Abstract
Purpose
To report preliminary clinical results and safety of 3D-printed patient-specific titanium radial head (RH) prosthesis in treatment of the irreparable RH fractures.
Material and methods
This multi-centric prospective study included 10 patients (6 men and four women, mean age 41 years (range, 25–64 years)). Three cases were classified as Mason type III and 7 cases as type IV. Patients were assessed preoperatively, intraoperatively, and at 1, 6, 12, 24, 36, and 48 weeks postoperatively. Range of motion (ROM), visual analog scale (VAS) score, Disabilities of the Arm, Shoulder and Hand (DASH) score, Mayo Elbow Performance Score (MEPS), radiology imaging, and laboratory blood and urine testing were evaluated.
Results
The prostheses were implanted utilizing cemented stems in 5 patients and cementless stems in 5 patients. Intraoperatively, well congruency of a prosthesis with capitellum and radial notch of ulna was observed in all cases. All patients had improvement of ROM, VAS score, DASH score, and MEPS during the postoperative follow-ups. At the final follow-up, mean elbow extension was 6.5° (range, 0°–30°), flexion 145° (range, 125°–150°), supination 79° (range, 70°–80°), and pronation 73.5° (range, 45°–80°). Mean VAS score was 0.3 (range, 0–3), DASH score was 12.35 (range, 1.7–23.3), and MEPS was 99.5 (range, 95–100). Postoperative radiographs demonstrated heterotopic ossification in 2 cases, periprosthetic radiolucency in 2 cases, and proximal radial neck resorption in 2 cases. No one had the evidence of capitellar erosion, implant failure, malpositioning, overstuffing, or symptomatic stem loosening. There was no significant alteration of laboratory results or adverse events related to the 3D-printed prosthesis implantation.
Conclusion
The preliminary results demonstrated that implantation of the 3D-printed patient-specific titanium RH prosthesis is safe and may be a potential treatment option for irreparable RH fracture.
Introduction
Radial head is one of the key structures of the elbow. It shares 60% of the load transferred across the elbow joint. The radial head also acts as the secondary stabilizer against valgus stress, posterior subluxation, and posterolateral rotatory instability of the elbow and as the primary contributor to longitudinal forearm stability.1,2
Radial head fractures are common intra-articular elbow fractures. Most fractures have been reported to occur in patients 20–60 years of age. Mason classified radial head fractures into 3 types: type I, non-displaced fractures; type II, marginal sector fractures with displacement; type III, displaced comminuted fractures involving the entire radial head. Johnston subsequently added type IV, which describes a radial head fracture with the dislocation of the elbow. The optimal treatment of the radial head fracture has been the subject of significant controversy. Due to an enhanced understanding of the importance of the radial head in elbow kinematics and the growing number of studies reporting better results of radial head replacement in comparison to excision and fixation, the use of a radial head prosthesis was advocated for irreparable fractures in Mason type III and type IV.1–3
Mismatch between a radial head prosthesis and the native anatomy reduces radiocapitellar contact areas and increased radiocapitellar contact forces which is potential for long-term articulating cartilage erosion or arthritic change. 4 Based on recent literatures, two leading causes of the mismatch were described. First, morphology of the proximal part of the radius is complex and highly variable. The precise replication of individual anatomy is difficult to achieve by using the commercially available prosthetic designs. 5 Second, it is impossible to consistently insert radial head prostheses anatomically. Frequent non-anatomic insertion of the prostheses was evidenced in several studies.6,7
Currently, patient-specific prosthesis is readily accessible thanks to the advancement of computer-aided design (CAD) and 3D printing technology.8,9 Based on the symmetry of the human skeleton, the 3D images of normal radial head can be reversed to manufacture the prosthesis of contralateral side which has the accurate shape and dimension for the missing part. 10 In addition, 3D preoperative planning can be used to generate the intraoperative guide in order to implant the prosthesis in the correct position. 8 A previous morphological analysis demonstrated that, in the setting of ideal prosthetic placement, the patient-specific implants generated from the contralateral intact radial head had less surface mismatch than the commercially available designs. 11
In this work, a 3D-printed patient-specific titanium prosthesis with customized intraoperative cutting guide was developed for the treatment of irreparable radial head fracture. The objective of this study is to report preliminary clinical results and safety of such prosthesis.
