Abstract
Introduction:
Cases:
Seventeen patients who ate the seeds of an unknown plant that grows in their gardens came to the emergency department with nausea, vomiting, and stomach ache complaints 4 h after ingesting the seeds. All patients were admitted to the critical care unit, monitored, and started on supportive treatment. After the treatment, patients’ vital signs and laboratory parameters were stable. Seven patients were discharged the next day upon the regression of symptoms. The remaining 10 patients were successfully discharged 2 days after the treatment. There are cases in medical literature where consumption of one-half of a
Conclusion:
Emergency physicians have to know the clinical course and medical methods regarding intoxication due to oral consumption of toxic plants. Severe toxicity and even mortality can be observed with the ingestion of the seeds of the plant. Similar symptoms (nausea, vomiting, and abdominal pain) were observed in all our cases, but no mortality was observed.
Keywords
Introduction
Known as the castor bean plant in Turkey,
The component that causes the toxication is ricin, which is a glycoprotein structure that consists of two chains (Chain A: protein synthesis inhibitor; Chain B: cell-surface bond function).
5
Ricin, especially when inhaled, is one of the most powerful and fatal toxic substances.
6
When inhaled, ricin can cause nausea, exhaustion, respiratory problems, coughs, and pulmonary edema that may lead to death. If ricin is taken orally, it can cause abdominal pain, nausea, vomiting, diarrhea, liver and kidney dysfunctions, and rare cases of hematemesis and melena. When injected into tissue, it can cause local pain, allergic reactions, liver and kidney dysfunctions, and necrosis.3,4,7,8 The lethal toxin dose is 3 to 5 µg/kg when inhaled and 20 mg/kg when taken orally. Unripe
In this presentation, 17 patients who ingested
Case presentation
Seventeen patients who ingested the seeds of an unknown plant that grows in their garden came to the emergency department (ED) with nausea, vomiting, and stomach ache complaints 4 h after ingesting the seeds. The first patient (a 9-year-old girl) ate a seed and liked its taste. She then offered the seeds to other family members, and all of them ate the seeds. Family members brought in plant samples, so we determined the plant. The plant had finger-like red leaves and red capsules with spines. We searched shape of the plant in a web search engine and we matched it with

The sample of the seeds that patients ingested.
All the patients were from the same family and had chewed and swallowed the seeds at the same time. None of the cases had complaints of diarrhea. The patients were three males and 14 females with an average age of 32.12 years (5–70 years). Each patient had consumed a minimum of one and a maximum of four seeds (2.18 on average). None of the patients, other than a 63-year-old woman with diabetes, hypertension, and coronary artery disease, had any other conditions.
The average systemic blood pressure of the patients was measured at 104/63 mmHg; each patient was at a normal level (the diastolic blood pressure of three child patients were not measured). The average heartbeat rate for patients 16 years and older was 93.3 beats per min. Two patients were observed with sinus tachycardia. For patients younger than 16 years old, the average heartbeat rate was measured at 103 beats per min. These rates were within the reference ranges that are normal for the patient ages. The average WBC (white blood cell) count of patients was calculated as 9,806 K/µL. The WBC values of nine patients were above the normal range. The urea/creatinine ratio, CRP (C-reactive protein), Na, K, and Cl tests of the patients showed no pathological conditions. The AST (aspartate aminotransferase) and ALT (alanine aminotransferase levels were above the normal range in one patient who had a history of diabetes, hypertension, and coronary artery disease. Results of the patient’s previous liver function tests were normal.
All patients were admitted to the critical care unit, monitored, and started on supportive treatment. Adult patients were given a metoclopramide 10 mg IV q8hr (every 8 h) and a normal saline infusion 100 cc/h IV as antiemetics. Patients younger than 16 years old were given a ½ normal saline + 5% dextrose infusion 50–70 cc/h IV and a metoclopramide 0.1 mg/kg IV q8hr. All patients were given orally 1g/kg of activated charcoal. The symptoms of the patients improved with antiemetic treatment. After the treatment, the patients’ vital signs and laboratory parameters were stable. Seven patients were discharged the next day upon the regression of symptoms. The remaining 10 patients were successfully discharged 2 days after the treatment. A 6-h asymptomatic period, stable vital signs, and laboratory parameters were considered as the criteria of discharge. All cases are presented in Table 1.
Personal characteristics, number of seeds, vital signs, laboratory findings, and admission information.
