Abstract
Formalin fixed paraffin embedded tissues blocks are used routinely to diagnose the economically important immunosuppressive infectious bursal disease virus (IBDV) in chickens. Immunohistochemical detection of viruses in tissue blocks has been done with varying results between laboratories. Extraction of IBDV RNA from tissue blocks allows IBDV strain identification at a molecular level. This allows correlation between virus identity and histological lesions present in the tissue. Experimentally reverse transcription-polymerase chain reaction (RT-PCR) detectable IBDV RNA could always be extracted from tissue blocks with acute +3 or higher histological lesion scores. However, many blocks from diagnostic field cases did not yield detectable IBDV RNA, in spite of having severe IBDV histological lesion scores. The reason for this can be the effect different formalin fixation conditions have on RNA detection from tissue blocks. To study the effect of various fixation parameters on RNA extraction and immunohistochemical detection of IBDV, bursas with maximum histological lesion score of 4 for IBDV were fixed in formalin under various conditions (different pH levels, temperatures, concentrations of formalin, and fixation duration). Only tissues fixed in formalin with a pH of 7.0, concentration of 5 or 10% formaldehyde, storage temperature of 25°C or less, and kept for up to 2 weeks in formalin yielded detectable IBDV RNA upon extraction. No RNA could be detected from tissues fixed under extreme temperature, pH, or formalin concentrations. Optimal fixation conditions for IHC detection of IBDV were 10% formalin concentration, pH 7.0, and temperature of 4°C, where maximum intensity of immunostaining was observed.
Keywords
Introduction
Classical infectious bursal disease (IBD), or Gumboro disease, is an acute, highly contagious viral disease of young birds characterized by severe necrosis, hypoplasia, or atrophy in the bursa of Fabricius, mainly observed at 3–6 weeks of age. 23 Infectious bursal disease has worldwide distribution, and the effects of the disease are economically significant to the commercial poultry industry. Highly virulent strains cause increased mortality rates in chickens. Survivors of classical IBD are immunosuppressed, which is also the outcome of IBDV infection with antigenic variant strains. Variant IBDVs differ from the classical IBDV strains in that they usually do not cause mortality and can infect chickens younger than 3 weeks of age. Variant viruses cause immunosuppression by viral induced apoptosis of lymphocytes in the bursa. Immunocompromised chickens have an increased susceptibility to other pathogens, such as Newcastle disease virus, infectious bronchitis virus, infectious laryngotracheitis virus, Salmonella spp, or to other opportunistic agents such as Escherichia coli. Furthermore, immunosuppressed birds may not respond properly to vaccination and may suffer severe or persistent postvaccinal reactions. 13
Infectious bursal disease virus (IBDV) is a member of the Birnaviridae family with a genome consisting of 2 segments of double-stranded RNA. Infectious bursal disease virus is a nonenveloped icosahedral virus with a diameter of 55–60 nm. The smaller segment of RNA encodes viral protein (VP) 1, which is the RNA polymerase of the virus. The large segment encodes a polyprotein that is processed into 3 structural proteins, VP2, VP3, and VP4. 2,10,19 The large segment also encodes the structural protein VP5 in another reading frame. 15 Of these different genes, VP2 gene encodes for the major antigenic protein and its hypervariable region is what confers variability among different IBDV strains.
The severity of IBDV infection depends on the virulence of the virus, age of bird, and level of circulating maternal antibodies. Cytotoxic T cells may exacerbate virus-induced cellular destruction by lysing cells expressing viral antigens. T cells may also promote the production of pro-inflammatory factors, such as nitric oxide, increasing tissue destruction. 18 Outbreaks with high mortality rates caused by very virulent IBDV (vvIBDV) strains have been reported in Europe, Asia, and Latin America. 21 Subclinical and immunosuppressive forms of the disease are prevalent in the United States, where reduction of body weight and lack of uniformity may be produced in mild outbreaks. 13
Real-time RT-PCR (rt-RT-PCR) has been used previously to detect IBDV in tissues of infected chickens that were either frozen or stored in phenol. 13,14 Infectious bursal disease viral RNA has also been detected by rt-RT-PCR from formalin-fixed, paraffin-embedded bursas. 17 Using this technique, formalin fixed paraffin embedded bursa of Fabricius from chickens experimentally infected with IBDV, and with histological lesion scores of +3 or more always yielded rt-RT-PCR detectable IBDV RNA. However, diagnostic field cases did not always yield rt-RT-PCR detectable RNA. Some samples (225 blocks over a 3-year period) that had acute +4 histological lesion scores were negative for IBDV with rt-RT-PCR. This finding occurred more often in diagnostic samples submitted from outside the United States than in those originating from the United States (paper to be published in Avian diseases). The reason why some infected samples tested negative by rt-RT-PCR has yet to be determined. It was recommended that these samples should be fixed in 10% buffered formalin and kept at room temperature, but pH analysis of formalin and HE artifacts indicated that some of the samples were fixed in more concentrated formalin, formalin with variable pH levels, or subjected to high temperatures during formalin fixation. These observations suggested that formalin fixation conditions might have an effect on rt-RT-PCR detectable RNA from tissue blocks.
