Abstract
Ventilator-associated pneumonia substantially increases mortality and imposes a heavy burden on healthcare resources. Patients undergoing endotracheal intubation and mechanical ventilation in emergency departments face a particularly high risk of ventilator-associated pneumonia. Compared with healthcare providers in intensive care units, those in emergency departments often lack sufficient awareness of timely and systematic ventilator-associated pneumonia prevention strategies, and existing intensive care unit-based care bundles are rarely tailored to emergency department-specific constraints. This gap leads to suboptimal implementation of ventilator-associated pneumonia preventive measures and increases the risk of ventilator-associated pneumonia. In this narrative review, we synthesize the current evidence on ventilator-associated pneumonia prevention and assess the feasibility of care bundles in resource-constrained emergency department settings. We propose the use of position, hand hygiene, airway, sedation, and equipment—a structured, multicomponent ventilator-associated pneumonia prevention strategy—in emergency departments with limited resources, supported by enabling strategies such as electronic checklists, multidisciplinary collaboration, and training to ensure adherence. Moving forward, efforts should focus on establishing consensus on emergency department-specific ventilator-associated pneumonia prevention protocols and optimizing nursing resource allocation to support implementation.
Keywords
Introduction
Mechanical ventilation (MV) is a life-sustaining intervention for patients with acute respiratory failure in the emergency department (ED) and intensive care unit (ICU). Inadequate or suboptimal management can lead to serious complications. Among these, ventilator-associated pneumonia (VAP) is notable because of its high prevalence and clinical burden. VAP—one of the most common hospital-acquired infections in patients receiving MV—is defined as pneumonia that arises ≥ 48 hours after intubation in patients without prior infection at the time of the procedure. 1 VAP development is closely linked to prolonged ventilator dependence and the use of invasive airway devices. Key contributing factors include microaspiration of contaminated secretions, immunocompromised hosts, and breaches in infection-control practices during airway management, such as suboptimal hand hygiene, inadequate sterilization of equipment, or improper suctioning techniques. 2 Surveys report that the mortality rate attributable to VAP is 8% in developed countries and up to 15.5% in developing countries, exerting considerable demand on acute-care beds, which constitute a key contributor to increased healthcare costs.1,3
VAP-associated risk begins at intubation and increases with the duration of ventilation. 4 Extended ED stays exacerbate exposure to modifiable VAP risks, such as suboptimal oral care and poor positioning management. Each additional hour of MV in resource-constrained ED settings may increase microaspiration and pathogen colonization, thereby accelerating the progression toward VAP. Therefore, initiating rigorous VAP prevention protocols immediately after ED intubation is critical. 5
Ventilator care bundles (VCBs) integrate evidence-based practices and effectively reduce the incidence of VAP in the ICU.6,7 However, unique challenges and complex resource constraints limit their full implementation in the ED. One key issue is human resource planning; determining adequate nurse staffing for the ED remains difficult, as multiple factors influence the ideal nurse-to-patient ratios. No existing model comprehensively incorporates all relevant variables to define an ED-specific staffing plan. Consequently, under prevailing staff shortages and a lack of dedicated equipment, nurses have less time and fewer resources for essential VAP prevention measures, such as frequent oral care and standardized airway management. 8 ICU settings in China report shortages of essential airway management devices (e.g. heated humidifiers and specialized subglottic suction cannulas), 9 which may suggest an even more severe situation in EDs, although it remains unassessed owing to scarce relevant surveys. EDs typically face more pronounced equipment constraints than ICUs due to insufficient availability, use of non-standard devices, rapid turnover, and cleaning and disinfection protocols that are often compromised under time pressures. 9 Standardized, specialized training on VAP prevention is notably less accessible to healthcare workers (HCWs) in the ED than in the ICU. The knowledge levels of ED nursing staff clearly reflect this gap in available training resources. For instance, a survey of 53 ED nurses revealed that none could correctly answer all questions, indicating significant deficiencies in their understanding of comprehensive VAP prevention strategies. 10 HCWs in high-volume centers report excessive workloads and emphasize that systemic changes are crucial to improve both working conditions and patient care. This is particularly true for EDs, where rapid patient turnover and unpredictable clinical evolution impede the reliable execution of complex multidisciplinary prevention protocols.11,12
Emergency nurses play a pivotal role in early VAP prevention by performing critical tasks such as assisting with intubation, patient positioning, oral care, and suctioning. 8 These procedures require strict adherence to best practices to minimize the risk of pathogen entry and aspiration. However, compliance with VAP prevention guidelines—known to reduce infection rates and improve patient outcomes—remains suboptimal. Nurses’ knowledge and adherence to VAP prevention measures are consistently reported as low to moderate. Even though VCBs are well-established and effective in the ICU, their implementation in the ED is often inadequate. 13 This gap may be attributed to multiple factors, including insufficient knowledge, the absence of structured VAP prevention protocols, inadequate feedback, lack of dedicated infection prevention teams, and limited training opportunities. 14 Moreover, the complexity of comprehensive ICU bundles often renders them misaligned with the clinical realities of the ED. This mismatch creates an urgent need for a dedicated, simplified VCB tailored to the ED, prioritizing the most feasible and high-impact interventions during the initial hours of MV.
