Abstract
Background:
In the elderly, diagnostic findings of pneumonia are often atypical. Computed tomography was recommended for the diagnosis of pneumonia in elderly patients. Recently, the usage of computed tomography as a screening tool for pneumonia in emergency departments has increased. Sarcopenia is defined as the loss of skeletal muscle mass and strength with aging. In this study, the association between sarcopenia and prognosis measured through computed tomography was evaluated compared to CURB-65.
Methods:
This study was conducted on patients diagnosed with pneumonia through computed tomography from 1 March 2018 to 31 March 2020. The paraspinous muscle size and attenuation were measured at a level located at the T12 pedicle level on axial computed tomography images. Paraspinous muscle size was presented as paraspinous muscle index. Differences in the prognostic performance among the paraspinous muscle size and attenuation, and CURB-65 were evaluated by the area under the receiver operating characteristic curve.
Results:
A total of 509 patients were included and 132 patients (25.9%) were admitted to the ICU, and 58 patients (11.4%) died in hospital. Paraspinous muscle index was the significant factor for predicting in-hospital mortality and ICU admission. The area under the receiver operating characteristic value of paraspinous muscle index for prediction of mortality was 0.738 and CURB-65 was 0.707. The area under the receiver operating characteristic of paraspinous muscle index and CURB-65 for predicting ICU admission were 0.766 and 0.704, respectively.
Conclusion:
As a method of measuring sarcopenia, paraspinous muscle index was superior to CURB-65 in elderly pneumonia patients. The use of computed tomography in predicting prognosis for elderly pneumonia patients will ease the economic burden.
Introduction
Pneumonia in elderly patients is associated with high mortality and morbidity due to infection It caused 6.8 million admissions and 1.2 million deaths among adults aged ⩾65 years in 2015.1,2 Elderly patients usually have comorbidities such as cancer, diabetes, chronic heart failure, chronic renal failure, and chronic respiratory disorders, which can lead to poor outcomes.3,4 Thus, these infections in the elderly constitute a socioeconomic burden for individual patients and healthcare systems. The diagnosis of pneumonia is based on the presence of clinical respiratory symptoms such as cough, sputum, dyspnea, fever, and new infiltration observed on chest radiography.5,6 In the elderly, the clinical symptoms of pneumonia are often atypical, such as masked fever due to a low-temperature threshold.7,8 Moreover, chest radiography is often difficult to interpret accurately, and abnormalities may be absent or nonspecific in elderly patients.9,10 Recently, some studies have shown the superior performance of computed tomography (CT) compared to chest radiography for the diagnosis of pneumonia and suggest that CT should be recommended for the diagnosis of pneumonia in elderly patients.11,12 Coronavirus disease 2019 (COVID-19), which was first reported in Wuhan City, has rapidly spread all over the world and has become a pandemic. 13 In recent studies, chest CT is recommended for COVID-19 diagnosis and has shown high sensitivity, 14 thereby leading to its increased use as a screening tool for pneumonia in emergency departments (EDs). 15
Decrease in respiratory muscle strength, poor cough reflex, and impaired swallowing functions contribute to pneumonia. 16 The diaphragm is the respiratory muscle that is primarily responsible for respiratory function. 17 Diaphragmatic atrophy can cause difficulty in breathing and weaken the ability to cough to expectorate sputum, which can lead to worsening of pneumonia. A previous study had reported that respiratory muscle training improved the coughing reflex and reduced the incidence of pneumonia in acute stroke patients. 18 The reduction in the strength of swallowing muscles is related to dysphagia. 19 Therefore, for effective treatment and prognostic prediction of pneumonia, muscle weakness in elderly patients must be considered. Some studies have demonstrated that sarcopenia is associated with respiratory muscle weakness and dysphagia in older people.20–22
Sarcopenia is defined as the loss of skeletal muscle mass and strength with aging. 23 Age-related loss of skeletal muscle and muscle strength causes a decrease in functional ability, which leads to a risk of falling and fracture, infections, and metabolic disorders, and reduces the quality of life and survival rate.23,24 Various methods are used for the diagnosis of sarcopenia, including dual-energy X-ray absorptiometry (DXA), bioelectrical impedance analysis (BIA), magnetic resonance imaging (MRI), and CT.
The use of CT has increased in EDs worldwide, 25 which has led to its increased contribution to the diagnosis of pneumonia. In this study, we examined the association of the assessment of sarcopenia via diagnostic CT with the prognosis of pneumonia patients. The assessment of sarcopenia in chest CT, which is used to diagnose pneumonia in elderly patients, has provided physicians with another prognostic tool. We further evaluated its superiority over CURB-65, a standard tool previously used to predict the prognosis of pneumonia.
Methods
Study design and setting
This study was conducted in the Department of Emergency Medicine at a tertiary-care university hospital with 50,000 annual emergency visits. This single-center retrospective study used the electronic medical records (EMRs) of patients who visited the ED. The study was approved by the institutional review board of our institution, and the requirement for written informed consent was waived.