Previous research has proved that formalin fixation of tissues is suitable for immunohistochemistry because formalin is a relatively mild fixative for preservation of antigenic reactivity. 22 However, formalin fixation can impair or block the antigenic reactivity of certain proteins. 1,4,16 This impairment may be owing to extensive cross-linking of proteins by formaldehyde, where amino groups become blocked and antibodies to those amino groups do not participate in IHC staining. 20
The objective of this study was to experimentally simulate field conditions in terms of storage time, temperature, pH of unbuffered formalin, and different concentrations of buffered formalin to test their effect on IBDV RNA extraction from paraffin embedded tissue for use in rt-RT-PCR and immuno-histochemical staining.
Materials and methods
Birds and experimental design
A total of 95 specific pathogen free (SPF) chickens were hatched from fertilized eggs a for this experiment. All birds were housed in Horsfall-Bauer isolation units in 19 groups of 5 and maintained under positive pressure. Food and water were provided ad libitum. At 3 weeks of age, the chickens were inoculated with IBDV Blen vaccine b which contains the 2512 strain. Inoculation routes of intranasal, oral, and intramuscular administration (1×104.0 EID50/route) were given simultaneously to each infected bird to ensure a high viral load in infected tissue. Five uninfected birds were maintained as uninfected controls. Bursal tissue was collected 5 days postinoculation and subjected to different fixation protocols as described in the experimental design shown in Table 1.
Histopathology and immunohistochemistry
A37% formaldehyde solution was diluted in either neutral phosphate buffered saline (PBS), distilled water, or was used undiluted for formalin fixation. The pH was adjusted with either 10% sodium hydroxide or 5% hydrochloric acid. Paraffin-embedded tissues were sectioned, mounted, stained with hematoxylin and eosin (HE) by using an automated stainer, c and examined for lesions by light microscopy. All sections were assigned a lesion severity score. For all samples, a lesion score of 1 represented no lesions; 2 represented mild reduction in overall follicle size; 3 represented moderate reduction in follicle size; and 4 represented either necrosis or follicle atrophy. 17 All procedures for immunohistochemistry (IHC) were done at room temperature. Five-micron-thick tissue sections were cut from paraffin-embedded samples and mounted on charged glass slides. c Paraffin was melted from slides (10 min at 65°C) and removed by immersion in Hemo-De d 3 times (5 min each). Slides were then air dried. Antigen retrieval was done by incubating slides with 10% proteinase K e for 5 min at room temperature. IHC staining was performed with an automated stainer f using a commercial kit g according to the manufacturer's recommendations. The primary antibody used was a mouse monoclonal antibody specific to and cross-reactive among all IBDV strains. h The secondary antibody h was a peroxidase labeled polymer conjugated to anti-mouse and anti-rabbit immunoglobulins. After IHC staining, sections were counterstained with hematoxylin, cover slipped, and examined by light microscopy. The degree of IBDV infection in each section was scored as follows: — = no infection; + = minimal infection (>10% of cells present); ++ = moderate infection (>10% and <30% of cells present); and +++ = intense infection (>30% of cells present). 17
Fixation conditions for bursal tissues from IBDV infected and control chickens.

Five individual bursas were tested per treatment.
This group was also used to represent 5 days duration in formalin.
Noninfected and infected controls were stored in 10% buffered formalin at 4°C to validate IHC procedure.
RNA extraction and quantification
RNA was extracted from formalin-fixed, paraffin-embedded bursas and examined for IBDV nucleic acid by rt-RT-PCR. 17 Sections totaling 50 μm in thickness were cut from each formalin-fixed, paraffin-embedded tissue block, deparaffinized in Hemo-De, e washed with 100% ethanol, and digested with 25 μg/ml proteinase K f for 1 hr at 50°C. RNA was extracted using Trizol i according to the manufacturer's recommendations, resuspended in 25 μl of 90% dimethyl sulfoxide, and frozen at −80°C until assayed. 17 Extracted RNA was quantified using eppendorf biophotometer j and 50–2,000 μl UVette. k
Real-time RT-PCR
The RNA extracted from tissue blocks was denatured at 95°C for 5 min and kept on ice. A rt-RT-PCR was performed using reagents from the LightCycler RNA Amplification SYBR Green I Kit l and the LightCycler machine m according to manufacturer's recommendations. The primers (B5 5′ TCTTGGGTATGTGAGGCTTG and B4 3′ GGATGTGATTGGCTGGGTTA) 17 were designed to amplify a 400-base pair (bp) segment of the hypervariable region of the IBDV VP2 gene (GenBank accession number DQ355819). Briefly, RT was performed at 55°C for 30 min, followed by denaturation at 95°C for 30 sec. Fifty-five PCR cycles were performed, consisting of denaturation (95°C for 1 sec), hybridization (55°C for 10 sec), and extension (72°C for 13 sec). A melting curve analysis was done with an initial denaturation at 95°C (for 1 sec). DNA melting was accomplished with an initial temperature of 65°C for 10 sec and a gradual temperature increase of 0.1 °C/sec until 95°C was reached. After which, there was a cooling segment and a gradual temperature decrease of 2°C/sec till the holding temperature 20°C was reached. The melting temperature of the expected 400-bp amplicon was between 82°C and 84°C. This estimated melting temperature was used to confirm the presence of IBDV specific products obtained using real-time RT-PCR. 17 A no template control (DEPC-water) and a positive IBDV control was used in each run. Electrophoresis of real-time RT-PCR products were run on 1.5% agarose gel and visualized by ethidium bromide staining, to correlate between melting curve peaks and size of amplified product. This was done initially to verify the specificity of the melting curve peaks seen in the rt-RT-PCR reaction printouts.
