Abstract
Background
While there is a clear trend towards less invasive spinal procedures in recent years, determining what specifically constitutes minimally invasive surgery (MIS) for spinal fusion procedures can be challenging as published definitions are heterogeneous and address only select factors.
Purpose
To utilize a Delphi Consensus Panel (DCP) process to build consensus among panelists regarding the definition of MIS for spinal fusion and define a consensus around best practices for adopting MIS technologies.
Methods
DCP is a widely accepted method of qualitative data collection and analysis used for generating consensus from panel members. The DCP consisted of 2 online questionnaires completed by 11 expert panelists. Round 1 included 7 multiple choice demographics and 11 open-ended questions. Round 1 responses were analyzed to inform the development of statements for further assessment in Round 2. Round 2 included 93 closed-ended, 5-point Likert scale questions and 11 open-ended questions. Consensus was defined as agreement among at least 8 out of 11 panelists.
Results
Consensus was reached on 56 out of 93 statements regarding spinal fusion and/or fixation procedures. There was a high degree of consensus around the role of reduced tissue trauma, patient recovery time, hospital length of stay, and post-operative pain for MIS spinal fusion procedures compared to open procedures; and the value of various technologies in performing MIS spinal fusion procedures.
Conclusion
A consistent definition of what constitutes MIS for spinal fusion may help drive consistency across surgeons, facilities, and payers to define clinical and economic implications for these approaches.
Keywords
Introduction
Over the past several decades, minimally invasive surgery (MIS) has been developed with the aim of providing similar surgical outcomes to traditional open techniques while reducing morbidity in terms of blood loss, tissue damage and recovery time.1–3 Traditional spinal fusion techniques require extensive incisions and may result in substantial muscle damage, leading to back pain, spinal instability, and loss of muscle strength. 4 MIS for spinal fusion aims to limit the impact of surgery, for example by reducing the size of the incision required and using tubular retractors to separate muscle tissue and limit damage.5,6 The benefits of less invasive spinal procedures over traditional open surgery have led to an upward trend in MIS for spinal fusion in recent decades.1,7
While MIS for spinal fusion is becoming more common, the attributes that define this approach are not well delineated, and published definitions are heterogenous. While existing definitions may capture the intention of MIS to reduce tissue damage and improve patient outcomes, they do not specify techniques, approaches, or other factors that would separate a minimally invasive procedure from a traditional open procedure. Furthermore, a spinal procedure that is “less invasive” than open surgery is not necessarily MIS, as “minimally invasive” implies that the approach used was the least invasive approach possible based on available techniques and technology. 8
Without a clear definition of what constitutes MIS for spinal fusion, it is difficult to compare outcomes for MIS with non-minimally invasive procedures, and to evaluate the potential benefits of MIS compared with non-minimally invasive techniques for spinal fusion. 8 For patients, these potential benefits include reduced blood loss during surgery, lower risk of infection, reduced pain, and a shorter recovery time. For healthcare providers, MIS for spinal fusion may lead to reductions in direct costs compared with open procedures, 9 including reductions in risk of surgical site infections, 10 patient length of stay, 11 and pharmacy costs. 11
As the field of MIS for spinal procedures continues to develop, it is important to establish consensus among healthcare providers regarding the definition of MIS for spinal fusion as well as best practices in these procedures. A consensus definition is required to be able to objectively measure and statistically validate outcomes in patients who undergo minimally invasive techniques compared with those undergoing traditional open procedures in spinal surgery. 8 To this end, we conducted a two-round Delphi Consensus Panel (DCP) with 11 spine surgeons with expertise in minimally invasive spinal fusion procedures to obtain consensus regarding the definition of MIS spinal fusion.
