Abstract
Background:
The effect of oxygen therapy in patients with suspected uncomplicated myocardial infarction and without hypoxemia at baseline was uncertain.
Objectives:
We aimed to perform a rigorous and comprehensive meta-analysis to robustly quantify the efficacy of oxygen therapy in patients with suspected uncomplicated myocardial infarction and without hypoxemia at baseline.
Methods:
We searched EMBASE, MEDLINE, Web of Science, and the Cochrane Library from inception to August 2018. Two researchers screened studies and collected the data independently. Randomized controlled trials were included. The main outcome was mortality at the longest duration of follow-up.
Results:
Seven studies involving 7732 patients (median = 157 patients; range = 72–6629 patients; interquartile range = 136–441 patients; oxygen therapy group = 3856 patients; conservative group = 3876 patients) were included. Compared with conservative therapy, oxygen therapy did not significantly reduce mortality at the longest follow-up (risk ratio = 0.97; 95% confidence interval = 0.68 to 1.40; z = 0.15; p = 0.88), at discharge (risk ratio = 1.04; 95% confidence interval = 0.48 to 2.26; z = 0.11; p = 0.91), 30 days (risk ratio = 1.08; 95% confidence interval = 0.78 to 1.49; z = 0.44; p = 0.66), and 6 months (risk ratio = 0.93; 95% confidence interval = 0.73 to 1.17; z = 0.66; p = 0.51) in patients with suspected uncomplicated myocardial infarction and without hypoxemia at baseline. Furthermore, there was no significant difference between two groups with regard to infarct size (standard mean difference = 0.15, confidence interval = –0.13 to 0.44; z = 1.07, p = 0.28), cardiac troponin levels (standard mean difference = 0.30, confidence interval = –0.09 to 0.69; z = 1.51, p = 0.13), in hospital reinfarction (risk ratio = 1.49, confidence interval = 0.80 to 2.78; z = 1.25, p = 0.21), and new-onset atrial fibrillation (risk ratio = 0.91, confidence interval = 0.69 to 1.18; z = 0.72, p = 0.47). Multiple subgroup analysis, sensitivity analysis, and trial sequential analysis were consistent with overall findings.
Conclusion:
Compared with conservative therapy, oxygen therapy did not decrease the mortality at the longest duration of follow-up, discharge, 30 days, and 6 months in patients with suspected uncomplicated myocardial infarction and without hypoxemia at baseline. Furthermore, large-scale, multicenter studies are needed to confirm our results.
Registration:
PROSPERO, number CRD42017078807.
Background
Myocardial infarction (MI) is one of the main causes of death and disability in the world, resulting in approximately 20% of all deaths in Europe.1,2 The annual incidences of new-onset MI are 550,000 in the United states, leading to 7.8% 30-day mortality. 3 An imbalance of substrate and oxygen supply and consumption during MI causes ischemia in myocardium and finally leads to cardiac cell death.
Since the report of oxygen therapy for angina by Steele more than a century ago, 4 oxygen therapy has been widely used in the early treatment of MI patients. The rationale for oxygen therapy is that supplemental oxygen may increase oxygen supply to the ischemic myocardium, hence limit myocardial injury and subsequent complications. This rationale is supported by some laboratory studies and small participants size non-randomized clinical trials.5–7
However, oxygen therapy may cause coronary vasoconstriction, increase coronary vascular resistance, decrease coronary blood flow, and increase the production of reactive oxygen species resulting in reperfusion injury.8–10 Although there was no debate on supplemental oxygen in patients with MI and hypoxemia at baseline, the effect of oxygen therapy in patients with suspected uncomplicated MI and without hypoxemia at baseline was uncertain.2,3 Many randomized controlled trials (RCT) found no benefits in patients with uncomplicated MI and without hypoxemia at baseline.11–17
Two recently published meta-analyses evaluated the effect of oxygen therapy in MI patients.18,19 However, one study 20 including MI patients with great proportion of hypoxemia at baseline was involved in both meta-analyses. Populations without hypoxemia at baseline are different from those with hypoxemia. Hence, the results of both previous meta-analyses may be biased due to including patients with hypoxemia at baseline. Therefore, we aimed to perform a rigorous and comprehensive meta-analysis to robustly quantify the efficacy of oxygen therapy in patients with suspected uncomplicated MI and without hypoxemia by investigating mortality rates and adverse effects.
Methods
We performed this study in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement 21 and guidelines described in the Cochrane Handbook for Systematic Reviews of Interventions. 22 The study protocol was published online (http://www.crd.york.ac.uk/PROSPERO/display_record.php?ID=CRD42017078807).
Study selection criteria
Types of studies
Randomized studies comparing oxygen therapy and conservative therapy in the treatment of suspected uncomplicated MI without hypoxemia were included. Conservative therapy was defined as titrated oxygen therapy to achieve an oxygen saturation 93%–96% or supplemental room air.
The exclusion criteria were case reports, animal studies, preclinical studies, and observational studies (retrospective studies, case-control studies, or prospective cohort studies).
Types of participants
Adult patients presenting to the hospital or emergency medical service (EMS) with suspected uncomplicated MI without hypoxemia as defined by the authors of each study were involved.
Hypoxemic patients with MI, that were defined as MI patients with an oxygen saturation of pulse oximetry less than 90%, were excluded. And patients younger than 18 years were also excluded.
Types of interventions
Patients in the control group received room air, and those receiving conservative oxygen therapy for achieving oxygen saturation 93%–96% were also included. Patients in the intervention group received liberal oxygen therapy or high concentration oxygen therapy.
Types of outcome measures
Our primary outcome was that all cause mortality at the longest duration of follow-up, the secondary outcomes included the following variables: mortality at discharge, 30 days, and 6 months; infarct size; cardiac troponin levels; in hospital reinfarction; and new-onset atrial fibrillation.
Data sources and search strategy
Researchers searched for relevant studies published in the EMBASE, MEDLINE, Web of Science, and the Cochrane Library. We also reviewed the references of relevant articles to avoid missing any studies. Gray data in the Chinese National Knowledge Infrastructure (CNKI) were also screened. We searched all relevant articles published from inception to August 2018. The details of the search strategy are shown in the supplemental files. There were no limitations on gender, patient sample size, or language.
Data extraction
Two researchers (X.L.Y. and R.Z.) independently and duplicately screened titles and abstracts to evaluate the potential studies. Disagreements were resolved by consensus or by discussion with a third author (J.W.). For the included studies, a full-text review was performed. Detailed study information, interventions, controls, and outcomes were extracted using a standardized data extraction form.
Quality assessment
The qualities of the included randomized studies were assessed according to the methods described in the Cochrane Handbook. 22
Statistical analysis
Our meta-analysis was performed on an intention-to-treat basis and involved all patients who were randomly assigned to any study group. Data were obtained by direct extraction or by indirect calculation.23–25
For binary outcomes, we calculated the risk ratios (RRs) and 95% confidence intervals (CIs). For continuous outcomes, the standardized mean differences (SMDs) and 95% CI were calculated. We graphically displayed the outcomes by forest plots and visually inspected the potential publication bias with a funnel plot.
We used DerSimonian–Laird random-effects models for pooling outcomes. The Mantel–Haenszel model was used for assessment of heterogeneity, with p < 0.05 and I2 > 50% indicating significant heterogeneity.
