Abstract
Introduction
Soft tissue defects are a devastating complication of prosthetic joint infections (PJI) after total knee arthroplasty (TKA). Rotational flaps are commonly utilized to address these defects with variable reports of success. This study aimed to identify predictors of poor outcomes in rotational muscle flap placement after prosthetic knee infections. The authors hypothesized that outcomes may vary based on infecting pathogen and treatment characteristics.
Methods
44 cases of rotational muscle flaps for prosthetic knee infection were retrospectively evaluated at a tertiary referral hospital from 2007 to 2020. Muscle flap types included 39 medial and four lateral gastrocnemius, and one anterior tibialis. Minimum follow-up was 1 year (median: 3.4 years). Primary outcome was flap-related complications. Secondary outcomes included recurrent infection requiring additional surgery, final joint outcomes, and mortality.
Results
One-year complication-free flap survivorship was 83.9%, recurrent infection-free survivorship was 65.7%, and amputation-free survivorship was 79%. Multivariable cox regression revealed that rheumatoid arthritis diagnosis (HR: 3.4; p = .028) and methicillin-resistant Staphylococcus aureus-positive culture (HR: 4.0; p = .040) had increased risk, while Coagulase-negative Staphylococcus infections had reduced risk for recurrent or persistent infection (HR: 0.2; p = .023). Final joint outcome was retained TKA implant in 18 (40.9%), amputation in 15 (34.1%) patients, and definitive treatment with articulating spacer in 10 (22.7%). 5-years survivorship from death was 71.4%.
Conclusion
Rotational muscle flaps for soft tissue coverage of the knee are often performed in limb salvage situations with poor survivorship from flap complications, reinfections, and amputation. When considering surgical options for limb salvage, patients should be counseled on these risks.
Introduction
Total knee arthroplasty (TKA) has become an increasingly common procedure to treat osteoarthritis, providing substantial improvements in pain, joint function, and overall quality of life. Although rare, the incidence of complications after TKA can be devastating. About 11% of patients will develop wound complications, with 0.33% requiring surgical intervention for early wound complications, which is critical to address since it increases the risk of deep periprosthetic joint infection (PJI).1–3 The incidence of PJI is estimated to range from 1 to 3% and accounts for 25% of revision TKA procedures.4–8 Treatment algorithms for PJI typically depend on the timing of symptom onset and the mechanism of infection, with acute PJI typically being treated with a combination of debridement, antibiotics, and implant retention (DAIR) and chronic PJI being treated with two-stage revision arthroplasty. Treatment failure occurs in 10-30% of patients undergoing two-stage revision procedures, and soft tissue defect further increases this risk of failure.9–14
Surgical options to mitigate this increased risk of failure when treating an infected TKA with a soft tissue defect include debridement of the skin edges with primary closure, negative pressure wound therapy with secondary healing, skin grafts at sites of epidermal deficit, and/or rotational or free tissue flaps. 15 Rotational medial gastrocnemius muscle flaps are most commonly used for anterior knee soft tissue defects following TKA due to its reliability and ease of harvest. 15 A secondary perceived benefit is increased local blood supply via the sural vasculature, which offers increased delivery of antibiotics and nutrients for healing. 15 Alternatives to rotational gastrocnemius muscle flaps include vastus lateralis or latissimus dorsi free tissue transfer. 16 Investigation thus far into outcomes following muscle flap coverage following infected TKA has been limited by small cohorts, lack of follow-up, and significant variability in reported outcomes and definitions of success.17–25 The purpose of this study is to evaluate the outcomes and rates of treatment failure for rotational soft tissue coverage in the course of surgical treatment for PJI following TKA.
Materials and methods
Patient selection
Institutional review board approval was obtained prior to data collection. The institutional electronic health record (EHR) database was queried for patients with a diagnosis of PJI receiving muscle flap surgeries after TKA between 2007 and 2020, identifying 59 cases. Cohort selection was performed as outlined in in the flowchart, Figure 1. Patients receiving flap reconstruction after megaprostheses for oncological resection (n = 5), chronic extensor mechanism deficiency (n = 3), and aseptic revision (n = 1) were excluded. Four patients requiring flap coverage after knee arthrodesis or resection arthroplasty after multiple failed treatment for PJI were also excluded. One latissimus dorsi free flap was removed. Ultimately, 44 patients with rotational muscle flaps after prosthetic knee infection and a minimum of 1 year of follow-up were included. Mean follow-up duration was 4.5 years (range: 1.0–12.5 years). Patient cohort selection flowchart.