Results
Histology and immunohistochemistry
Out of 90 infected birds, 89 bursas had lesion scores of 4, the remaining bursa had a lesion score of 3. All 5 chickens from the uninfected negative control group had lesion scores of 1. All of the IBDV uninfected negative control slides were negative using immunohistochemistry. All 5 IBDV infected positive control slides demonstrated strong immunoreactivity for IBDV. This demonstrated the specificity of the IHC technique used (Fig. 1, Table 2).
Histological evaluation of 5 bursas from each treatment group demonstrated that for IBDV infected bursas fixed in different concentrations of formalin, the optimal formalin concentration for formalin fixed tissues used for IHC staining was 10% formalin (Table 2). The use of 5% formalin for formalin fixation produced a light nonspecific background staining of erythrocytes and fibroblasts. Infectious bursal disease virus antigen was not detected in slides that came from paraffin embedded tissues fixed in 20% or 37% formalin (Table 2). One area in one of the 5 samples fixed in 37% formalin had lymphoid cells with positive staining. Light staining of apoptotic bodies, epithelial cytoplasm and vacuoles were present and was interpreted as nonspecific background staining. Hematoxylin and eosin staining showed severe lymphoid depletion, atrophy in follicles, and increase in interfollicular stroma. Artifacts seen in tissues fixed with 20% or 37% formalin concentrations included granularity of interplical mucin, and mucin in epithelial cysts. Tissues fixed in 37% formalin had diffuse eosinophilic staining.
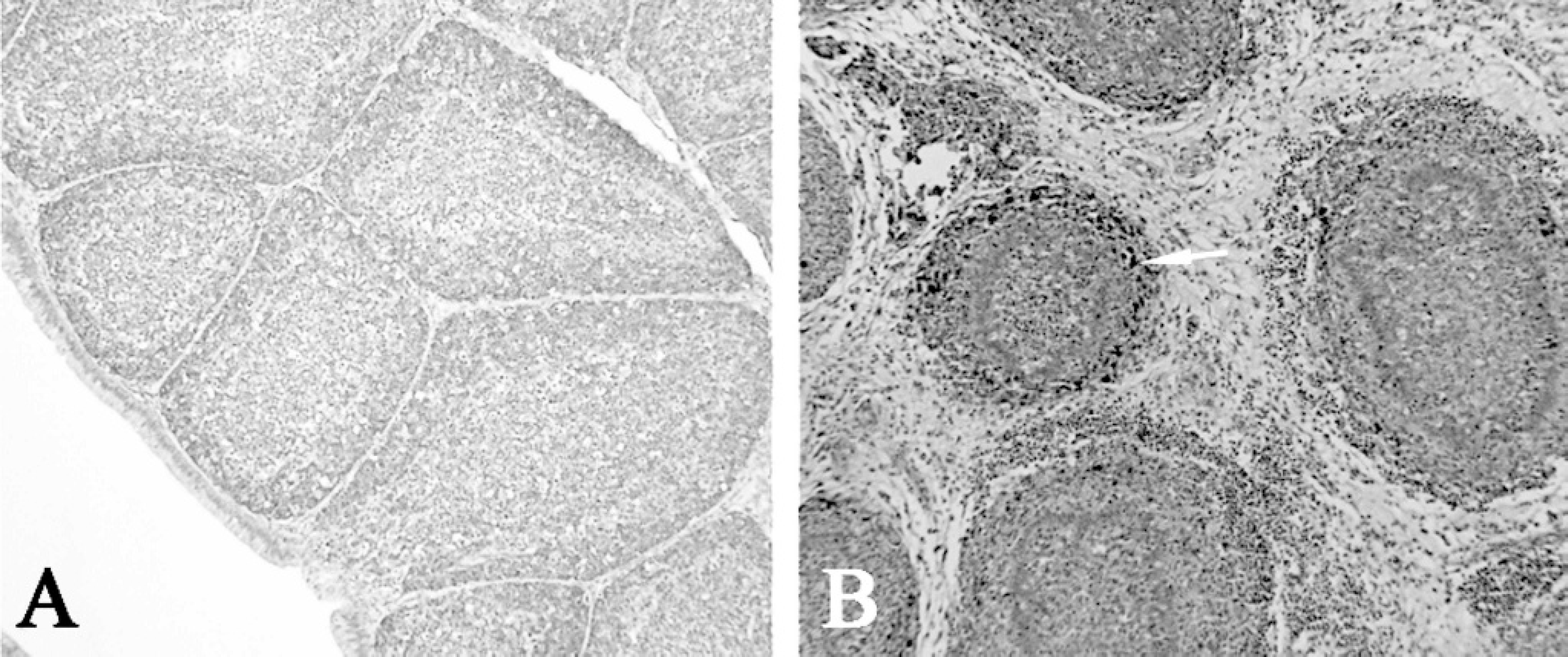
Photomicrograph of bursa of Fabricius. (IHC technique specificity control for IBDV infected and noninfected tissues).
