Abstract
Objective
To determine the effects of applying a natural polymer solution (shellac) to dry gangrene, and thereby prevent infection and progression to wet gangrene, in diabetic patients.
Methods
This randomized, controlled, pilot study included patients with type 2 diabetes mellitus who had unreconstructable peripheral vascular disease and were awaiting autoamputation. Eligible patients were contraindicated for surgery, had a limited life expectancy and/or had refused surgical amputations. All patients received the best possible medical treatment and were randomized to receive either topical shellac or antiseptic application of 10% povidone-iodine (PVP-I) solution to gangrenous areas.
Results
A total of 26 diabetic patients were eligible for inclusion, of whom 23 completed the study: 13 were treated with shellac and 10 with PVP-I. The amputation rate was higher in the conventional treatment group versus the shellac group (60.0% versus 46.2%, respectively), although this difference was not statistically significant.
Conclusions
Shellac is a natural solution that may be used to embalm gangrenous tissue in selected diabetic patients. More studies are needed to assess the long-term outcomes of this technique.
Introduction
The pathophysiology of gangrene in the diabetic foot is particularly complex and is influenced by interactions between arterial insufficiency, neuropathy, ulceration and infection.1,2 Gangrene usually represents a late manifestation of peripheral limb ischaemia in patients with diabetes mellitus,3,4 often requires amputation4–6 and may result in death.3,7 Gangrene caused by peripheral arterial disease (PAD) is commonly aseptic or noninfected and has a ‘dry’ appearance, whereas gangrene caused by bacterial infections or sepsis has a ‘wet’ appearance.1,2 Dry gangrene is usually less severe than wet gangrene and may result in autoamputation (i.e. spontaneous detachment from the body and elimination of a gangrenous part), whereas wet gangrene may lead to cellulitis, loss of the extremity, septicaemia and death.1,3
Diabetic ischaemic peripheral gangrene most commonly affects the toes/digits, feet and in some cases the entire leg. 2 PAD in patients with diabetes involves macro- and microangiopathy components,3,8 the management of which generally focuses on the major causative component. 1 PAD in patients with diabetes usually affects the medium and small arteries throughout the lower limb, particularly those in the leg and foot, whereas diabetic microangiopathy affects arterioles in the skin. 3 Many patients with diabetes develop gangrene caused by occlusive PAD, which is not amenable to revascularization procedures.9,10
In most cases, aseptic dry gangrenous tissue is left for full demarcation and subsequent autoamputation. 10 Surgical debridement and amputation are usually reserved for infected or septic wet gangrene. 1 Wet gangrene can develop from dry gangrene, however, often as a result of primary infection in immunocompromised patients with diabetes. 3 Thus, every effort must be made to prevent the infection of dry gangrenous tissue and its resulting transformation to wet gangrene.
For many patients with peripheral gangrene caused by long occlusive infrapopliteal atherosclerotic lesions, which are not amenable to standard vascular/endovascular reconstructive procedures, physicians often take a conservative approach and allow autoamputation. 10 Geriatric, bed-ridden patients with several comorbidities and patients with a short life expectancy, particularly those on mechanical ventilation in intensive care units, may also require conservative management, with nonsurgical interventions.5–7 Moreover, patients in certain cultures, particularly in Saudi Arabia, refuse amputations for religious or cultural reasons. 3 Other patients are temporarily contraindicated for any surgical intervention, including major amputations. 1 In such cases, the attending physicians must endeavour to prevent the transformation of dry to wet gangrene, while awaiting improvements in the patient’s condition. 10
Shellac is natural polymer of animal origin derived from the hardened secretion of the lac insect, Laccifer (Tachardia) lacca (order Homoptera, family Coccidea). These are scaly insects that grow on specific trees in China, India, Burma, Thailand, Laos, Cambodia and Vietnam. The resin is secreted to cover the insect larvae.11,12 Shellac is made up of aliphatic, aleuritic, shelloic, and jalaric alicyclic hydroxy acids and their polyesters. It is not water soluble, but is soluble in alcohol and alkaline solutions. 11 Shellac is widely used in the food industry for sealing and glossing as it forms a natural superficial layer that coats and protects foods. It is also used in the pharmaceutical industry where it is often used, as an alternative to synthetic polymers, as an enteric coating for phytopharmaceuticals and food additives.13,14 Shellac is commonly used at the authors’ institution to preserve and embalm dead animals and cadavers for teaching gross anatomy. The early results of using shellac as a nontoxic preservative for human embalming were promising. 11
The present clinical study investigated, for the first time, the topical application of shellac, compared with standard treatment with povidone-iodine (PVP-I), for preventing the progression of dry gangrene to wet gangrene and extension of gangrene to adjacent healthy tissues, in selected patients with diabetes mellitus who were offered the nonsurgical option of waiting for autoamputation. It was assumed that topically applied shellac would work as a natural polymer film that would protect the superficial tissues, embalm dead tissues and prevent tissue infection in patients undergoing conservative treatment while awaiting autoamputation. It was hypothesized that this approach would reduce the amputation rate in patients with unreconstructable PAD, in those who refused amputation for cultural reasons, in those who were unsuitable for immediate amputation and/or were waiting for improvements in their condition to undergo revascularization, and in elderly bed-ridden patients with a short life expectancy, contraindicated for surgery.