DCP is a widely accepted method of qualitative data collection and analysis and is recommended by the Food and Drug Administration (FDA) as a method for generating consensus from a panel of experts on a given topic. A panel of experts is used to increase the qualitative strength of recommendations or consensus. 12 Other key features of a DCP are anonymity, repeated iterations of knowledge elicitation, resolution of differences, advocation of refined opinion and group feedback. 13 It is believed that opinions of individuals collected separately can be more accurate than opinions reached through face-to-face discussion, avoiding potential for groupthink or individual dominance, and allowing each panelist freedom of expression without outside pressure or influence. 14
Materials and methods
Study design
The two-round DCP was administered as two semi-structured web-based questionnaires. While a two-round DCP is not always sufficient to establish consensus, there was a sufficient literature base to draw upon when formulating the questions to be included in the Round 1 questionnaire. 15 A targeted literature search was conducted to identify potential definitions for MIS spinal fusion procedures to inform the Round 1 questionnaire. Gaps were identified in existing MIS definitions and used to inform the Round 1 questionnaire, which consisted of 7 multiple choice demographic questions and 11 open-ended questions (questionnaire was deployed from December 2021 to April 2022). Round 1 panelist responses were analyzed, identifying key themes and concepts to inform the development of consensus statements for further assessment in Round 2.
Panelists were provided a high-level summary of the Round 1 questionnaire results as they completed the Round 2 questionnaire. The Round 2 questionnaire included 93 consensus statements presented as closed-ended, 5-point Likert scale questions (Strongly disagree, Disagree, Neutral, Agree, Strongly agree) and 11 open-ended questions (questionnaire was deployed from May 2022 to June 2022). Round 2 questions were grouped into 3 sections with multiple sub-sections (Figure 1). Round 2 questionnaire structure, developed based on the findings of the Round 1 questionnaire.
Expert panel recruitment
In line with conventional DCPs, which involve panelists who have been selected for their knowledge on a specific topic, spine surgeons with experience in MIS for spinal fusion procedures in the U.S. were invited to the panel using a contact list provided by the sponsor. The surgeons were contacted via a phone and email campaign. A physical mailing campaign was also conducted using a publicly available database of approximately 160 physicians available from the Society for Minimally Invasive Spine Surgery (SMISS). Surgeons with substantial experience in MIS fusion procedures were targeted to ensure the panel was comprised of experts in this field.
Physicians that expressed an interest in participating in the DCP were asked to complete a screening questionnaire and qualifying surgeons were asked to participate in the panel. In order to meet the study inclusion criteria, panelists were required to have a medical degree, be practicing in the U.S., be post-residency or board certified in orthopedic surgery or neurosurgery, and perform at least 4 MIS fusion procedures per month. Panelists were geographically diverse (with representation from 10 states), practiced in a range of institution types (for-profit, not-for-profit, and academic medical centers), and performed MIS fusion procedures in a mix of settings (hospital inpatient and outpatient).
Panelists were offered an honoraria payment of $100 after completing the Round 1 questionnaire and $350 after completion of the Round 2 questionnaire. Nine of the 11 panelists received the total honorarium amount, and two panelists did not request payment.
Data analysis
Round 1 questionnaire responses were analyzed by 2 reviewers using NVivo version 1.6.1 (QSR International, Burlington, MA) qualitative coding and analysis software to identify key themes and concepts. The Round 2 questionnaire 5-point Likert scale question (Strongly disagree, Disagree, Neutral, Agree, Strongly agree) responses were analyzed using Microsoft Excel 2016 (Microsoft Corporation) to calculate the percent of panelists that agree/strongly agree with each of the 93 consensus statements. Prior to the launch of the Round 1 questionnaire, consensus was defined as >70% of participants selecting “Agree/Strongly Agree” for a specific statement.
Results
Panelists
Panelist demographics.