This study aims to provide HCWs in resource-limited EDs with targeted and feasible VAP prevention strategies and analyze barriers to implementing these preventive measures in these settings.
Methods
In this narrative literature review, we analyzed the available evidence regarding VAP prevention. The search was conducted from November 2024 to February 2025 using PubMed, Cochrane Library, Medline Ovid, Web of Science Core Collection, Scopus, and China National Knowledge Infrastructure databases, without restrictions on publication date. The initial search employed medical subject headings terms including “ventilator-associated pneumonia AND prevention.” Based on the identified prevention strategies, we performed targeted searches to evaluate the impact of individual interventions on VAP outcomes.
We included publications in English and Chinese, guidelines, empirical studies, and review articles to contextualize the findings. After removing duplicates, two researchers independently screened the abstracts of the remaining articles. To further contextualize and interpret the findings, we also selected relevant references—including those identified through snowball sampling—that focused on fundamental evidence-based nursing practices for VAP prevention. This review was guided by the principles of the Scale for the Assessment of Narrative Review Articles. 15 Through systematic screening and analysis of the existing evidence, this review identified modifiable factors for VAP prevention and excluded interventions with no clear benefit.
Results
Description of the included literature
In Figure 1, we present the literature selection process for this review. A total of 1823 records were identified and screened, resulting in the inclusion of 45 studies (42 English, three Chinese) based on the predefined inclusion and exclusion criteria. The included studies comprised 18 high-level evidence sources (e.g. systematic reviews and guidelines), 24 interventional and observational studies (e.g. randomized trials and cohort studies), and three supportive syntheses (e.g. scoping and narrative reviews).

Search strategy flowchart.
PHASE bundle components
Based on a synthesis of current evidence, the proposed PHASE bundle consolidates fundamental, evidence-based nursing measures for VAP prevention, including positioning, hand hygiene, airway care, sedation minimization, and equipment management. Among these, airway care includes the majority of specific interventions, such as oral care, cuff pressure monitoring, suctioning, subglottic secretion drainage (SSD), early gastrointestinal decompression, and early enteral nutrition (EN). The protocol is summarized in Figure 2 and detailed in Table 1. The key evidence for each measure is analyzed in the following sections.