Surgical treatment
In this cohort, patients presenting to a tertiary care referral hospital with prosthetic knee infection were treated with debridement, antibiotics, and implant retention (DAIR) for acute PJI or 2-stage exchange for chronic infections. In brief, DAIR involved excisional debridement of necrotic or devitalized tissues and irrigation of the retained implant and joint with antibiotic impregnated solution, 3% acetic acid, diluted betadine, and normal saline. Metal components were scrubbed with a soft brush using chlorhexidine. After modular components were exchanged, the wound was irrigated again with normal saline and dissolvable antibiotic beads (broad spectrum antibiotics added to calcium sulfate) were placed intra-articularly. Jackson-Pratt (JP) surgical drains were placed within the joint (at the discretion of the orthopaedic surgeon) before the arthrotomy was closed as completely as possible, prior to same-surgery handoff to the plastic surgery team performing the flap reconstruction. Decision for flap placement was made by the orthopaedic surgeon preoperatively based on anticipated soft tissue availability, with notification of possible flap requirement to the plastic surgery team. Final decision was made intraoperatively after direct assessment of soft tissue coverage. Patients undergoing 2-stage revision had flap placement during explantation of the implant or prior spacer, or during reimplantation after spacer removal. Of 31 patients treated with an antibiotic spacer, 15 (48.4%) were treated with an articulating spacer (antibiotic cement, primary femoral component, all polyethylene tibial component) and 16 (51.6%) were treated with a static spacer (bulk cement in joint interface with stabilizing device selected by size of bone defect). Intra-operative cultures of the soft tissue, synovial fluid, and bone were collected for culture. For patients receiving flap reconstruction during second stage reimplantation, cultures obtained during prior component removal were reported. All flaps were performed by plastic surgeons. Medial and lateral gastrocnemius flaps were released from the Achilles tendon and the contralateral head, and assessed for vascularity before rotation over the anterior aspect of the knee to cover the defect. In one patient, the gastrocnemius had undergone 100% fat atrophy and a joint decision was made by the orthopedic and plastic surgery teams to perform a rotational anterior tibialis flap. In patients with remaining skin defect over the rotated muscle flap, a split-thickness skin graft (STSG) was harvested from the patient’s ipsilateral. Finally, a wound vacuum-assisted closure (V.A.C.) was placed over the skin graft. Three patients received STSG within 1 week after discharge. All patients received tailored antibiotic treatment under guidance of infectious disease physicians, which typically consisted of 6 weeks of parenteral antibiotic treatment and 6 weeks of oral antibiotic treatment, depending on the isolated microorganism and patients’ response to treatment All patients were allowed to weight bear as tolerated (WBAT) after their procedure except for patients with static spacers who were allowed to WBAT in a knee immobilizing orthosis. After discharge, patients were followed by both a dual orthopaedic-infectious disease musculoskeletal infections clinic and the plastic surgeon.
Study variables
The institutional EHR was manually reviewed and data was collected on patient age, sex, race, comorbidities including smoking status, diabetes, obesity, peripheral vascular disease, rheumatoid arthritis, and overall Elixhauser comorbidity score, wound location and size, prior surgical history, flap surgery, surgical treatment for PJI, and microbiological culture results. Primary outcome was flap-related complications. Flap related complications included any wound complications (eschar, dehiscence), flap failure, and focal necrosis requiring revascularization and debridement. Secondary outcomes were recurrent or persistent infection requiring surgery, amputation, death, flap-related complications, orthopaedic complications, and 90-days ED visits and readmissions.
Statistical analysis
Descriptive statistics comparing 2-stage and DAIR were performed using univariable statistics. Normality of continuous variable distributions were determined by the Shapiro-Wilk normality test and visually via histogram representation. The t test or Mann-Whitney U test were used as appropriate for comparison of continuous variables, and chi-square or Fisher’s exact test was performed for categorical variables. Multivariable cox (proportional hazards) regression analysis, controlling for age, smoking status, Elixhauser score, and PJI treatment type, was performed for calculating hazard ratios for various risk factors for primary (reinfection) and secondary outcomes (amputation and death). p-values reported on survival curve graphs were computed from log rank tests. Post hoc comparison of flap outcomes by flap type was performed using the chi-square test. All statistics were performed using RStudio v 1.1.463 (R Foundation, Vienna, Austria). Findings were considered statistically significant if p-value was <.05.