Material and methods
Demographic details of the patients.

F, female; M, male; R, right; L, left; AMS 2 CF, anteromedial subtype 2 coronoid fracture; TS 1 CF, tip subtype 1 coronoid fracture; TS 2 CF, tip subtype 2 coronoid fracture; MCL, medial collateral ligament; LCL, lateral collateral ligament.
Design and manufacturing process of 3D-printed prosthesis and surgical guide
The patient-specific radial head prosthesis was a monoblock with 2 portions: head and stem. The computerized prosthetic model was generated for subsequent manufacturing process. Each model was designed with a close collaboration between surgical team and engineering team. A high-resolution CT scanning (Philips Brilliance 64 CT scanner, Cleveland, Ohio, USA; voxel size 0.3 × 0.3 × 0.3 mm, 120 kV, 150 mAs, pitch 0.5) of both elbows was performed and reconstructed into 3D images. The 3D implant model was constructed using CAD and image processing techniques. The rendered 3D model of the normal side was reversed and aligned with the 3D model of the fractured side by registration the surface of intact portion of the proximal radius to identify correct profile of the head portion. The additional thickness of cartilage was also augmented to the model using the cartilage-reproducing image reconstruction method.
12
An impressed mark was made at the base of the head portion to be used as a reference for implant placement. A stem portion had a straight cylindrical feature with cone-shaped distal tip. It was designed in conforming to the individual alignment of intramedullary canal (Figure 1(a)–(d)). The stem diameter was adjusted to press fit into the canal while the stem length was determined to achieve a cantilever quotient of 0.35. The stem surface was set to touch the inner cortex of the redial neck and intramedullary canal of proximal radius in the first four patients and switched to overlap for 0.2–0.4 mm in the remaining patients. Medial and lateral flutes had also been added to the stem to allow rotational stability. The lateral flute was formed in a straight line with an impressed mark of the head. To determine structural strength of each implant, finite element analysis with ANSYS mechanical software (Ansys Inc., Canonsburg, Pennsylvania, USA) was conducted. After the computerized model was completed, the manufacturing process was conducted by the engineering organization (Meticuly Co. Ltd, Chulalongkorn University, Thailand) which is ISO 13,485 certified for the development and production of the orthopedic devices. The prosthesis was fabricated from medical grade titanium alloy (Ti-6Al-4 V) powder using selective laser melting 3D-printed machine (Mlab cusing 200 R, Concept Laser, Germany). Testing and standard compliance including full biocompatibility test according to ISO10993 standards and 3D printing standards according to USFDA on additive manufacturing ASTM F3001 were performed. The processes of sterilization according to ISO 17,665, Packaging according to ISO 11,607, and labeling according to ISO 15,223 were executed. Prosthetic design. (a) The rendered 3D model of the normal side was reversed and aligned with the 3D model of the fractured side by registration the surface of intact portion of the proximal radius to identify correct profile of the head portion. (b) The additional thickness of cartilage was also augmented to the model using the cartilage-reproducing image reconstruction method (yellow arrow). (c) An impressed mark was made at the base of the head portion to be used as a reference for implant placement (blue arrow). (d) A stem portion was designed in conforming to the individual alignment of intramedullary canal.