Study population and data collection
Between 1 March 2018 and 31 March 2020, we enrolled patients aged 65 years and over who visited the ED and were diagnosed with pneumonia through a CT scan. The exclusion criteria included metallic prostheses, which can lead to the metal artifact on CT and incomplete EMR data owing to patients leaving against medical advice or being transferred to another hospital. Board-certified radiologists diagnosed pneumonia with images stored in a picture archiving and communication system (PACS), and one board-certified emergency physician retrieved data from the EMRs of the diagnosed patients. Patients’ demographics, initial vital signs, results of laboratory tests (including the complete blood count (CBC) and blood chemistry tests), and clinical findings were retrospectively collected.
Measurement of muscle size and attenuation
Chest CT scans were performed using a 64-slice, multidetector CT scanner (Somatom Perspective ONE Dynamic Volume CT, Siemens Healthineers Corporation, Erlangen, Germany). Images stored in the PACS software were obtained and used to independently measure paraspinous muscles by two board-certified emergency physicians who were unaware of the patients’ baseline characteristics or medical outcomes. Paraspinous muscle size and attenuation (measured in Hounsfield units (HU) 26 ) were measured using a freehand technique based on measuring regions of interest at the T12 pedicle process level (Figure 1), similar to other studies for sarcopenia.27,28 The measurements were performed bilaterally, and the average of both sides was calculated. Paraspinous muscle size was presented as paraspinous muscle index (PMI)—the total paraspinous muscle area (mm2) divided by the square of the patient’s height (m2)—which helps to normalize the measured paraspinous muscle area for the height of the patient.
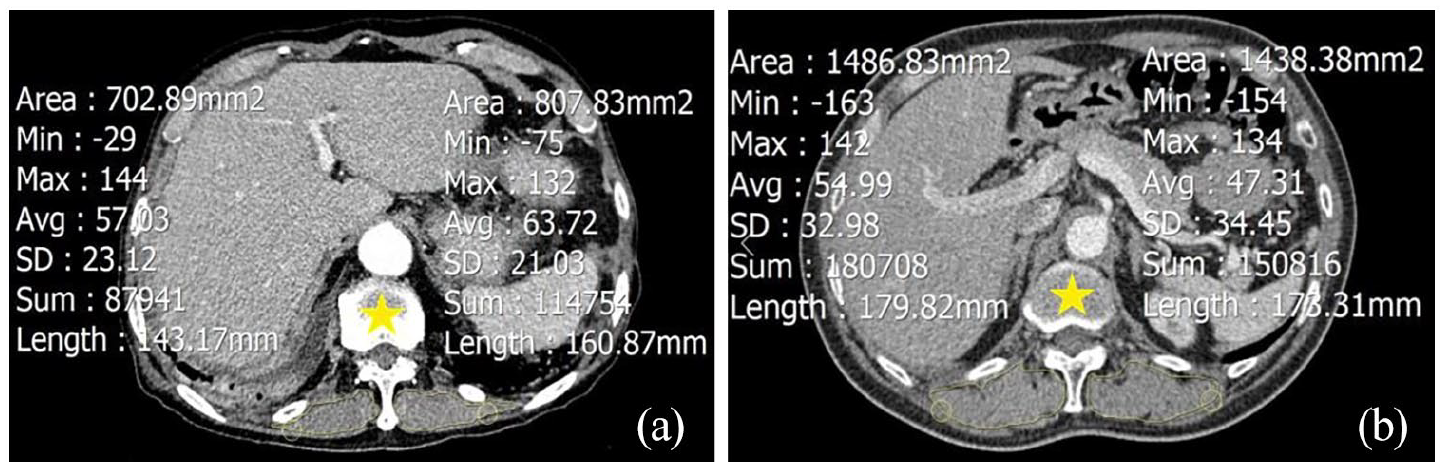
Two different patients. Axial chest CT images outlining the paraspinous muscles bilaterally were obtained at L12 level (yellow stars). (a) 76-year-old man, image shows a cross-sectional area (yellow outline) measuring the left and right sides, which were 807.83 and 702.89 mm2, and mean muscle attenuation measuring the left and right sides, which were 63.72 HU (SD: 21.03) and 57.03 HU (SD: 23.12), respectively. (b) 67-year-old man, image shows a cross-sectional area (yellow outline) measuring the left and right sides, which were 1438.38 and 1486.83 mm2, and mean muscle attenuation measuring the left and right sides, which were 47.31 HU (SD: 34.45) and 54.99 HU (SD: 32.98), respectively.
CURB-65 calculation
CURB-65 score is based on confusion, urea (>7 mmol/L), respiratory rate (⩾30/min), low systolic (<90 mmHg) or diastolic (⩽60 mmHg) blood pressure, and age (⩾65 years). The state of consciousness, blood pressure, respiratory rate, and serum blood urea nitrogen (BUN) levels at the time of the ED visits were used for the calculation of the CURB-65 score. The lowest score for all target patients was 1 point because this study was targeted at patients over the age of 65 years.
Outcomes
The primary outcome of the study was to assess whether the paraspinous muscle could be used to predict mortality and the need for intensive care in pneumonia patients. The secondary outcome was to compare the predictive performance of the paraspinous muscle to that of the reference diagnosis tool, CURB-65.