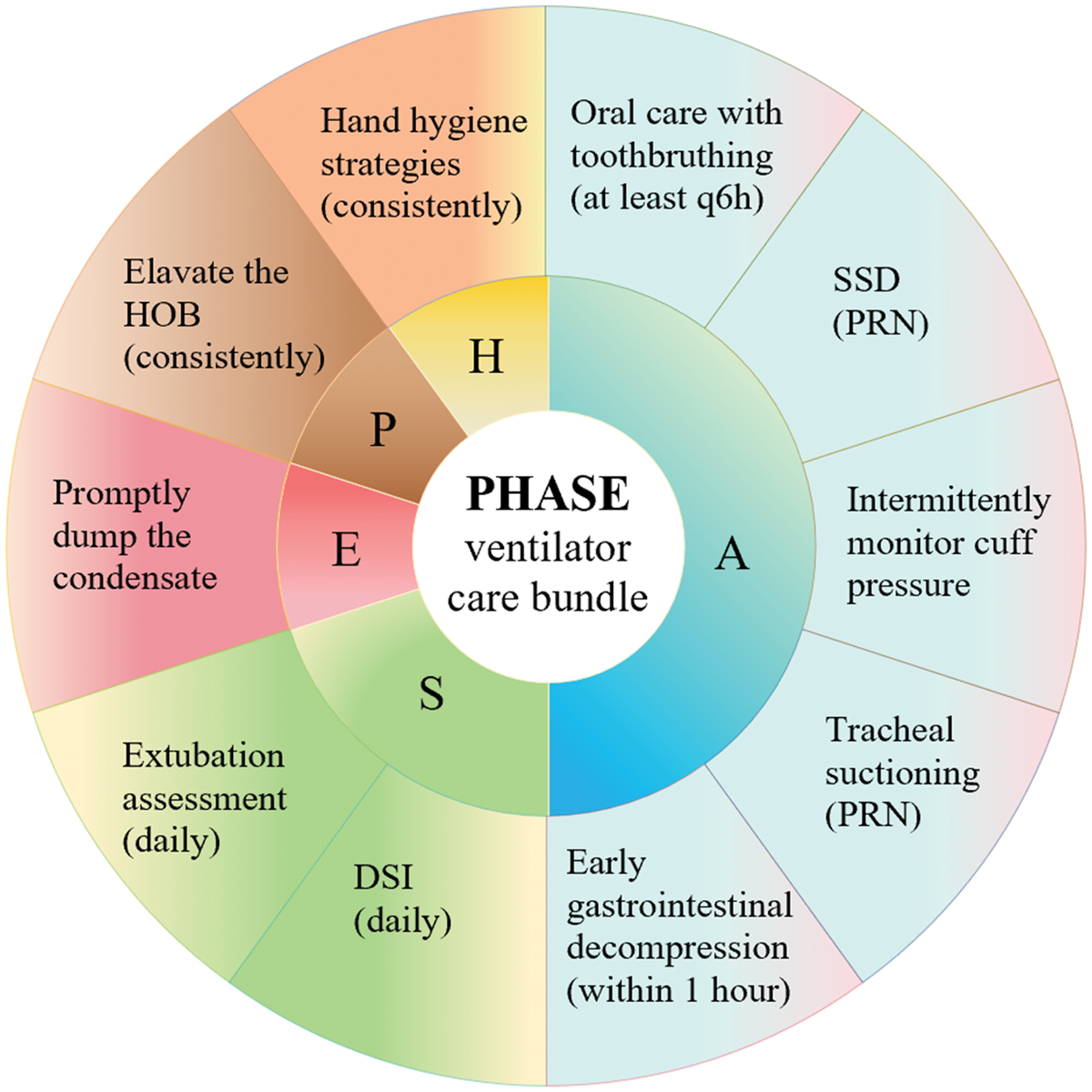
PHASE VCB for preventing VAP in the ED. P stands for position; H stands for hand hygiene; A stands for airway; S stands for sedation; E stands for equipment. VCB: ventilator care bundle; VAP: ventilator-associated pneumonia; ED: emergency department; SSD: subglottic secretion drainage; HOB: head of the bed; DSI: daily sedation interruption; PRN: pro re nata; “Within 1 hour” refers to the first hour following intubation.
Prevention of VAP in patients with MV PHASE VCB in the ED.

ED: emergency department; PHASE: P, position; H, hand hygiene; A, airway; S, sedation; E, equipment; HOB: head of the bed; MV: mechanical ventilation; SSD: subglottic secretion drainage; EN: enteral nutrition; PRN: when necessary.
Specific objective criteria must be established based on actual circumstances.