A surgical guide was created with a similar workflow. The computerized model of the guide was set on the dorsal radial aspect of the proximal radius. Its undersurface was designed to match with underlying bone surface and its posterior edge was in conforming to the lateral edge of the radial tuberosity. A platform was built on the proximal edge of the guide to specify the location and plane of bone cut according to the plan. A hole for k-wire fixation had been made to stabilize the guide. In addition to be used for radial neck osteotomy, a surgical guide also provided the intraoperative references for accurate placement of the prosthesis. An anatomic model of the fractured proximal radius with a cutting mark was also created in assistant the surgeon to be sure that the surgical guide was placed correctly (Figure 2(a) and (b). The guide and anatomic model were manufactured by Fused Deposition Modeling (FDM) printer (Zortrax, Poland) using certified medical grade material (polyamide) filament. The guides and models were sterilized using low-temperature hydrogen peroxide gas plasma (Sterrad system, Advanced Sterilization Products, Div. of Johnson & Johnson Medical Inc., Irvine, California, USA) prior to surgery. Surgical guide design. (a) The guide was set on the dorsal radial aspect of the proximal radius with its posterior edge in conforming to the lateral edge of the radial tuberosity. (b) A platform was built to specify the location and plane of bone cut (blue arrow). A hole for k-wire fixation has been made to stabilize the guide (yellow arrow).
The production time of 3D-printed implant and surgical guide was 5 days after receiving the CT data.
Operative technique
With the patient in supine position, a straight lateral or a straight posterior midline longitudinal skin incision was made. A Kocher’s approach (between anconeus and extensor carpi ulnaris) was done to access the radial head. Careful attention was directed to preserve the radial collateral ligament if it is intact. The annular ligament was incised longitudinally. The radial head fragments were identified and removed. The forearm was fully pronated to provide ample exposure of the radial tuberosity. A surgical guide was placed on the proximal radius using the radial tuberosity as a reference. After the guide seated properly, a 1.2 mm K-wire was inserted through a premade hole to fix with the near cortex. The radial neck was osteotomized using an oscillating saw. The saw blade was aligned with the platform of cutting guide. An electrocautery was used to mark the first reference on the cutting surface of radial neck in line with the reference on the platform of cutting guide. The k-wire and cutting guide were removed. A mark from K-wire insertion was used as the second reference for prosthetic placement. All radial head fragments were assembled on a table to ensure that the whole head had been resected. Preparation of the intramedullary canal was performed. Size broaches were used to obtain the planning size. The prosthesis was introduced into the canal with its impressed mark and lateral flute of the stem aligned with the first and second