Statistical analyses
Among baseline characteristics, continuous variables were presented as means with standard deviation, whereas categorical variables were presented as counts with percentage. Statistical analysis was performed using the independent t-test or the Mann–Whitney U-test for continuous variables and Pearson’s chi-square test or Fisher’s exact test for categorical variables. To determine independent factors affecting the patient’s prognosis, we performed multivariate logistic regression of statistically significant variables by univariate analysis. Multivariate analysis yielded the adjusted odds ratios (ORs) and 95% confidence intervals (CIs).
The predictive accuracy was determined by using the area under the receiver operating characteristic (AUROC) curves. An optimal cutoff value was presented as the point at which the value of “sensitivity + specificity-1” was maximized (Youden’s index). 29
Intraclass correlation coefficients (ICCs) were used to determine the reliability between two reviewers’ measurements. ICCs between 0 and 0.20 were considered poor agreement, 0.21–0.40 fair agreement, 0.41–0.60 moderate agreement, 0.61–0.80 good agreement, and 0.81–1.00 excellent agreement. 30 All analyses were performed using SPSS version 26.0 (IBM Corp., Armonk, NY). The AUROC curve analysis was performed using the DeLong et al. 31 method with the MedCalc version 19.4.1 (MedCalc Software Ltd, Ostend, Belgium). The significance level was set at p < 0.05.
Results
Patients’ characteristics
A total of 509 patients were included in this study (Table 1). The mean age of the enrolled patients was 78.9 ± 7.3 years; 55.5% were male, and there were 63 patients (12.4%) with altered mental status. The total number of admitted patients was 389 (76.4%) of which 132 (25.9%) were admitted to the intensive care unit (ICU), and 58 (11.4%) patients died during the clinical study.
Baseline characteristics of patients (total n = 509).
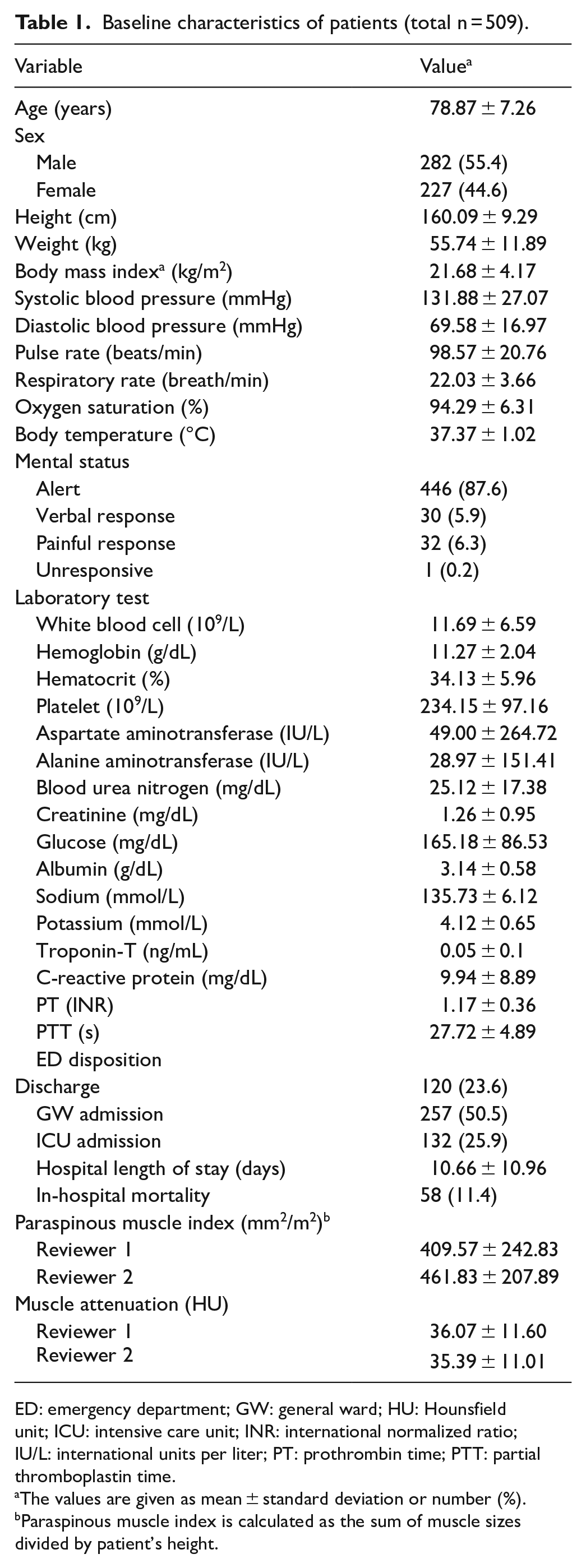
ED: emergency department; GW: general ward; HU: Hounsfield unit; ICU: intensive care unit; INR: international normalized ratio; IU/L: international units per liter; PT: prothrombin time; PTT: partial thromboplastin time.
The values are given as mean ± standard deviation or number (%).
Paraspinous muscle index is calculated as the sum of muscle sizes divided by patient’s height.