We performed prespecified subgroup analyses for the primary outcomes, including study types, study settings, confirmed MI, and room air or not in conservative group.
We also used trial sequential analysis to estimate the reliability of our meta-analysis by examining for sufficient data to avoid type I (false-positive) and type II (false-negative) errors. Trial sequential analysis was performed using TSA software (version 0.9.5.9 Beta; Copenhagen Trial Unit, Copenhagen, Denmark), The Lan–DeMets approach was used for construction of the O’Brien-Fleming monitoring boundaries and the optimal information size, which was set to an alpha of 0.05 with a two-sided beta of 0.80 and relative risk reduction of 10%. The mortality incidence in the control group was set according to previously published studies.11–17
Sensitivity analysis
Sensitivity analyses were conducted on the mortality outcomes to test the robustness of the results by the following methods: changing to a fixed-effect model, changing to use of the Sidik–Jonkman method for random effects, shifting to the Biggerstaff–Tweedie method, and removing the highest weighted study.
We used Review Manager Software (Version 5.3, The Cochrane Collaboration, Copenhagen, Denmark), SPSS 13.0 (IBM Corp., Armonk, NY, USA), and TSA software (version 0.9.5.9 Beta, Copenhagen Trial Unit, Copenhagen, Denmark) to conduct the statistical analysis. The Grading of Recommendations Assessment, Development, and Evaluation (GRADE) Guideline Development Tool (GRADEpro; McMaster University 2014, Hamilton, Canada) was used to evaluate the quality of evidence for each outcome. 26 The quality of evidence was stratified into four grades: high, moderate, low, or very low; p < 0.05 was considered statistically significant.
Results
Our study identified 1366 relevant publications. After removing duplicate results and screening the titles and abstracts, 831 publications were rescreened for titles and abstracts. Finally, 26 studies were obtained for full-context review. A total of 19 studies were excluded due to not meeting the inclusion criteria. The details of the excluded studies and reasons for their exclusion are shown in the supplemental files, Table S1. Finally, we included seven studies11–17 with a total of 7732 patients (median = 157 patients; range = 72–6629 patients; interquartile range (IQR) = 136–441 patients; Oxygen therapy group = 3856 patients; conservative group = 3876 patients) in this meta-analysis. Of the seven included studies, the mean age of patients was 66.1 years, of whom 69.9% (range = 62%–84%; IQR = 66%–79%) were men and 30.1% were women (range = 16%–38%; IQR = 21%–34%). The flow rates of oxygen in oxygen therapy group were shown in Table 1. In six of the seven included studies, patients in conservative group received mainly room air, except the study by Ranchord and colleague. The selection process of the eligible studies is shown in Figure 1. The included study characteristics and baseline patient characteristics are shown in Table 1 and Table 2.
Basic characteristics of the included studies.
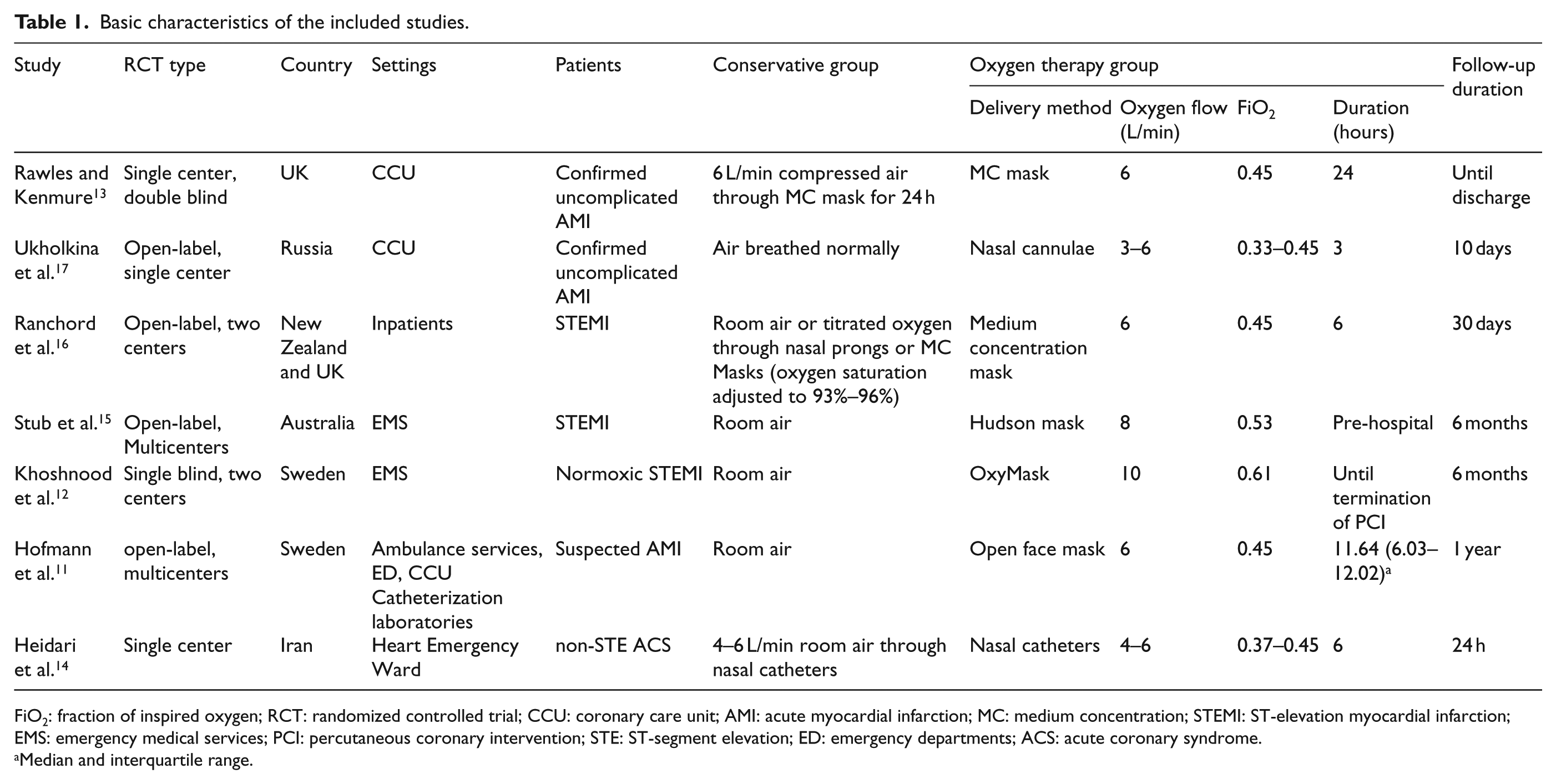
FiO2: fraction of inspired oxygen; RCT: randomized controlled trial; CCU: coronary care unit; AMI: acute myocardial infarction; MC: medium concentration; STEMI: ST-elevation myocardial infarction; EMS: emergency medical services; PCI: percutaneous coronary intervention; STE: ST-segment elevation; ED: emergency departments; ACS: acute coronary syndrome.
Median and interquartile range.
Baseline patient characteristics of the included studies.