Elevating the head of the bed
Elevating the head of the bed (HOB) is a cornerstone intervention for reducing aspiration risk in patients undergoing MV. Based on its simplicity, cost-effectiveness, ease of implementation, and proven efficacy in significantly reducing VAP incidence, current guidelines strongly recommend maintaining an HOB elevation of 30°–45° unless contraindicated. 14 A randomized controlled trial (RCT) demonstrated that an HOB elevation below 30° significantly increases aspiration risk, whereas a semi-recumbent position (45°) reduces VAP incidence (P = 0.022) by minimizing the gravitational reflux of oropharyngeal secretions. 16 However, clinical decision-making should balance this benefit against the potential risks of hemodynamic instability and pressure injuries. 17
Hand hygiene
The latest Chinese VAP prevention guidelines provide specific recommendations for hand hygiene, including optimizing infrastructure to ensure easy access to handwashing facilities for HCWs, emphasizing critical moments for hand hygiene, and clarifying that wearing gloves does not replace thorough handwashing. 18 Furthermore, hand hygiene compliance (HHC) monitoring technologies significantly reduce healthcare- and invasive device-associated infections. 19 These automated systems employ auditory alerts, flashing lights, and tactile feedback to remind HCWs to perform hand hygiene and transmit the corresponding data to a management platform for analytical review. Although the standard code of conduct mandates HHC among HCWs, achieving high compliance remains challenging. A systematic review summarizing findings from multiple studies reported that HHC rates typically range from 60% to 70%. 20
Oral care
Evidence indicates that daily oral care with tooth brushing significantly lowers VAP rates, shortens the duration of MV, and reduces ICU length of stay. 21 Regarding oral care solutions, a systematic review concluded that povidone iodine (risk ratio (RR) = 0.69, 95% confidence interval (CI): 0.50–0.95), hydrogen peroxide (RR = 0.38, 95% CI: 0.15–0.96), and nitrofurazone (RR = 0.29, 95% CI: 0.14–0.58) each significantly reduced VAP incidence compared with saline or placebo. In contrast, neither Listerine nor sodium bicarbonate demonstrated a significant preventive effect. 21 These conclusions were based on studies assessed as having a low risk of performance bias. Research has reported an association between chlorhexidine use and increased mortality (odds ratio (OR) 2.32–2.92) in lower-risk patients; however, a recent RCT found that discontinuing its use did not affect VAP incidence or mortality.22,23
Cuff pressure monitoring
Guidelines and literature recommend maintaining endotracheal cuff pressures (Pcuff) between 20 and 30 cmH2O (1 cmH2O = 0.098 kPa), with careful checks every 6 hours and following any change in patient position.24,25 Elevated Pcuff (>30 cmH2O) can directly injure the trachea, potentially causing complications such as tracheoesophageal fistula or stenosis. Conversely, insufficient Pcuff (<20 cmH2O) allow oropharyngeal or gastric secretions to leak past the cuff, promoting microaspiration and increasing the risk of VAP. 26 Although Chinese practice widely recommends maintaining Pcuff of 25–30 cmH2O, the evidence supporting this specific target remains limited. 18 A single-center prospective trial found no clinical benefit of more frequent over less frequent Pcuff pressure monitoring. 27 Importantly, routine manual adjustment of Pcuff often leads to significant pressure loss and potential fluid leakage. 28 In contrast, automated systems reduce the rate of underinflation to 18% (vs. 63% with manual adjustment, P < 0.001) and lower VAP rates. They may also decrease human error and improve compliance in high-acuity settings. 29
Endotracheal suction
Clinical guidelines in China widely recommend closed suction systems to reduce procedural contamination and occupational exposure for HCWs. 18 Although these systems may cause fewer transient physiological disturbances—such as declines in oxygenation and increases in heart rate and blood pressure—compared with open suctioning, the clinical significance of these effects remains uncertain. Systematic reviews and meta-analyses have indicated no statistically significant difference in VAP incidence between closed and open suction methods. 30 The most recent meta-analysis suggests that normal saline instillation before suctioning enhances secretion clearance; however, it is also associated with adverse events, including oxygen desaturation, tachycardia, and a higher rate of VAP. 31 Given these risks and the insufficient evidence of net benefit, guidelines discourage routine saline instillation.14,18
Early gastrointestinal decompression and EN
Research has demonstrated that gastrointestinal decompression within the first hour following endotracheal intubation significantly reduces intra-abdominal pressure, improves pulmonary compliance, and enhances oxygenation in patients with MV, a finding with potential implications for VAP prevention. 32 Guidelines recommend early EN within the first 48 hours of hospital admission, a strategy supported by high-quality evidence. 14 By stimulating normal gut function, early EN reduces the risk of bacterial translocation to the lungs, thereby lowering the incidence of VAP. A propensity score-matched study showed that patients receiving early EN had a significantly lower incidence of VAP (12.9% vs. 25.8%, P = 0.026) and a shorter hospital stay (21 vs. 24 days, P = 0.015) compared to those receiving late EN. 33 Compared with gastric feeding, post-pyloric feeding significantly reduces aspiration events and pneumonia incidence and shortens the duration of MV, ICU stay, and overall hospitalization (all P < 0.001). 34 However, post-pyloric tube placement requires specialized expertise that may not be available in all clinical settings and can lead to delays in initiation. Given that gastric feeding more closely mimics physiological digestion, guidelines recommend reserving post-pyloric feeding for patients with gastric feeding intolerance or those at high risk of aspiration. 14
SSD
A meta-analysis showed that the use of endotracheal tubes with SSD reduced the incidence of VAP by 44%. 35 A single-center RCT comparing continuous and intermittent SSD reported no statistically significant difference in VAP rates between the two methods. Nonetheless, the continuous drainage group exhibited clinically meaningful benefits, including shorter duration of MV and reduced ICU length of stay. 36 These findings should be interpreted in light of the trial’s limited sample size and the exclusion of patients with early extubation, which may limit generalizability to short-stay populations. Additionally, the unblinded design raises the possibility of detection bias in outcome assessment.