Results
Patient, PJI, and flap characteristics
Patient, PJI, and flap characteristics stratified by PJI surgical procedure.
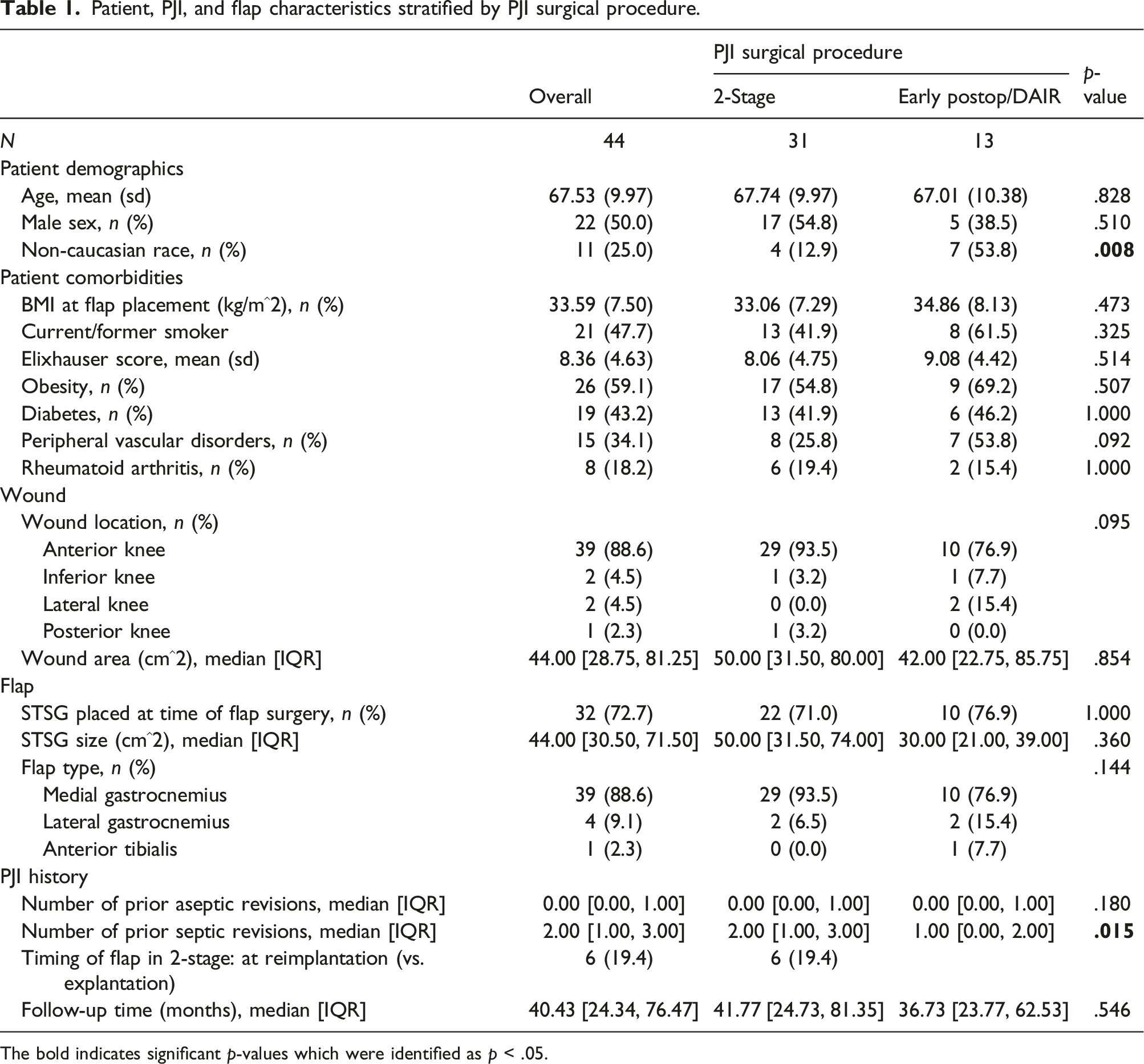
The bold indicates significant p-values which were identified as p < .05.
