Abstract
Background:
With the flourishing application of botulinum toxin cosmetically and therapeutically is the emergence of iatrogenic botulism, a new type of botulism in addition to the traditional ones.
Objectives:
We aim at a comprehensive review of the clinical characteristics of iatrogenic botulism. Methods: The available publications are retrieved and studied.
Results:
Botulinum toxin blocks cholinergic transmission in the neuromuscular junctions and autonomic ganglia. The blockade can spread from the site of tissue injection to adjacent or sometimes far off structures, resulting in inadvertent disabling or even lethal effects. On literature review, weakness and dysphagia are the commonest complications of iatrogenic botulism, whereas ophthalmological and oropharyngeal symptoms are more prevalent in the cosmetic group and dyspnea in the therapeutic group. Antitoxin therapy is required in about 20% of the patients. Diagnosis of iatrogenic botulism is primarily clinical and should not be confused with the neurological diagnoses possessing similar clinical manifestations. Vigilance to the drug formulation, dosage, and administration during botulinum toxin injection are part of the preventive measures in minimizing the occurrence of iatrogenic botulism.
Conclusion:
While overlapping with the traditional types of botulism, iatrogenic botulism carries its unique clinical characteristics.
Introduction
Botulism is a neuroparalytic disease caused by the toxins of Clostridium botulinum and less commonly toxin producing strains of Clostridium baratii and Clostridium butyricum. It is classified with respect to the route of acquisition as food, wound, infant, adult infectious, and inhalation botulism. This classification was added the sixth, iatrogenic, following medical application of botulinum toxins to humans in the last couple of decades. The medical values of botulinum toxin can be dated back to the 1817 publication by Christian Andreas Justinus Kerner, who first recognized that botulinum toxin paralyzed skeletal muscles and parasympathetic function and proposed its use as a therapeutic agent. 1 However, the therapeutic potential of botulinum toxin was not recognized until the reports of its injection into the eye muscles to correct strabismus in monkeys and humans in 1980 and 1981 respectively.2,3 In 1984, botulinum toxin was first reported to be successful in treating blepharospasm. 4 In 1987, Jean Carruthers noted that after botulinum toxin treatment for blepharospasm, the frown lines disappeared. She and her husband published the first report on botulinum toxin application for cosmetic purposes in 1996. 5 Since then, the therapeutic (e.g. limb dystonia, axillary and palmar hyperhidrosis, over-contractile bladder, achalasia) and cosmetic (e.g. forehead and face wrinkles, leg slimming) application of botulinum toxin flourishes ceaselessly, and the toxin is an indispensable tool in modern aesthetic practice. In parallel with its popularity are reports of iatrogenic botulism. From May 2016 to September 2019, at least 18 cases of iatrogenic botulism have been under investigation by the Department of Health of Hong Kong.6–14
Botulinum toxin
The Clostridium botulinum produces multiple immunologically distinct serotype toxins: A, B, C1 (C2 is not a neurotoxin), D, E, F, and G. The toxins A, B, E, and rarely F cause human botulism, and C1 and D affect non-human mammals, birds, and fish. The human toxic metalloproteases’ intracellular longevities in cultured neurons and the duration of neuromuscular blockade in rodents and humans are all in descending order of A, B, F, and E. 15 In foodborne botulism, type A infection is more likely to progress to respiratory paralysis that requires endotracheal intubation than type B and followed by type E.16–18
Monkeys and humans respond very similarly to the same dosage of botulinum toxin A injected into the eye muscles to correct strabismus.19,20 Extrapolation from monkey studies suggests that the minimum intramuscular systemic toxic dose and lethal dose of botulinum toxin A for humans are 33 and 38 U/kg, respectively, 19 while 1 unit denoting the median intraperitoneal LD50 for mice.