references. The stem was advanced until the prosthetic neck fully contacted on the cutting surface. When the press-fit stability of stems was felt to be insufficient, the implant was retrieved, and the low-viscosity bone cement (CMW 3; DePuy, Warsaw, Indiana, USA) was injected into the intramedullary canal before reinserted the stem. The position of prosthesis, joint congruity, and elbow stability were assessed using direct visualization and fluoroscopic imaging. The annular ligament was repaired. The LCL and MCL were reapproximated to the bone using either transosseous suture or suture anchor when necessary (Figure 3(a)–(f)). Operative technique. (a) A surgical guide was placed and a K-wire was inserted through a premade hole. (b) The radial neck was osteotomized. (c) A mark was made on the cutting surface of radial neck in line with the reference on the platform of cutting guide using an electrocautery. (d) Preparation of the intramedullary canal was performed. (e) The prosthesis was introduced into the canal with its impressed mark and lateral flute of the stem aligned with the cautery mark and K-wire hole. (f) The stem was advanced until the prosthetic neck fully contacted on the cutting surface.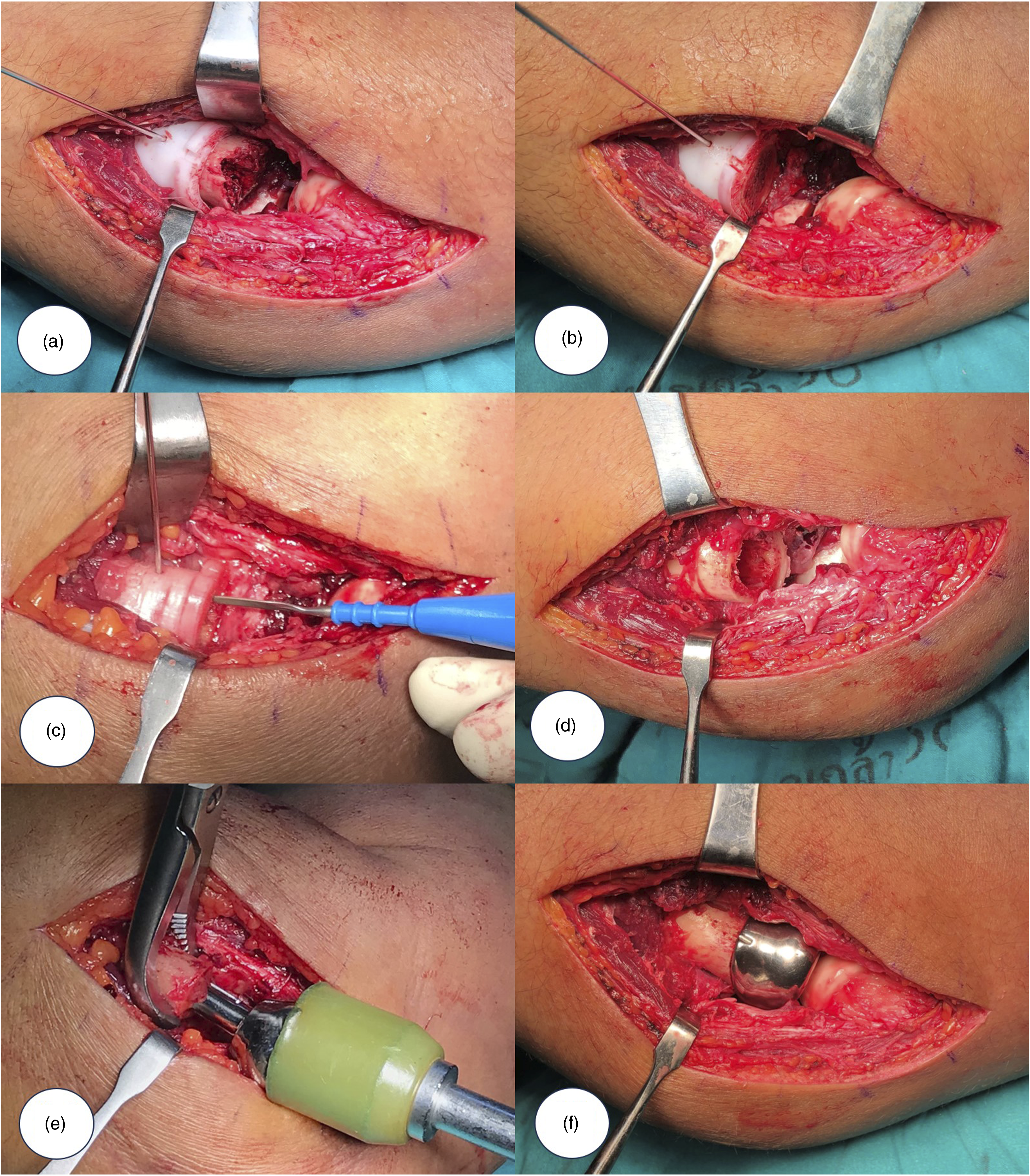
Postoperative management
The elbow was immobilized in 90° of flexion and neutral forearm rotation until the first postoperative visit. Patients were advised to perform the active-assisted range of motion (ROM) exercises in 7–10 days after the surgery. Active and gentle passive motion exercises began after 3 weeks. Stretching and strengthening exercises were initiated 6 weeks postoperatively. Prevention of heterotopic ossification (HO) formation with irradiation or medications was not used in this study.
Evaluation
The preoperative and postoperative evaluation consisted of clinical and functional assessment, radiographic examination, and laboratory tests. The clinical and functional assessment including ROM of the elbow, visual analog scale (VAS) score, Disabilities of the Arm, Shoulder and Hand (DASH) score, and Mayo Elbow Performance Score (MEPS) was performed preoperatively, and at 6, 12, 24, 36, 48 weeks postoperatively. The adverse events and serious adverse events were documented. The anteroposterior, lateral, medial oblique, and lateral oblique radiographs of the elbow were taken for evaluation preoperatively and postoperatively in every visit. The presence or absence of implant failure, implant malpositioning, overstuffing, capitellar erosion, periprosthetic radiolucency, heterotopic ossification, and proximal radial neck resorption was recorded. Laboratory tests preoperatively and at 12, 48 weeks postoperatively included complete blood count, serum inflammatory markers (erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP)), coagulation test, renal function test, liver function test, serum electrolyte, serum calcium, serum phosphate, serum aluminum, and urine analysis. For the assessment of serum metal concentration in association to the titanium alloy, only the serum aluminum level was done because the titanium and vanadium level blood tests were not available.
