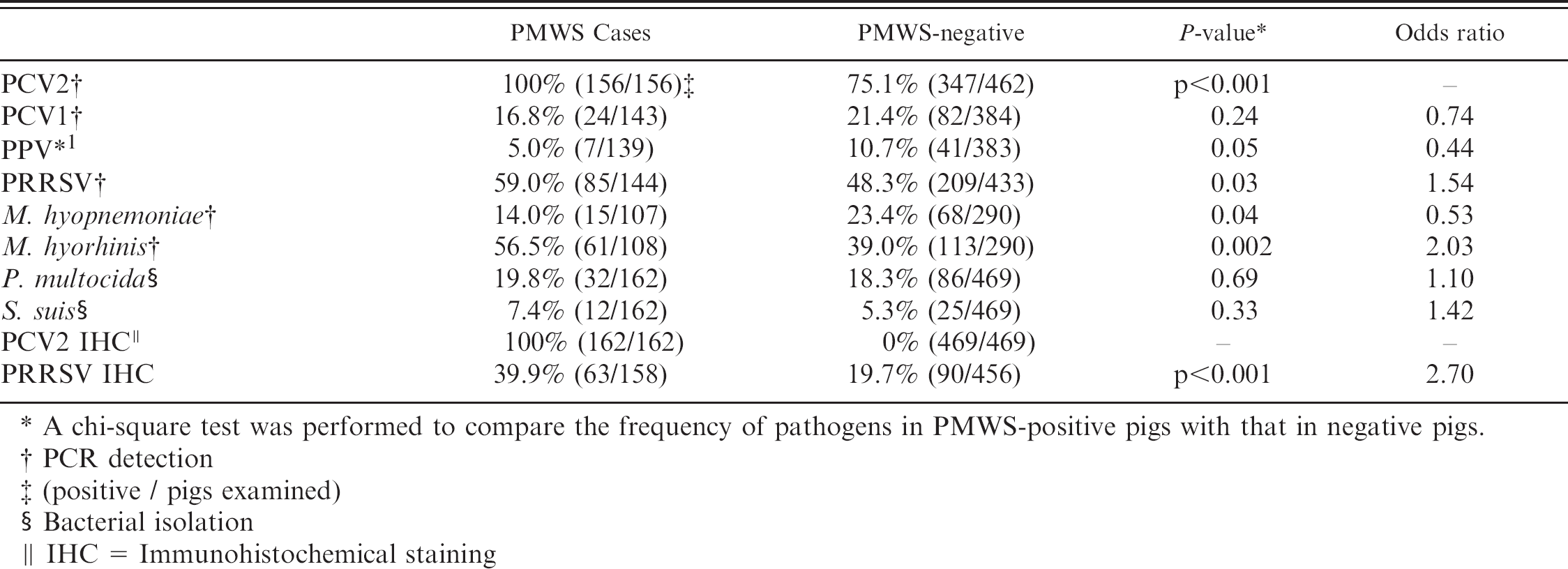
A chi-square test was performed to compare the frequency of pathogens in PMWS-positive pigs with that in negative pigs.
PCR detection
(positive / pigs examined)
Bacterial isolation
IHC = Immunohistochemical staining
Prevalence and mortality of PMWS in farms and association with PRRS
The prevalence of PCV2 and PMWS in swine farms between 2000 and 2003 is summarized in Table 4. The total proportion of farms positive for PCV2 was 96.7% (119/123), with a range of 86.2-100% by year. The total proportion of farms positive for PMWS was 50.4% (65/129), with a range of 34.5-65.7% by year. The proportion of farms with a high frequency of cases (defined as a diagnosis of PMWS in more than 50% of animals examined) was 22.5% (29/129), with a range of 16.7-27.6% by year.
The total proportion of farms positive for PRRS was 46.5% (60/129), while that of farms positive for PRRS was 58.5% of PMWS-positive farms (38/65) over the entire period. Comparison of the proportion of farms positive for both PMWS and PRRS (58.5%) to that positive for PRRS but negative for PMWS (34.4%, 22/64) showed a significant difference between the groups (P = 0.006), and the odds ratio for the strength of the association for both PMWS and PRRS was 2.7 (95% confidence interval = 1.3, 5.5).