Measurement of paraspinous muscle
The paraspinous muscle size and attenuation were measured twice in all 509 patients by two reviewers. The left, right, and average (mm2) muscle sizes measured by reviewer 1 were 636.6, 636.5, and 636.6, respectively, and the mean attenuation (HU) values were 36.0, 36.1, and 36.1, respectively. The left, right, and average (mm2) muscle sizes measured by reviewer 2 were 598.0, 592.3, and 595.1, respectively, and the mean attenuation (HU) values were 35.3, 35.4, and 35.4, respectively. The PMI (mm2/m2) calculated for both reviewers’ measurements were 409.6 and 461.8. The reliability for muscle size and attenuation between reviewers 1 and 2 was excellent. The ICCs for the muscle size measured by the two reviewers were 0.877 [95% CI: 0.854‒0.897] and 0.886 [95% CI: 0.864‒0.904] for the left and right muscles, respectively. The ICCs for the muscle attenuation measured by the two reviewers were 0.968 [95% CI: 0.962‒0.973] and 0.966 [95% CI: 0.960‒0.971] for the left and right sides, respectively.
Clinical factors associated with mortality of pneumonia patients
Clinical factors affecting the in-hospital mortality of pneumonia patients are shown in Table 2. The clinical and laboratory data of survival groups and mortality groups were compared by univariate analysis. Among the patients’ general characteristics and vital signs, age, systolic blood pressure, respiratory pressure, oxygen saturation, and altered mental status were independent risk factors for mortality. Among laboratory tests, white blood cell (WBC), hemoglobin (Hb), hematocrit, BUN, creatinine, albumin, sodium, Troponin, C-reactive protein (CRP), prothrombin time (PT), and partial thromboplastin time (PTT) were independent risk factors for mortality. Among the measured paraspinous muscle, only PMI, not attenuation, showed a significantly lower in the mortality group. Based on these factors, multivariate regression analysis showed that RR, creatinine, albumin, troponin, CRP, PT, and PMI were significant predictors of mortality.
Logistic analysis of mortality predictors.
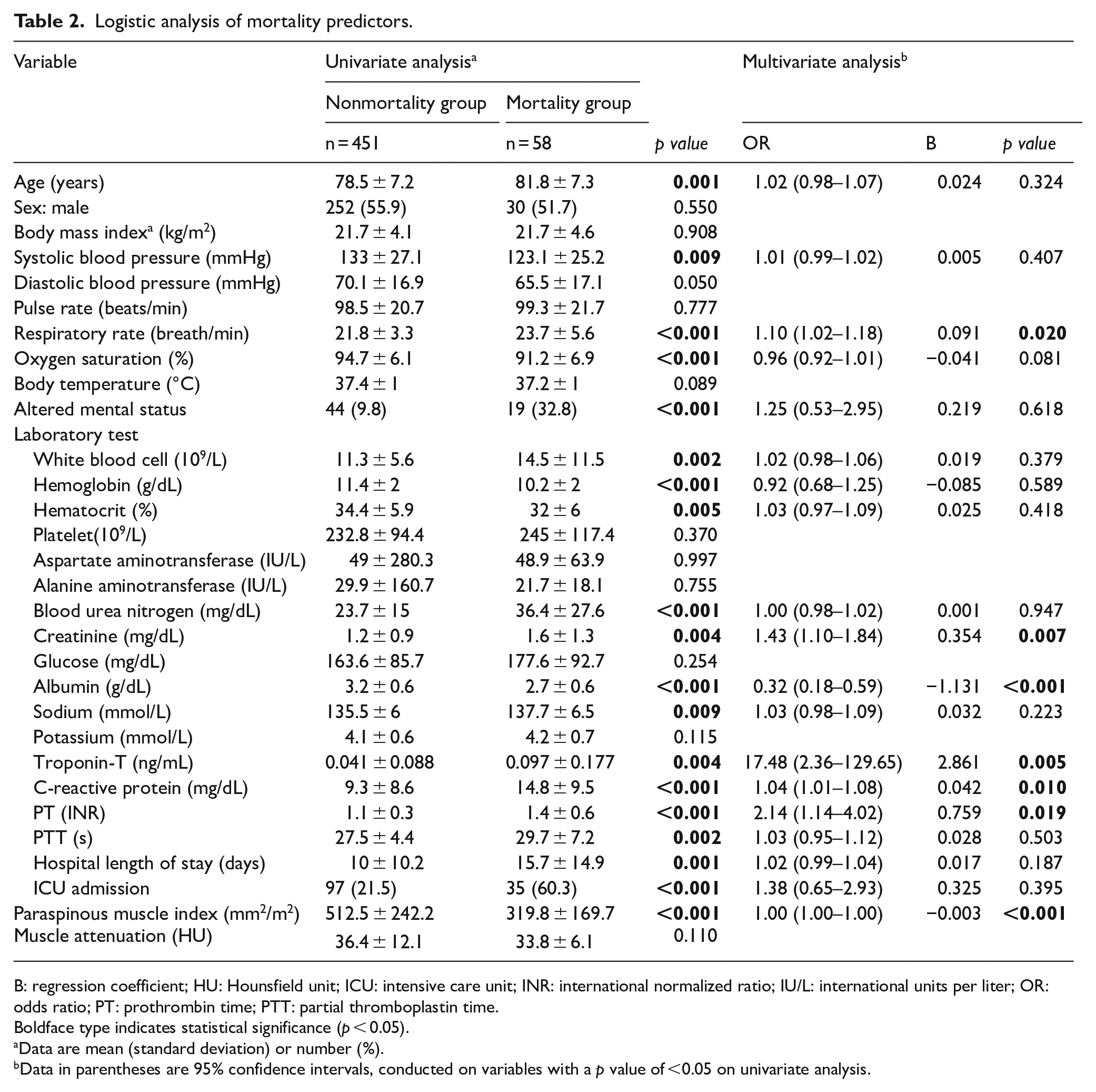
B: regression coefficient; HU: Hounsfield unit; ICU: intensive care unit; INR: international normalized ratio; IU/L: international units per liter; OR: odds ratio; PT: prothrombin time; PTT: partial thromboplastin time.
