Abstract
Introduction:
Administration of botulinum toxin is an increasingly popular procedure in the medical and aesthetic field. There is emerging evidence that it can influence fibroblast activity and minimise tension around the scar by virtue of muscular chemoimmobilisation. This review aims to explore the current evidence base behind the treatment of keloid scars with botulinum toxin.

Methods:
A detailed literature review was conducted using PubMed Medline, Embase and Web of Science databases. Manuscripts were appraised and classified in accordance with the Joanna Briggs Institute Levels of Evidence by an independent consultant in evidence synthesis. The results of this search are presented in descending order of evidence for botulinum toxin as a primary management agent as well as a secondary adjunct following extralesional keloid excision.
Discussion:
On the basis of level 1 evidence, botulinum toxin appears to be equivalent to triamcinolone in producing a short-term reduction in keloidal volume, height and vascularity. A number of level 1 and 2 studies also suggest that botulinum toxin may be particularly helpful in alleviating symptoms of keloid associated pain and itch. There are currently limited studies appraising the value of botulinum toxin in the postoperative management of keloid scars.
Conclusion:
Botulinum toxin may represent a promising agent in the management of keloid scars. However, further research involving large-scale studies with comparative designs and long-term follow-up is warranted to delineate the value of this therapeutic modality in scar management protocols.
Lay Summary
Botulinum toxin (BXT) is a popular product used in a variety of medical and aesthetic interventions. Its mechanism of action relies on temporarily paralysing the muscles in the target area as well as influencing the activity of a variety of cell types. We undertook this study to ascertain whether BXT is a helpful agent for the management for keloid scars. We conclude that there is some supporting evidence that BXT can be as helpful as steroids in improving the troublesome characteristics of keloid scars including bulk and redness. Additionally, it appears to have a notably beneficial effect in alleviating the problematic symptoms of pain and itch. The evidence behind the role of BXT for the management of keloids after surgical removal is very limited at present.
Introduction
Keloid scars represent a challenging clinical entity and are believed to represent the extreme spectrum of scar hypertrophy due to a number of pathophysiological processes including fibroblast hyperactivity; they differ from hypertrophic scars in that excessive tissue proliferation occurs beyond the margins of the original wound, while no spontaneous regression occurs over time.1,2 A number of different management strategies have been described in the literature including intralesional/extralesional excision,3,4 corticosteroid injection,5,6 irradiation 7 and cryotherapy; 8 however, the ideal therapeutic approach has not been unequivocally reached.
Botulinum toxin (BXT) is a protein neurotoxin produced by the anaerobic spore-forming bacterium Clostridium botulinum, 9 which has been utilised for various indications including blepharospasm, strabismus, hyperhidrosis and facial lines. 1 While seven immunologically distinct serotypes exist, BXT-A remains the most widely used in clinical practice. In exerting its paralytic effects, BXT-A prevents synaptic acetylcholine vesicle neuroexocytosis via the proteolytic cleavage of presynaptic membrane associated synaptosomal nerve-associated protein 25. 1 One of the emerging applications of BXT includes scar management, with a number of proposed beneficial actions including the alleviation of muscular tension alongside interference with fibroblast activity.