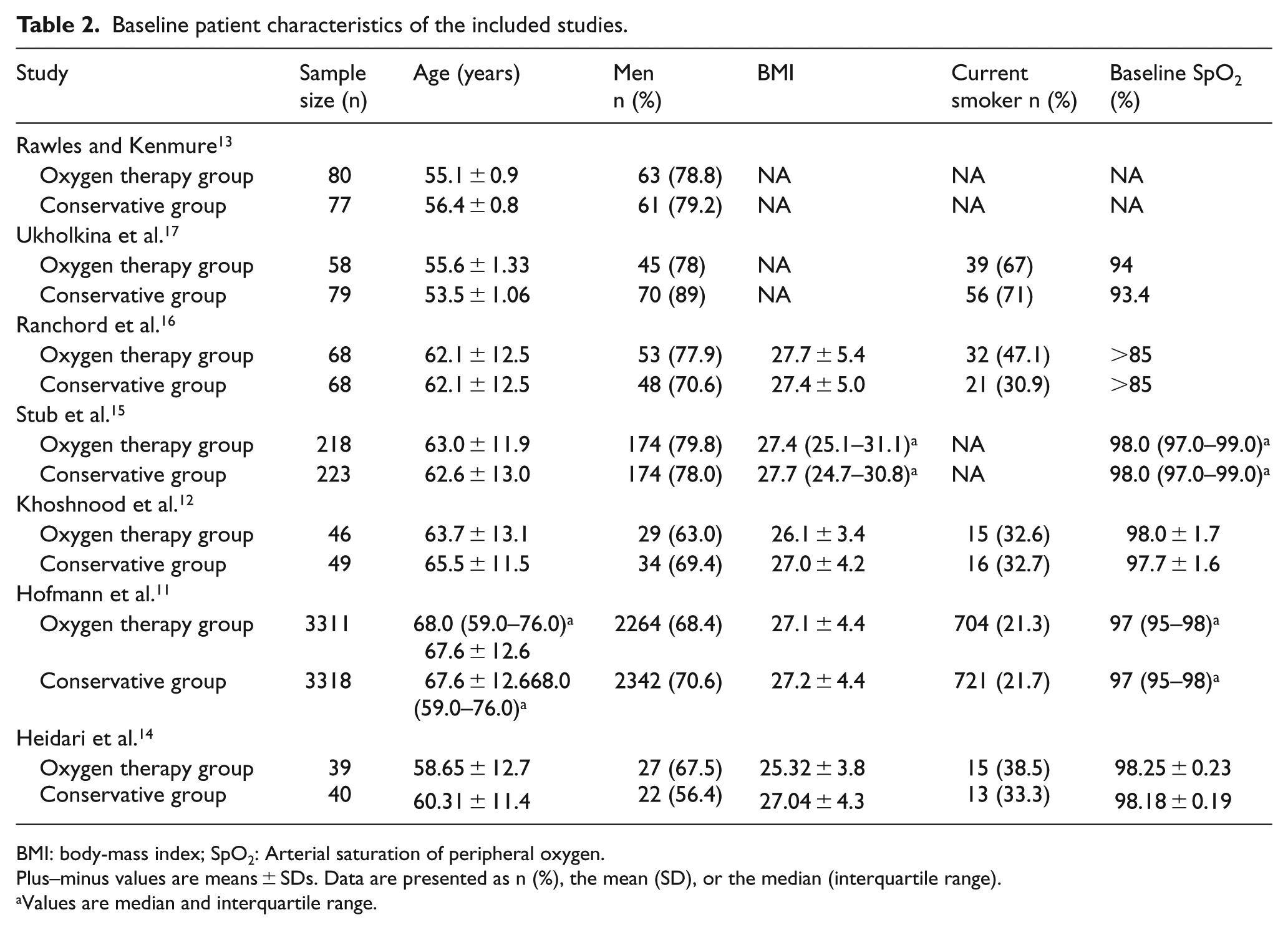
BMI: body-mass index; SpO2: Arterial saturation of peripheral oxygen.
Plus–minus values are means ± SDs. Data are presented as n (%), the mean (SD), or the median (interquartile range).
Values are median and interquartile range.
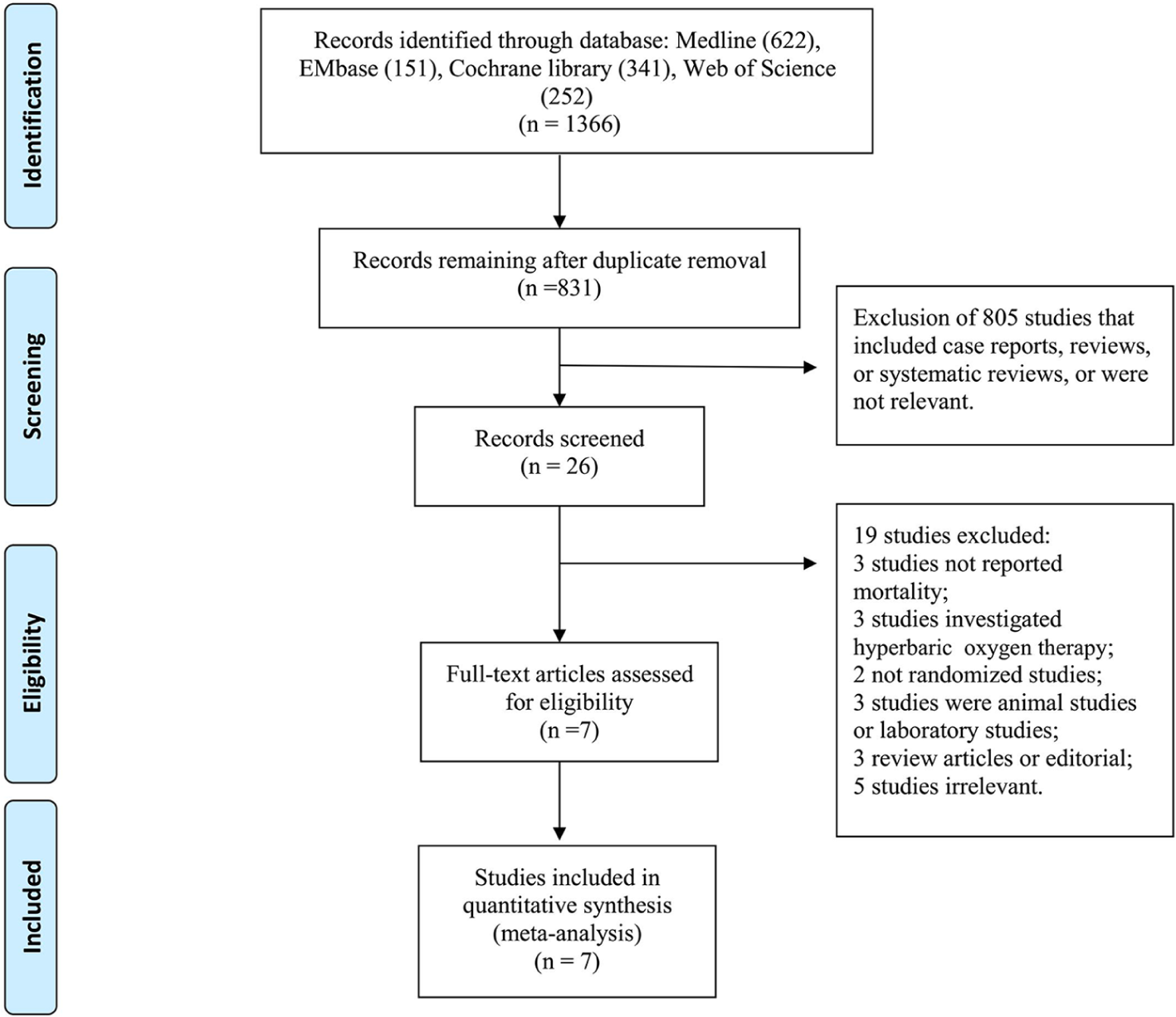
Study selection flow chart.