Registered botulinum toxins in Hong Kong
In Hong Kong, eight commercial products of botulinum toxin A are registered under the name of Botox 100 and 200 units, BTXA 50 and 100 units, Dysport, SIAX 100 units, and Xeomin 50 and 100 units. No toxin B like Myobloc is registered. 21
Dose conversion and variation across botulinum toxin preparations
Given the non-standardization of the mouse potency assay across laboratories and thus the possible inherent potency difference among the units of different botulinum toxin formulations, a unit of a particular formulation may not be biologically equal to that of another. To date, clinical studies on therapeutic outcomes are the most informative tool for making practical comparisons of botulinum toxin products. A systematic review of randomized trials on blepharospasm, hemifacial spasm, and cervical dystonia concluded that a Botox to Dysport unit ratio of 1 to 3 was essentially bioequivalent. 22 A later study using Botox and Dysport in 1 to 3 unit ratio found that forehead muscle activity on electromyography was significantly weaker in the Dysport group from 10 weeks onward after the injection, hinting that the bioequivalent unit ratio between the two commercial formulations might be less than 1 to 3. 23 In Germany, the recommended ratio for the treatment of glabellar lines is actually 1 unit Botox to 2.5 units Dysport. 23 Trials on blepharospasm, glabellar lines, and cervical dystonia demonstrated that 1 unit of Botox therapeutically corresponded with 1 unit Xeomin.24–26
The dosage of botulinum toxins is injection site dependent and may also be formulation dependent as illustrated by the followings. The recommended dose of Botox and Dysport for lower limb spasticity is 300 to 400 units and 10 to 15 units/kg divided among selected muscles, respectively, and the dose for glabellar lines is 20 units and 50 units in five equal aliquots, respectively.27,28 The maximum retreatment dosage is 400 units in 3 months for Botox and 1000 units in 12 weeks for Dysport.27,28 Regarding Xeomin, the dose in treating glabellar line is 20 units and should be redosed not sooner than 3 months and in relieving upper limb spasticity is 400 units and repeated not earlier than 12 weeks. 29
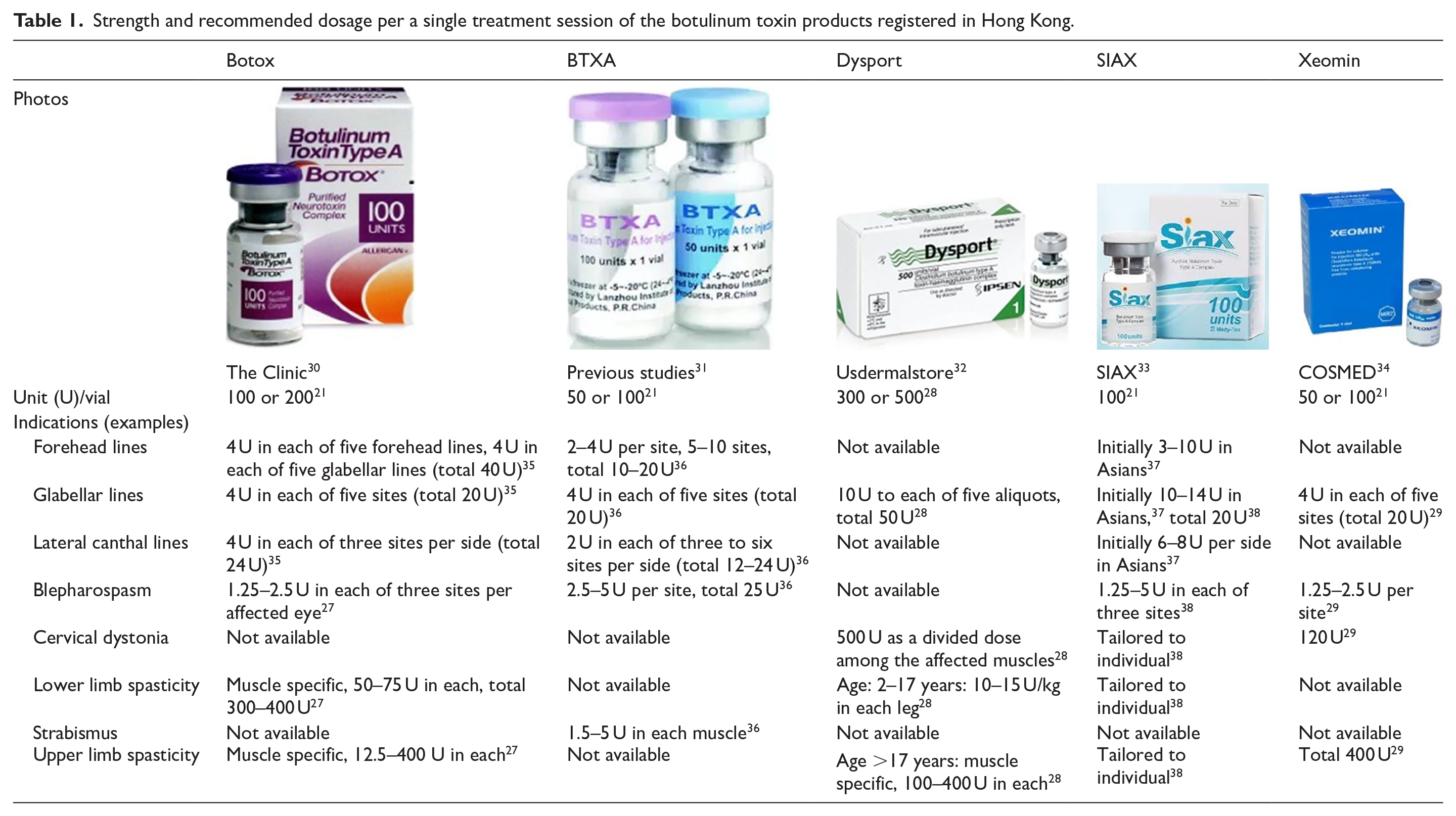
Table 1 lists out mainly the recommended dosage of the botulinum toxins registered in Hong Kong. Of these various commercial products, heterogeneity exists with respect to not only their doses, but additionally the indications’ availability, and the applicable age and ethnicity of recipients. The dosage of a single toxin has to be selected very carefully and the predicted effect of a treatment may also have to be relied on prior experience and individual factors.21,27–38
Strength and recommended dosage per a single treatment session of the botulinum toxin products registered in Hong Kong.