Minimize sedation
The DSI protocol involves completely or partially stopping sedative medications for a defined period (typically up to 5 hours) while maintaining a target Richmond Agitation-Sedation Scale score between 0 and -2. 24 Multimodal strategies, such as nurse-driven sedation protocols for targeted light sedation and DSI (e.g. spontaneous awakening trials) for patients without contraindications, are employed to achieve this. DSI is contraindicated in patients who are intubated within 24 hours, require escalated sedation dosages, receive neuromuscular blockers, have elevated intracranial pressure, present with low mean arterial pressure, require high fraction of inspired oxygen and high positive end-expiratory pressure support, exhibit high peak inspiratory pressure, or experience seizures. 24 Both DSI and nurse-driven protocols facilitate assessable and adjustable sedation management, maintaining a balance between comfort and alertness. A multicenter randomized trial comparing non-sedation and light sedation under DSI found no difference in ventilator-free days, ICU length of stay, or 90-day mortality. 37 A meta-analysis reported that nurse-driven sedation protocols significantly reduce VAP incidence (RR = 0.438; 95% CI: 0.292–0.657; P < 0.001), in-hospital ICU mortality, and sedation-related adverse events among patients receiving MV. 38 Current evidence remains insufficient to favor one specific sedation-minimizing protocol over another.
Implement a ventilator liberation protocol
Guidelines strongly recommend implementing a structured ventilator liberation protocol based on daily sedation interruption. 14 This protocol involves daily assessment of readiness for extubation in suitable patients, primarily through spontaneous breathing trials. Evidence indicates that using such protocols in appropriate patients can shorten the median duration of MV by 1 day compared with non-protocolized care. 39 Furthermore, combining ventilator liberation protocols with daily sedation interruption may produce synergistic effects, facilitating earlier extubation.
Ventilator circuit management
A key preventive measure involves the timely drainage of ventilator circuit condensate, as stagnant fluid can serve as a reservoir for pathogens. To mitigate this risk, nurses should routinely drain the fluid and position the collection cup at the lowest point of the circuit to prevent retrograde flow into the patient’s airway. 14 Regarding circuit change frequency, guidelines recommend changing ventilator circuits only when they are visibly soiled or malfunctioning, rather than on a fixed schedule that prompts frequent, unnecessary changes. A network meta-analysis showed that changing circuits every 3 days significantly reduced VAP incidence compared with changing circuits every 1, 2, 4, or 14 days. Probability rankings further identified that a 3-day interval had the highest likelihood of being the optimal intervention, followed by 5 and 7 days. 40 In contrast, another meta-analysis of six studies reported no significant differences in VAP incidence or mortality between 3- and 7-day circuit changes. 41 Therefore, although a single optimal interval remains unclear, evidence suggests that, compared with very frequent changes (1–2 days), less frequent changes (3–7 days) do not increase VAP risk.
Discussion
Patients are at risk for VAP from the moment of emergency endotracheal intubation. While evidence-based care bundles are effective in the ICU, the dynamic and resource-constrained ED environment presents unique challenges for implementing complex ICU protocols directly. 42 Therefore, this narrative review proposes the PHASE bundle—a feasible, context-specific set of interventions designed for early use in the ED. Its primary aim is to bridge the preventive gap before ICU transfer by elevating awareness and initiating core measures at the earliest point of risk. Although the PHASE bundle may not represent the optimal combination, its core innovation lies in establishing the paradigm that VAP prevention must begin in the ED. It provides a foundational framework to inform future research aimed at optimizing emergency care–specific prevention strategies.
