Abstract
Introduction
Symmetrical peripheral gangrene (SPG) is characterized by symmetric acral necrosis in the absence of large vessel obstruction. Since being described in 1891 by Hutchinson, it has been associated with septicemic conditions. 1 The occurrence has been related to disseminated intravascular coagulation (DIC), and up to 85% of patients with SPG have been reported to have consumptive coagulopathy. 2 Previous studies have proposed risk factors for SPG development, but only a limited number have been identified.3,4
The common data model (CDM) was used to standardize medical records and data for multicenter collaborative studies. Since CDM provides medical information related to patient care such as diagnosis, medication, surgery, and examination, this big data source offers suitable clinical information for conducting this study. Furthermore, since patients’ personal information was anonymized, it did not violate research ethics guidelines. CDM also has advantages for evaluating complications of rare diseases. Many benefits have been found in CDM-related research, and it is gaining popularity in various medical fields.
This study aimed to identify the predictors of SPG using the established CDM and propose a new scoring system for predicting hospitalized patients at risk of developing SPG.
Materials and methods
This study was approved by our institutional review board in accordance with the Declaration of Helsinki (1989) by the World Medical Association.
Data source
This retrospective observational study was conducted using the Observational Health Data Sciences and Informatics (OHDSI) open-source software and the OMOP CDM version 5.3 database. The database contained de-identified EMR data converted to a standard format using the OMOP CDM system. Over two million patients’ EMR data spanning nearly 15 years, beginning in 2006, were transformed and collected in our hospital’s CDM database.
Registry-based patient selection
In this retrospective registry-based case series study, we investigated a cohort of patients who received intravenous noradrenaline, epinephrine, and vasopressin injections to treat hypovolemic conditions between January 2011 and December 2020 using the CDM database. In total, 81,535 patients were included in the registry. The cohort registry collects information on patient demographics. Among the patients included, those who had previously undergone amputation ( Flow diagram of the study.
Variables
We conducted a literature review to identify all probable risk factors for SPG. Variables that could not be identified in the EMR or converted to CDM were excluded. We included the following clinical data from the CDM database: demographic variables (age and sex), comorbidities, and laboratory values (complete blood cell counts, albumin, cholesterol, erythrocyte sedimentation rate, and C-reactive protein). Using SNOMED-CT, comorbidities were detected in the CDM database. Laboratory measurements were identified using concept IDs defined by Logical Observation Identifiers Names and Codes (LOINC), an international standard for identifying medical laboratory observations. We identified amputation as a salvage treatment for SPG.
Statistical analyses
Statistical analysis was performed using the R software, version 4.0.4 (R Foundation for Statistical Computing, Vienna, Austria). Descriptive data are expressed as mean ± standard deviation (SD), median (min-max), or number and frequency, where applicable. Chi-square and Fisher exact tests were performed for categorical variables, with SPG as the dependent variable and other variables as independent variables. Age was analyzed as a categorical variable by splitting the intervals by 10 years. T-tests were used to analyze continuous variables. We used a multivariate logistic regression model to identify risk variables for SPG and calculated the odds ratios (ORs) and 95% confidence intervals (CIs). To avoid selection bias, the results were further validated by propensity score matching (PSM) for age and sex using the “MatchIt” package in R. Statistical significance was set at
Statistically significant risk factors were then divided by each of their Exp(b) values to simplify the numerical value without reducing its proportional significance. Receiver operating characteristic (ROC) analysis was performed to assess the sensitivity and specificity of the novel scoring system.
Results
Baseline and demographic data
Patients’ demographic data.