Toxicological mechanism
Botulinum toxin acts on cholinergic neurons like the neuromuscular junction and the autonomic ganglia. It consists of a heavy and a light chain. The C-terminal region of the heavy chain irreversibly binds to receptors at the presynaptic neurons (toxin A binds to GT1b gangliosides, toxin B to GD1a and GQ1b gangliosides).39–41 The complex is then taken up by endocytosis. The disulphide bond between the two chains is cleaved in the cytoplasm. The light chain cleaves the proteins (toxins A and E cleave synaptosomal associated protein-25; toxins B, F, and G cleave synaptobrevin/vesicle-associated membrane protein) that are needed for the fusion of acetylcholine vesicles with the cell membrane to form the exocytotic SNARE (soluble N-ethyl-maleimide-sensitive factor attachment protein receptor) complex and inhibiting acetylcholine release from the nerve terminals. 42 (Figure 1)
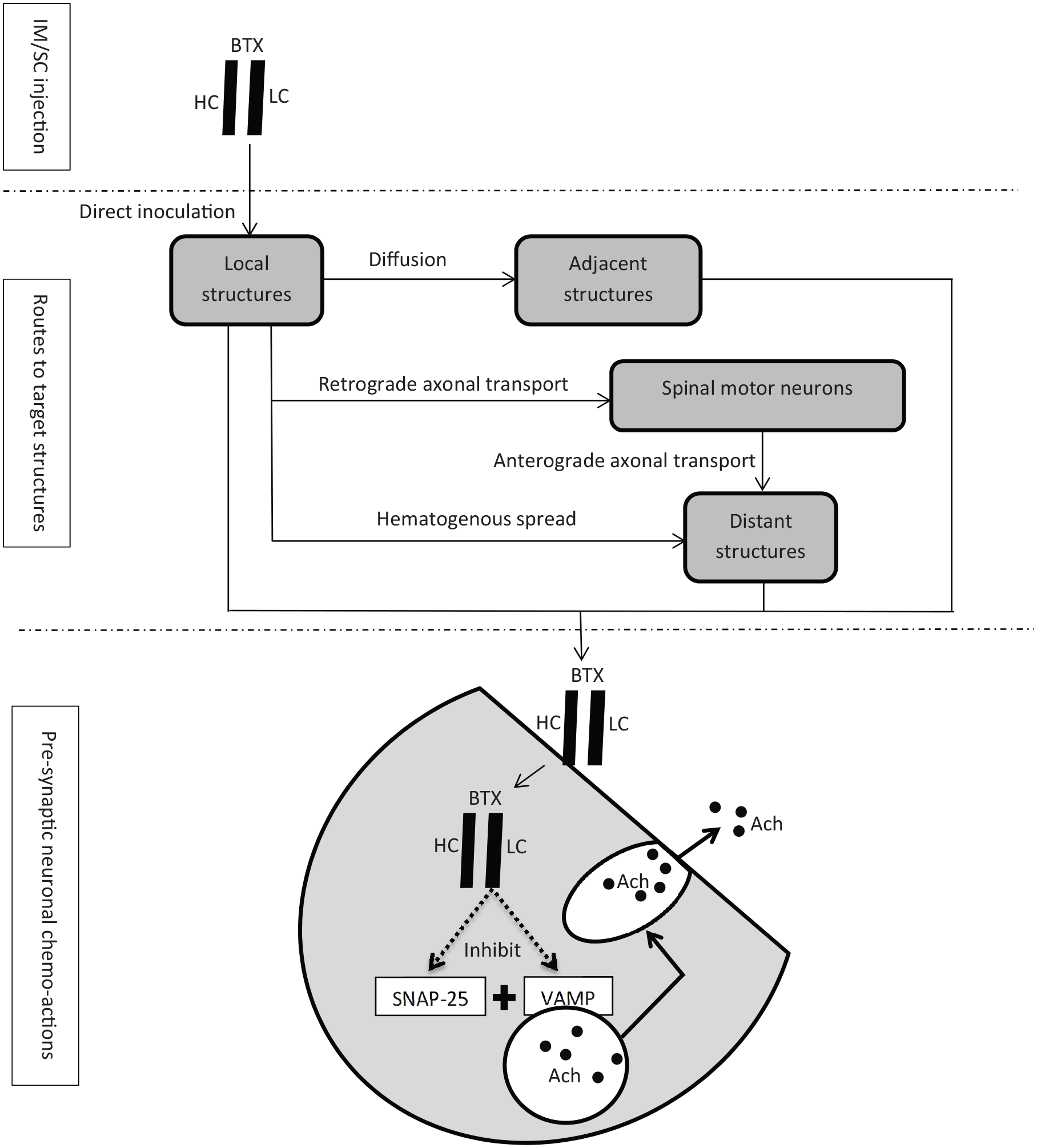
The toxicological mechanism of iatrogenic botulism.