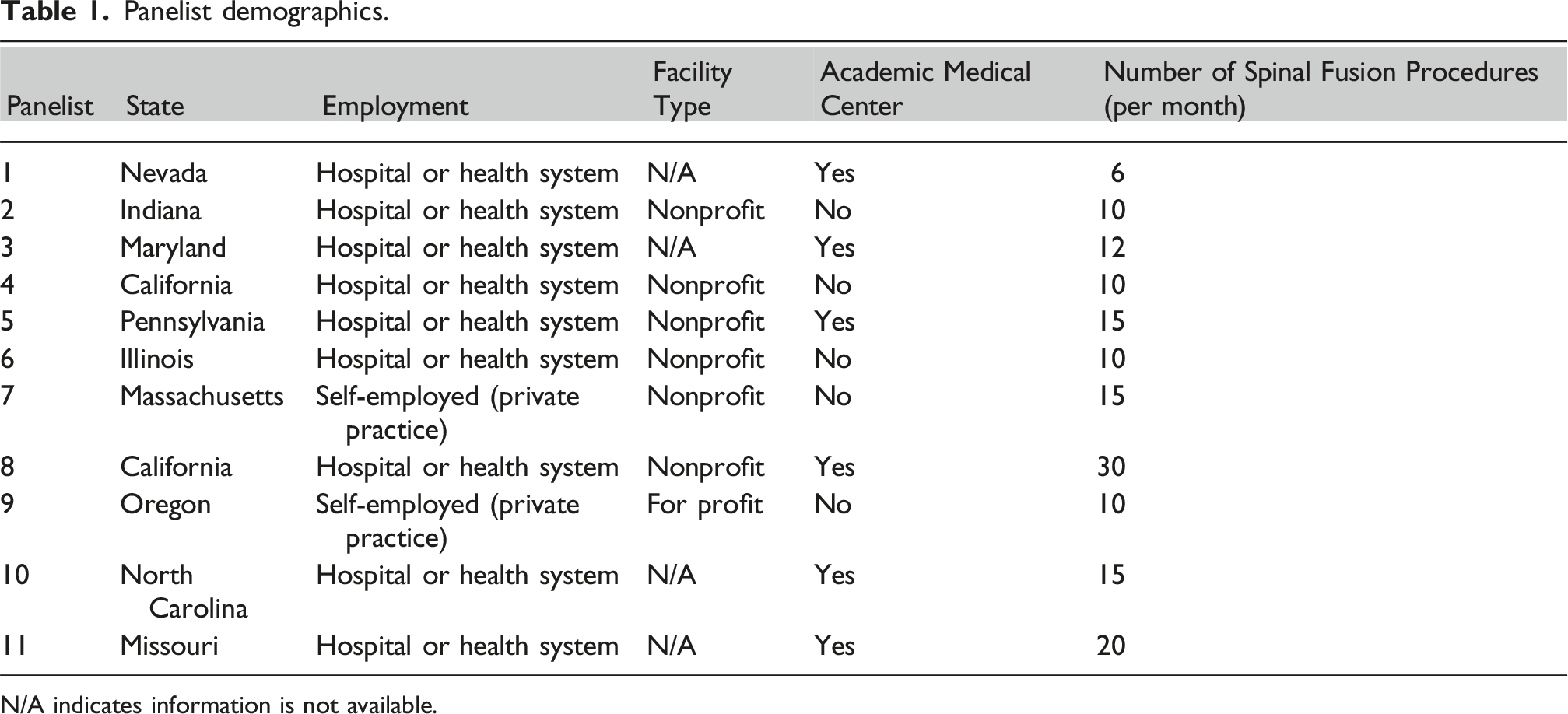
N/A indicates information is not available.
Round 1
A total of 93 consensus statements were identified from panelist’s open-ended responses to the Round 1 questionnaire. Consensus statements fell into the three broad categories: defining spinal fusion MIS, valuable technologies in spinal fusion MIS, and MIS adoption for spinal fusion procedures.
Round 2
Consensus was reached by panelists on 56 out of the 93 statements regarding MIS spinal fusion procedures.
Defining MIS spinal fusion
Tissue trauma and surgical approach consensus statements.

MIS indicates minimally invasive surgery
Anterior Lumbar Interbody Fusion = ALIF; Posterior Lumbar Interbody Fusion = PLIF; Transforaminal Lumbar Interbody Fusion = TLIF; eXtreme Lateral Interbody Fusion = XLIF; Oblique Lateral Interbody Fusion = OLIF.