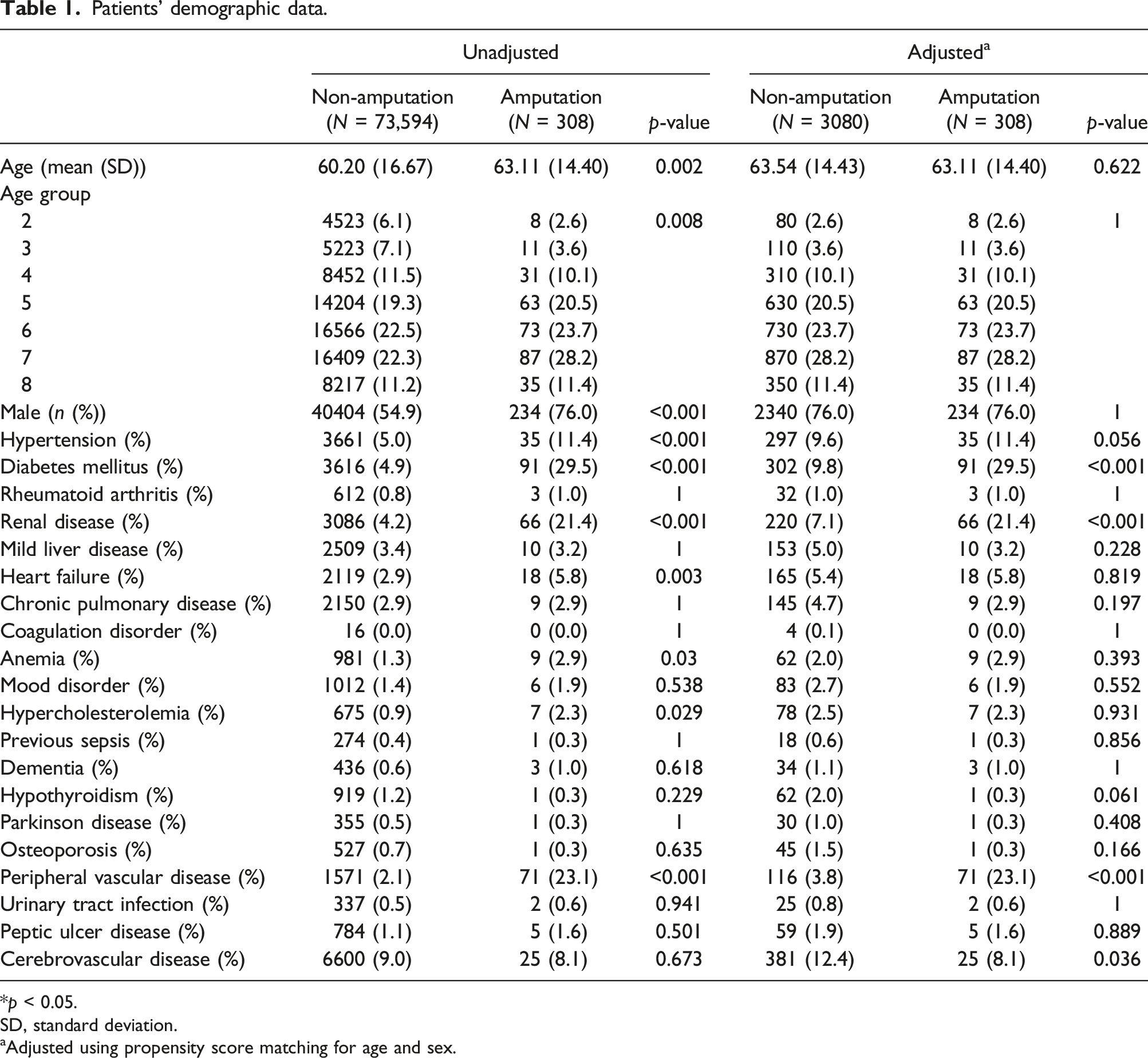
*
SD, standard deviation.
aAdjusted using propensity score matching for age and sex.
Laboratory values as risk factors for SPG.

aAdjusted using propensity score matching for age and sex.
Logistic regression analysis.