Results
The prostheses were implanted utilizing cemented stems due to suboptimal interference press-fit (Figure 4(a)–(d)) in 5 patients (patient 1, 2, 3, 4, and 10) and cementless stems (Figure 5(a)–(d)) in 5 patients (patient 5, 6, 7, 8, and 9). Intraoperatively, well congruency of a head portion with capitellum and radial notch of ulna was observed in all cases. 3D-printed prosthesis with cemented stem in treatment of a radial head fracture (Patient 4). (a) Preoperative images. (b) 3D rendering of fractured radial head and computer-aided design of prosthesis. (c) Postoperative images at 48-week follow-up. (d) Postoperative images demonstrated the range of motion. 3D-printed prosthesis with cementless stem in treatment of a radial head fracture (Patient 6). (a) Preoperative images. (b) 3D rendering of fractured radial head and computer-aided design of prosthesis. (c) Postoperative images at 48-week follow-up. (d) Postoperative images demonstrated the range of motion.

Preoperative and postoperative clinical and functional outcomes.

VAS: visual analogue scale; MEPS: Mayo Elbow Performance Score; DASH: Disabilities of the Arm, Shoulder and Hand.
Clinical and functional outcomes of cemented and cementless groups at the final follow-up.
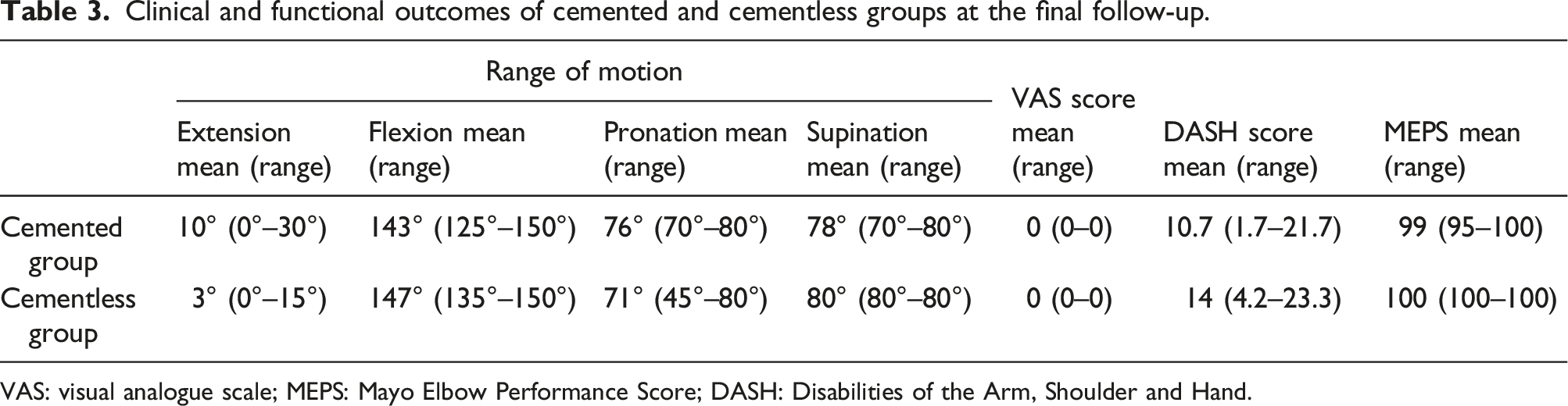
VAS: visual analogue scale; MEPS: Mayo Elbow Performance Score; DASH: Disabilities of the Arm, Shoulder and Hand.