Tension across a healing wound/scar is considered a key factor in facilitating formation of keloids. By virtue of paralysing the neighbouring muscle mass, BXT has the potential to reduce the tensile forces created during the process of wound healing, which, according to mechanobiology principles, can attenuate the inflammatory response/associated cellular metabolic activity believed to underpin scar hypertrophy. 10
A number of studies have provided evidence for the role of BXT in the modulation of fibroblast activity through a variety of mechanisms including:
Decreased levels of transforming growth factor (TGF)-β1 and connective tissue growth factor (CTGF) causing inhibition of fibroblast proliferation.11 –13
Altered expression of genes involved in keloidogenesis including S100A (upregulation), TGF-β1, VEGF, MMP-1 and PDGFA (downregulation). 14
Modulation of the fibroblast cell cycle. 15
Prevention of fibroblast-to-myofibroblast differentiation. 16
Applications of botulinum toxin in wound healing and scarring
There are a number of studies in animals and humans that provide supporting evidence for the use of BXT in the management of scars. One of the first animal studies investigated the effects of BXT on the cosmetic appearance of unfavourably oriented scars on the foreheads of six Cynomolgus macaque monkeys. 17 An assessment performed by a panel of blinded facial plastic surgeons at three-month follow-up revealed a statistically significant superior aesthetic outcome in favour of toxin-treated wounds as evaluated on a 10-cm Visual Analogue Scale (VAS; P < 0.01). 17
With regards to clinical use in humans, a number of studies have focused on the application of BXT-A on healing in facial wounds. A split-scar, double-blinded randomised control trial (RCT) of 14 patients undergoing facial reconstructive surgery revealed that the early postsurgical administration of BXT-A (compared with 0.9% normal saline) yielded narrower facial surgical scars at six-month follow-up (P = 0.001). 18 In a further two studies, the postoperative administration of BXT-A was found to produce a statistically significant improvement in median VAS score compared to placebo at 6- and 12-month follow-up (P = 0.003 and P < 0.001, respectively).19,20 Another study of 30 patients receiving BXT-A injection within five days after primary closure of vertical forehead lacerations in comparison to placebo showed an improvement in Vancouver Scar Scale (VSS) scores (P = 0.003) and a reduction in wound width (P = 0.007); these findings were further supported by improved quantitative scar discolouration measurements (P < 0.0001) at six-month follow-up. 21
The beneficial effects of BXT-A on scar quality have been similarly reported in the cleft lip field. One double-blinded RCT of 58 patients with lip scars reported the administration of BXT-A to result in an improvement in VAS score and reduced width in postsurgical scars (compared with normal saline; both P < 0.001). 22 Another study involving 60 patients revealed that the immediate postoperative administration of BXT-A produced cleft lip scars ranking more highly with regards to quality and aesthetic appearance based on six-month postoperative photography. 23
In addition, the efficacy of BXT-A in the surgical management of hypertrophic scars has been similarly demonstrated in a double-blinded RCT of 17 median sternotomy patients. Results at six-month follow-up showed an improvement in VSS score and scar width measurement in comparison to normal saline (P < 0.05). 24
In another split-scar double-blinded RCT involving 15 patients with fresh thyroidectomy scars, the use of BXT-A within 10 days of surgery significantly improved the quality of postsurgical scars as measured via a modified Stony Brook Scar Evaluation Scale in comparison to 0.9% saline (P < 0.001). 25 A preliminary report on the use of BXT-A in hypertrophic scar healing has also identified a favourable effect. 27
A meta-analysis involving the collation of nine RCTs on the use of BXT-A for the prevention of hypertrophic scarring in the maxillofacial/neck area has also been identified in our literature search. 26 A statistically significant difference was identified in favour of toxin-treated wounds in terms of scar width (three articles; P = 0.003), patient satisfaction (two articles; P = 0.006) and VAS score (four articles; P < 0.00001). It should be noted that several of the study findings within this meta-analysis have been presented within the preceding section of our review.19,20,22,25 In considering the emerging value of BXT in the management of scars, this work aims to evaluate the current available evidence on the efficacy of BXT in the management of keloid scars.
Methodology
A detailed literature review was conducted in PubMed Medline, Embase and Web of Science databases using the following keywords: ‘botulinum AND scar’ and ‘botulinum AND keloid’. Our search retrieved 87 citations; all abstracts were screened by both authors to ensure relevance. A total of 11 studies (nine English and two Chinese) were identified in accordance with the inclusion criteria (human studies involving patients presenting with one or more keloid scars). The two Chinese articles were translated via a professional academic translation service before inclusion in the study. The selected papers were appraised and classified according to the Joanna Briggs Institute Levels of Evidence with the help of an independent research consultant in evidence synthesis. Collectively, the findings of this review are presented separately for BXT as a primary agent (Table 1) and as a postsurgical adjunct (Table 2) in descending order of evidence.
Botulinum toxin as a primary management agent.

IL, intralesional; VAS, Visual Analogue Scale; VSS, Vancouver Scar Scale.