Patients and methods
Study population and design
This prospective, randomized, controlled 1-year trial was designed and conducted at the multidisciplinary Diabetic Foot Research Clinic at King Abdulaziz University Hospital, Jeddah, Saudi Arabia, over an 18-month period (commencing January 2010). Because there is no specific system to arrange the patients’ medical records in the clinic, patients were sequentially allocated, based on every second medical record, to receive either topical application of PVP-I (conventional treatment) or shellac solution to the necrotic and adjacent tissue. Both groups received the best possible conventional treatment throughout the study. Treatment assessment was not blinded as the two products are different in many properties.
Inclusion criteria for the study were as follows: patients with type 2 diabetes who presented with peripheral, dry, well-demarcated gangrene in their feet and who were offered the option to wait for nonsurgical autoamputation; elderly, bed-ridden patients with diabetes who refused amputation and/or were contraindicated for revascularization or surgery; patients who had recently received initial antibiotic therapy (these patients could enter the study 1 week after complete cessation of such therapy). Patients who presented with any evidence of wet or infected gangrene, evidence of osteomyelitis or those currently on antibiotics were excluded from the study.
The study received ethical approval from the Bioethical Research Committee of King Abdulaziz University (Approval No. 299-09). All eligible conscious patients and relatives of unconscious patients were informed about the protocol and gave written informed consent to participate in the study. Written consent was obtained from the patient and/or patients’ relatives before photographing their lesions.
The trial was registered on the ISRCTN register and was assigned the following number: ISRCTN72459118. Details of the trial can accessed at the following link: http://www.controlled-trials.com/ISRCTN72459118.
Preparation of shellac
Shellac was prepared once at the beginning of the study, as described previously. 11 For embalming procedures, 80 kg of dry shellac resin was purchased from a local market and placed in a 250 -l plastic tank. In a separate tank, 120 l of ethanol and 80 l of sterile distilled water were mixed. Diluted ethanol was then added to the dry shellac, causing a reaction between the components which resulted in formation of a wax on the bottom of the tank. After 2 h, a semitransparent solution was observed and most of the hard wax had deposited on the bottom of the tank. The refined shellac solution was collected and considered ready for use. It was tested using standard microbiological techniques before clinical use and was sterile (i.e. lacking bacterial contaminants or antibacterial activity against various bacterial strains commonly isolated in diabetic foot infections). The shellac preparation used in this study has been patented for use in the treatment of gangrenous tissue in diabetic foot gangrene and in the treatment of chronic wounds by the Turkish Patent Institute (Patent No. a201002903).
Study treatment and procedures
The shellac was applied by soaking a 4 × 4-cm sterile gauze in shellac solution for 30 s, which was then used to swab the gangrenous tissue. The tissue was then left to dry for ∼5 min. An alcohol swab was used to clean the shellac from healthy tissue adjacent to the gangrenous tissue. A light, dry, dressing was applied 10 min later and left undisturbed for 24 h. After 24 h, the dressing was removed and the gangrenous area was swabbed again with ethanol, to remove the shellac. The shellac was reapplied after 5 min. In the conventional treatment group, the same steps were applied, but the gauze was soaked in 10% PVP-I instead of shellac. The procedure was repeated once daily in the conventional treatment group and additionally (as required), such as after showering or removal of dressing in the shellac group. All patients were asked to visit the clinic every month or when signs of inflammation or fever were observed.