Boldface type indicates statistical significance (p < 0.05).
Data are mean (standard deviation) or number (%).
Data in parentheses are 95% confidence intervals, conducted on variables with a p value of <0.05 on univariate analysis.
Clinical factors associated with ICU admission of pneumonia patients
The associations between the ICU admission and clinical and laboratory data are presented in Table 3. Univariate analysis revealed that systolic and diastolic blood pressures, RR, oxygen saturation, body temperature, and altered mental status were significant risk factors for ICU admission. Among laboratory tests, WBC, Hb, hematocrit, aspartate aminotransferase, BUN, creatinine, albumin, troponin, CRP, and PT were independent risk factors for ICU admission. Among the paraspinous muscle measurements, only PMI was an important factor that was found to be lower in the ICU admission group. Multivariate logistic regression analysis was performed for these factors. As a result, oxygen saturation, body temperature, altered mental status, Hb, hematocrit, PT, and PMI were found to be significantly associated with ICU admission.
Logistic analysis of ICU admission predictors.
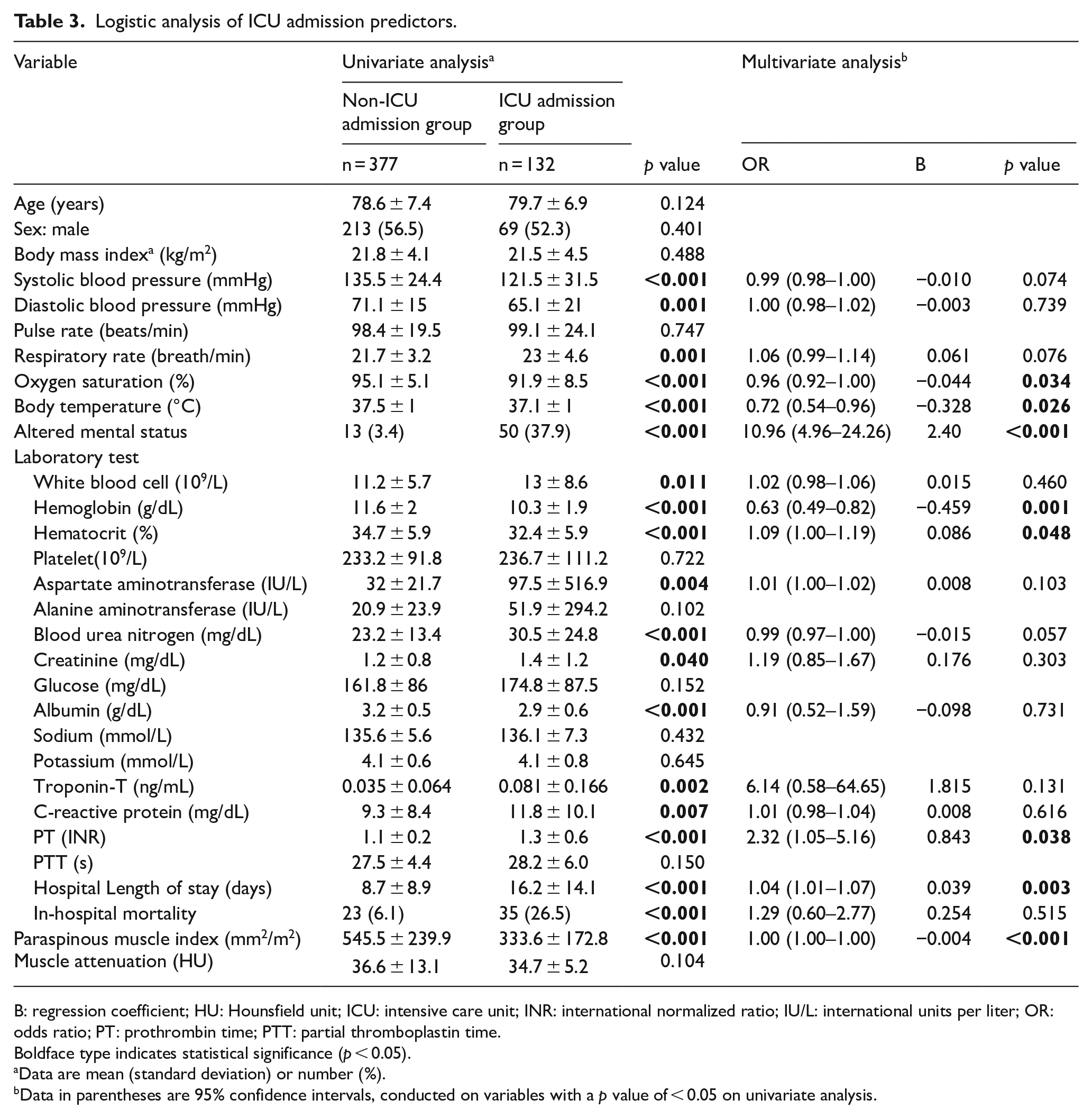
B: regression coefficient; HU: Hounsfield unit; ICU: intensive care unit; INR: international normalized ratio; IU/L: international units per liter; OR: odds ratio; PT: prothrombin time; PTT: partial thromboplastin time.