Following focal injection of botulinum toxin, effects can be extended from local to adjacent and distant structures. Diffusion of botulinum toxin from the injected muscles to the adjacent muscles has been well documented, even they are separated by fasciae. 43 Diffusion of botulinum toxin to contralateral muscles has also been reported. Animal studies have shown that botulinum toxin can spread to a distance of 30–45 mm from the injection site. 44 Distant effects may be attributed to hematogenous spread of the toxin or retrograde axonal transport to spinal motor neurons followed by anterograde transport to the other motor units. 45 A rat model study demonstrated retrograde transport of catalytically active botulism toxin by central neurons and motor neurons from peripheral sites and subsequent transcytosis to various synapses. 46 An electromyographic research showed that changes in electromyography at remote sites were observed 3 to 13 days after focal injection of botulinum toxin A 47 (Figure 1).
As revealed by rodent experiments, botulinum toxin receptors exist in the central nervous system and a small amount of botulinum toxin crosses the blood–brain barrier, possibly by retrograde spread. Whether the toxin can induce centrally mediated effects has to be confirmed.47,48
Overall safety profile of botulinum toxin injection
A meta-analysis on randomized controlled trials contrasting Botox (N = 1425) and placebo (N = 884) identified that only focal weakness was significantly commoner (114/582, 19.6% vs 12/383, 3.1%) in the Botox-treated patients suffering from blepharospasm and cervical dystonia. 49 As a therapy for hemifacial spasm, botulinum toxin A was separately associated with upper lid ptosis and focal weakness on the side of injection in 5/232 (2.2%) and 3/232 (1.3%) treatment sessions. 50 Dsyport therapy for glabellar lines is ensued by ptosis of mild to moderate degree in 46/3861 (1.2%) treatment exposures. 51 A review on 758 children (94% sustaining spastic cerebral palsy, the rest with other neurological diseases) who received a total of 1594 Dysport injection sessions found out 16 (1%) incidents of focal muscle weakness and generalized muscle weakness in six children. A dose >1000 IU was a significant predictor of adverse outcome. 52 The above studies support a low likelihood of severe poisoning induced by injection of botulinum toxin.
Clinical presentations
Table 2 presents the available data on gender, age, toxin dosage, and time course of 54 patients from a total of 211 patients who are reported with symptoms and signs of iatrogenic botulism following cosmetic (number = 172) and therapeutic (number = 39) administration of botulinum toxins (Table 3).53–75 A comprehensive analysis other than symptoms and signs is limited by the non-standardization of the data across the reports, for example, absence of data items in some reports, some data presented in ranges instead of single discrete values, inconsistent range of values, and diverse measuring units.
Gender, age, toxin dosage, and time course of the iatrogenic botulism cases.

Median.
Inter-quartile range.
Range.