Tissues fixed in 10% buffered formalin at different fixation temperatures showed variable IHC staining. Fixation at 4°C yielded the best staining. The number of IHC staining positive cells decreased as the fixation temperature increased (Table 2). Hematoxylin and eosin artifacts observed in tissues stored at 50°C included streaming and smudging of nuclei, loss of follicular architecture and very plump fibroblasts in stroma of atrophied plica.
Histopathology and IHC mean staining scores of 5 bursas of Fabricius from chickens infected with IBDV and subjected to different fixation conditions.
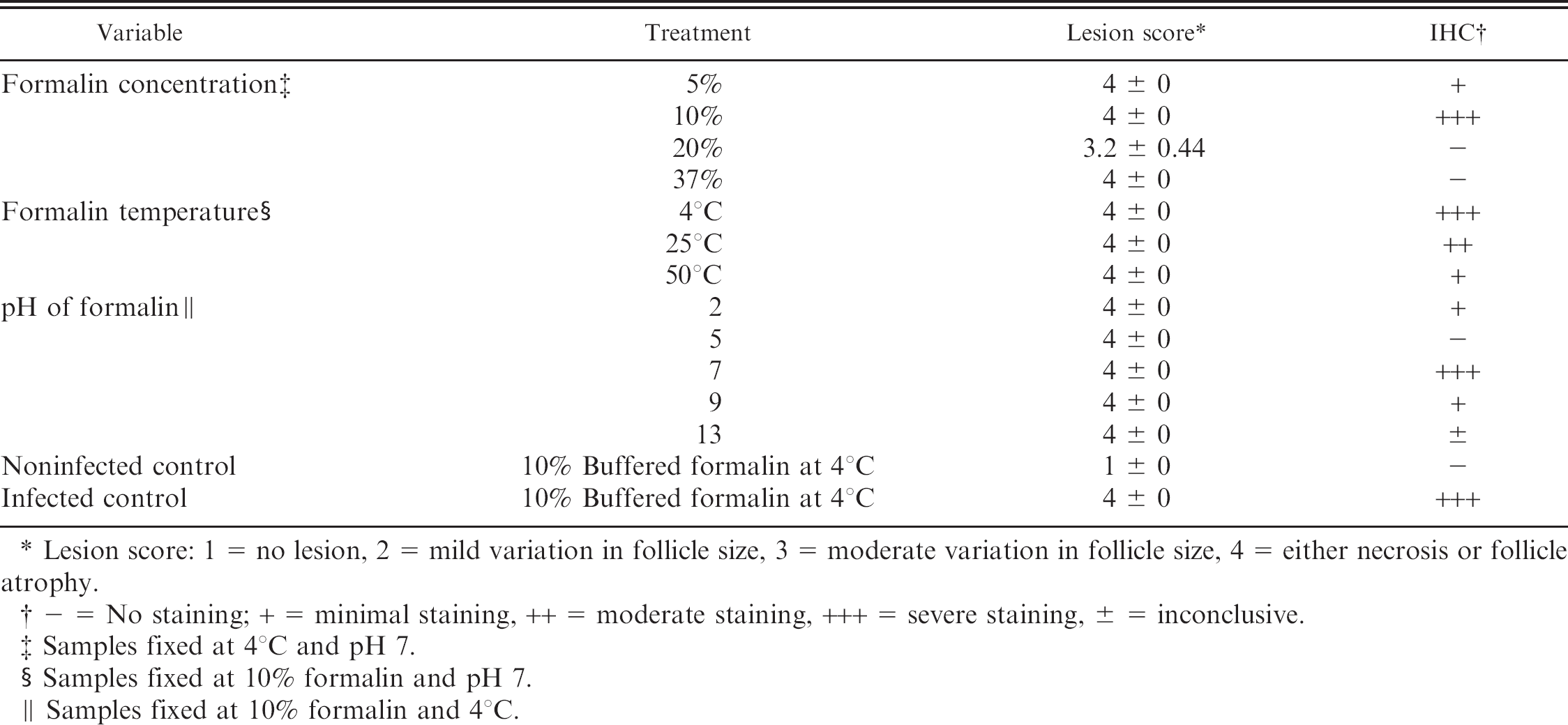
Lesion score: 1 = no lesion, 2 = mild variation in follicle size, 3 = moderate variation in follicle size, 4 = either necrosis or follicle atrophy.
— = No staining; + = minimal staining, ++ = moderate staining, +++ = severe staining, 6 = inconclusive.
Samples fixed at 4°C and pH 7.
Samples fixed at 10% formalin and pH 7.
Samples fixed at 10% formalin and 4°C.