To examine whether PMWS consistently decreased productivity on each swine farm, mortality in those aged 30–120 days was investigated at the time of collection. This age range was selected from the observation that the majority (98.1%) of cases were located in animals aged 30–120 days. There was a wide variation in mortality among PMWS-positive farms, ranging from 0.1 to 32.0%. Farms were almost equally distributed by herd size between PMWS-positive and -negative farms. Among PMWS-positive farms (n = 65), 26.2% were farms with <100 sows, 49.2% had 101–500 sows, 13.8% had 501–1,000 sows and 10.8% had <1,001 sows. Among PMWS-negative farms (n = 64), 32.8% had <100 sows, 46.9% had 101–500 sows, 12.5% had 501–1,000 sows, and 7.8% had < 1,001 sows. No significant difference was seen in average total mortality between PMWS-positive (8.8 ± 6.4%) and -negative farms (7.6 ± 6.8%). When farms with a PMWS frequency of more than 50% (PMWS [>50%]) were extracted from the PMWS-positive farm group (n = 28), mortality (10.9 ± 7.2%) was significantly higher than in PMWS-negative farms and farms negative for both PMWS and PRRS (n = 42, 6.9 ± 5.8%). Mortality in the PMWS-positive and PRRS-negative farm group (n = 27, 10.3 ± 8.0%) was higher than that in farms negative for both PMWS and PRRS. Mortality in farms positive for PMWS (<50%) and negative for PRRS (n = 12, 12.5 ± 8.7%) was higher than that in farms negative for PMWS and in farms negative for both PMWS and PRRS. No significant differences were seen in mortalities in farms positive for both PMWS and PRRS (n = 27, 7.7 ± 4.8%) and in farms positive for both PMWS (50%,) and PRRS (n = 16, 9.7 ± 6.0%) compared with any other subgroups. Regarding another production parameter, the average number of days to market was evaluated in each group but no significant difference was seen among them.
Prevalence of PCV2 and PMWS in farms.
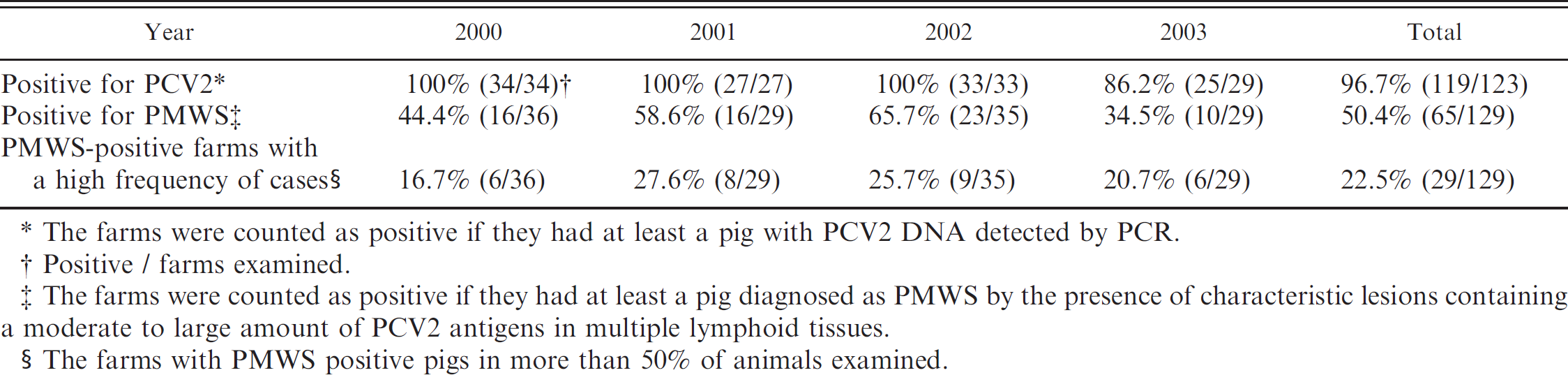
The farms were counted as positive if they had at least a pig with PCV2 DNA detected by PCR.
Positive / farms examined.
The farms were counted as positive if they had at least a pig diagnosed as PMWS by the presence of characteristic lesions containing a moderate to large amount of PCV2 antigens in multiple lymphoid tissues.
The farms with PMWS positive pigs in more than 50% of animals examined.