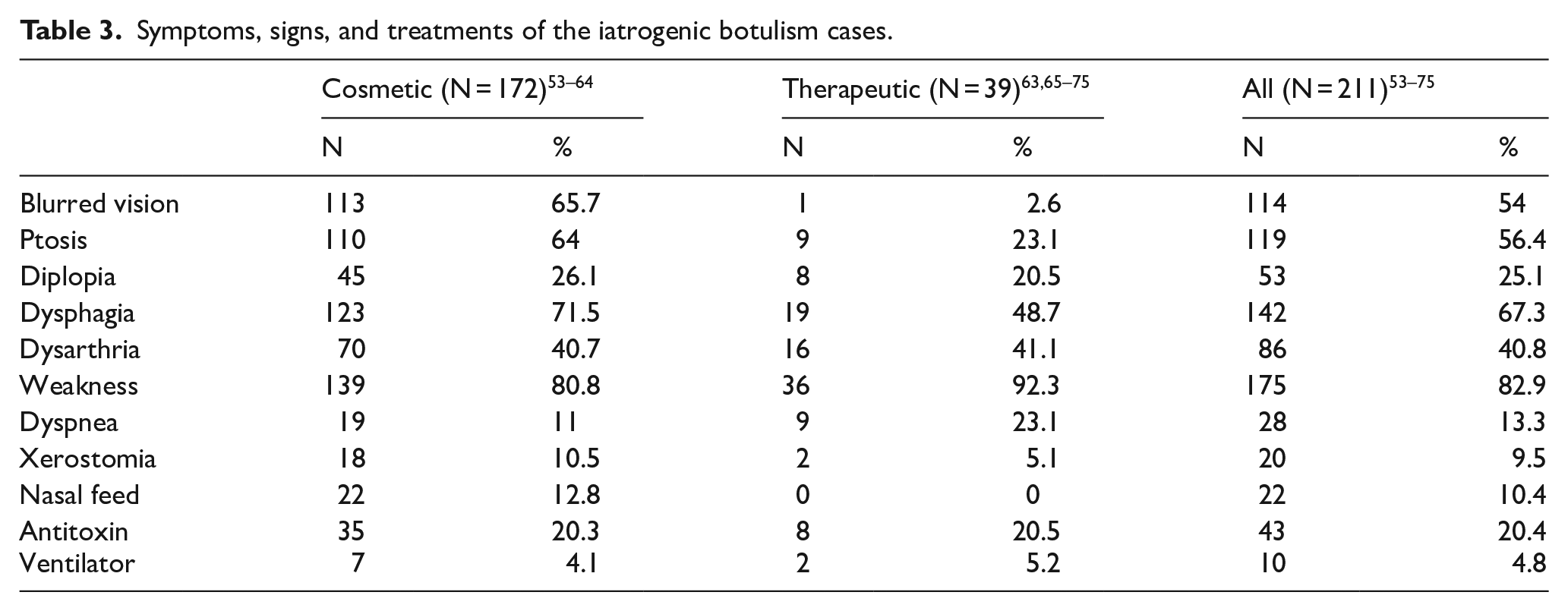
Symptoms, signs, and treatments of the iatrogenic botulism cases.
Botulinum toxin is a recognized medication to treat muscle spasticity. Thus, the therapeutic group is not surprised to have a wider range of age, both genders, and a larger number of units delivered. Of all the 211 subjects, 128 are known to be administered botulinum toxin A (Botox in 13, Dysport in 12, Neuroxin in 7, unknown brand name in 96), 1 administered toxin B (Myobloc, not tabulated 76 ), while the toxin serotype was not specified in 82. All the 128 patients were given doses under the minimum systemic or fatal doses extrapolated from monkey study except a pediatric patient who developed systemic botulism as a result of a severe overdose of the injected toxin A (40 U/kg) with unconfirmed trade name. The child developed dysphagia and weakness after a latency of 10 days. Supportive treatments were provided and she made a complete recovery after 3 months. 70 The Myobloc case was a 10-year boy with spastic quadriparesis, received 19,000 U, that is, 812 U/kg divided among four limbs, suffered from ptosis, generalized weakness, and dyspnea 1 week afterwards. Following a period of non-invasive ventilation, gastrotomy feeding and other supportive measures, it took 6 months for a full resolution of his weakness. 76
Four patients each received advertently 20 µg research grade botulinum toxin A in 100 µg/vial preparations. The strength in the 100 µg/vial preparation far exceeds that in the 5 ng-vial intended to be supplied for human use. All the four patients survived after antitoxin therapy and prolonged ventilation. 53
In general, the onset of symptoms is about 7 days for our iatrogenic botulism cases. In contrast, the incubation period of foodborne botulism is 12–36 hours (minimum 4 hours and maximum 8 days), 77 wound botulism 4–18 days, 78 and inhalation botulism 1–3 days. 77
More patients in the cosmetic group are injected botulinum toxin in the head and neck region than the limbs, while the reverse is true in the therapeutic group.
Weakness and dysphagia are the commonest toxicities in both the cosmetic and therapeutic groups. Blurred vision, ptosis, dysphagia, and xerostomia are more frequent in the former and dyspnea is more frequent in the latter. While dysphagia is one of the commonest symptoms in both groups, none of the patients in the therapeutic groups, in contrast with 12.8% of the cosmetic group patients, required nasal feed. The need of antitoxin and ventilator support was similar in both groups.
Classical botulism develops stereotypically as a descending bilateral paralysis starting from cranial nerve palsy and progressing down to the limbs. The earliest symptoms are blurred vision and diplopia. Other symptoms may follow in the sequence of dysphagia and dysarthria, weakness of the upper limbs and then lower limbs. Respiratory paralysis occurs in the severe cases. In iatrogenic botulism, the body tissues next to the injection site may be the only, most, or earliest affected because of the local diffusion spread of the toxin. Gastrointestinal symptoms are obvious in foodborne, and infant and adult infectious botulism yet absent or mild in the other forms, as in our reviewed cases. 77
Recovery involves sprouting of the affected axon, growth of new motor endplates, and regeneration of the cleaved SNARE proteins.79,80 In a mouse model, functional muscle recovery occurs after the sprouting of new motor nerve terminals, which reaches a peak after 5–10 weeks. 81 In our patients, full recovery needs approximately 2 months.