*Bold text indicates result reaching consensus.
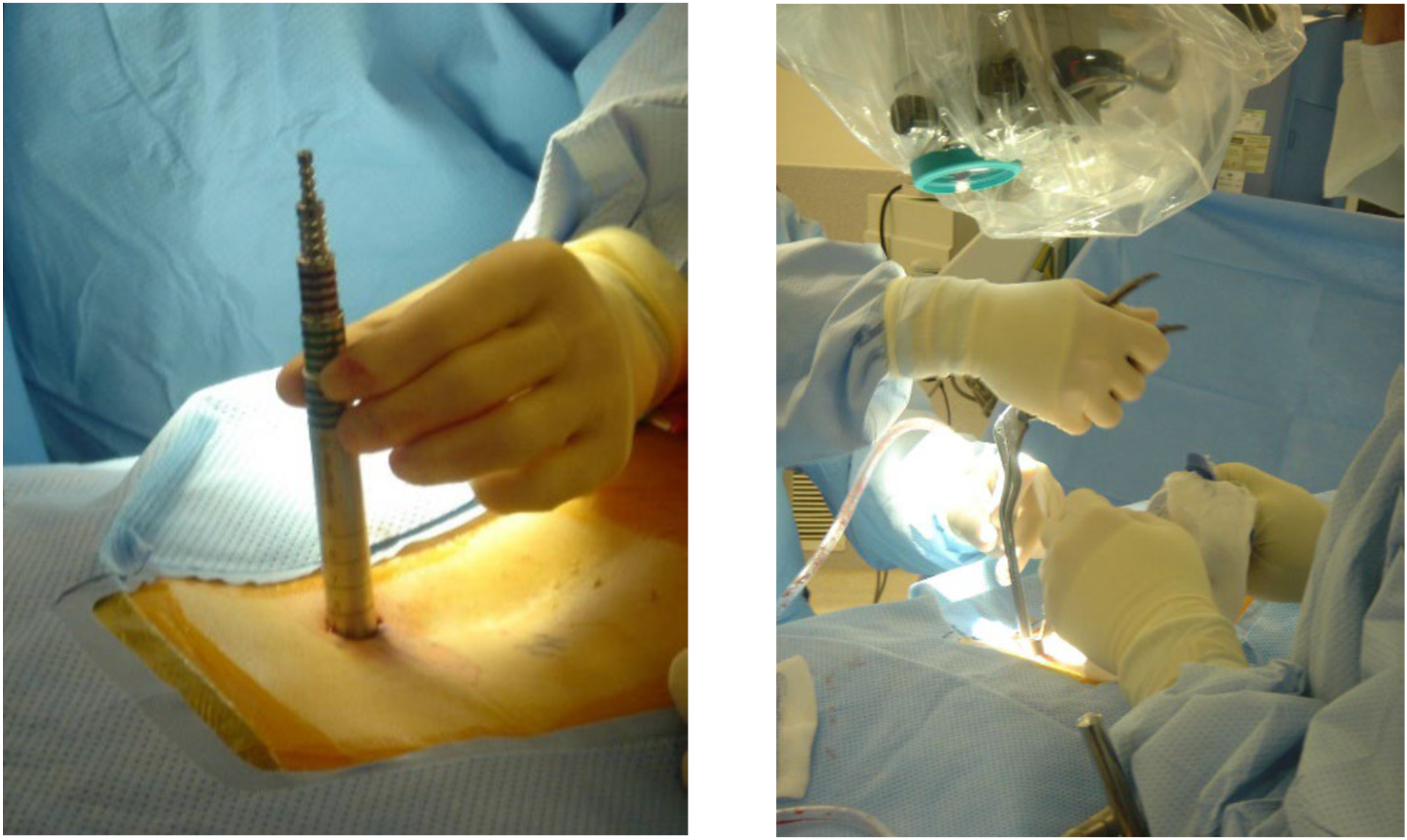
Surgical approach was identified as an important factor for determining whether a spinal fusion and/or fixation procedure can be considered MIS (73%). Specifically, mini-posterior lumbar interbody fusion (mini-PLIF), mini-transforaminal lumbar interbody fusion (mini-TLIF), extreme lateral interbody fusion (XLIF) and oblique lateral interbody fusion (OLIF) were identified as MIS approaches (Table 2). However, there was no consensus regarding whether anterior lumbar interbody fusion (ALIF), posterior lumbar interbody fusion (PLIF), transforaminal lumbar interbody fusion (TLIF), posterior or posterolateral, or posterolateral gutter fusion approaches are MIS approaches. The panel agreed that the method of screw insertion/placement (e.g., percutaneous vs direct vision) was also considered important in determining if a spinal fusion procedure can be classified as MIS (82%). Figures 2 and 3 help illustrate the differences between MIS and non-MIS approaches.16,17 Steps involved in traditional open surgery. Reprinted with permission from: OrthoInfo. https://orthoinfo.aaos.org/en/treatment/minimally-invasive-spine-surgery/. Tubular retractor and operating microscope used during minimally invasive lumbar surgery.