All seven included RCT studies were considered to be at low risk of bias as evaluated by the Cochrane risk of bias tool (see supplemental files, Figure S1 and S2). 22
Mortality
Mortality data were available from all seven included studies with various follow-up durations.11–17 In the study by Hofmann and colleagues, the duration of patient follow-up was 1 year, which was the longest. In the studies by Stub et al. 15 and Khoshnood et al., 12 the duration of patient follow-up was 6 months. In the studies by Heidari et al., 14 the follow-up duration was 24 h. In the studies by Ukholkina et al. 17 and Ranchord et al., 16 the follow-up duration were 10 days and 30 days, respectively. When these data were pooled together, there was no heterogeneity among studies (χ2 = 0.36, p = 0.36, I2 = 9%, Figure 2) and the oxygen therapy group did not show a significant mortality reduction compared with that of the conservative group (RR = 0.97; 95% CI = 0.68 to 1.40; z = 0.15; p = 0.88; Figure 2).
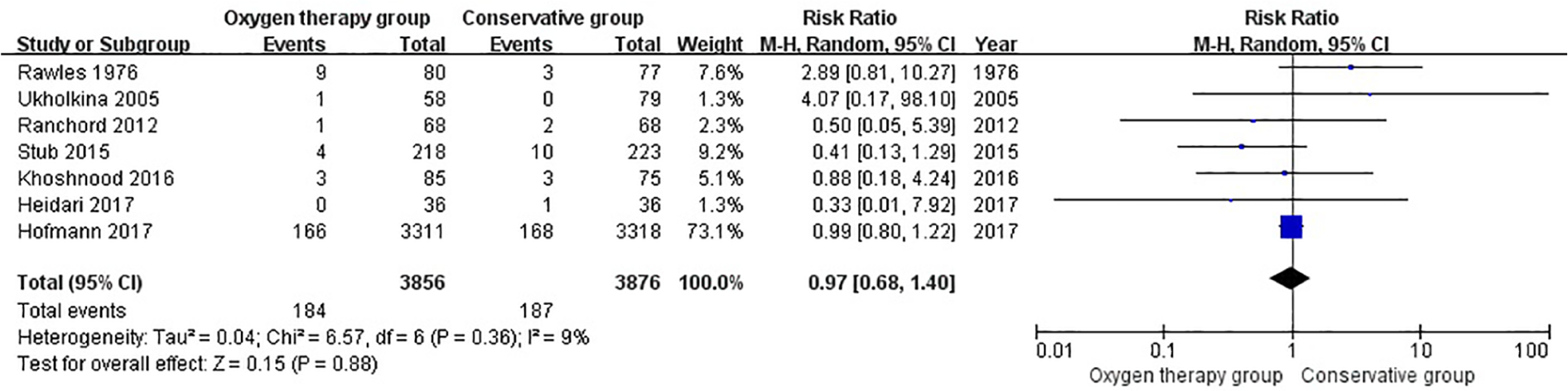
Comparison of mortality between oxygen therapy group and conservative group at the longest follow-up.
Subgroup analyses demonstrated no significant interactions with study type (single center RCT RR = 2.31 (95% CI = 0.77 to 6.98) vs multicenter RCT RR = 0.96 (95% CI = 0.78 to 1.17), pinteraction = 0.12), study settings (coronary care unit (CCU) RR = 3.03 (95% CI = 0.93 to 9.83) vs non-CCU RR = 0.95 (95% CI = 0.78 to 1.17), pinteraction = 0.06), confirmed MI (confirmed MI RR = 0.94 (95% CI = 0.41 to 2.14) vs suspected MI RR = 0.99 (95% CI = 0.80 to 1.22), pinteraction = 0.90) and room air in conservative group (room air RR = 0.99 (95% CI = 0.61 to 1.60) vs not room air RR = 0.50 (95% CI = 0.05 to 5.39), pinteraction = 0.58) (Table 3, Figure S3–S6).
Subgroup analysis for mortality outcomes (displayed with risk ratio).
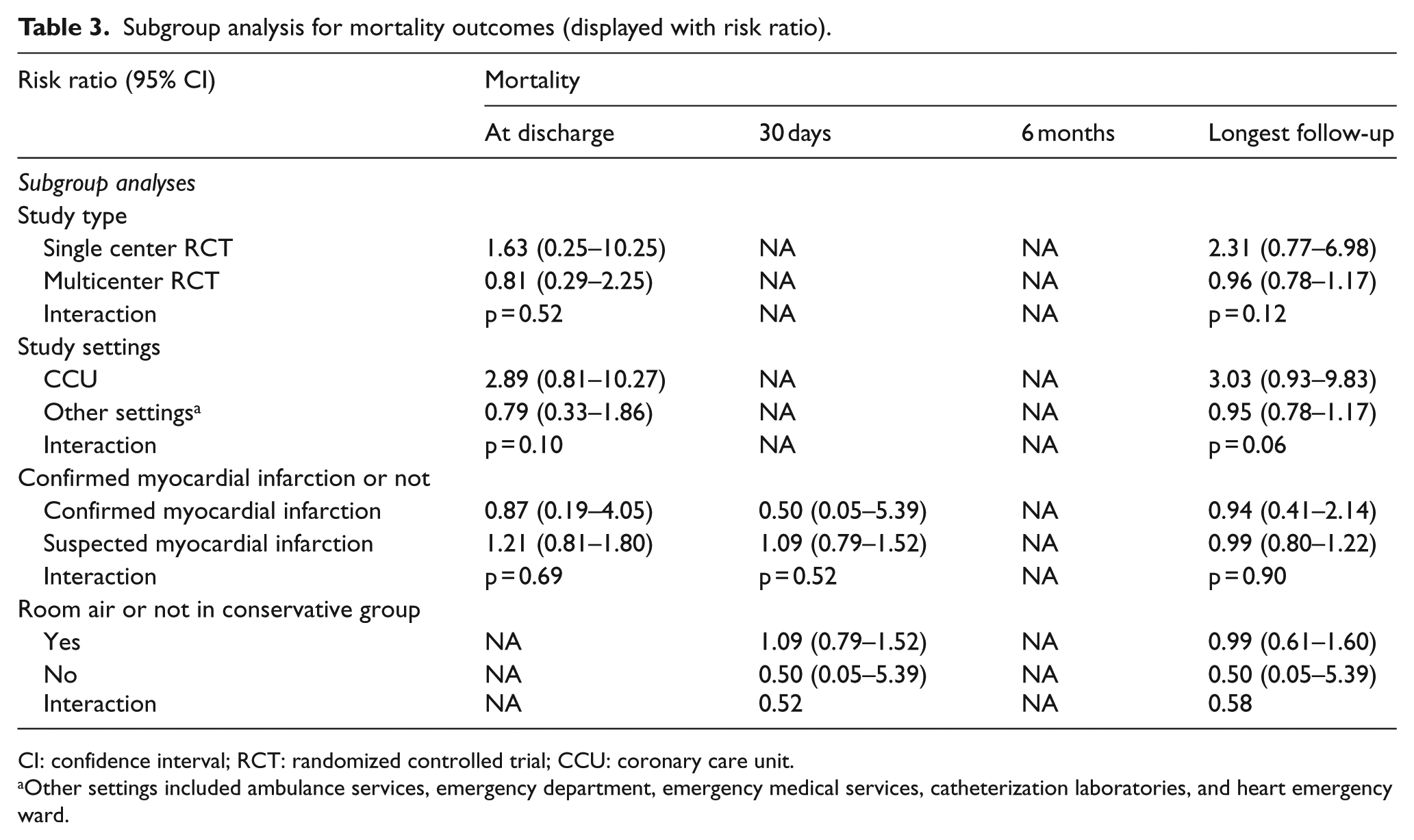
CI: confidence interval; RCT: randomized controlled trial; CCU: coronary care unit.