The pH of formalin used for fixation had the most variable effects on IHC staining. Optimal staining was observed when infected bursal tissue was fixed at pH 7.0. At pH 2.0 minimal IHC staining of apoptotic bodies within the center of the follicles and very rare moderate staining of mononuclear cells was observed in all 5 bursas of this group. At pH 5.0 there was a complete loss of staining. All tissues fixed in formalin at pH 9.0 and 13.0 exhibited nonspecific staining of apoptotic bodies and cellular debris. Hematoxylin and eosin staining artifacts at pH 2.0 included tinctorial variations of epithelial cells, while at pH 5.0 there was cell shrinkage. At pH 9.0 there was streaming of epithelial nuclei and at pH 13.0 there were epithelial shrinkage and compressed fibroblasts.
IBDV RNA extraction and real time RT-PCR
The amount of extracted RNA extracted from tissue blocks ranged from 159.92-1,194.66 μg/ml. Specificity of rt-RT-PCR melting curve analysis was verified by running amplicons on a 1.5% agarose gel stained with ethidium bromide (Fig. 2). Detectable IBDV RNA was extracted from all 5 bursas stored in pH 7.0 as well as 1 of the 5 bursas stored in pH 13.0. All tissues fixed in pH 2.0, pH 5.0, or pH 9.0 had a histopathological lesion score of 4 but none yielded detectable IBDV RNA that was identifiable by rt-RT-PCR melting curve. (Table 3). Real-time RT-PCR melting curve identifiable RNA was extracted from bursas stored at temperatures 4°C and 25°C but not from 50°C (Table 3). Real time RT-PCR detectable IBDV RNA was extracted from bursas of Fabricius stored in 5% or 10% buffered formalin, but not from those tissues stored in 20% buffered formalin or 37% unbuffered formalin (Table 3). Storage time for up to 2 weeks had no effect on IBDV RNA extraction and identification, as melting curve peaks specific for IBDV were present at all fixation times studied. However, storage times longer than 24 hours increased the melting peak by 1°C; and after 2 weeks it was increased by 2°C. (Fig. 3).
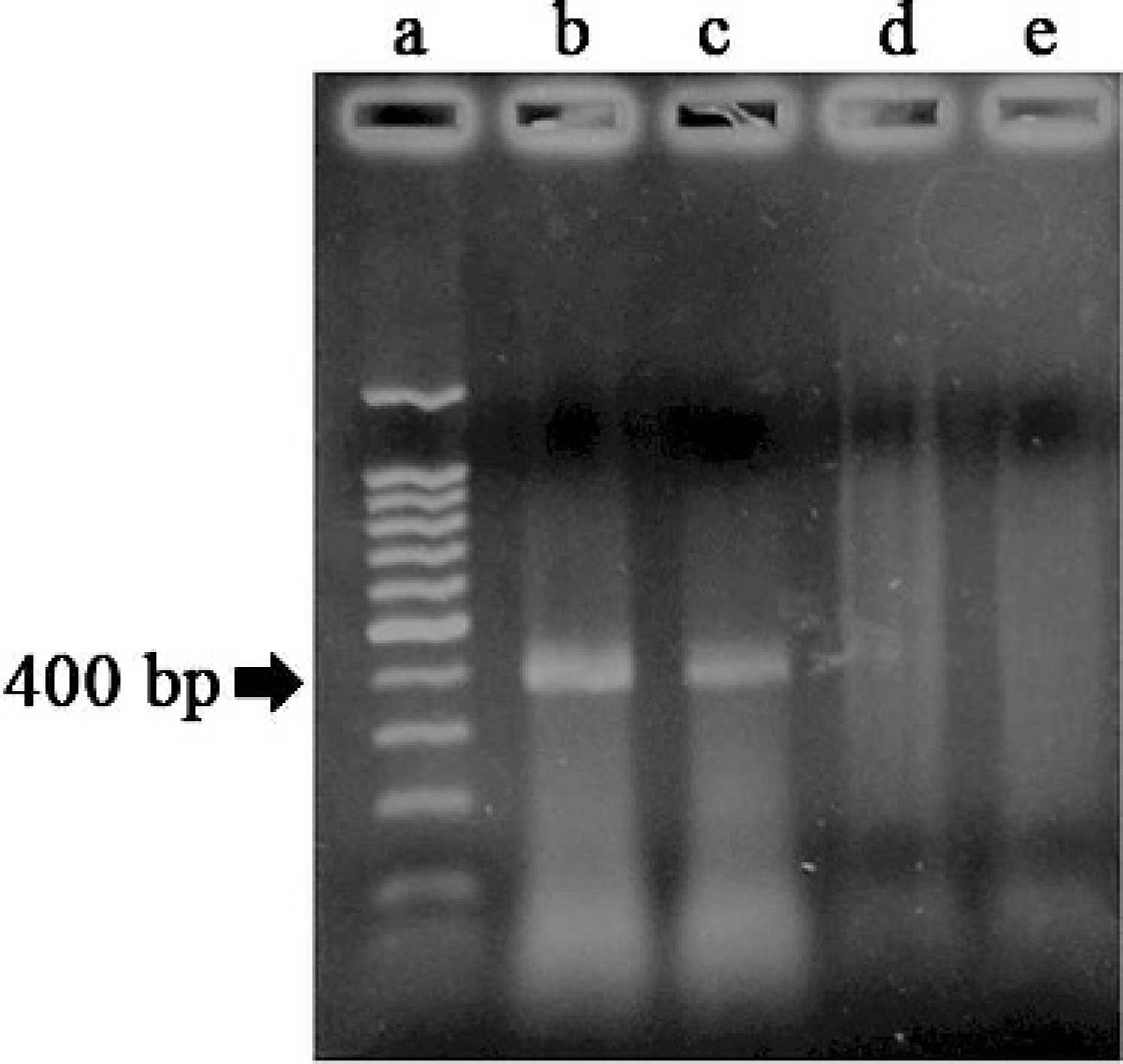
RT-PCR product of amplified VP2 fragment: lane a, molecular size marker; lane b, amplified VP2 fragment from IBDV control positive; lane c, amplified VP2 fragment from RNA extracted from a tissue block, lane d, negative RT- PCR control (primers with water); lane e, negative extraction control from RNA extracted from an uninfected tissue block.