Most wound defects were located on the anterior aspect of the knee (n = 39/44, 88.6%) and median defect size was 44.0 cm2 [IQR: 28.8–81.3]. A split-thickness skin graft (STSG) was performed at the time of flap reconstruction in 72.7% of cases. Median STSG size was 44.0 cm2 [IQR: 30.50–71.50]. The most common muscle utilized for flap reconstruction was the medial gastrocnemius (n = 39/44, 88.6%), followed by lateral gastrocnemius (n = 4/44, 9.1%), and anterior tibialis (n = 1/44, 2.3%).
The median number of prior septic revisions for patients receiving flaps during 2-stage treatment was 2 [IQR: 1–3] compared to 1 [IQR: 0–2] for patients treated with DAIR (p = .015). For patients who underwent 2-stage exchange arthroplasty, most patients had flap placement during component removal or spacer exchange (n = 25/31, 80.6%) as opposed to during component reimplantation (n = 6/31, 19.4%).
Short- and long-term outcomes after index PJI treatment and flap placement
Short- and long-term outcomes after index PJI treatment and flap placement.
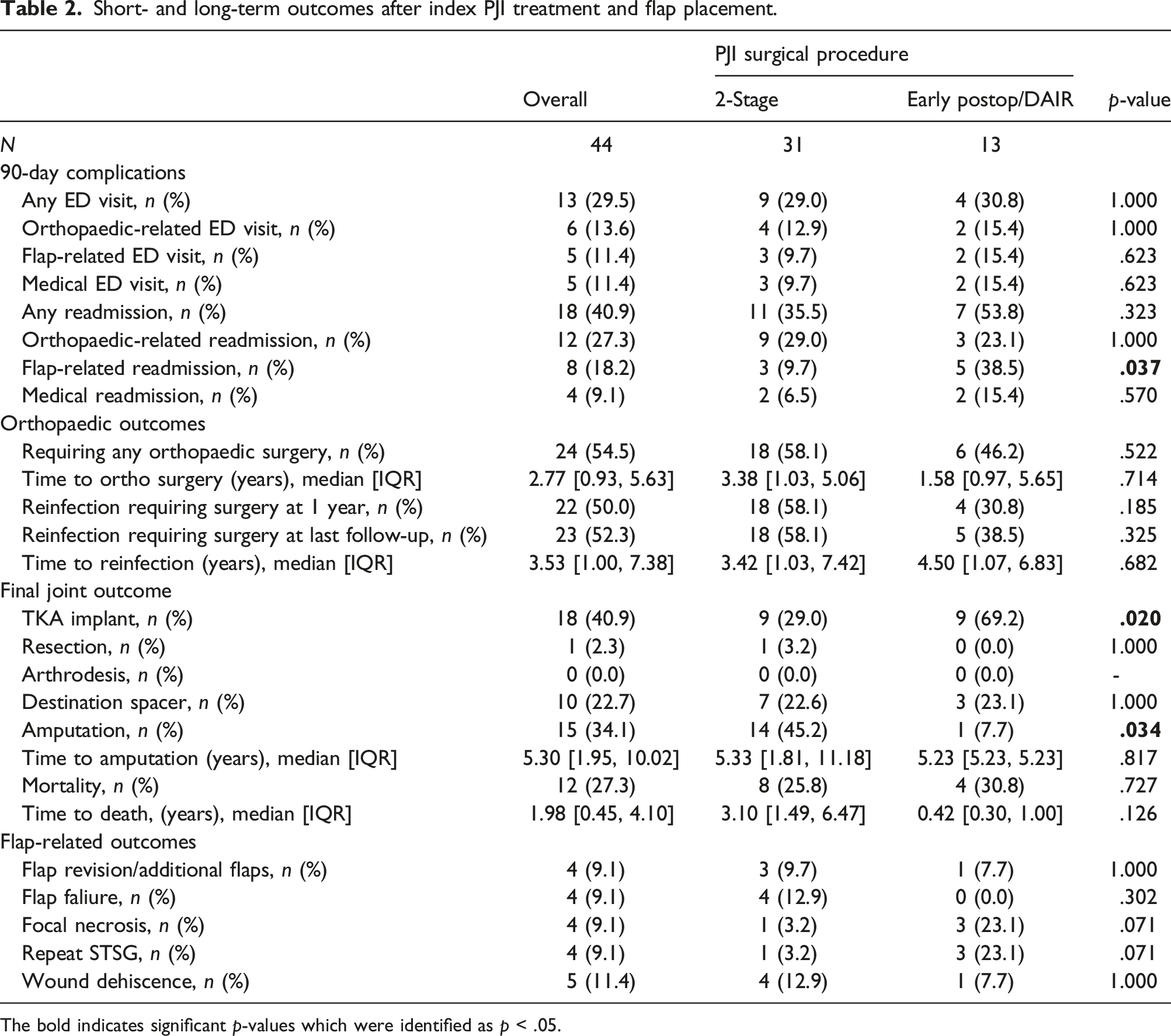
The bold indicates significant p-values which were identified as p < .05.