Other settings included ambulance services, emergency department, emergency medical services, catheterization laboratories, and heart emergency ward.
The results were robust to sensitivity analyses, including changing to a fixed-effect model, Sidik–Jonkman effect model, or Biggerstaff–Tweedie effect model and removing the highest weighted study (Table 4, Figure S7). However, for the primary outcome, trial sequential analysis showed that the cumulative Z curve did not reach the O’Brien-Fleming monitoring boundary, futility boundary, and the optimal sample size (Figure 3). Hence, a decisive conclusion should be made cautiously.
Sensitivity analysis of the mortality outcomes.
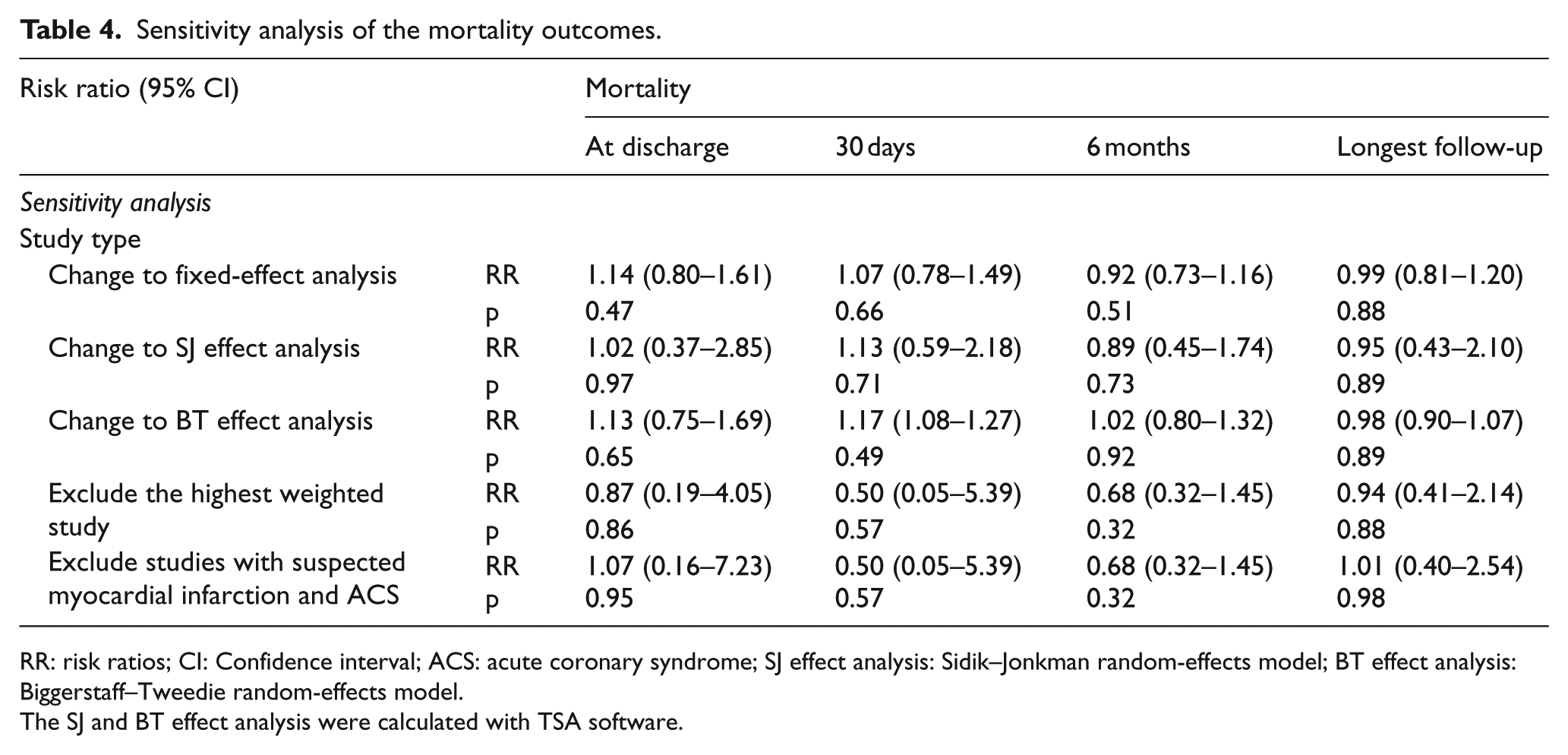
RR: risk ratios; CI: Confidence interval; ACS: acute coronary syndrome; SJ effect analysis: Sidik–Jonkman random-effects model; BT effect analysis: Biggerstaff–Tweedie random-effects model.
The SJ and BT effect analysis were calculated with TSA software.