No mortality is recorded in the iatrogenic botulism reports. For the other types of botulism, the overall mortality is 3%–5% but is >60% in the old days without antitoxin. 82
Diagnosis
The diagnosis of iatrogenic botulism is mainly based on clinical presentation. Recent outbreaks, injection from an unlicensed source, and a short time gap from the last dose add to the clues. Investigations are usually unnecessary but may be indicated in selected cases, for example, sporadic case, clinically doubtful, and need to rule out other treatable causes.
Electro-diagnostic studies
Post-exercise or post-high frequency repetitive nerve stimulation compound muscle action potential increment, which implies neuromuscular junction facilitation, is classically considered as highly specific and sensitive in human botulism. 83 Yet, it has been mentioned only in a single patient with iatrogenic botulism, who received a low dose of Botox (30 U) for hyperhidrosis. 67
Conversely, decrement after low-frequency repetitive nerve stimulation, which is considered unusual in human botulism, 84 has been found in several articles on iatrogenic botulism, although consistently being small (up to 25%).66,67,75,85
The compound muscle action potential values are typically decreased in botulism.75,83,84 In case reports with iatrogenic botulism, compound muscle action potential values were low or marginally below the normal limits.67,72,86
Spontaneous activity at electromyography in iatrogenic botulism patients consists of denervation potentials known as positive sharp waves and fibrillations.66,67,75,85 Motor unit action potentials are often reduced in duration and amplitude, with unstable and polyphasic morphology as a result of denervation.66,67,75,85,87
Increased jitter (the time for end-plate potential of neuromuscular junction to reach the action potential threshold) and blocks revealed by single-fiber electromyography are present in botulism and less commonly in iatrogenic botulism.66,75,84,85,87,88 Interestingly, exaggerated jitter and blocking were also detected in asymptomatic subjects following therapeutic botulism toxin A injections, which indicates single-fiber electromyography is sensitive in detecting subclinical iatrogenic botulism, but poorly discriminating the differential diagnosis of neuromuscular junction pathologies. 89
Motor and sensory nerve conduction studies are normal.
Toxin detection
Toxin detection in patient specimen is not widely available and is facilitated by specimen collection at the time close to symptom onset. In vivo mouse lethality bioassay is the routine standardized method to detect the presence of botulinum toxin in clinical specimen. Although the toxin serotypes in iatrogenic botulism are potentially only A and B, it can be confirmed by using mouse bioassay studies with antitoxin neutralization. 90 In vitro proof of iatrogenic botulism can be established with detection of toxin in the patient’s blood by enzyme-linked immunosorbent assays and mass spectroscopy to identify the toxic protein and its serotype.91,92
Biopsy
A 45-year-old woman developed limb weakness after Dysport injection in her neck muscle for cervical dystonia. A biopsy of her left triceps muscle showed changes consistent with denervation. It showed that local botulinum toxin injections can inhibit neuromuscular transmission in distant muscles and cause botulism. 75
Functional magnetic resonance imaging
Functional magnetic resonance imaging (fMRI) performed on nine patients who received illegal cosmetic injections of highly concentrated botulinum toxin detected the following abnormalities when compared with healthy controls: the regional homogenicity values in the left posterior lobe of the cerebellum extending to the right anterior lobe of the cerebellum as well as in the right anterior lobe of the cerebellum extending to the right para-hippocampal gyrus and right posterior lobe of the cerebellum were significantly attenuated. There was also weakened amplitude of low-frequency fluctuation values in the right anterior lobe of the cerebellum extending to the left anterior lobe of the cerebellum and right posterior lobe of the cerebellum, as well as in the right anterior lobe of the cerebellum. These brain activities on MRI concur with studies suggesting retrograde transport of botulinum toxin from the peripheral sites, yet the exact pharmacology behind and the clinical significance need further elucidation. 56
Treatment
In our case series, 20.4% of the patients were treated with antitoxin. Botulinum antitoxin should be administered in an attempt to prevent neurological deterioration in suspected or confirmed iatrogenic cases of more than mild severity or progressing in severity. It can shorten the duration of mechanical ventilation for the respiratory compromised patients. Patients with wound botulism who received antitoxin within 12 hours of presentation have a lower incidence of respiratory failure and shorter duration of mechanical ventilation. 93
Botulinum antitoxin neutralizes circulating toxin and it cannot reverse the established toxicities. 94 But the experience of antitoxin therapy to against iatrogenic botulism is limited and there are reports contradicting failure of toxicity reversal. In a report, when trivalent (A, B, and E) antitoxin was administered from one to three vials to five patients, marked improvement was observed in the two patients who received two and three vials, respectively, and mild improvement in the one vial group. The one vial group was repeated another vial 12 h later, and the response was impressive. 65 Antitoxin may hasten recovery by more than binding of the intravascular toxin: accelerating neuromuscular conduction through increase of acetylcholine release from nerve terminals and redistribution of the toxin from its receptors before the irreversible endocytosis of the light chain. 65