Conventional treatment was defined as the protocol used to manage ulceration or gangrene in patients with diabetes, and included review of the patient’s history, medical examination, investigations and basic treatments. All patients in both groups received the same medications, as specified in the practice guidelines adopted by the clinic. The only difference between the two groups was the application of PVP-I or shellac.
Visits were scheduled monthly for the first 6 months, although patients were asked to visit the clinic whenever necessary for 1 year. Photographs of the gangrenous and adjacent tissue were taken every month by nurses and were assessed by an attending researcher. Swab cultures and white blood cell (WBC) counts were also determined every month.
Study assessments and endpoints
Outcomes were considered good if the patient’s WBC count was normal, local and systemic cultures were negative for infection, and body temperature and local skin temperature, measured using a digital thermometer, were normal. Outcomes were considered bad if the WBC was elevated, local or systemic cultures were positive, and body temperature (≥37.8℃) or local skin temperature (difference of ≥2.2℃) were abnor mal.
Progression of gangrene was defined as the extension of gangrene into healthy tissue by 1 cm in horizontal or vertical directions. Dry gangrene was defined as dry, blackish, full-thickness patches of skin and or whole toe or forefoot gangrene without evidence of bacterial infection, as confirmed by analysis of swabs taken for microbiological assessment. Wet gangrene was defined as wet or purulent lesions, or a tissue with an offensive smell and evidence of bacterial infection, as confirmed by analysis of swabs taken for microbiological assessment. Transformation to wet infected gangrene was considered as a clear indication of failure. If wet gangrene developed, patients usually proceeded to surgical amputation and/or debridement.
Minor amputations included autoamputation or surgical amputation of the toes or trans-metatarsals. Major amputation included surgical amputation above or below the knee.
The primary study endpoint was the extension of gangrene to adjacent healthy tissue and progression from dry to wet gangrene. Secondary endpoints included patient death, autoamputation, surgical amputation and/or debridement, and performance of avvascular revascularization procedure.
Statistical analyses
Statistical analyses were performed using the SPSS® software package, version 16.0 (SPSS Inc., Chicago, IL, USA) for Windows®. The sample size of this pilot study was expected to be small; consequently, the differences between the two groups were not expected to be statistically insignificant. Nevertheless, Fisher’s exact test was used to compare parameters where the expected value in one or more of the cells in 2 × 2 contingency tables was <5. A P-value of <0.05 was considered to be statistically significant.