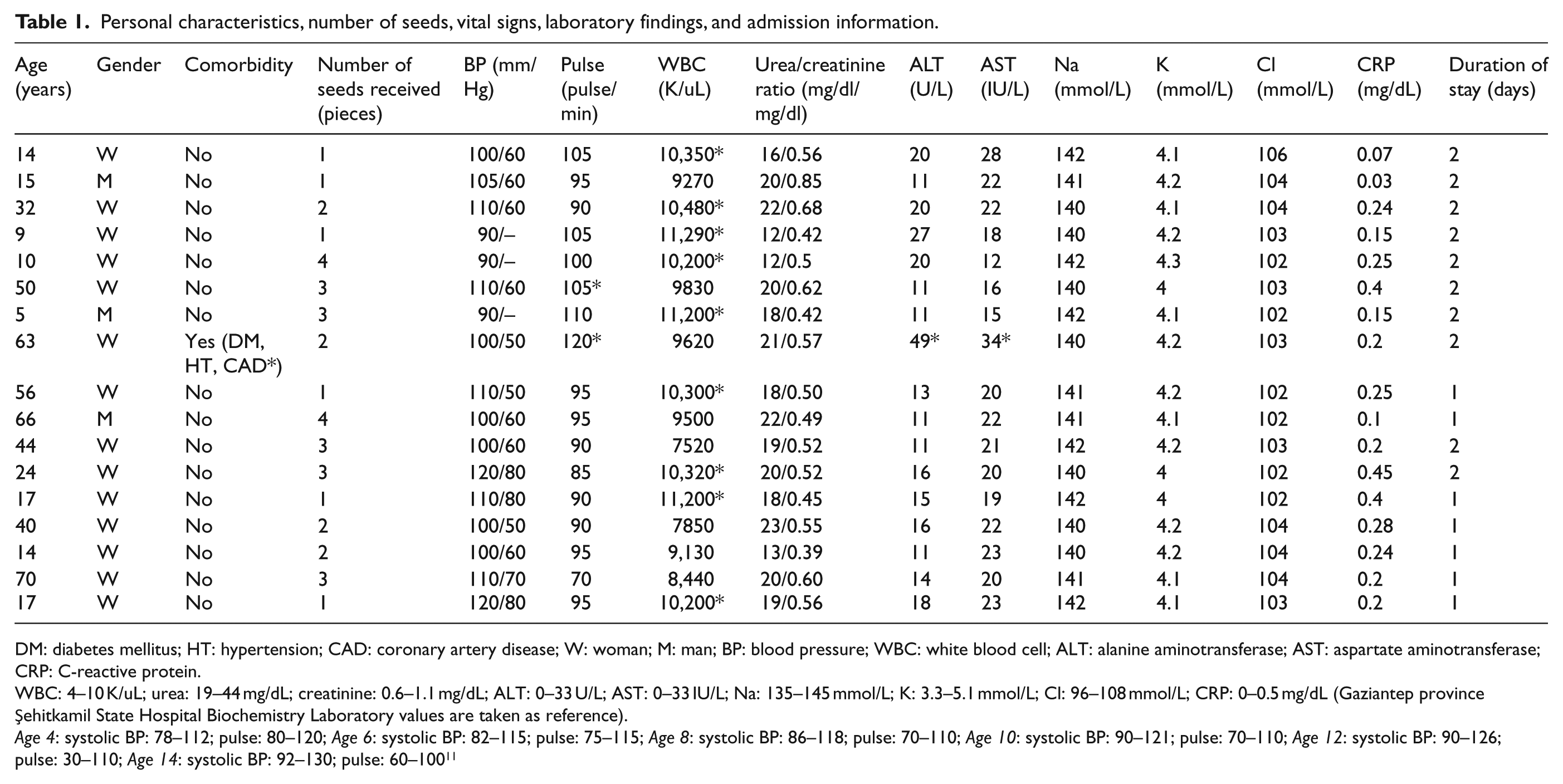
DM: diabetes mellitus; HT: hypertension; CAD: coronary artery disease; W: woman; M: man; BP: blood pressure; WBC: white blood cell; ALT: alanine aminotransferase; AST: aspartate aminotransferase; CRP: C-reactive protein.
WBC: 4–10 K/uL; urea: 19–44 mg/dL; creatinine: 0.6–1.1 mg/dL; ALT: 0–33 U/L; AST: 0–33 IU/L; Na: 135–145 mmol/L; K: 3.3–5.1 mmol/L; Cl: 96–108 mmol/L; CRP: 0–0.5 mg/dL (Gaziantep province Şehitkamil State Hospital Biochemistry Laboratory values are taken as reference).
Discussion
The clinical course of the intoxication due to oral consumption of toxic substances depends on the quantity of the substance consumed, the time passed after consumption, and the patient’s age, weight, and comorbidities. In medical literature, clinical symptoms of
In cases of
The medical literature states that the oral consumption of
Some studies have reported cases of leukocytosis (WBC), increased liver function test (AST/ALT) results, increased kidney function test (BUN = blood urea nitrogen) results, and electrolyte disorders due to
There is no known antidote of the toxic substance, ricin.6,7 Stabilization of vital signs, fluid replacement, and symptomatic supportive care have been suggested to be the main treatment of ricin intoxication.6,7 In the literature, gastric lavage and activated charcoal have been recommended to reduce toxic absorption with consideration for time passed after the oral consumption. 6 In our presentation, gastric lavage was not used as the patients came to the ED approximately 4 h after oral consumption (gastric emptying time was considered to have been exceeded). However, in accordance with the literature, 1 g/kg of active charcoal was given orally to patients to reduce the GI absorption of toxic ricin. Cases with plasma exchange have been reported; 12 however, because our patients responded to symptomatic treatments, plasma exchange was not required.
Previous studies have highlighted that the length of patient follow-up depends on whether the symptoms last or not. Some publications have announced the average follow-up length as 17 to 37.5 h. 10 However, there are reported cases with follow-up lengths of up to 90 days. 7 In our presentation, 10 out of 17 patients were tracked for 2 days, and seven patients were tracked for 1 day in the critical care unit. The average time spent in the unit was 38.1 h, which is normal according to the literature. No patients were pronounced dead.
Conclusion
Contrary to popular belief, green plants can cause intoxication cases that are as deadly as chemical-synthetic substances. Learning about the plants that can lead to intoxication, raising public awareness, and helping to protect public health are physician’s duties. These 17 cases are the first
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
The data used to support the findings of this study are included within the article.
Ethical approval
Ethical committee approval was obtained from Hasan Kalyoncu University (Desicion no: 2018/31; Date: 20 November, 2018).
Human rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Written informed consent was obtained from the patients.