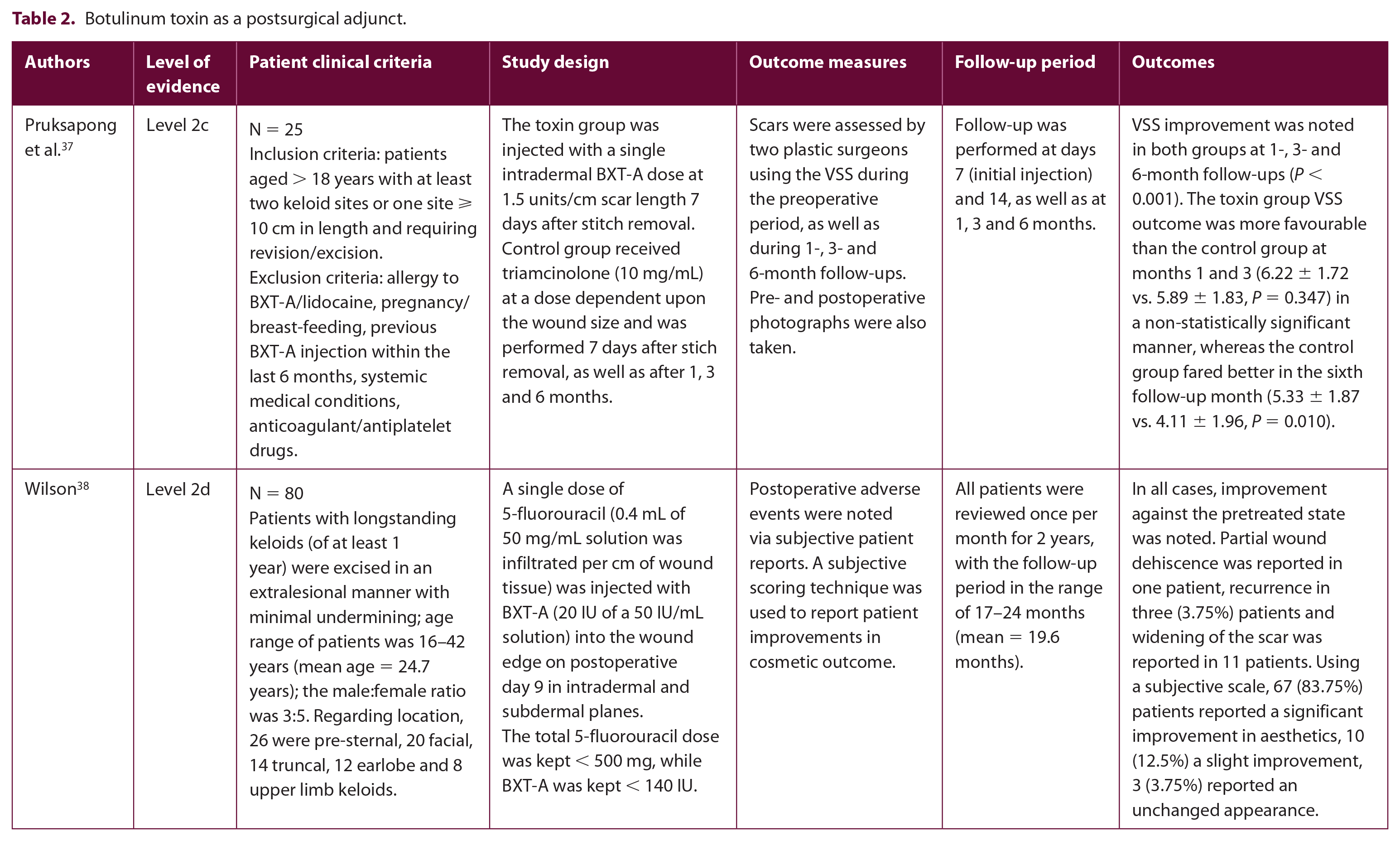
Botulinum toxin as a postsurgical adjunct.