Discussion
These findings clearly demonstrate that PCV2 and PMWS are common in pig populations throughout Japan. Indeed, 82.7% of pigs examined (556/672) were positive for PCV2 DNA and 23.4% (162/692) were positive for PMWS. Almost all (96.7%) of the 123 farms in this study were positive for PCV2 DNA, and over half (50.4%, 65/129) had at least 1 pig with PMWS. These results also indicate that PMWS is widespread in Japan and may exist in variable forms in swine farms, including a subtle endemic or sporadic form.
There was no difference in average mortality between PMWS-positive (8.8 ± 6.4%) and -negative farms (7.6 ± 6.8%). This was unexpected, because a characteristic feature of farms with PMWS is an increased number of uneconomical growing pigs and secondary infections. 14 One possible explanation for these observations is that a considerable proportion of PMWS may be expressed as an endemic or sporadic disease that causes no apparent reduction in farm productivity. This consideration arose on the basis of findings of the high prevalence of PMWS in pigs and farms and the occurrence of PMWS in farms with very low mortality, the minimum of which was 0.1%. We speculate that the presence of PRRS might confound the comparison of mortality because it is a significant factor in worsening mortality among pigs in the growing phase. 39 In the present study, mortality in farms positive for PRRS but negative for PMWS was high (9.0 ± 8.3%). Although PRRS to some degree influenced the comparison of mortality between PMWS-positive and -negative herds, this confounding effect was not present in the subgroup of farms positive for PMWS and negative for PRRS.
Indeed, mortality in this sub-group (10.3 ± 8.0%) was significantly higher than in farms negative for both PMWS and PRRS (6.9 ± 5.8%). From another standpoint, mortality in the subgroup of farms with a high frequency (>50%) of pigs affected with PWMS (10.9 ± 7.2%) was significantly higher than that in PMWS-negative farms. Moreover, mortality in the subgroup of farms with a high frequency of pigs affected with PWMS (>50%) but negative for PRRS (12.5 ± 8.7%) was significantly higher than that in farms negative for both PMWS and PRRS. These farms may be thought of as having an epidemic or outbreak of PMWS with an apparent increase in uneconomical growing pigs. On the other hand, the proportion of farms with a low frequency of pigs affected with PMWS (<50%) accounted for 55.4% of PMWS-positive farms (36/65 farms). Subclassification of PMWS into epidemic and endemic forms may be needed in the case definition of PMWS, and the frequency of individual pigs diagnosed with PMWS among examined pigs may be useful in examining the diagnosis of herd cases as well as in the increased mortality in the farms. Taken together with observations in pigs with PMWS lesions in Japan in 1989, 29 the spread of PMWS in Japanese pig populations, including farms with extremely low mortality rates and with a low frequency of the disease, suggests that not only PCV2 infection but also PMWS existed sporadically before the PMWS outbreaks in the 1990s. Swine farming significantly changed in Japan in the 1990s due to a drastic increase in the number of large-scale farms. This environmental change in swine farming or epidemics of other pathogenic microorganisms may have promoted the recent appearance of PMWS.
A previous serological study in Japan found antibodies to PCV2 in 94.6% of clinically healthy pigs and in 96.6% of farms. 19 Similar high prevalence rates of PCV2 infection have been reported in other countries. 9,27,46 In the present study, PCV2 DNA was detected in 75.1% of pigs without histological lesions of PMWS. porcine reproductive and respiratory syndrome, M. hyopneumoniae, or bacterial infections were major factors in the poor growth of these PMWS-negative pigs, as shown in Table 3. However, lesions histologically similar to those of PMWS (e.g., lymphocyte depletion, granulomatous inflammation, and eosinophil infiltration) were observed in some PMWS-negative pigs. Lymphocyte depletion or granulomatous inflammation in PMWS-negative pigs may be caused by physical distress, nutritional influences, or other infectious agents. 40,45 Although eosinophil infiltrations in lymphoid tissues were seen in experimental infections with PCV2, 11,20 the diagnostic value of this sign in PMWS may be lower than those of the other characteristic lesions because of its low frequency in PMWS-positive pigs. These observations strongly suggest that a single examination based on clinical appearance, viral detection, or histological lesions is not sufficient for a diagnosis of PMWS. The results of this study therefore corroborate the current normative guidelines for the diagnosis of PMWS, which require the fulfillment of three criteria: presence of clinical signs, characteristic histological lesions, and detection of PCV2 within the lesions. 44 Immunohistochemical examination and in situ hybridization have been used to detect PCV2 in tissue sections 2,38 and are essential to the diagnosis of PMWS. In the present study, however, a more limited definition, namely the characteristic lesions in multiple lymphoid tissues and a moderate to large amount of PCV2 antigen by IHC within the lesions was used in the diagnosis of individual pigs in addition to the current normative guidelines on the basis that lymphoid lesions in a single tissue or the presence of a small amount of PCV2 antigen was observed in pigs which developed subclinical infections in experimental inoculations with PCV2. 4,11,20,26,35 Further, lesions caused by PCV2 infection in extralymphatic tissues may be classified as other diseases rather than PMWS. 43 Accordingly, we excluded 41 pigs positive for the lesions in a single lymphoid tissue and with a small amount of PCV2 antigen by IHC, 15 pigs negative for PCV2 antigen in lymphoid tissues but with antigen in lesions in lung or kidney, and 5 pigs positive for PCV2 antigen but having no lesions in lymphoid tissues as not PMWS-positive. The interpretation of the finding of pigs with PCV2 antigen but no lymphoid lesions is difficult; possibilities may include that these pigs are in a convalescence stage or have a very mild infection of the virus.