Effect of fixation conditions on extraction of RNA from bursas of Fabricius from chickens infected with IBDV.
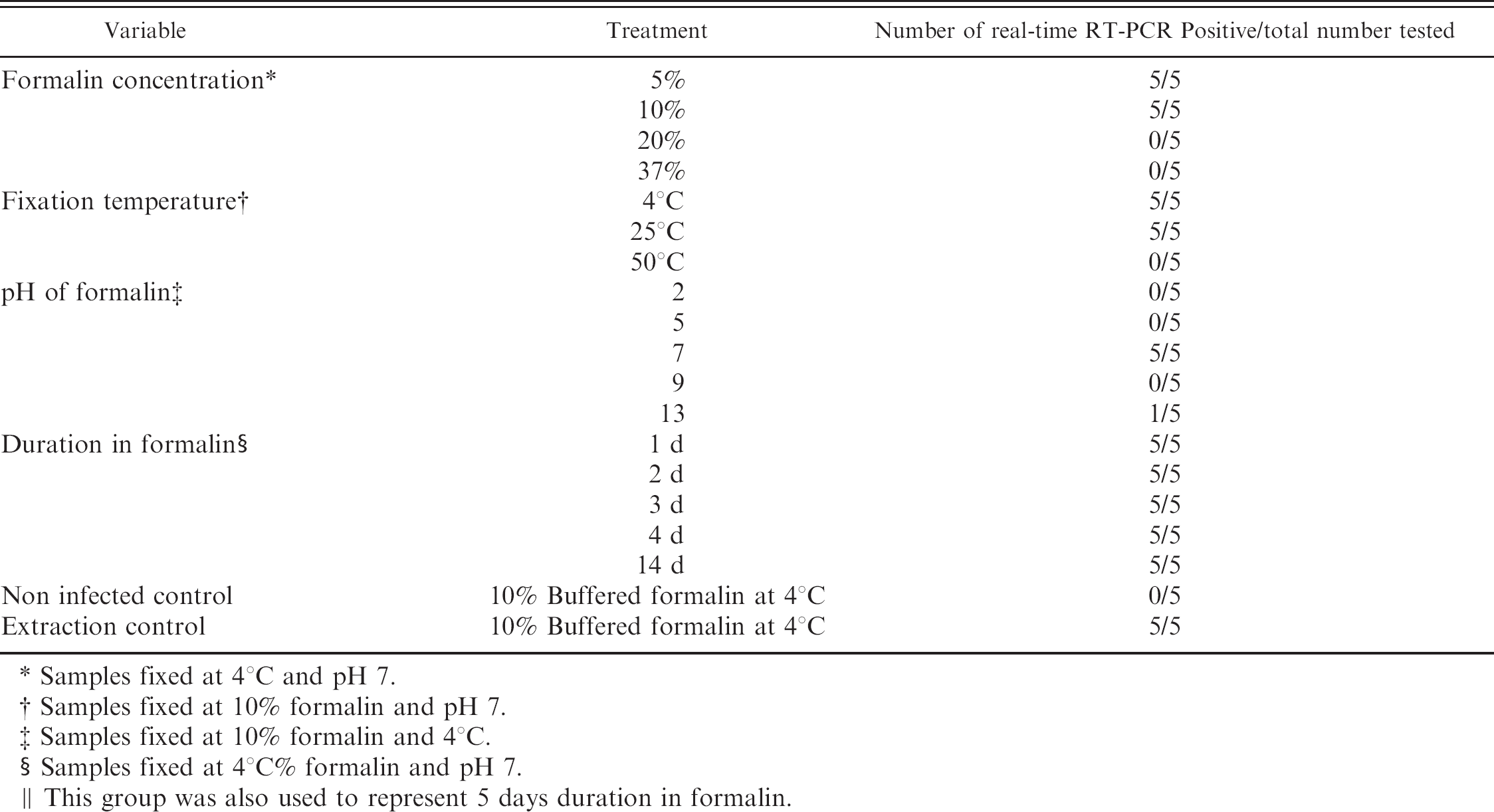
Samples fixed at 4°C and pH 7.
Samples fixed at 10% formalin and pH 7.
Samples fixed at 10% formalin and 4°C.
Samples fixed at 4°C% formalin and pH 7.
This group was also used to represent 5 days duration in formalin.