This review identifies a core set of interventions with a consistent evidence base for VAP prevention. These measures—including HOB elevation, hand hygiene, oral care with toothbrushing, cuff pressure monitoring, sedation minimization, and prompt drainage of ventilator circuit condensate—collectively constitute the PHASE bundle for VAP prevention.6,14,21 They form a core of high-strength, concordant evidence supported by international guidelines, multicenter RCTs, and meta-analyses. These measures merit designation as essential core components not only because of their proven efficacy in reducing VAP incidence but also because of their high feasibility—they are relatively low-cost, simple, and readily integrable into protocolized care bundles—and their alignment with universal infection control principles such as hand hygiene and oral care. Moreover, by focusing on essential nursing interventions and omitting non-evidence-based or indirectly related measures, such as routine use of chlorhexidine for oral care and prophylaxis for deep vein thrombosis (DVT) and peptic ulcer disease (PUD), this bundle reduces human and material resource demands. This streamlined, easy-to-train structure significantly enhances its feasibility for clinical adoption.
The Institute for Healthcare Improvement VCB comprises four key elements: (1) HOB elevation to 30°–45°, (2) daily sedation interruption and assessment of readiness to extubate, (3) PUD prophylaxis, and (4) DVT prophylaxis. 43 The PHASE bundle excludes PUD and DVT prophylaxis. These prophylactic measures can significantly reduce thromboembolic and gastrointestinal bleeding risks in ICU populations; 44 however, their direct or indirect contribution to VAP prevention remains unclear. The guidelines do not recommend their use specifically for VAP prevention. 14 To elucidate their role, future studies should address two questions: first, whether these prophylactic measures complement or potentially conflict with other VAP prevention strategies; and second, whether their integration into VAP-specific care bundles provides additive value compared with their isolated use. The proposed emergency PHASE bundle is a nurse-led clinical pathway designed specifically to prevent VAP in patients who are already intubated and receiving MV; nursing staff can implement it independently. Consequently, while current guidelines strongly recommend avoiding intubation and employing non-invasive positive pressure ventilation (NIPPV) for VAP prevention, 14 these approaches were excluded from the bundle because EDs frequently manage critical scenarios, such as respiratory arrest, that necessitate immediate intubation.
The implementation of certain measures for interventions in which the optimal method remains uncertain or context-dependent warrants careful evaluation. A prominent example is the use of chlorhexidine for oral care, for which researchers have reported inconsistent evidence regarding its efficacy.22,23 This heterogeneity may stem from variations in patient populations (e.g. baseline rates of multidrug-resistant organism colonization), differences in intervention protocols (e.g. concentration, frequency), or inconsistencies in outcome definitions. Given the lack of conclusively proven benefit for VAP prevention and emerging concerns regarding potential harm, we align with current guidelines in advising caution against its routine use. 14 The decision to use chlorhexidine should involve multidisciplinary team agreement on an individual basis and follow local hospital prescribing policies. Aspiration of oropharyngeal secretions is a primary contributing factor to VAP, as patients receiving MV exhibit diminished cough reflexes and impaired mucociliary clearance due to endotracheal intubation. 45 The physiological rationale for prevention derived from ICU evidence remains valid in the ED; however, its effective and safe application requires adaptation to this distinct context. Meta-analyses suggest that SSD may reduce the incidence of VAP; however, the evidence remains insufficient to confirm its impact on critical outcomes such as duration of MV, length of stay, or mortality. The scarce availability of specialized endotracheal tubes in some regions further limits the pragmatic adoption of SSD in the ED. 9 Moreover, current guidelines recommend SSD primarily for patients expected to require more than 48–72 hours of MV and for in-hospital patients undergoing emergency intubation or preoperative patients at elevated risk of prolonged intubation. 14 This targeted guidance aligns well with the ED context, where emergency intubation is common, and a subset of high-risk patients may derive benefit. Consequently, the PHASE bundle designates SSD as an optional intensification measure, enabling selective application based on individualized risk assessment and local resource availability.