Results from experimental co-infections 3,16,41 have suggested that infection with PRRSV is a co-factor related to the exacerbation of PMWS. In this study, the frequency of PRRSV as determined by PCR in PMWS-positive pigs was significantly higher than in PMWS-negative pigs (P = 0.03) and the odds ratio was 1.54. On the other hand, the immunohistochemical presence of PRRSV in lung was significantly higher in PMWS cases (P < 0.001). The difference in the frequency of PRRSV between positive and negative cases was wider by immunohistochemical presence than with PCR detection, with an odds ratio for the former of 2.7. The proportion of pigs with PRRS pneumonia was significantly higher in PMWS-positive (58.5%) than -negative farms (34.4%) (P = 0.006). The odds ratio for the strength of association for PMWS and PRRS in farms was 2.7. These data indicate that the manifestation of PRRS pneumonia or proliferation of PRRSV in lung is a risk factor for the occurrence of PMWS, rather than the presence of PRRSV infection. Interestingly, mortality on farms with both PRRS and PMWS was not significantly different from that on farms positive for PMWS but negative for PRRS. Other factors may be related to the exacerbation of PMWS. Comparison between M. hyorhinis-positive and -negative pigs revealed that this bacterial infection was also a risk factor (P = 0.002, odds ratio; 2.03). Mycobacterium hyorhinis is considered part of the normal flora of the upper respiratory tract and a secondary pathogen in porcine pneumonias. 6 In this study, however, there was no significant difference for other secondary respiratory bacteria such as Pasturella multocida or treptococcus suis. A relationship between M. hyorhinis infection and PRRSV in a case of severe pneumonia has been reported. 18 The role of M. hyorhinis infection in PMWS warrants further study.
In conclusion, this study indicates that there is a high prevalence of PCV2 infection and PMWS in Japanese pig farms. Mortality in farms with PMWS-affected pigs varied widely. These findings suggest that PMWS manifests in not only epidemic but also endemic forms with no apparent increase in mortality in swine farms.
Acknowledgements
We wish to thank the following members of the Working Group for the Diagnosis of PWMS for their microbiological and histopathological examinations in pigs and investigations of mortality on farms: Kodama Y, Sato N, Seki Y, Onuma M, Abe Y, Takahashi K, Oikawa T, Hosokawa M, Miyano H, Hamaguchi N, Murayama S, Hayashi M, Kikuchi T, Iizima Y, Higuchi A, Arai M, Endo S, Michigoshi K, Yoshimura R, Kira T, Nakai T, Inoue M, Yasuda W, Okazaki N, Yamamoto Y, Takahashi S, Ando M, Toujou H, Yamada M, Risho Y, Kawanabe M, Ichimaru H, Arishima T, Yamamoto K, and Kudaka M. We gratefully acknowledge the financial support provided by the Ministry of Agriculture, Forestry and Fisheries of Japan. We would also like to thank Dr. Steven D. Sorden of Iowa State University for his constructive suggestions.
Footnotes
a.
Nichirei Biosciences Inc, Tokyo, Japan.
b.
Qiagnen K.K., Tokyo, Japan.