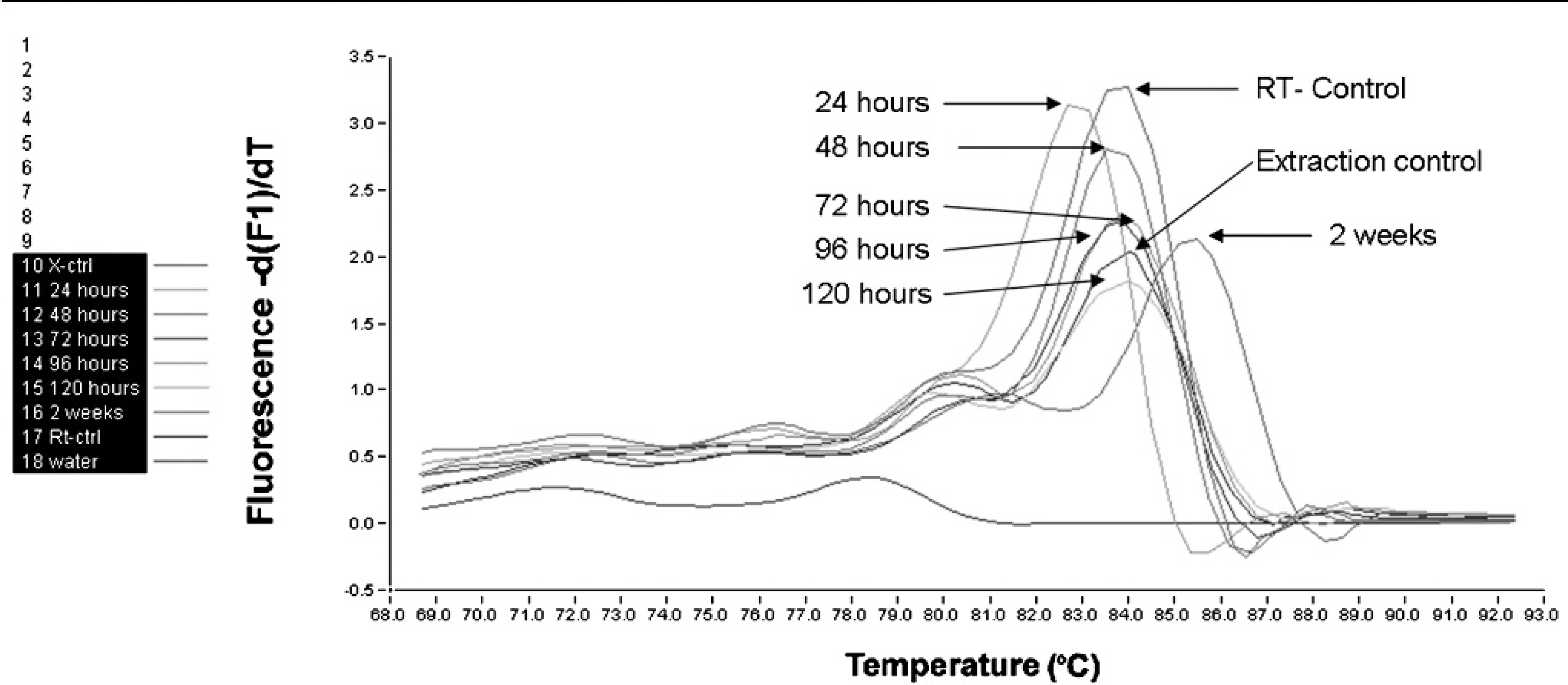
Real time RT-PCR melting curve analysis for IBDV amplicons extracted from tissues fixed in 10% buffered formalin at 4°C and submitted to different fixation durations (hours). Melting curve peak after 24 hours storage was 83°C, but increased to 84°C after 48–120 hours of storage, and after 2 weeks of storage, to 86°C, indicating changes in nucleotide sequences. Reverse transcriptase-control is the reverse transcriptase PCR control for a known IBDV and extraction control is a known paraffin embedded block with extractable IBDV genetic material.
Discussion
Formalin fixation is a common method for tissue preservation in preparation for histological evaluation of tissues. For immunohistochemistry, small (10 × 10 × 3 mm) tissue fragments fixed promptly in neutral buffered formalin for 6–24 hours generally had adequate cytological preservation and immunolocalization, with minimal antigen masking. 9 Variations in fixation times or conditions cause the majority of false negatives in immunohistochemistry. 7 It was reported that lack of consistency in formalin fixation protocols among laboratories, influences the outcome of staining in immunohistochemistry. 12 Formaldehyde fixes tissue by reacting primarily with basic amino acids to form cross-linked “methylene bridges.” Formalin fixation conditions have a direct effect on number of methylene bridges. 7 The number of methylene bridges and which amino groups are cross-linked in the tissue are important variables in the outcome of immunohistochemical staining, and could also be an important factor in the ability to extract rt-RT-PCR detectable RNA from tissue blocks.