Visualization consensus statements.

MIS indicates minimally invasive surgery.
*Bold text indicates result reaching consensus.
There was no consensus regarding whether spinal fusion procedures performed using intraoperative fluoroscopic image guidance, intraoperative Computed Tomography (CT) computer assisted navigation (CAN), fluoroscopic CAN, robotic navigation, or intraoperative neurophysiologic monitoring (IONM) are minimally invasive.
Panelists were also asked whether they agree with statements related to patient recovery and MIS spinal fusion. There was 100% consensus that MIS spinal fusion should result in reduced patient recovery time, hospital length of stay, and duration of patient postoperative pain in comparison to open surgery. There was 82% consensus that MIS spinal fusion should result in reduced infection rates in comparison to open surgery. There was no consensus regarding MIS spinal fusion and decreased neurologic-complications, cerebrospinal fluid (CSF) leaks, return to emergency room (ER), or pseudarthrosis in comparison to open surgery.
Valuable technologies in MIS spinal fusion
Technology value consensus statements.

MIS indicates minimally invasive surgery; OR, operating room; CT, computed tomography; CAN, computer assisted navigation; IONM, intraoperative neurophysiologic monitoring.
*Bold text indicates result reaching consensus.
There was 100% consensus that patient pathology dictates whether MIS spinal fusion is an appropriate treatment option, and that navigation is more valuable/needed for MIS spinal fusion and fixation procedures in patients with complicated anatomy than in patients with no spinal anomalies. There was consensus that patients with severe spinal deformities are less likely to be a candidate for a MIS spinal fusion procedure (73%); patient goals and preferences dictate whether MIS spinal fusion and/or fixation surgery is an acceptable treatment option (91%); and patient pathology influences the value of CAN when performing MIS spinal fusion procedures (82%).
MIS adoption for spinal fusion
MIS adoption consensus statements.

MIS indicates minimally invasive surgery.
*Bold text indicates result reaching consensus.
Discussion
While the definition of MIS spinal fusion procedures is complex and multi-faceted, panelists reached consensus on several patient-centric statements (e.g., the amount of tissue trauma, patient recovery time, post-operative pain, etc.). Conversely, no consensus was reached on a number of surgery-centric factors (e.g., visualization and navigation) indicating there is not a single set of surgical parameters that define MIS.
Consistent with existing definitions for MIS spine fusion procedures, the overarching consensus was that tissue trauma is an important factor for defining whether a spinal fusion and/or fixation procedure is MIS. 19 Specifically, based on this DCP, the definition of a MIS spine fusion procedure may be expanded to include requirements of less soft tissue damage, less muscle damage, and less blood loss than open surgery. While a number of MIS definitions focus on reduced incision length for MIS,20–22 the DCP indicates that overall tissue trauma is more important to the definition of MIS for spinal fusion procedures than incision length, either of a single incision or combined length of multiple incisions.