Relatively few high-quality interventional studies have focused specifically on VAP prevention in the ED setting. This evidence gap necessitates reliance on inferences from ICU studies and underscores the urgent need for ED-targeted research. Future studies should prioritize determining the minimum effective frequency of key nursing interventions (e.g. oral care, cuff pressure checks) within the ED’s rapid workflow to guide feasible protocols and improve nursing adherence. Additionally, the current diagnosis of VAP is subjective and nonspecific. 46 Differences in definitions, documentation, and analytical approaches compromise the reliability of reported outcomes, making it difficult to determine whether an observed decline in VAP incidence reflects genuine clinical improvement. 47 It is also important to note that EDs function primarily as the starting point influencing risk rather than as the diagnostic location. To circumvent these diagnostic and setting-specific challenges, future research should validate the impact of bundles like PHASE using objective, VAP-relevant outcomes in the ED, such as duration of MV.
Effective implementation of the PHASE bundle requires a multidisciplinary approach. 48 Within this framework, physicians, nurses, respiratory therapists, and infection prevention specialists assume distinct and complementary roles to ensure all bundle components are executed. Physicians are responsible for core medical decisions and invasive procedures, including formulating DSI and spontaneous breathing trial protocols and prioritizing NIPPV when appropriate. Nurses act as lead executors and coordinators of the bundle; in addition to implementing basic nursing measures, they monitor patient status, facilitate communication, and ensure timely execution of interventions, serving as the central hub of the prevention process. To sustain adherence amidst high acuity and turnover, providing educational support and using concise, time-stratified checklists tailored to the ED’s brief, episodic workflow are critical.49,50 Clinical teams should also actively monitor adherence through chart or bedside audits and dynamically adjust audit frequency based on compliance levels. 14
This study has some limitations. As a narrative review, it lacks a systematic protocol and explicit risk-of-bias assessment, which may introduce selection bias. Furthermore, the simplified design of the bundle, crucial for feasibility, may be insufficient for managing complex or special cases. Future research should prioritize targeted, high-quality interventional studies in the ED to evaluate the impact of the entire PHASE bundle on patient outcomes.
Conclusions
This narrative review synthesized existing evidence to develop a nurse-led, ED-specific bundle for the early prevention of VAP. We propose that the PHASE bundle, which integrates five core measures—positioning, hand hygiene, airway care, sedation minimization, and equipment management—is both feasible and critical for mitigating VAP risk prior to ICU transfer. To sustain its effectiveness, the PHASE strategy requires tailoring to local clinical settings and ongoing evaluation using objective compliance and outcome measures. The development of ED-focused monitoring tools is essential to maintain protocol adherence and support continuous quality improvement. Future initiatives should focus on building a consensus on ED-specific VAP prevention practices and optimizing nursing resource allocation.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261417865 - Supplemental material for A potential bundle for preventing ventilator-associated pneumonia in the emergency department: A narrative review
Supplemental material, sj-pdf-1-imr-10.1177_03000605261417865 for A potential bundle for preventing ventilator-associated pneumonia in the emergency department: A narrative review by Jia Di, Guo Xuan, Xu Shanshan and Yang Jianzhong in Journal of International Medical Research
Footnotes
Acknowledgments
We sincerely thank the experts in VAP prevention and infection control for their valuable guidance, which was instrumental in defining the core theme of this narrative review and refining the critical appraisal framework. Their insights greatly strengthened the academic rigor and focus of this review. We also thank the reviewers of The Journal of International Medical Research for their thorough evaluation and thoughtful feedback during the revision process, which significantly improved the quality of this manuscript. Finally, we acknowledge Editage (![]() ) for providing writing support.
) for providing writing support.
Author contributions
JD conducted literature screening, selection, and critical appraisal, drafted the original manuscript, and developed visualizations. GX performed the literature screening and study selection, provided overall supervision throughout the project, and critically revised the manuscript for important intellectual content. XSS contributed to the methodological design, provided supervisory guidance, offered key recommendations for data interpretation, and critically reviewed and edited the final manuscript. YJZ (corresponding author) conceived and designed the narrative review, supervised the overall process, critically reviewed & edited the manuscript, and acquired funding for this work. All authors have approved the final version and agree to submission.
Data availability statement
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Funding
This work was supported by the Clinical Research Fund of the Talent Cultivation Project for Outstanding Hospital Talents and Innovative Teams of the First Affiliated Hospital of Xinjiang Medical University (grant number: CXT D202408).
Originality
This manuscript is original, has not been published elsewhere, and is not under consideration by any other journal.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