Tissues fixed in 20% or 37% formalin undergo shrinkage 10 and may suffer from alteration of epitope conformation. The IBDV epitopes detected by the monoclonal antibodies used in this study are conformationally dependent; 5 thus fixation conditions such as formaldehyde concentration or pH that may cause changes in epitope conformation rendering them undetectable by these monoclonal antibodies. Maximum tissue fixation, i.e., cross-linking, occurs in the pH range of 4–5.5, 9 therefore, the presence of too many cross-links or cross-links of certain amino groups may cause antigen masking. 20 This may be the reason why there was minimal or no immunostaining in tissues fixed in pH ≤ 5.0. Inconclusive IHC staining at pH ≥ 9.0 may also be caused by tissue shrinkage or alteration of reactive epitopes.
Immunohistochemical staining was best in tissues fixed at 4°C. Increased temperature reduced specific IHC staining, but did not eliminate it. The reduction in staining may be caused by tissue shrinkage at temperatures higher than 4°C. 8 This result is in agreement with Key's findings; however the mechanism for this effect is unknown. 12
Real-time RT-PCR melting curve analysis of infected and noninfected controls validated the RNA extraction procedure. Real-time RT-PCR on known IBDV genomic material was performed as a positive rt-RT-PCR control and on DEPC-water as a negative rt-RT-PCR control. Extracting genomic RNA for identification of IBDV from bursas of Fabricius stored at acidic pH (pH 5.0 and pH 2.0) was unsuccessful in all 10 tissue blocks. Previous research which found DNA damage in cells at low pH, 11 may explain the lack of extracted genomic RNA in this experiment. Unbuffered formalin oxidizes to formic acid and an acidic environment causes degradation of nucleic acids because the β-glycosidic bonds in the purine bases are hydrolyzed at pH 4. 4 Loss of some amino group cross-links occurs at low pH and may result in loss of RNA from the fixed tissue. 3 Extraction of genomic RNA at a basic pH (pH 9.0 and 13.0) was also unsuccessful and the reason for this is unknown.
The thermodynamic stability of a nucleic acid duplex strongly depends on the denaturation temperature of the duplex at atmospheric pressure. 6 Denaturation of genomic RNA may have occurred when tissues were formalin fixed at 50°C for 5 days and might explain the absence of rt-RT-PCR detectable RNA from these blocks.
At formalin concentrations of 20% or 37%, the extraction of RNA from tissue blocks yielded no rt-RT-PCR detectable RNA. This may be owing to excessive crosslinking of amino groups resulting from the acidic nature of formalin at higher concentrations; indeed, maximum tissue fixation, i.e., cross-linking, occurs in the pH range of 4.0-5.5, 9 thus hindering genomic RNA extraction.
Fixation time had no effect on RNA extraction or subsequent identification (Fig. 3), but longer fixation times are not recommended because of the higher frequency of nonreproducible sequence alterations. This is demonstrated by the increase in melting-peak temperature over time. Formalin may cause cross-linking of cytosine nucleotides on either strand. As a result, in PCR the Taq-DNA polymerase fails to recognize the cytosine and incorporates an adenine in the place of a guanosine, creating an artificial C-T or G-A mutation. 22 There was a direct correlation between amount of total RNA extracted and favorable storage conditions. Tissue blocks stored in pH 7.0 and 5% or 10% formalin at 4°C or 25°C yielded the most RNA (972.31-1,194.66 μg/ml). Tissues stored in the other conditions studied yielded less amount of RNA (159.92-528.64 μg/ml). Amount of RNA yield may be affected by number of crosslinks of fixed tissue.
In summary, for optimal IBDV RNA extraction and sequence identification, tissues should be fixed in formalin at a pH of 7.0, concentration of 5% or 10% and stored at 4° C to 25°C for 7 days at most. Extremes in temperature, pH, or formalin concentration have a negative effect on tissues used for RNA extraction. Longer tissue storage times in formalin may cause inaccuracies in sequencing results. Optimal conditions for fixation of tissues infected with IBDV that will be used for IHC evaluation are 10% formalin concentration, pH 7.0, and temperature of 4°C. For practical purposes, fixation temperature of 4° C is recommended for both procedures. This can be achieved practically by placing formalin jars or whirlpaks in a cooler with a cardboard separating the ice from the samples.
Acknowledgements
The authors would like to thank Dr. Stanley Kleven and Linda Purvis for reviewing this manuscript.
Footnotes
a.
Sunrise Farms, Catskill, NY.
b.
IBD-Blen™ vaccine, Merial Select, Gainesville GA.
c.
Leica AutoStainer XL, Nussloch, Germany.
d.
Superfrost/Plus; Fisher Scientific Pittsburgh, PA.
e.
HemoDe Fisher Scientific, Pittsburgh, PA.
f.
Proteinase K: DAKO, Carpinteria, CA.
g.
Leica ST 5050, Nussloch, Germany.
h.
DAKO Envision System; DAKO Carpinteria, CA.
i.
Monoclonal antibody #HB9490, ATCC, Manassas, VA 20108.
j.
Trizol: Life Technologies, Inc., Gaithersburg, MD.
k.
Eppendorf biophotometer, Eppendorf AG, Hamburg, Germany.
I.
Uvette, Eppendorf AG, Hamburg, Germany.
m.
ROCHE Molecular Biochemicals, Indianapolis, IN.