Boldface type indicates statistical significance (p < 0.05).
Data are mean (standard deviation) or number (%).
Data in parentheses are 95% confidence intervals, conducted on variables with a p value of < 0.05 on univariate analysis.
Comparison of predictive performance for in-hospital mortality and ICU admission
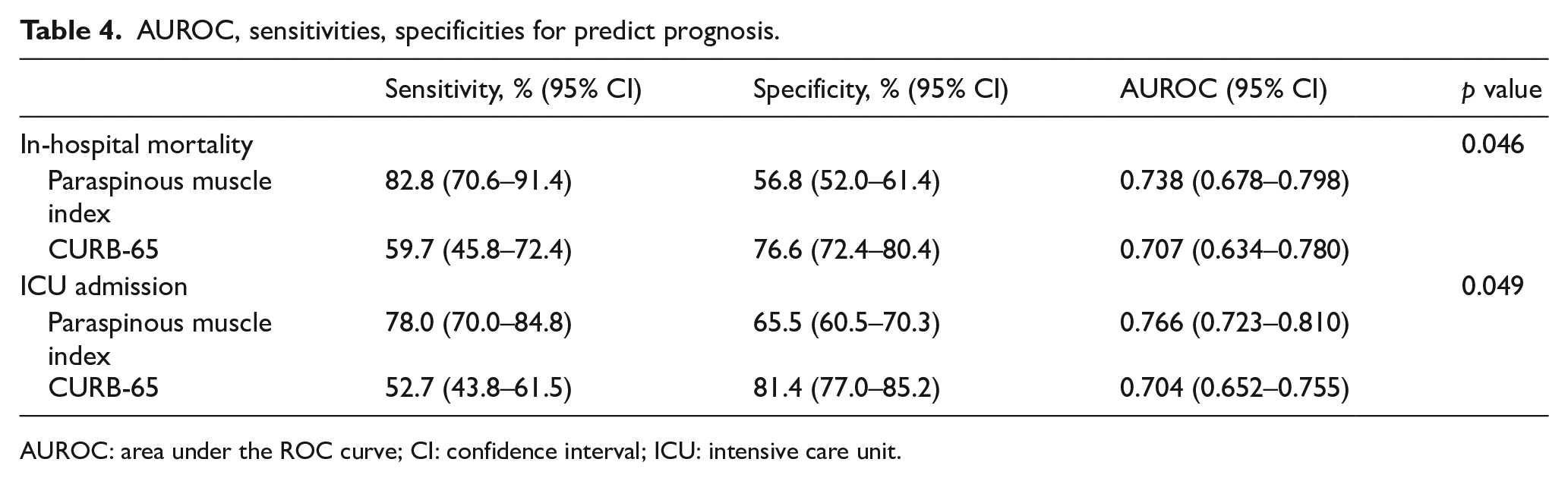
Multivariate regression analysis revealed PMI to be an important factor for predicting in-hospital mortality and ICU admission. The AUROC curves for the prediction of mortality are shown in Figure 2. The AUROC value of PMI for the prediction of mortality was 0.738 (95% CI: 0.678–0.798), and the cutoff value was 464.40 with 82.8% sensitivity and 56.8% specificity. The AUROC value of CURB-65 for the prediction of mortality was 0.707 (95% CI: 0.634–0.780), and the cutoff value was 3 with 59.7% sensitivity and 76.6% specificity (Table 4). When compared between the two criteria, they were statistically significantly different (p value: 0.046).