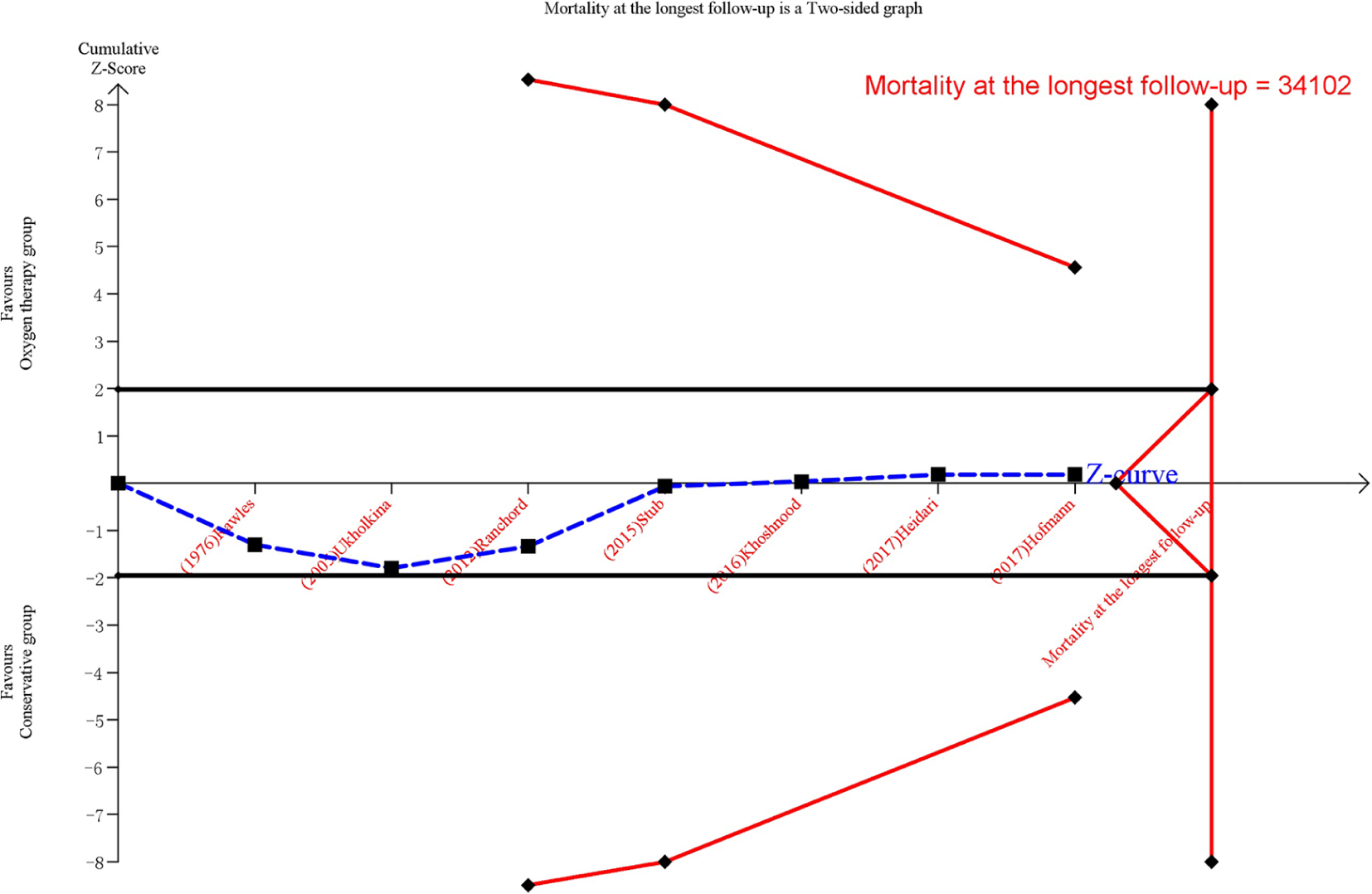
Trial sequential analysis for mortality at the longest follow-up.
Oxygen therapy also did not significantly decrease the risk of death compared with conservative therapy at discharge (RR = 1.04; 95% CI = 0.48 to 2.26; z = 0.11; p = 0.91; Figure S8), 30 days (RR = 1.08; 95% CI = 0.78 to 1.49; z = 0.44; p = 0.66; Figure S9), and 6 months (RR = 0.93; 95% CI = 0.73 to 1.17; z = 0.66; p = 0.51; Figure S10). Subgroup analysis also demonstrated there were no significant interactions with regard to study types, study settings, confirmed MI and room air, or not in conservative group. Trial sequential analysis were consistent with overall findings (Table 3, Figure S11–S15). Sensitivity analysis and trial sequential analysis did not change the overall findings for mortality outcomes (Table 4, Figure S16–S21).
Infarct size
Three studies reported the infarct size according to the cardiac magnetic resonance imaging. When the results were pooled together, there was moderate heterogeneity among studies and there was no significant difference between two groups (SMD = 0.15, CI = –0.13 to 0.44; z = 1.07, p = 0.28; I2 = 31%, Figure 4). The heterogeneity might be due to various detecting time points and treatment measures.
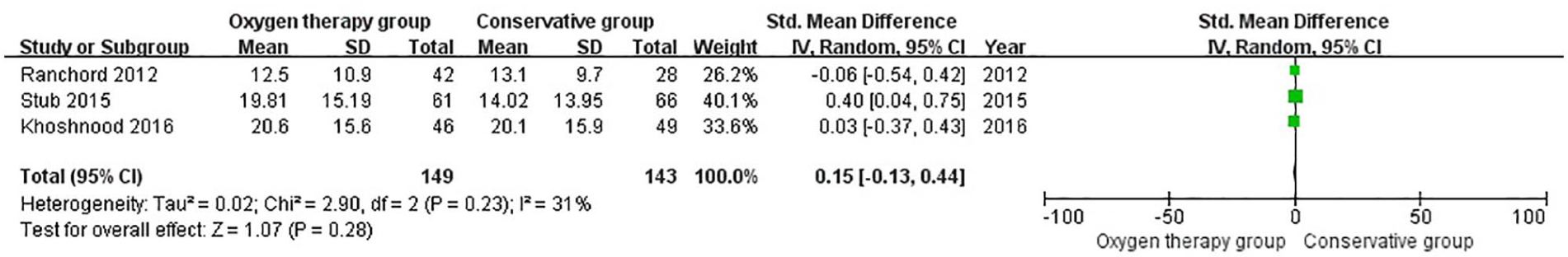
Comparison of infarct size between oxygen therapy group and conservative group according to MRI in suspected uncomplicated myocardial infarction without hypoxemia.
Cardiac troponin levels
Of the seven included studies, five studies reported the cardiac troponin levels. When the results were pooled together, there was no significant difference between two groups (SMD = 0.30, CI = –0.09 to 0.69; z = 1.51, p = 0.13, Figure 5). However, this result should be interpreted with great caution due to significant heterogeneity among studies (χ2 = 55.07, df =4, p < 0.001, I2 = 93%). The heterogeneity might be due to various detecting time points and clinical treatment measures among the involved studies.
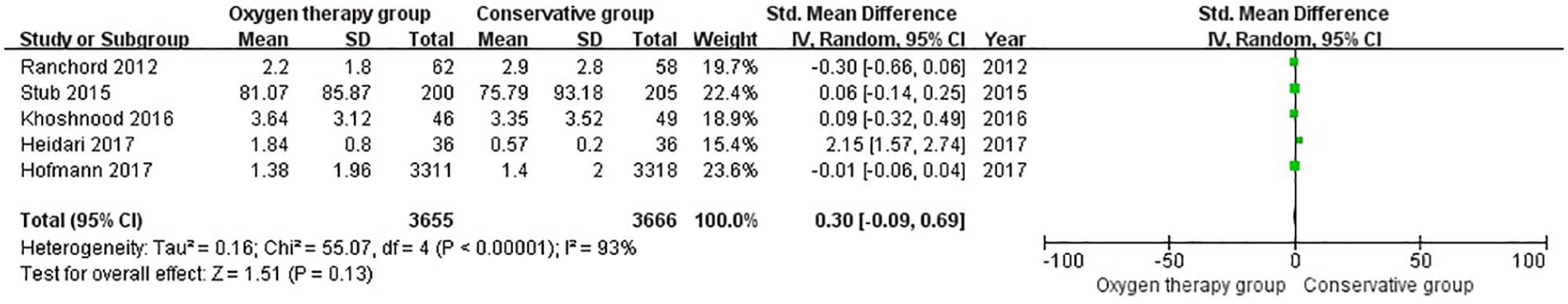
Comparison of cardiac troponin levels between oxygen therapy group and conservative group in suspected uncomplicated myocardial infarction without hypoxemia.