Results
Botulinum toxin as a primary management agent
Level 1
Shaarawy et al. 28 performed a randomised double-blinded comparative study in 24 female patients to evaluate the effects of repeat BXT-A injection versus intralesional (IL) corticosteroid therapy on keloid scars (level 1c). Patients were randomised via a computer-generated random sequence into two groups: group A, receiving IL steroid (10 mg/mL triamcinolone) repeated every four weeks for six sessions, and group B, receiving IL BXT-A at 5 IU/cm3 repeated every eight weeks for three sessions. The total treatment duration was six months with a non-significant difference between groups with regards to baseline age, disease duration, Fitzpatrick skin type and keloid volume (P = 0.201, 0.957, 0.673 and 0.940, respectively). Assessment was performed via the recording of objective parameters (hardness, elevation and redness), subjective complaints (itching, pain and tenderness) on a scale of 0 (none) to 3 (maximum complaint), as well as measurements of keloid volume. Patient satisfaction was evaluated (unsatisfied, satisfied and highly satisfied) and photographs were taken before and seven months after treatment. A reduction in keloid volume in all patients against baseline was reported (82.7% for group A, 79.2% for group B; P < 0.01), with a similar trend observed with respect to a decrease in height and redness (P < 0.01); however, no statistically significance difference was noted between groups. Interestingly, softening of all patients’ lesions was reported at seven-month follow-up, albeit to a greater extent in group A (hardness score of 0.167 ± 0.389 vs. 1.25 ± 0.621 for groups A and B, respectively, P < 0.01). Results regarding the subjective improvement of itching, pain and tenderness assessment were more favourable in group B and were noted having started two months after the initiation of treatment (P < 0.01). On completion of the study, the percentage of highly satisfied patients was greatest in those receiving BXT-A (75% vs. 50%). Overall, treatment was generally well tolerated by patients in both groups. Skin atrophy and telangiectasia were evident in three patients receiving IL steroids during the follow-up period (25%), while no patients receiving BXT-A suffered post-study side effects.
Level 2
Rasaii et al. 29 performed a study with 23 patients (a total of 40 skin lesions), comparing the effects of IL triamcinolone acetonide injection plus placebo-normal saline (group 1) vs. IL triamcinolone acetonide plus BXT-A (group 2) (level 2c). A maximum of 40 mg triamcinolone (20 mg/mL) and 20 U BTX-A was injected per session for all patients, and a single injection was given every four weeks for a total of 12 weeks (three injections in total). To assess the effects of BXT-A, keloid height, vascularity, pigmentation and pliability were assessed via a blinded investigator at baseline, sessions 1–3 and at one-month follow-up. All variables were scored using the VSS apart from pain and itch severity, which were measured using a VAS. No statistically significant difference against baseline was reported between groups with regards to lesion height, vascularisation or pliability score; nevertheless a significant decrease in pain and pruritis score was noted (P < 0.001). Although these data appear to indicate that IL triamcinolone alone and in combination with BXT-A yield similar short-term beneficial effects with regards to a reduction in keloidal height, redness and pliability, the statistically significant influence on symptomatic relief is of particular interest. However, the very short follow-up period employed represents a major limitation.
In a similar study, Li et al. 30 conducted a randomised single-blinded study of 32 patients with a total of 85 skin lesions with dimensions ⩽ 4 cm in diameter and ⩽ 2 cm in height. The three limbs of this study comprised: group A, receiving IL betamethasone with BXT-A; group B, receiving IL betamethasone with fluorouracil; and group C, receiving IL betamethasone alone (level 2c). Steroid was injected at a volume of 1 mL (containing 6.43 mg of betamethasone dipropionate and betamethasone 2.63 mg sodium phosphate), while fluorouracil was administered as 0.5 mL of 2.5 g/L and BXT-A delivered at volumes of 0.5 mL. In the combination groups, all agents were mixed before loading into a high-pressure needleless injector. Injection spots were spaced 1 cm apart and three treatments were performed every two weeks. Of particular note, group A mixtures included BXT-A (max dose 100 U) only in the first treatment, while the remainder were steroid based alone. Keloid volume was measured via the creation of lesion-specific alginate impression moulds, and hardness was assessed via a durometer (average of three measurements). Itch and pain were measured using a VAS and lesion appearance was graded using a patient subjective scale. Keloid photographs were also obtained. On study completion (six weeks), no statistically significant difference was noted among groups with regards to lesion volume, hardness and appearance (P > 0.05). Although statistical significance was demonstrated between groups A and C for pain and itch (P ⩽ 0.05), this was not observed between groups A and B, or B and C (P > 0.05). The authors concluded that the combination of betamethasone with BXT-A resulted in a more favourable effect on the sensory symptoms associated with keloid scars when compared to betamethasone alone.