Results
A total of 26 patients were enrolled and randomized into the study; 13 received shellac and 13 received PVP-I (conventional) treatment. Three patients allocated to conventional treatment withdrew 3 weeks after enrolment. The remaining patients fulfilled one of the stated endpoints. An example of application of shellac in an 82-year-old diabetic male with end-stage renal disease and unreconstructable PAD, who refused surgical amputation, is shown in Figure 1.
Plantar (a) and lateral (b) views of the foot of an 82 year-old male with type 2 diabetes who refused surgical amputation. The patient had end-stage renal disease, unreconstructable PAD and had undergone coronary artery bypass grafting 7 years previously. The patient died 8 months after starting shellac use without undergoing major amputation (written consent was obtained from the patient and his son for photography of his lesions to be included).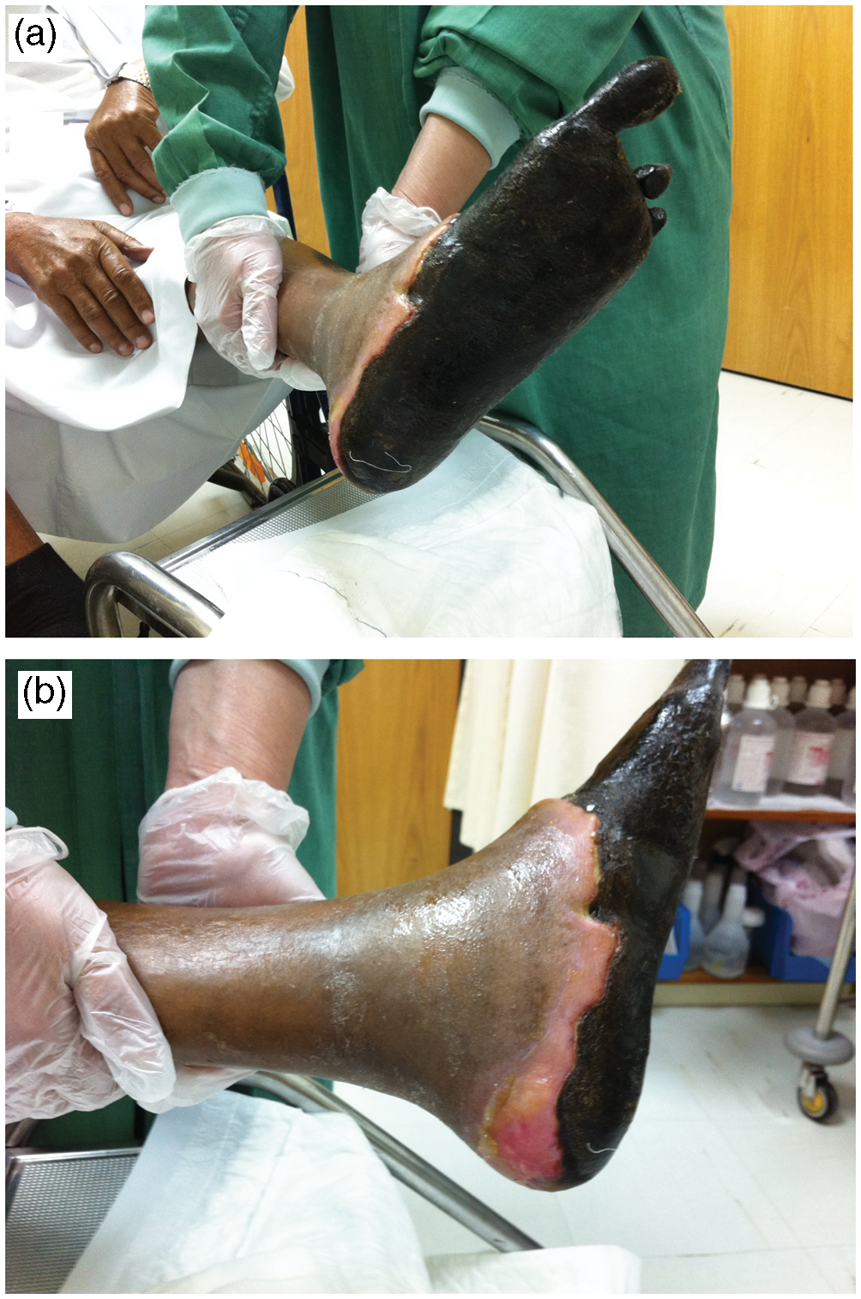
Baseline characteristics and study outcomes at 12 months in diabetic patients with unreconstructable peripheral vascular disease (n = 23), randomized to receive treatment with topical shellac or antiseptic application of povidone-iodine solution (conventional treatment) on gangrenous areas of the foot.

Data presented as n, n (%) of patients or mean ± SD.
There were no statistically significant between group differences (P ≥ 0.05) in study outcomes at 12 months; Fisher’s exact test.
The overall rate of amputation was 52.2%: six patients in each group lost parts of their extremities. All of those who underwent surgical amputation had evidence of spreading infection. The ratio of major to minor amputation was 1 : 1 and included: six digit autoamputations that did not require surgical excision and were considered positive outcomes; six major amputations with three above-knee amputations in the conventional group, and two above-knee and one below-knee amputations in the shellac group. The overall rate of surgical amputations was higher in the conventional treatment group than in the shellac group (60.0% versus 46.2%, respectively), although this difference was not statistically significant.
In the shellac group, three patients died during the study, compared with one patient death in the conventional treatment group. All patients died due to comorbidities that led to cardiorespiratory failure; none of these deaths were directly related to the patients’ lower extremity clinical condition. At the time of death, each patient had preserved extremities without undergoing surgical amputation. The remaining 19 patients (82.6%) were still alive at the 12-month follow-up visit and, of these, only six underwent major amputation (31.6%) (Table 1). None of the patients treated with shellac developed an allergic reaction.