To simplify the numerical value without reducing its proportional significance, the value of each Exp(B) was divided by the least significant value of Exp(B). Therefore, each patient could be scored between 0 and 14.34, depending on the summation of their DM (3.59 points), RD (2.59 points), PVD (7.76 point), or CVD (0.40 point). ROC analysis was performed to assess the sensitivity and specificity of this novel scoring system, showing an area under the ROC curve (AUC) greater than 0.65 (Figure 2). Using a numerical threshold of 1.49, the specificity and sensitivity of the scoring system were determined to be 51.3% and 75.0%, respectively. Receiver operating characteristic (ROC) curve.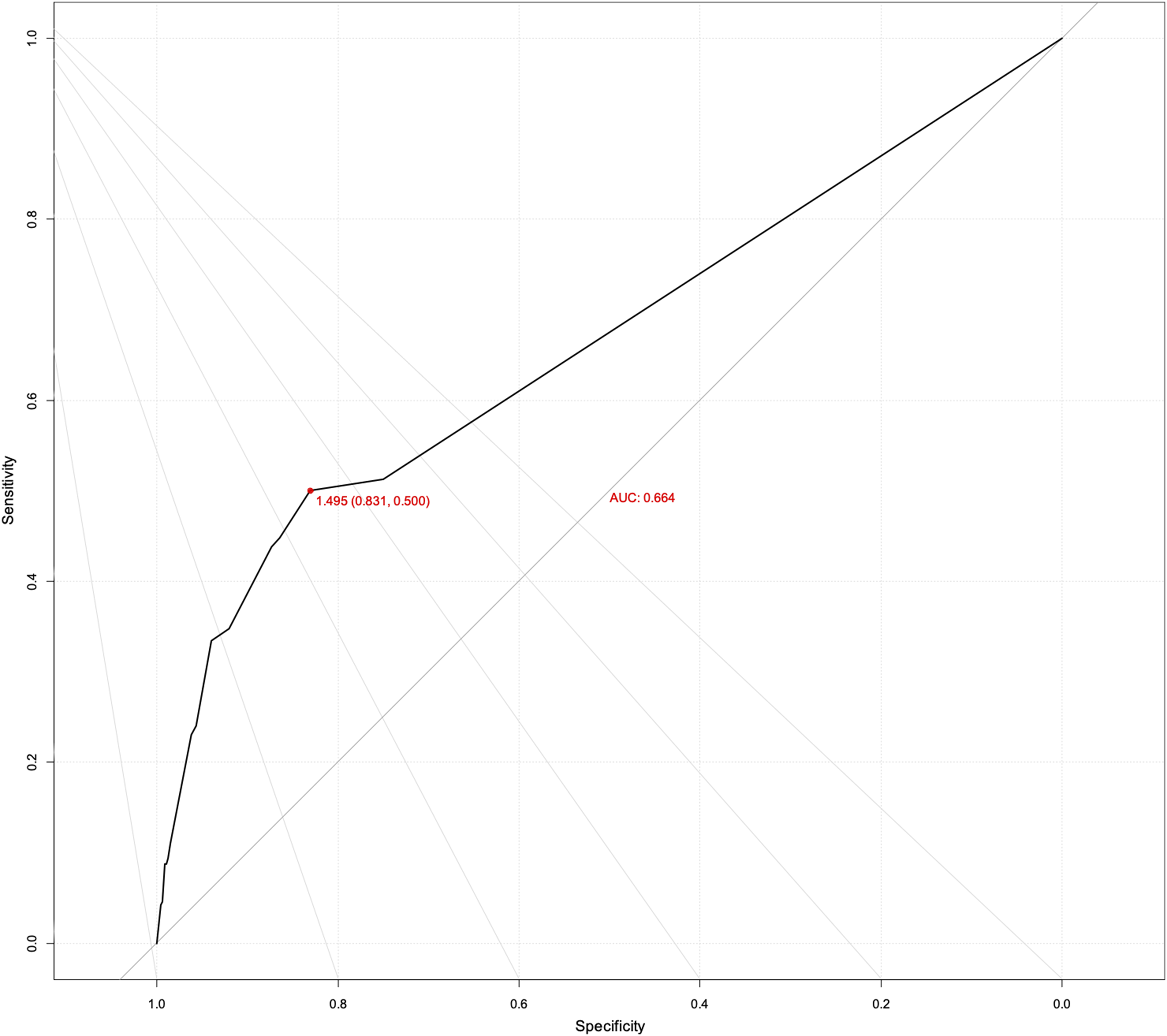
Discussion
This study was performed on a large cohort using the CDM database and PSM technique to minimize bias. Several risk factors have been found to be related to the development of SPG, such as age, sex, hypertension, DM, RD, heart failure, anemia, hypercholesterolemia, and PVD. Laboratory markers such as albumin, eosinophils, hematocrit, lymphocytes, monocytes, neutrophils, ESR, aPTT, creatinine, and BUN were also found to be correlated with SPG and should be cautiously monitored. The scoring system based on these risk factors can be used as a tool to identify hospitalized patients at a high risk of developing SPG.