Most patients (n = 24/44, 54.5%) required additional orthopaedic surgery (excluding planned reimplantation in the 2-stage group). 50% required surgery for reinfection at 1 year post-operatively, and 52.3% (n = 23/44) were reinfected at last follow-up. Final joint outcome was successful TKA reimplantation in 18 (40.9%), amputation in 15 (34.1%), definitive treatment with an articulating spacer in 10 (22.7%), and resection arthroplasty in one (2.3%). Overall mortality at final follow-up was 27.3% (n = 12/44).
The most common flap complication was wound dehiscence (n = 5/44, 11.4%). Other flap complications occurred at the same rate (n = 4/44, 9.1%): flap failure, additional flaps, focal necrosis, and repeat STSG.
Overall survivorship from reinfection, amputation, death, and flap complications
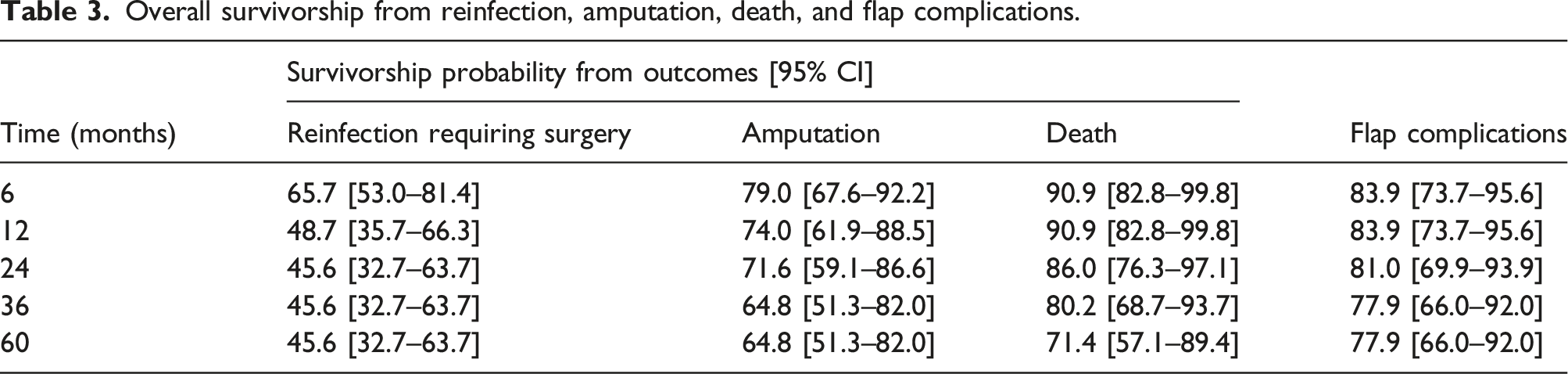
Overall survivorship from reinfection, amputation, death, and flap complications.
Risk factors for reinfection requiring surgery, amputation, death, and flap complications
Multivariable cox regression of risk factors for unfavorable outcomes.

The bold indicates significant p-values which were identified as p < .05.

Survivorship from reinfection requiring surgery after MRSA PJI.