In another study, Popescu et al. 31 performed a placebo-controlled study of 42 patients with lesions in the range of 5–11 cm in length (level 2c). For each patient, one half of a keloid scar was injected with BXT-A (2 U in each of multiple points, each separated by 2 cm), while the other half was injected with normal saline. Scar appearance was evaluated by blinded observers at three and six months and patient satisfaction assessed using a VAS. On examination, the tendency to form keloid scars was maintained in the regions of scar tissue injected with normal saline. Conversely, areas treated with BXT-A demonstrated more pliable scar formation, and a significant aesthetic improvement at both three- and six-month follow-up (P < 0.017 and P < 0.0222, respectively). Subjective patient satisfaction was additionally rated as being either good or excellent for the BXT-A scar segments. Overall, the authors suggested that the use of BXT-A may represent a suitable alternative to conventional keloid treatments, using low BXT-A doses. However, no follow-up period beyond six months was employed to assess the longevity of the observed effect as well as keloid scar recurrence.
Zhou et al. 32 conducted a study with 58 patients to compare the additive effects of using BXT-A in conjunction with IL betamethasone and topical hyaluronic acid in the treatment of a single 1–5 mm thick lesion (level 2c). Participants were divided into two groups: group 1, receiving IL betamethasone with BXT-A and topical hyaluronic acid (joint treatment); and group 2, receiving IL betamethasone and topical hyaluronic acid (control). Betamethasone (a mixture of betamethasone dipropionate and sodium phosphate) was injected at a concentration of 0.2 mL/cm3 (maximum 1 mL/lesion) once every four weeks for 12 weeks in total, while BXT-A (100 U/mL) was injected around the keloid lesion at 1-cm intervals (4 U per point); hyaluronic acid was applied daily on the keloid surface. Adverse reactions and photographs were recorded both before and after treatment, while the severity of pain and itching were recorded using a VAS once per month. Keloid thickness was also assessed via colour Doppler ultrasonography (average of three measurements); the re-emergence of itching and/or keloid thickening was assessed subjectively by the patient once a week and then at the three-month post-injection follow-up. On completion, a greater reduction in VAS score was noted in the joint treatment group (51.3% reduction) at three-month follow-up, while a statistical significance was identified between both groups at the one-, two- and three-month assessments (P < 0.05). Additionally, the joint treatment group was found to fare better with regards to the reduction of scar thickness (P < 0.05). Of note, subjective complaints were reported as having recurred in control group patients at two weeks after injection, but not in the joint treatment group. Although patients in the control group and seven patients in the joint treatment group suffered an adverse reaction (including telangiectasia, hyper/hypopigmentation and purpura), this incidence was not statistically significant between the two groups (P > 0.05); self-limiting localised subcutaneous congestion at the site of injection was reported in one patient receiving BXT-A.
Zhibo et al. 33 performed a prospective, uncontrolled study in 12 patients to evaluate the effects of BXT-A for the treatment of keloid scars (level 2d). IL BXT-A at 35 U/mL (with the total dose in the range of 70–140 U per session) was administered at three-month intervals for a maximum of nine months in total. Response to therapy was assessed via a patient satisfaction 5-point scale ranging from no improvement to excellent, alongside photographic records. Additionally, observations were made by an independent investigator with regards to keloid size, height, induration and flattening; assessments were performed at the start of BXT-A injection (day 1), at one, three and twelve month follow-up. Overall, therapeutic outcomes with BXT-A were deemed excellent in three, good in five and fair in four patients (i.e. no patients experienced failure of therapy). Keloid regression from the periphery, lesion flattening and a reduction in keloid size were noted in all patients, while one-year follow-up revealed no signs of relapse. However, despite the authors having concluded towards BXT-A being an effective and safe treatment for the management of keloids, the study itself comprised a small sample size and lacked an appropriate negative control.