In the literature, the etiology of SPG is reported to be multifactorial, including renal disease, septicemic conditions, 1 DIC, ergotism, Raynaud’s phenomenon, 2 use of inotropics,5–7 diabetes mellitus, 7 para-neoplastic syndrome, 8 previous history of cold injury, 9 asplenia, 10 protein C deficiency, 11 and atherosclerosis. 12 Antiphospholipid syndrome has also been frequently mentioned,13–16 with one extensive case of gangrene induced by warfarin treatment for the syndrome. 17 Our findings align with these previous studies. Clinically, the presence of these risk factors or laboratory findings should be communicated to patients and their families to inform them about the potential for developing SPG.
Treatment for SPG includes warming of the extremities and the use of eglandin. Recent case reports have also shown success with agents such as botulinum toxin, pentoxifylline, antiplatelet agents, systemic hyperbaric oxygen therapy, epoprostenol, tissue plasminogen activator, sildenafil citrate, and topical nitric oxide ointment therapy. However, amputation often remains the last resort. This outcome is devastating for patients and should be prevented with early administration of vasodilating agents.
Previous studies have attempted to identify risk factors associated with amputation after vasopressor administration. One study investigated 24,365 adults with sepsis, of whom 26 underwent amputation. A higher risk was associated with a higher Sequential Organ Failure Assessment (SOFA) score, elevated serum lactate, and bacteremia at the onset of sepsis. 4 Another study found a relationship between the peak dose of noradrenaline, dopamine, and epinephrine and the occurrence of SPG. The SOFA score, which measures the severity of organ dysfunction across six systems, was also a predictor of SPG development.3,18 Despite its high correlation with SPG, the SOFA score is complex, with different values corresponding to various grades in the classification system.
The scoring system developed in this study is a practical tool for identifying hospitalized patients at risk of developing SPG. The elements considered include DM, renal disease (RD), peripheral vascular disease (PVD), and cerebrovascular disease (CVD). Points are assigned as follows: DM (3.59 points), RD (2.59 points), PVD (7.76 points), and CVD (0.40 points), resulting in a score range from 0 to 14.34. Higher scores indicate a higher risk of developing SPG. The ROC curve analysis determined a threshold of 1.49, meaning that a score above 1.49 indicates a high risk of developing SPG.
The CDM, using the OHDSI database, facilitates reproducible, large-scale observational research. CDMs manage large amounts of data in the medical field. We used CDM coding algorithms to assess the effectiveness of our methodology. Tertiary centers using CDM query their databases to extract relevant data.
One limitation of this study is the small number of amputation surgeries as the outcome. The threshold of 1.49 means that having only DM, RD, or PVD places a patient in the high-risk group. However, this scoring system is the first to quantitatively assess the risk of hospitalized patients receiving intravenous noradrenaline, epinephrine, and vasopressin developing SPG. Another limitation is the binary outcome used, with only “amputation” and “no amputation” considered. Mild SPG cases were not included, and no linear relationship between the scoring system and disease severity was established. Further research is needed to confirm this relationship.
In conclusion, this study demonstrates that the CDM can be used to conduct studies on SPG and serves as a reference for future CDM-based risk factor analyses. Mitigating the identified risk factors may help predict the occurrence of SPG and reduce the need for amputation surgery in patients with SPG.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical Data-driven Hospital Support Project through the Korea Health Information Service (KHIS), funded by the Ministry of Health & Welfare, Republic of Korea; and the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HR22C1832).