In hospital reinfarction
Five studies reported the recurrence of MI during hospital stay. There was no significant difference between the two groups (RR = 1.49, CI = 0.80 to 2.78; z = 1.25, p = 0.21, Figure 6). And there was no significant heterogeneity among studies (χ2 = 5.63, df = 4, p = 0.23, I2 = 29%).
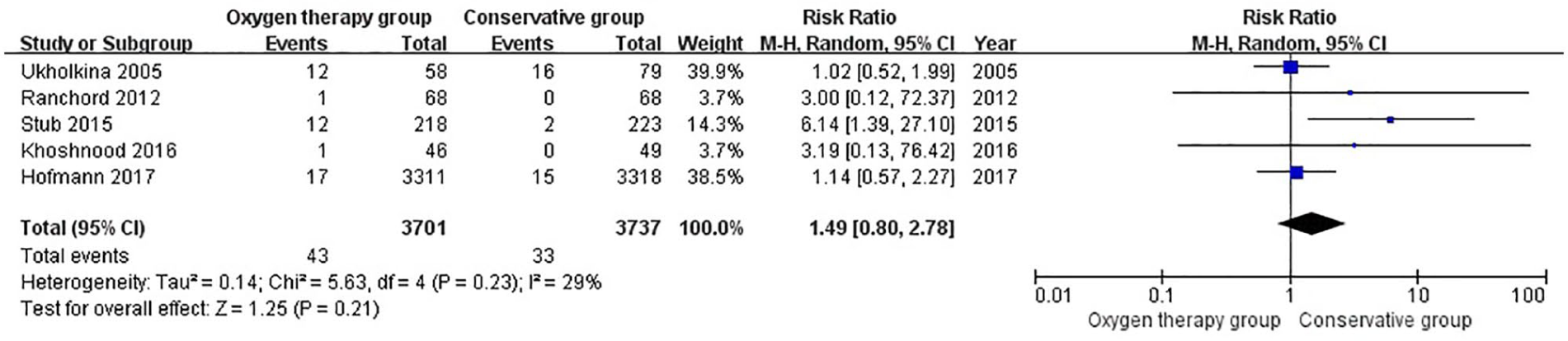
Comparison of in hospital reinfarction between oxygen therapy group and conservative group in suspected uncomplicated myocardial infarction without hypoxemia.
New-onset atrial fibrillation
New-onset atrial fibrillation was reported as complications of suspected MI in three studies. When the results were aggregated, there was no significant difference between the two groups (RR = 0.91, CI = 0.69 to 1.18; z = 0.72, p = 0.47, Figure 7).
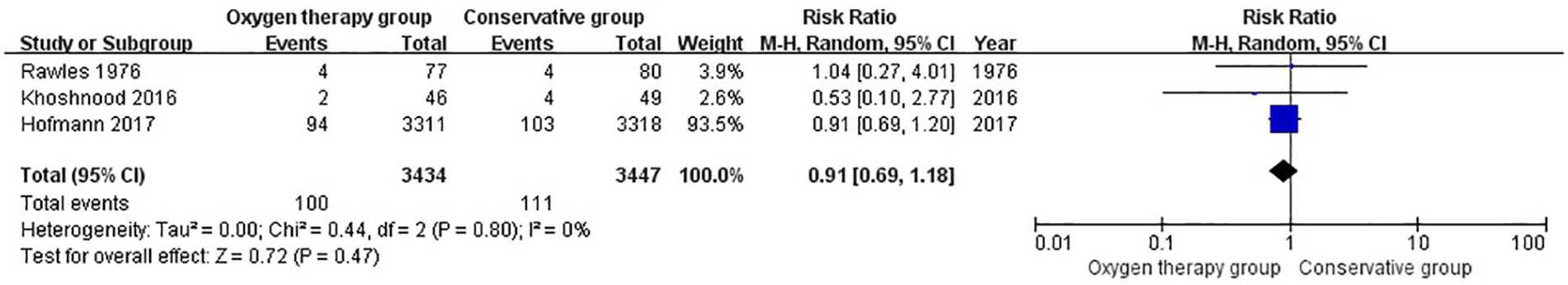
Comparison of new-onset atrial fibrillation between oxygen therapy group and conservative group in suspected uncomplicated myocardial infarction without hypoxemia.
Visual inspection of the funnel plot did not show any publication bias (Figure S22–S29). Summary of findings with GRADE system were shown in Table 5.
Summary of findings.
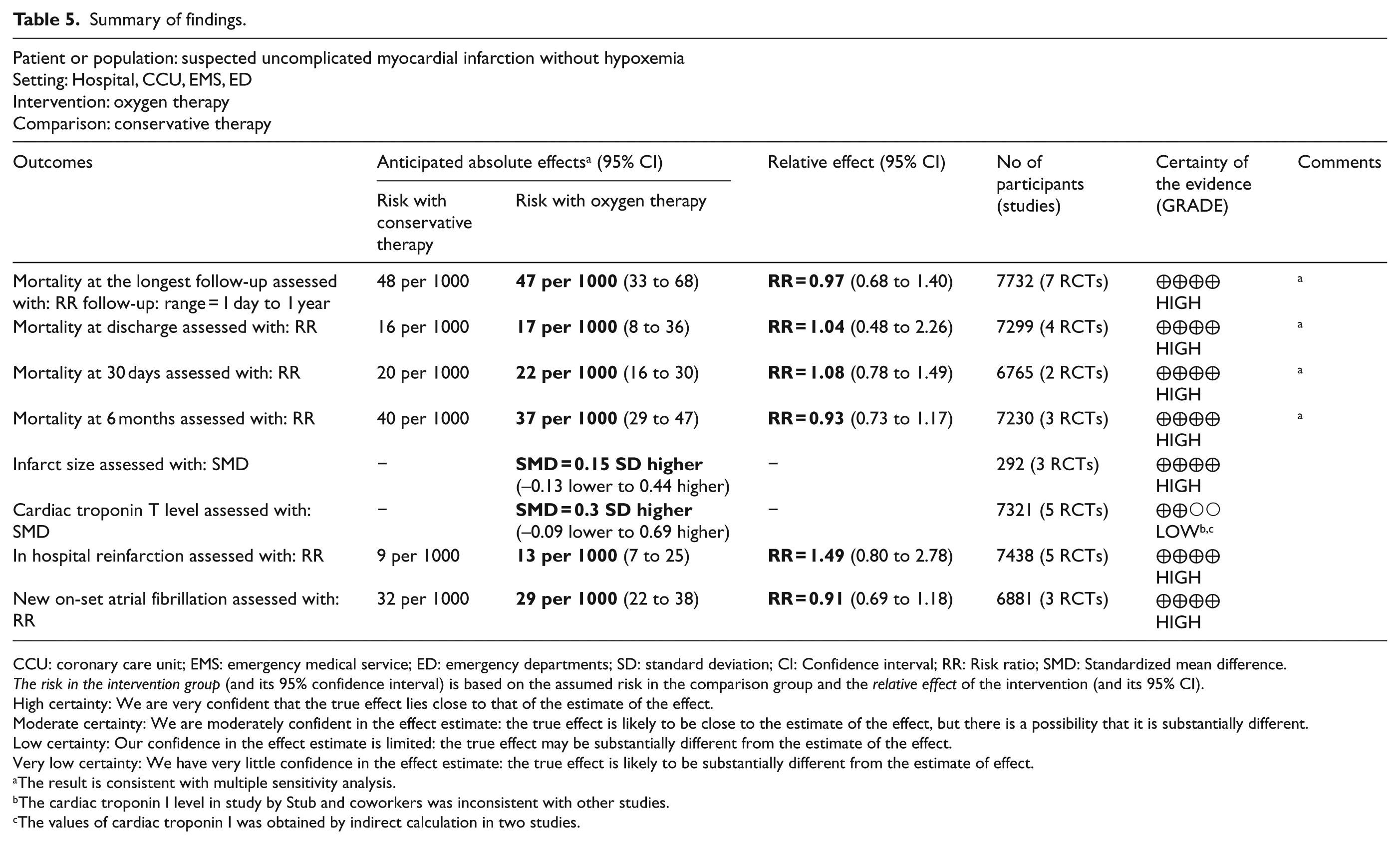
CCU: coronary care unit; EMS: emergency medical service; ED: emergency departments; SD: standard deviation; CI: Confidence interval; RR: Risk ratio; SMD: Standardized mean difference.