Botulinum antitoxin is best delivered early to achieve the maximal effects. A woman with iatrogenic botulism after botulinum toxin A injection did not respond to antitoxin 6 days after symptom onset. 95 Yet, reports on benefits of delayed antitoxin therapy for iatrogenic botulism do exist. Two women who were each treated with two doses and four doses of monovalent botulinum type A antitoxin 9 and 7 days after their symptom onset showed clinical improvement. 54 Prolonged toxemia as possible explanation for a delayed therapeutic efficacy is illustrated by two cases of food borne botulism in which toxemia existed up to 12 days in one patient and 25 days in another. Intestinal toxin colonization as a culprit for the prolonged toxemia was unlikely because their stool samples had become negative for botulinum toxin by that time; instead sustained toxemia is more likely resulted from ingestion of high doses of toxin. 96 Notably, the pharmacokinetics of botulinum toxin in iatrogenic botulism has been rarely researched and not been formally compared with that in foodborne botulism. Among our cases, only three of them were measured blood toxin levels which were profoundly high: from 12 to 24 mouse LD50/mL after taking a huge 20 µg dose. Given the vast majority of our reviewed patients receiving merely therapeutic doses of botulinum toxin and assuming the elimination half-life 230–260 minutes of the toxin in mice and rats being similar to that in humans, lengthy toxemia in iatrogenic botulism appears not commonly expected. 53
The Hospital Authority of Hong Kong has now replaced the trivalent botulinum toxin (against serotypes A, B, and E) by the heptavalent botulinum antitoxin (against serotypes A to G). The existing antitoxin is a F(ab′)2 compound. It carries a low risk of immediate hypersensitivity and serum sickness. Of the 249 heptavalent botulinum antitoxin-treated patients in an analysis, rash happened in 2% of them and hemodynamic instability in one patient. 97 Monovalent botulinum antitoxin A is available in Mainland China and is serotype specific for most of the present botulinum toxin preparations.
Apraclonidine is a α2 adrenergic receptor agonist and a weak α1 adrenergic receptor agonist. The former action which contracts Muller’s muscle or superior tarsal muscle leads to the adoption of apraclonidine eye drops as a treatment for upper eyelid ptosis following botulinum toxin injection. Six subjects who developed botulinum toxin induced ptosis were each administered two drops of 0.5% apraclonidine eye drops to the eye with ptosis, four showed >50% improvement, and two had complete or almost complete recovery after an observation of 20–30 minutes. A common scheme for apraclonidine to maintain the therapeutic effects is one or two drops thrice daily until ptosis resolves. 98
Respiratory compromise is the primary cause of death. Clinical evaluation, pulse oximetry, spirometry, and blood gas are paramount in monitoring the ventilation integrity. Mechanical ventilation should be initiated at the earliest time indicated. For our patients, 10 out of 211 patients (4.8%) were put on ventilator and four cases were documented the duration of ventilation from 36 to 44 days and up to 171 days in 1 case.
Differential diagnoses
The differential diagnoses of botulism encompass other neuromuscular disorders including myasthenia gravis (MG), Lambert–Eaton myasthenic syndrome (LEMS), Guillain–Barré syndrome (GBS), and Miller Fisher syndrome (MFS). 84
Unlike botulism, MG does not present with a directional progression of paralysis. Patients with botulism may positively respond to anticholinesterase drugs such as edrophonium.99,100 Typically the response, if any, is weak and is only observed in the early phase of poisoning. However, patients with mild botulism may mimic MG with a dramatic response to anticholinesterase agents and signs of clinical pathological fatigue. 84
LEMS which is a presynaptic autoimmune disease shares common clinical features with botulism such as weakness and autonomic disturbance. Compared to botulism, it is characterized by first absent or minimal ophthalmological involvement and weakness of lower limbs dominant over upper limbs, second a greater increment after high frequency repetitive nerve stimulation (100% plus vs 20%–60%), and third the existence of IgG auto-antibody against N-type calcium channel on presynaptic nerve terminal in 90% of cases. 101
The pattern of descending weakness is a clinical hallmark of botulism and distinguishes it from the classical form of GBS, which usually presents with ascending weakness. 16 MFS, with ocular and bulbar abnormalities, may present a more difficult diagnostic challenge but the pupils in botulism may be dilated and fixed. Paresthesia is absent in botulism and present in GBS and MFS. Cerebrospinal fluid protein level is normal in botulism and is elevated in both GBS and MFS. It has been estimated that 90% of MFS with ophthalmoplegia have autoantibodies to GQ1b in acute phase sera. 102
Table 4 summarizes the classical differences between botulism and its differential diagnoses.