Patient pathology, goals and preferences may dictate whether MIS spinal fusion surgery is an acceptable treatment option, and panelists commented that technologies can help expand the number of patients that are candidates for MIS procedures. There was also consensus around a number of patient-centric factors associated with MIS including reduced patient recovery time, hospital length of stay, infection rates, and duration of patient postoperative pain versus open surgery, possibly as a result of reduced tissue trauma, a defining feature of MIS.23,24
While certain visualization and navigation technologies are considered valuable to MIS spinal fusion procedures, consistent with other studies, panelists also agreed that the additional costs (e.g., costs of new technologies) impede adoption of MIS for spinal fusion procedures. 25 Similarly, panelists agreed that challenges with reimbursement for MIS procedures (e.g., no additional reimbursement for MIS procedures compared to open procedures) may also impede adoption of MIS for spinal fusion procedures. 26
The intensive learning period, perceived gaps in clinical evidence comparing MIS with open surgery, paired with cost and reimbursement barriers 26 can make adoption of MIS for spinal fusion procedures challenging regardless of the potential benefits of MIS. It is important to note that the DCP was composed of only MIS-practicing physicians, and findings related to adoption of MIS for spinal fusion procedures may not reflect barriers to adoption for non-MIS-practicing physicians. Developing a common definition of what constitutes MIS spinal fusion procedures would allow for more objective measurement and statistical validation of outcomes in patients who undergo minimally invasive techniques compared with those undergoing traditional open spinal fusion procedures. Over time, this can support more appropriate reimbursement for these procedures, and is an important step to help remove barriers to adoption.
In addition to identifying areas of consensus, the study was able to highlight areas where there is less certainty in the field. Further investigations are potentially warranted to confirm these findings. As with all small sample sizes, the responses of the selected expert panel may not be representative of all physicians who perform MIS spinal fusion procedures. Similarly, while it is possible that recruitment of SMISS members may result in a bias towards aspects of the MIS definition, the diversity in background and geography hopefully counters any potential effect.
Conclusion
Panelists agreed that overall tissue trauma was one of the most important factors in defining MIS spine fusion procedures. Surgical approach was also considered an important factor for determining whether a spinal fusion and/or fixation procedure can be considered MIS; panelists agreed that mini-PLIF, mini-TLIF, XLIF, and OLIF approaches are minimally invasive. Panelists identified a number of technologies that are valuable to MIS; however, the utilization of these technologies during a procedure does not necessarily define whether or not the procedure itself is minimally invasive, further supporting the conclusion that there is not a single set of surgical parameters (e.g., surgical approach, technologies employed during surgery, etc.) that define MIS. While there is consensus regarding a number of benefits associated with MIS (e.g., reduced length of stay, decreased duration of patient postoperative pain, and decreased infection rates in comparison to open surgery), panelists also agreed that there are a number of significant barriers to adoption of MIS for spine fusion procedures. Only 55% agree on what the major risks and complications are in MIS spinal fusion procedures, a surprisingly low level of agreement for a field where precision and safety are paramount. However, without a shared understanding of the most common or serious risks, it is difficult to develop standardized protocols, training and address the learning curve needed in MIS surgery. Surgeons can approach the same procedure with vastly different expectations and techniques hence MIS requires specialized skills and time to master. Emphasizing structured training, mentorship, and simulation could reduce risks and complications identified as key for patient safety whilst achieving MIS proficiency. The ability to define which spinal fusion practices should be considered MIS can inform future study designs and support evidence collection to help demonstrate the value of MIS going forward.
Footnotes
Acknowledgments
The authors would like to acknowledge the support of Dr Amit Jain and Caroline Smith for their contributions to the analysis of the results of the DCP. We appreciate the time commitment and expertise of the panelists that participated in the DCP. The authors would like to thank Kristi Skorija (Market Access Consulting & HEOR, Fortrea) and Mihalina Georgallis (Market Access Consulting & HEOR, Fortrea) for their valuable advice, critical reading of the manuscript, and help with submission.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by DePuy Synthes Spine.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