There was no significant adverse event related to the 3D-printed prosthesis implantation. One patient had a revision of ligament repair because LCL and MCL re-rupture with elbow subluxation occurred from immature use of the arm pushing the body up from the bed at 1 week postoperatively (patient 1). The elbow joint was congruent following reattached the re-ruptured ligaments. Another patient sustained the motorcycle accident at 30 weeks after the surgery (patient 3). The fractures of ipsilateral humerus and 4th metacarpal bone were detected but the elbow joint and prosthesis were intact. Open reduction and plate fixation of the humerus and closed treatment of the metacarpal were performed. Returning to the normal activities was successful at 48-week follow up for radial head replacement.
Radiographic examination postoperatively revealed that the radial head implant was in the alignment as planned preoperatively in all cases. No patient had the evidence of subsequent capitellar erosion, implant failure, implant malpositioning, and overstuffing. Heterotopic ossification appeared radiologically in 2 patients. Ossification occurred around the radial neck, near the medial collateral ligament and anterior aspect of the ulnohumeral joint in 1 case (patient 1), and at the anterolateral aspect of radial neck in 1 case (patient 9). Radiolucent line between 0 and 1 mm surrounding the cementless stem in 1 case (patient 5) and cemented stem at the bone cement interface in 1 case (patient 10) was found. Proximal radial neck resorption was observed in 2 cases (patients 2 and 3) (Figure 6(a)–(c)). Summary of clinical, functional, and radiographic findings at the final follow-up are shown in Table 4. One patient who developed HO had 45° of pronation (patient 9) and 1 patient who had proximal radial neck resorption had 120° of flexion (patient 2) which is less than functional ROM. Postoperative radiographic findings. (a) Heterotopic ossification (yellow arrow) (patient 9). (b) Radiolucency at the bone cement interface (white arrow heads) (patient 10). (c) Proximal radial neck resorption (white arrow) (patient 3). Summary of clinical, functional, and radiographic findings at the final follow-up. VAS: visual analogue scale; MEPS: Mayo Elbow Performance Score; DASH: Disabilities of the Arm, Shoulder and Hand.

The laboratory evaluation showed no significant alteration of the results of complete blood count, serum inflammatory markers, coagulation test, renal function test, liver function test, serum electrolyte, serum calcium, serum phosphate, serum aluminum, and urine analysis after prosthesis implantation. All patients had the serum aluminum level below the predictive value of aluminum toxicity (6 µg/dl). The laboratory results are available online in supplement material.
Discussion
This preliminary study evaluated on the clinical outcomes and safety of the 3D-printed patient-specific titanium prosthesis.
Several implant options are currently available in treatment of the radial head fracture. Monopolar metallic prosthesis is a frequently used type of implant due to its biomechanical load-bearing abilities and effectiveness in stabilizing the radiocapitellar joint. 13 A recent systematic review and meta-analysis demonstrated the differences across different implants in functional scores, certain complications, and revision rates. 14 However, very limited available information for the patient-specific radial head implant has been published by medical journal to date. The possible shortcoming of using the 3D-printed patient-specific implants in clinical practice is the complex manufacturing processes for each individual which may lead to prolong the waiting time for surgery. 15 A systematic review showed that the patients who had radial head replacement acutely tend to have better results than in a delayed fashion. 16 Although the 3D-printed implant and cutting guide required 5 days of production, this doesn’t seem to impact on the outcome of treatment. The results in this study revealed that the mean elbow extension was 6.5°, flexion was 145°, supination was 79°, pronation was 73.5°, DASH score was 12.35, and MEPS was 99.5 points at the final follow-up. These clinical and functional outcomes compare favorably with other studies that have evaluated the commercially available monopolar metallic radial head prosthesis in the treatment of irreparable radial head fractures.17–20 Most of the studies of radial head implants have analyzed the clinical results and reported the range of motion achieved at the final follow up, without providing information on the recovery.17–21 A recent prospective analysis in treatment of complex elbow injury demonstrated that the critical period to obtain the functional elbow was the first 6 months. 21 The improvement was slower thereafter and ROM remained the same from 1 year onwards. In our study, the time for recovery of functional ROM in 5 patients was 6 weeks, in 3 patients was 1 weeks and in 1 patient was 24 weeks postoperatively.