The classical differences between botulism and its differential diagnoses.
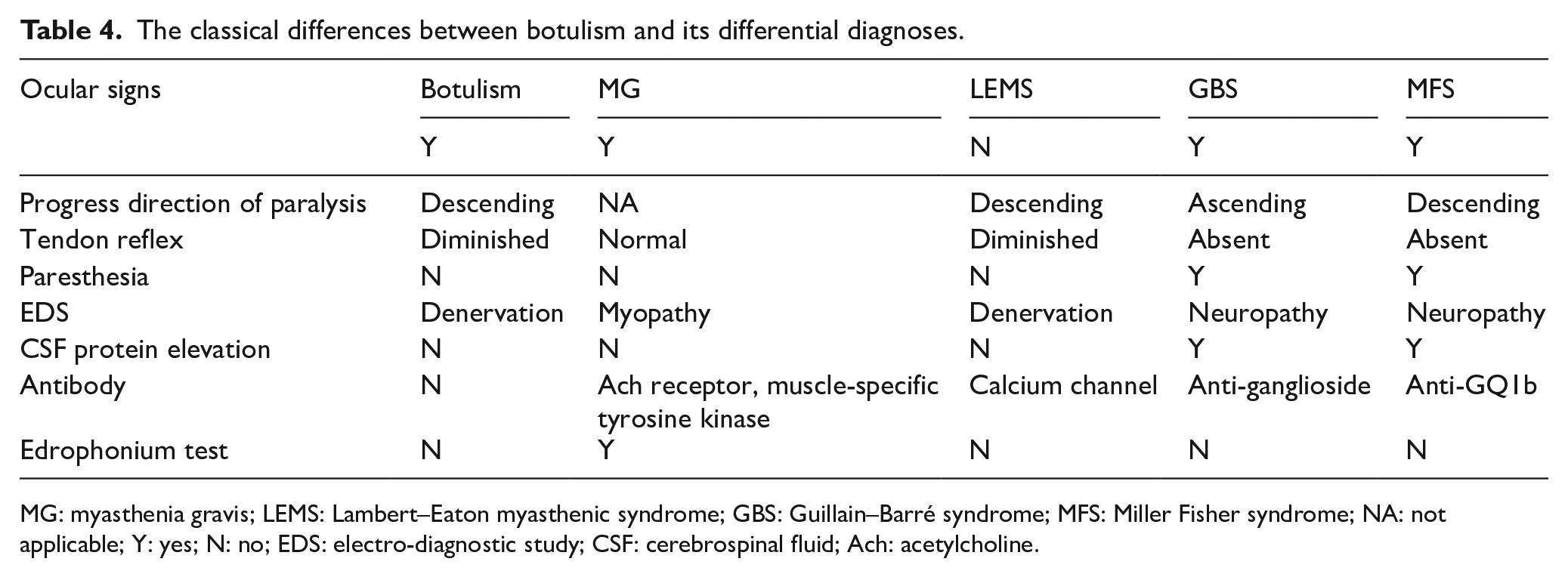
MG: myasthenia gravis; LEMS: Lambert–Eaton myasthenic syndrome; GBS: Guillain–Barré syndrome; MFS: Miller Fisher syndrome; NA: not applicable; Y: yes; N: no; EDS: electro-diagnostic study; CSF: cerebrospinal fluid; Ach: acetylcholine.
Prevention
Caution should be exercised regarding the appropriate doses specific to the formulation and the sites of injection, concentration after reconstitution, time interval between treatment sessions, injection into the muscle rather than the adjacent tissues, as well as previous treatment response. In poor responders, excessive doses should be avoided. Antibody development to botulinum toxin A may be a reason of poor response after prior multiple injections, and toxin B can be considered. 103 Patients with neuromuscular diseases such as MG, Lambert–Eaton syndrome, and myopathies are particularly susceptible to adverse events of botulinum toxin. Patients with anterior horn disorders are also reported to have a hypersensitivity to botulinum toxin. Botulinum toxin is absolutely contraindicated in patients with known hypersensitivity to the components of the formulation and relatively contraindicated in patients with disorders of the neuromuscular junction and co-administration with medications that interfere with neuromuscular transmission, for example, aminoglycoside antibiotics, streptomycin, quinidine, and cholinesterase inhibitors. 44
Footnotes
Author contributions
H.T.F. is responsible for concept and design, data collection, drafting up the text body, and proof reading the final version. K.M.C. is responsible for revising the text body. S.K.T.L. is responsible for revising the text body.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
Data are from case reports, series and media report and materials from relevant literatures.