The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
High certainty: We are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: We are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low certainty: Our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect.
Very low certainty: We have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect.
The result is consistent with multiple sensitivity analysis.
The cardiac troponin I level in study by Stub and coworkers was inconsistent with other studies.
The values of cardiac troponin I was obtained by indirect calculation in two studies.
Discussion
This meta-analysis, including 7732 MI patients (Oxygen therapy group = 3856 patients; conservative group = 3876 patients), demonstrated that compared with conservative therapy, oxygen therapy did not significantly reduce mortality at the longest follow-up (RR = 0.97; 95% CI = 0.68 to 1.40; z = 0.15; p = 0.88), at discharge (RR = 1.04; 95% CI = 0.48 to 2.26; z = 0.11; p = 0.91), 30 days (RR = 1.08; 95% CI = 0.78 to 1.49; z = 0.44; p = 0.66), and 6 months (RR = 0.93; 95% CI = 0.73 to 1.17; z = 0.66; p = 0.51) in patients with suspected uncomplicated MI and without hypoxemia at baseline. Furthermore, there was no significant difference between two groups with regard to infarct size (SMD = 0.15, CI = –0.13 to 0.44; z = 1.07, p = 0.28), cardiac troponin levels (SMD = 0.30, CI = –0.09 to 0.69; z = 1.51, p = 0.13), in hospital reinfarction (RR = 1.49, CI = 0.80 to 2.78; z = 1.25, p = 0.21), and new-onset atrial fibrillation (RR = 0.91, CI = 0.69 to 1.18; z = 0.72, p = 0.47).
Previous animal and human studies have demonstrated that high concentration oxygen can increase vasoconstriction, promote inflammation, and produce oxidative stress on multiple organs, such as cardiovascular, pulmonary, and neurological systems.8–10 These adverse effect may offset the benefits of supplemental oxygen in patients with suspected uncomplicated MI and without hypoxemia. Furthermore, some studies showed that supplemental oxygen might decrease the vigilance of medical staffs and delay identification of deterioration of diseases due to pseudo-normalization of peripheral blood oxygen saturation.27,28
Our results are consistent with previously published meta-analysis,18,19 demonstrating no benefits with oxygen therapy in patients with suspected uncomplicated MI and without hypoxemia at baseline. Furthermore, our study had advantages over previous meta-analysis by adopting more rigorous inclusion criteria and focusing on patients without hypoxemia at baseline. In the study by Wilson and coworkers, which was included in both of the previously published meta-analysis, there was 48% patients with moderate hypoxemia (SpO2 < 90%) and 19% patients with severe hypoxemia (SpO2 < 80%) at baseline. 20 Therefore, results of the previously published two meta-analysis might be biased. After carefully reviewing and discussion, the study by Wilson and coworkers was excluded in our study.
The results of this meta-analysis were robust. We performed an up-to-date and comprehensive search and adopted rigorous inclusion criteria. The methodology in our study was rigorous. And the findings with regard to mortality outcomes were consistent in multiple subgroup analysis. Furthermore, the results were supported by multiple sensitivity analysis. However, for the primary outcome, trial sequential analysis showed that the cumulative Z curve did not reach the O’Brien-Fleming monitoring boundary, futility boundary, and the optimal sample size (Figure 3). Hence, a decisive conclusion should be made cautiously. Furthermore, large-scale, multicenter studies are needed to confirm our results.
There were some limitations in our study warranting consideration. First, there were variations in the duration of oxygen therapy among the included studies, which could affect our results. To address this problem, subgroup analyses and multiple sensitivity analyses were performed. And the subgroup results remained consistent with the overall findings. Multiple sensitivity analysis including changing effect models, excluding the highest weighted study, did not change the overall results (Table 4). Second, the settings of included studies varied (Table 1). However, a subgroup analysis of this study did not find any interactions with regard to the study settings (Table 3). Furthermore, the variations in the RCT settings in our study enabled our results to have general external validity. Finally, the populations in the present meta-analysis are patients with suspected uncomplicated MI and without hypoxemia at baseline. It means that the populations involved in this study are different from those with hypoxemia or cardiac shock. So the findings of our study should be only applied in the type of populations the present meta-analysis studied.
Conclusion
In this meta-analysis, compared with conservative therapy, oxygen therapy did not decrease the mortality at the longest duration of follow-up, discharge, 30 days, and 6 months in patients with suspected uncomplicated MI and without hypoxemia at baseline. Furthermore, there was no significant difference between two groups with regard to infarct size, cardiac troponin T level, in hospital reinfarction, and new-onset atrial fibrillation. Furthermore, large-scale, multicenter studies are needed to confirm our results.
Supplemental Material
Supplemental material for Oxygen therapy versus conservative therapy in suspected uncomplicated myocardial infarction without hypoxemia: A meta-analysis of randomized controlled studies
Supplemental material, Supplemental_material for Oxygen therapy versus conservative therapy in suspected uncomplicated myocardial infarction without hypoxemia: A meta-analysis of randomized controlled studies by Rui Zhang, Youfeng Zhu, Min Zhang, Haiyan Yin and Jianrui Wei in Hong Kong Journal of Emergency Medicine
Footnotes
Acknowledgements
We acknowledge all the people who helped us perform this systematic review and meta-analysis.
Authors’ Note
Jianrui Wei is also affiliated with Guangzhou Women and Children’s Medical Center, Guangzhou, China.
Author contributions
All authors conceived the study and contributed to the study design. H.Y. and R.Z. collected and extracted the data. Y.Z. and J.W. performed the analyses. Y.Z., H.Y., and M.Z. performed the literature review. All authors contributed to the writing of the draft and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (NSFC81871585) and the Natural Science Foundation of Guangdong Province (2018A030313058).
Availability of data and material
The data sets are available from the corresponding author upon reasonable request.
Informed consent
Written informed consent was not necessary because no patient data have been included in the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