Discussion
Patients with diabetes are at high risk of developing foot complications, which may lead to tissue loss or gangrene. In the authors’ clinical practice, a large proportion of patients present late with overt gangrene.3,15 In these advanced cases, little can be done other than to provide the best available medical treatment, which includes antibiotic therapy 9 and treatment of any underlying macrovascular disease. The final option in these patients with diabetic foot disease is usually planned surgical amputation.1,5,6,8,15 In patients with sterile dry gangrene, however, a nonsurgical option is to wait for autoamputation. 10 This approach is often welcomed by patients in certain cultures where amputation is not acceptable for cultural or religious reasons. Other patients may also be offered this relatively conservative approach – particularly those with gangrene caused by severe unreconstructable PAD involving the foot and digital arteries, including patients with diabetes on haemodialysis for end-stage renal disease.5,7,9 Another group of patients who wait for autoamputation includes elderly bed-ridden patients with comorbidities that contraindicate open surgical vascular bypass or percutaneous endovascular procedures. 8 In some of these patients, immediate surgical amputation may substantially increase mortality, with perioperative mortality rates ranging from 7–17%.3,7,15 It is also of concern that gangrene may persist in some patients with diabetes, critical limb ischaemia and end-stage renal disease, despite successful grafting of a patent vascular bypass.5,7 Thus, preserving the gangrenous tissue and preventing infection represent transient and/or permanent objectives in managing these groups of patients. Conservative treatment in these cases usually involves topical application of antiseptic solutions, such as PVP-I, as used in the present study.
The present prospective, randomized,controlled, pilot study tested a natural complementary and alternative medicine, shellac, compared with a routinely used chemical antiseptic (PVP-I). This approach was based on the reported preferences of patients with diabetes for using complementary and alternative medicines in Saudi Arabia and in other countries.16,17 Shellac is already patented for such indications in Turkey and is used as superficial protective film in the food and pharmaceutical industry.12–14 Shellac is also used as an ingredient in edible fruit coatings to limit water loss and prevent desiccation and weight loss, and to prevent pathogen entry. Shellac forms a barrier that is fairly impermeable to oxygen and water, and reduces gas exchange. The reduction in oxygen levels reduces the respiration rate of fruits and vegetables and prolongs their shelf-life by delaying oxidative breakdown of tissue.12,18 Although, the whole mechanism of the preservation process is not yet known, the authors believe that shellac provides distinct and substantial chemical properties for embalming in clinical situations, similar to the effects observed in the preservation of foods. Shellac is also used for embalming dead animals, and was used by the current research group as a nontoxic preservative for human embalming. 11 Das and Jacob reported a heightened prevalence of allergic contact dermatitis in areas where cosmetics that included shellac were applied. 19 Such side-effects were not observed in the present study. The antibacterial effects of shellac were also suggested in a report discussing on the nonclinical application of shellac. 20
Shellac was applied as a natural barrier in the present study to protect the underlying sterile gangrenous tissue in order to prevent desiccation and microbial infection. Unlike antiseptics commonly used in the management of gangrene, shellac exerts no antibacterial activity against bacteria commonly isolated from diabetic foot disorders. The results were encouraging, as shellac showed superiority over PVP-I solution in terms of a lower rate of surgical amputation, although this difference was not statistically significant.
There were some limitations to this preliminary study, including the small numbers of patients treated in both groups and the short duration of follow-up. These limitations indicate that a larger study, which follows patients for longer time periods, is required. Another limitation was that, unlike in cadaver embalming, shellac was used as a topical protective barrier and does not penetrate the deeper tissue. 11 The authors are currently developing a pressure chamber to enable shellac to penetrate deeper tissue under air pressure.
In conclusion, shellac is a natural polymer solution that may be used to embalm gangrenous tissue in selected patients with diabetes who refuse amputation or who are offered a nonsurgical option to wait for autoamputation. It may also be used to embalm gangrenous tissue in patients with a limited life expectancy, as well as bed-ridden patients and patients contraindicated for surgical procedures. Shellac may reduce rates of surgical amputation compared with antiseptic preparations. Additional larger studies are needed, to determine the long-term outcomes and safety of shellac.
Footnotes
Acknowledgements
The authors thank the Mohammad Hussein Al-Amoudi Chair for Diabetic Foot Research for funding this study, Dr Hossam Shoaib and Dr Amr Khayat for assistance with data collection, and Dr Adel Ibrahim for help performing the statistical analyses.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
Funding this study was provided by the Mohammad Hussein Al-Amoudi Chair for Diabetic Foot Research.