The commercially available radial head implants are currently manufactured by the milling method. Berschback et al. demonstrated that the levels of serum metal ion in patients with radial head prosthesis were not greater than in ones with a well-functioning total hip arthroplasty. 22 Based on the literature review, there is no published data regarding serum metal ion concentration in association with radial head prosthesis manufacturing with 3D printing method. In the present study, the prostheses were printed from the titanium alloy powder. Their metal composition includes titanium, aluminum, and vanadium (Ti-6Al-4V). 23 Titanium has been the material of choice in fabrication of several orthopedic implants due to its excellent biocompatibility and mechanical properties. The occurrence of titanium toxicity is rarely reported. Titanium toxicity is bone loss due to inflammatory reactions, hypersensitivity to titanium and allergic reactions, and yellow nail syndrome. 24 A pilot study in the rabbit model demonstrated that the organ and serum titanium levels were significantly higher in subjects implanted with milled and 3D-printed titanium implants compared to the control group. However, there was no significant difference in subjects implanted with traditionally fabricated and printed implants. 25 Toxic effects of aluminum include painful osteopenia, microcytic anemia, cholestatic hepatotoxicity and hypoparathyroidism. 26 The biologic problems of vanadium including cytotoxic and causing allergic reactions were described. 27 In our study, no statistically significant difference between the preoperative and postoperative serum aluminum was detected. None had the serum level above the predictive value of toxicity. The titanium and vanadium levels were not directly evaluated. However, no evidence of such metal toxicity, allergic reaction or disease was observed from the clinical, laboratory and radiologic follow-up.
In the present study, the patient-specific surgical guide has been used intraoperatively. The use of 3D-printed cutting guide is reproducible surgical technique that offers the precise results of osteotomy in several bones.28–30 Positioning of the 3D-printed guides is a critical step of this procedure. 31 To our knowledge, the surgical guide for placement of the radial head prosthesis has never been described. In the present study, the radial tuberosity was used to determine correct position of the cutting guide. The length, width, thickness, and geometry of the cutting guide can be properly adjusted with computer-aided software preoperatively to minimize the area of dissection. No evidence of the injury of important surrounding structures including biceps tendon or posterior interosseous nerve was found from placement the guide in this location. In addition to representing the cutting plane and level of radial neck, the guide provided the rotation reference in placement of the prosthesis. The surgical guide in the present study made from polyamide filament using FDM technique. Polyamide is commonly used material for fabricating the surgical guide because of the durability to withstand the heat of sterilization and contact with surgical instruments including saw and k-wire.32,33
The patient-specific radial head implant in this study was monopolar type which was fabricated in a monoblock fashion. The location and alignment of head-stem junction was designed individually in order to set the radial head portion in an anatomic position. This feature is different from the commercially available monopolar prosthesis which has fixed head-stem angle. Moon et al. observed the alteration of radial head tilt from implanting the fixed head-stem prosthesis when the neck cut needed to be done in the different level and when the different stem length was selected. 34 Apart from the capability to adjust the alignment of head-stem junction, the head portion and stem length of the patient-specific prosthesis could be determined preoperatively. Shukla found that the risk of instability press-fit prosthesis is dependent on cantilever quotient which is the ratio of length of radial head portion divided by length of total implant. A CQ of 0.35 was predictive of stability whereas a CQ of 0.6 or greater was predictive of instability. 35 In the present study, we could maintain the CQ of the prosthesis to be less than 0.35 with decrease the head portion length from minimizing the additional bone cut, and with increase the stem length from expanding the straight stem aligned to the intramedullary canal. The diameter of the press-fit stem was also determined preoperatively; however, optimal interference press-fit was not achieved in 5 of 10 patients. Insufficient press-fit of the stem in the first 4 patients possibly occurred from CAD setting the stem surface simply touch with the inner cortex. After change the setting to be overlapping, interference fit was successful in 5 of 6 cases. Planning error from the ill-defined margin of the inner cortex, technical error from intraoperative canal broaching, or individual bone quality may also contribute to suboptimal press-fit. In these cases, bone cement was used to obtain a satisfactory fixation. The guideline of using bone cement for insufficient intraoperative press-fit stability is similar to that used in a commercially available prosthesis with press-fit design. 36 In a retrospective study, there were no differences in survivorship between cemented versus uncemented stems. 17