Level 4
Gauglitz et al. 34 investigated the effects of IL BTX-A for the treatment of four patients with therapy-resistant keloids (level 4c). The total dose of IL BXT-A was in the range of 70–140 Speywood units (Azzalure – approximately equivalent to 28–56 U BXT based on a 2.5:1 Dysport to BXT ratio) per session every two months for a maximum of six months (three injections in total). A 3D optical profiling system was used to objectively measure differences in keloid height and volume at baseline as well as after each injection session. In the present study, repeat BXT-A injection was not shown to significantly alter the macroscopic appearance, morphology or keloid size/height. In line with these clinical data, expression of the proliferating cell nuclear antigen marker following BXT-A therapy was not observed, and the transcriptional activity of genes excessively activated in keloid scars was unchanged.
In another study, Robinson et al. 35 investigated the effects of IL BXT-A for the treatment of 12 patients with resistant keloids over a five-year study period (level 4c). This study involved the use of 20–100 IU of toxin (dose dependent on lesion size and location) with eight patients receiving concurrent alternating intradermal triamcinolone therapy alongside BXT-A. Overall, the observation of a completely flattened keloid scar was reported after an average of 11 months course of BXT-A injections (range = 2–43 months). Interestingly, reported side effects included keloid recurrence (two patients), skin atrophy with ulceration (due to concurrent steroid therapy; one patient) and ulcer formation resulting in keloid recurrence (after intense pulse light therapy; one patient).
In another study, Uyesugi et al. 36 reported a single case of a large anterior chest wall keloid (level 4d)treated with a total of 100 U of BXT-A injected throughout the scar in a fan like distribution. At five-week follow-up, the patient reported having experienced a significant improvement in subjective symptoms and substantial reduction in pain. Interestingly, visual inspection revealed no change in keloid appearance; nevertheless light touch examination revealed reduced allodynia of the scar and surrounding skin.
Botulinum toxin as a postsurgical adjunct
Level 2
Pruksapong et al. 37 performed a prospective controlled study in 25 adult patients presenting with at least two keloid sites or one site exceeding 10 cm in length, requiring scar revision/excision (level 2c). The scars were excised completely under local anaesthetic and primary wound closure was performed with subcuticular closure or simple interrupted stitches in higher tension areas. To evaluate the effects of BXT-A, each patients’ two scar sites were randomised to either toxin or control group. The toxin group was injected with a single BXT-A intradermal dose (1.5 U/cm scar), seven days after stitch removal, while the control group was injected with triamcinolone acetonide (10 mg/mL) seven days after stitch removal (initial injection), repeated in the first, second and third month thereafter. Follow-up was performed at day 7 (initial injection) and day 14, as well as one-, three- and six-month periods using a VSS by two blinded plastic surgeons. No statistically significant difference was found in the mean preoperative VSS between the control and toxin groups and both groups showed a statistically significant improvement with both interventions at all follow-up timescales. The VSS outcome in the toxin group was reported as more favourable than the control group at months 1 and 3 in a non-statistically significant manner (6.22±1.72 vs. 5.89±1.83, P = 0.347), whereas the control group fared better in the sixth follow-up month (5.33 ± 1.87 vs. 4.11 ± 1.96, P = 0.010). However, a number of limitations exist within this study including the protocol design, which incorporates a single dose of BXT-A in one arm and repeated steroid injections in the control arm.