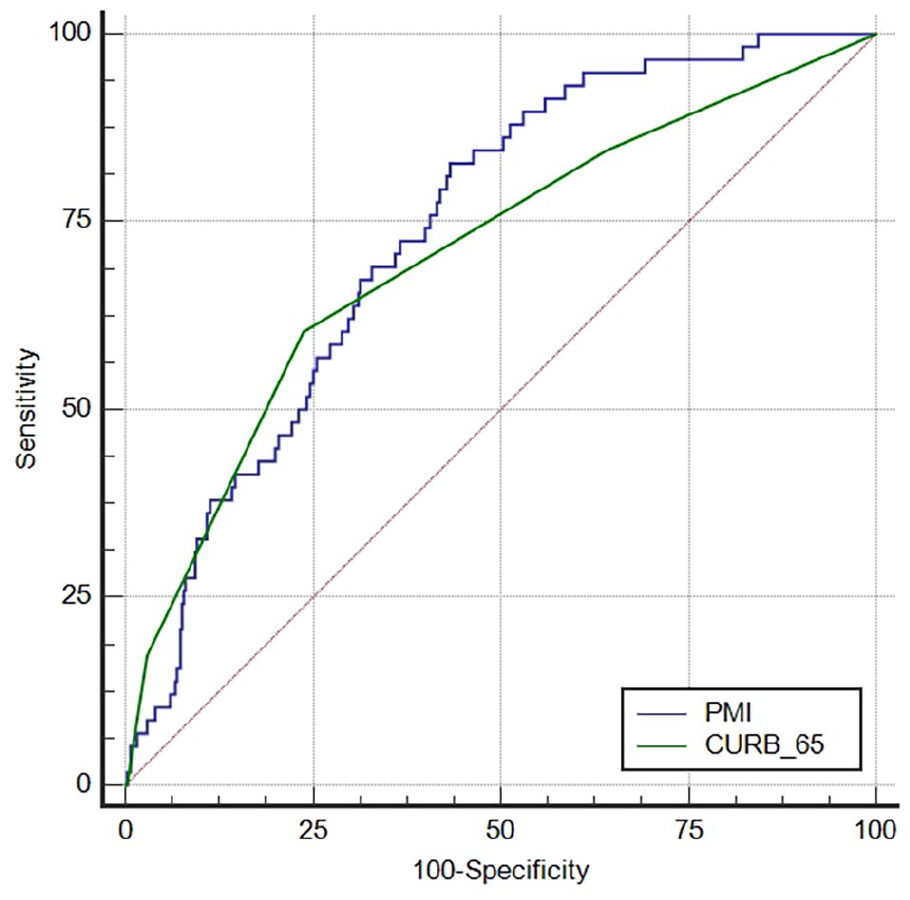
Comparison of the AUROCs for the prediction of mortality.
AUROC, sensitivities, specificities for predict prognosis.
AUROC: area under the ROC curve; CI: confidence interval; ICU: intensive care unit.
The AUROC curves of PMI and CURB-65 for the prediction of ICU admission are shown in Figure 3. The AUROC values of PMI and CURB-65 for predicting ICU admission were 0.766 (95% CI: 0.723‒0.810) and 0.704 (95% CI: 0.652‒0.755). For PMI, the cutoff value was 456.71 with 78.0% sensitivity and 65.5% specificity. For CURB-65, the cutoff value was 3 with 52.7% sensitivity and 81.4% specificity (Table 4). The comparison between each criteria showed statistically significant differences (p value: 0.049).
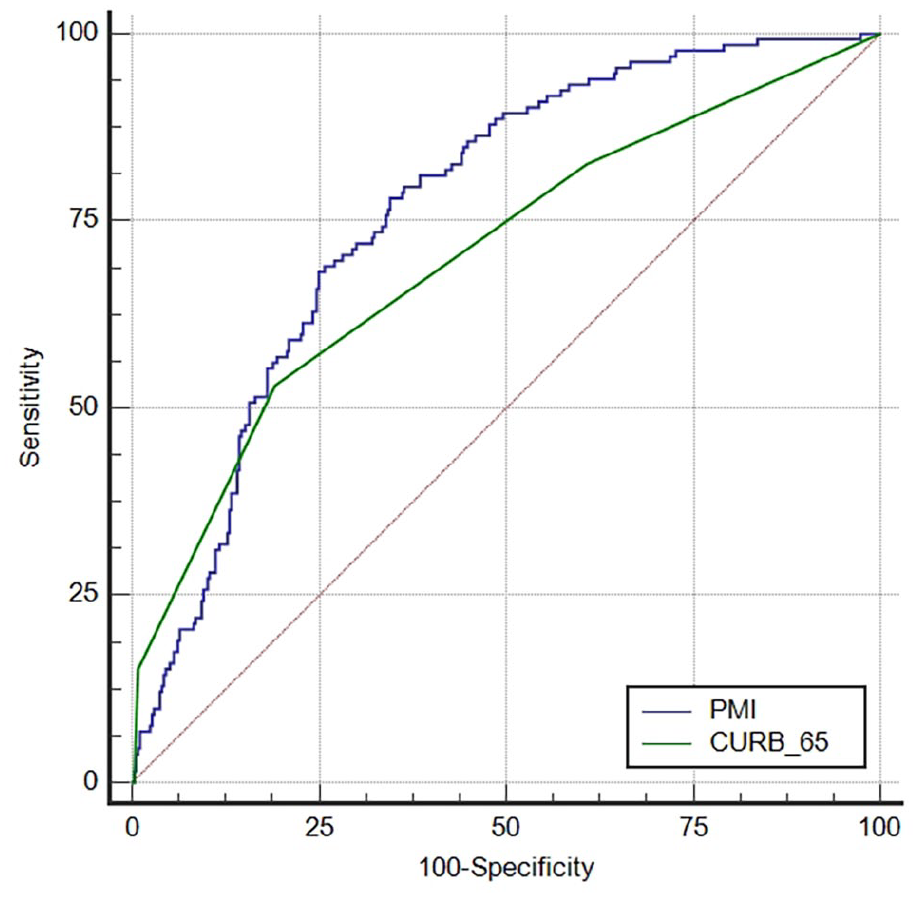
Comparison of the AUROCs for the prediction of ICU admission.