According to the literature review, several complications were described following the radial head replacement. 37 Overlengthening due to high implantation of the prosthetic head causes overloading of distal humerus with subsequent capitellar erosion and arthritic change. Overlengthening is not a rare condition, being at least responsible for 13%–23% of radial revisions. 38 For the patient-specific prosthesis presented in this study, the final position was planned preoperatively. With the fixed level of bone cut and prosthetic height, no patient had the clinical and radiographic signs of overlengthening postoperatively. Periprosthetic radiolucency was commonly observed in radial head prosthesis with both cementless and cemented stem designs with varying clinical presentation. 22 Many studies showed that the radiographic signs of radiolucency were not correlated with poor surgical results.19–21 The radiolucent lines greater than 1 mm indicating loosening did not have any major negative effect in terms of clinical-functional outcomes and quality of life.39,40 On the other hand, symptomatic stem loosening of radial head implants necessitating implant removal or revision arthroplasty was reported. 41 A loose radial head prosthesis can cause proximal radial forearm pain regardless of whether the prosthesis is cemented or cementless. In the present study, periprosthetic radiolucency was found in two cases. Both patients did not have the clinical presentation of symptomatic loosening; however, longer follow up is required. Proximal radial neck resorption was found in 2 cases while restricted elbow flexion was observed in 1 patient. We hypothesized that this finding was the stress shielding phenomenon. Lee et al. demonstrated that stress shielding did not affect the clinical results. 42 In the present study, postoperatively HO was found in 2 of 10 cases. The occurrence of HO was reported at the variable rate following the radial head replacement.19,20,43 Bowman et al. observed that the patients who developed HO after radial head replacement had a statistically significant postoperative ROM deficits. 43 In our study, one of the patients who developed HO had restricted forearm pronation. Elbow instability with radial head prosthesis is often seen in the early postoperative period. Biomechanical studies have shown that radial head prosthesis alone could not stabilize the elbow to normal. 44 In our study, a patient who had associated ligament injury sustained a dislocation of the elbow postoperatively from premature weight bearing of the arm. This event was not referred as the complication related to the prosthesis because it occurred from failure of the ligament repair and congruity of the elbow was restored following reattached the re-ruptured ligaments.
This study was a multi-centric prospective study with the clinical, radiographic and laboratory evaluation at the baseline and follow-ups. However, it has some limitations including small sample size, absence of the controlled group, and short clinical follow-up. Although literature analysis suggests that if the clinical and radiographic results are positive after a 6 to 12-month follow-up, good outcomes can be also expected at a medium- or long-term follow-up, 45 a minimum follow-up of 2–3 years was advocated to fully evaluate the complications, such as symptomatic loosening and stress shielding, after radial head arthroplasty.42,46
Conclusion
The clinical, radiographic and laboratory outcomes in this preliminary study demonstrated that implantation of the 3D-printed patient-specific titanium radial head prosthesis is safe and may be a potential treatment option for irreparable radial head fracture.
Supplemental Material
Supplemental Material - Radial head replacement with the 3D-printed patient-specific titanium prosthesis: Preliminary results of a multi-centric prospective study
Supplemental Material for Radial head replacement with the 3D-printed patient-specific titanium prosthesis: Preliminary results of a multi-centric prospective study by Suriya Luenam, Arkaphat Kosiyatrakul, Chris Charoenlap, Chedtha Puncreobutr and Boonrat Lohwongwatana in Journal of Orthopaedic Surgery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was in-kind supported (including manufacturing of titanium implants) by Meticuly Co. Ltd, Chulalongkorn University, Thailand.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