Another study in this category of evidence (level 2d) reported the effects of a single prophylactic 5-fluorouracil dose (0.4 mL of 50 mg/mL per cm wound with a maximum dose < 500 mg) in combination with BXT-A (20 IU with a maximum dose < 140 IU) nine days following keloid excision.38 In total, 80 patients presenting with longstanding keloids of at least one year in duration and a variety of underlying aetiologies were studied, including surgery, trauma, cosmetic piercing and cutaneous infections. All keloids had previously undergone therapeutic attempts involving triamcinolone injection (72 patients), topical silicone gel (32 patients), previous surgical excision (30 patients), laser resurfacing (four patients) as well as irradiation (34 patients), and were followed up once monthly for a mean period of 19.6 months (range = 17–24 months). A subjective post-treatment improvement was noted in most cases, with 83.75% of patients reporting significant improvement, 12.5% slight and 3.75% no improvement. Scar widening was identified in 11 (13.75%) patients alongside 3 (3.75%) cases of recurrence. Transient local adverse reactions, including pruritus, pain, burning and residual post-inflammatory hyperpigmentation, were noted in 26.3% of patients during post-injection weeks 2–4.
Discussion
The rationale for the application of BXT in the management of keloid scars rests on its potential to minimise tension around the scar as well as influence the activity of fibroblasts, which are implicated in the pathogenesis of scar formation. Our work investigated the current knowledge basis pertinent to BXT as a potential primary management agent for keloid scars. There is level 1 evidence to suggest that BXT-A may be equivalent to triamcinolone in terms of producing a reduction in volume, height and redness of keloid scars following a seven month follow-up period. Additionally, BXT appears to be associated with more favourable outcomes in terms of pain and itch control, alongside subjective patient satisfaction scores when compared to steroid injections. 28 Three level 2 studies have also suggested there may be additive value to the use of BXT-A with IL steroids in terms of alleviating pain and itch compared to steroid injections alone,29,30,32 as well as of an effect on the reduction of keloid scar volume. 32 Two further studies in this category, one intraindividual intervention/placebo and one prospective uncontrolled, appear to point towards toxin monotherapy rendering pliability to keloid scars at six-month follow-up.31,33 However, one of the main shortcomings of all studies is the short follow-up periods employed (12 months maximum), which provides little supporting evidence with regards to the long-term therapeutic benefit. Another notable limitation is the interexperimental variability in BXT-A and steroid dose used, as well as of the thickness and width characteristics of the keloid scars selected for study inclusion; these factors may account for the observed differences in reported outcomes. However, one of the proposed advantages of BXT-A as a primary agent appears to be its potential association with a reduced incidence of side-effects frequently observed with local steroid injections;32,33 this may form a sensible rationale for the inclusion of toxins in management protocols for keloid scars. Given that the majority of data is currently based on small-scale trials, caution should be taken when interpreting these results.
There are only two level 2 studies appraising the value of BXT-A as a postoperative keloid management adjunct. In the first study, a single intradermal injection of BXT-A seven days after suture removal was found to be inferior to four injections of triamcinolone at six-month follow-up using VSS scores. 31 The second reported encouraging results following the use of a single combined injection of 5-fluorouracil and BXT-A on the ninth postoperative day after extralesional excision of keloid scars. However, despite the long follow-up period employed (mean = 19.6 months), no comparative arm was used. 37 Furthermore, the lack of undermining following extralesional scar excision (as a tension relieving strategy) may be postulated to have contributed to less favourable outcomes given the pivotal role of tension relief in the surgical management of keloid scars. For these reasons, it is very challenging to draw valid conclusions about the value of BXT-A as a postoperative adjunct in keloid treatment at present.
Conclusion
The rationale for the inclusion of BXT in keloid scar management protocols rests on its ability to confer chemoimmobilisation to the underlying musculature and attenuate fibroblast activity, both of which play a role in the pathogenesis of keloid scars. Results so far appear to provide some evidence that BXT may have an equivalent value in improving the height, volume and vascularity of keloid scars, as well as confer a notable symptomatic benefit when compared to steroid injections. However, the currently available data on BXT as a postoperative adjunct after extralesional excision is extremely limited. Significant shortcomings of studies in the field include the small cohort sizes appraised, the short follow-up periods employed as well as of the heterogeneity of outcomes measured. At present, the use of BXT for scar management is perhaps best supported in the context of research trials. Further high-quality studies are therefore awaited to further define the role of BXT in keloid scar management protocols.
Footnotes
Acknowledgements
The authors would like to acknowledge Ewelina Rogozinska, research consultant in evidence synthesis, for her assistance with the categorisation of the included studies into levels of evidence.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