Discussion
In this study, the PMI showed superiority in prognostic prediction compared to CURB-65, a reference diagnostic tool of pneumonia. An important initial assessment of pneumonia is to predict the severity of the disease for providing appropriate treatment options. The pneumonia severity index (PSI), introduced in 1997 to classify the severity of pneumonia, uses 20 variables to evaluate the severity of pneumonia. 32 Although the PSI has been widely used, it uses too many variables to be adopted in the ED. Thus, CURB-65 was proposed by the British Thoracic Society for the prognostic assessment of community-acquired pneumonia. 33 CURB-65 is more useful than PSI because it uses only five variables and shows no difference in its predictive performance in pneumonia patients. 34 However, in elderly patients, as age-related variables are all applicable over the age of 65 years, they will eventually be reduced to four variables, and among these variables, confused mental status is likely to be subjectively judged by physicians. In addition, one study found that the performance of CURB-65 for the prediction of mortality deteriorated with increasing age. Notably, the death of patients with low scores increased with age. 35
In 2016, sarcopenia was adopted in an International Classification of Diseases-10 code as a muscle disease. 36 Various methods are available for the diagnosis of sarcopenia, including different methods for the measurement of muscle mass such as bioelectrical impedance analysis, dual-energy X-ray absorptiometry, MRI, and CT. BIA devices are economical and portable; however, their accuracy depends on the patient’s hydration status and the types of equations used for appendicular skeletal mass estimation.37,38 DXA devices are not portable, making it difficult to measure muscle mass in patients who receive oxygen or who require clinical monitoring. CT scan is recommended for a more accurate diagnosis than chest radiography in elderly pneumonia patients. 11 Measuring muscle mass and strength through CT taken for diagnosis of pneumonia may help predict sarcopenia at no additional cost and time.
In this study, only PMI, not attenuation, was found to be associated with prognosis in elderly pneumonia patients. However, this does not mean that muscle mass alone is related to sarcopenia. Another study conducted on hip fracture patients showed that only muscle attenuation, not PMI, was associated with the patient’s prognosis. 39 There are many definitions and measurements of sarcopenia; however, in infectious diseases such as pneumonia, muscle atrophy may play a more important role. Indeed, PMI was shown to be a prognostic factor for the patients enrolled in this study. Aged patients often have repeated inflammation—chronic and acute inflammation. 40 Inflammation causes the production of proinflammatory cytokines, which in turn induce muscle atrophy by proteolysis. 41 This process consists of two steps. First, the initial myofibrillar protein is cleaved by calpains and/or capase-3, followed by further degradation through the ubiquitin-proteasome system.41,42 The second step is autophagy, which causes muscle proteolysis during muscle atrophy in sepsis or hypoxia.43,44 Sarcopenia is originally defined as the loss of skeletal muscle mass and strength with aging. Some studies have shown that levels of inflammatory cytokines are negatively correlated to muscle mass and strength.45,46 Recently, sarcopenia has been related to high serum inflammation parameters, suggesting that its cause is inflammation. 47
This study has some limitations. First, this retrospective study was performed in a single-center institution and was not a multicenter study. This limits the degree to which results can be generalized. Multicenter and prospective studies would be required to generalize the results of this study to patients with gastrointestinal bleeding. Second, we only included patients with pneumonia diagnosed by CT, which may have caused a potential selection bias. Finally, muscle size and attenuation were measured manually, giving rise to errors or incorrect values. However, we tried to minimize this bias by ensuring independent measurements by two researchers.
In this study, low PMI was associated with poor outcomes in elderly pneumonia patients. PMI outperformed CURB-65, a widely used prediction tool. In an aging society, the use of diagnostic CT for predicting prognosis in elderly pneumonia patients will ease the economic burden due to this disease. Moreover, measuring muscle size using PACS software does not require much effort and does not incur any additional costs.
Footnotes
Acknowledgements
We would like to acknowledge our emergency department staff for their support.
Author contributions
S.H.L. contributed to conception and design. S.H.L. and S.J.B. contributed to acquisition, analysis, and interpretation of data. S.J.B. and K.K. contributed to drafting the article for intellectual content. S.J.Y. contributed to statistical analysis. All authors reviewed, revised, and approved the article for submission. S.H.L. contributed to study supervision.
Availability of data and materials
The data sets used in this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
This study was retrospective using patient medical data, so the need for patient consent was waived.
Ethical approval and ethical number provided by the review board
This study was approved by the institutional review board of Ewha Womans University Mokdong Hospital, and the requirement for written informed consent was waived (IRB No 2021-02-036).