Survivorship from reinfection requiring surgery after CoNS PJI.
Patients undergoing DAIR for surgical treatment of their PJI had reduced risk for amputation (HR: 0.11 [0.01–0.99]; p = .043) but increased risk for flap complications (HR: 4.94 [1.36–17.96]; p = .015) compared to 2-stage treatment in the multivariable analysis. Current or former smoker status was associated with increased risk for amputation (HR: 4.53 [1.15–17.81]; p = .031). Older age was associated with increased risk for death after flap placement (HR: 1.11 [95%CI: 1.03–1.20]; p = .005).
Discussion
We found acceptable survivorship of rotational flaps after prosthetic knee infections from flap-related complications but poor 1-year and 5-years survivorship from recurrent or persistent infection of 48.7% and 45.6%, respectively. Large open wounds after prosthetic knee infection are challenging problems for orthopaedic surgeons.26–28 Full-thickness defects that leave exposed bone and prosthesis significantly increases the risk for deep periprosthetic joint infection and may lead to prosthesis resection, arthrodesis, or limb loss.15,26 Rotational muscle flaps for defect coverage have been described with large variability in reported success rates for prosthesis and limb salvage.19,29–31 Although patients presenting with wound defects have significant comorbidites and prior surgical history of the joint, risk factors for successful limb salvage with muscle flaps are not well understood.16,32–34 The present study identifies several risk factors for poor outcomes following flap placement after prosthetic knee infection.
Our results on survivorship from reinfection lie within the range (42.3–63%) reported by prior studies of rotational flaps for prosthetic knee infection at various endpoints.19,32,35 The largest study of rotational flaps after TKA by Houdek and colleagues included flaps for aseptic causes of soft tissue defects, such as during primary TKA (n = 18/83), and reported the highest revision survival rate of 68% at 10 years. 33 Although flap reconstruction is utilized in various causes of soft tissue defects, PJI-induced defects have a considerably worse prognosis. 32 Notably, our amputation rate at final follow-up was higher at 34.1% (n = 15/44) than previously reported rates of 3.7–23.1% while our rates of other functional limb loss outcomes (i.e., resection or arthrodesis) were lower (n = 1/44).19,32,35 At our tertiary referral hospital, patients were offered rotational flaps in the setting of multiply-operated limb salvage after recalcitrant PJI, with a median of two prior septic revisions. The majority of amputations in our cohort were performed in patients undergoing either implant explantation or reimplantation during 2-stage revision (45.2% vs 7.7% in DAIR), who also had more prior septic revision surgeries than the DAIR group. This indicates that the 2-stage group had failed more prior PJI treatments. Despite the higher rate of amputation in the 2-stage group, mortality rates were similar for the 2-stage and DAIR groups (25.8 vs 30.8%), which may reflect more aggressive but appropriate treatment decision-making to achieve infection eradication by the surgeon and patient. Our cohort’s overall mortality rate of 27.3% is corroborated by the 26% reported by a prior study, which also supported the notion of advanced age as an independent risk factor. 35 Mortality rates are significant after soft tissue reconstruction for PJI, thus. While rotational muscle flaps can fill defects and provide a vascularized wound bed for antibiotic delivery, risk for recurrent infection remains particularly high for PJI patients, and functional limb loss may be unavoidable.
Infection clearance after PJI was associated with a multitude of factors including host status, appropriate treatment method, and pathogen.36,37 In this study, CoNS was the most common organism identified (n = 10, 21.7%), and MRSA (n = 5) and MSSA (n = 4) were the next most common (Supp. Table 2). Our culture results are consistent with three small studies identifying Staphylococcus spp. most frequently, and one study that found CoNS, specifically, to be the most common pathogen identified (n = 16, 37%).25,31,32,35 Resident gram-positive skin bacteria comprise a majority of pathogens identified in PJIs with wound complications. Interestingly, our results demonstrated an increased risk of recurrent infection for patients with MRSA-positive PJI and a reduced risk of reinfection for CoNS PJI patients. MRSA has been well-established to have infection recurrence despite appropriate surgical treatment, and although CoNS has recently gained increasing attention for its virulence with rising rates of methicillin-resistance, our cohort highlights MRSA as the pathogen with worse outcomes in the setting of flap reconstruction.37,38 The relatively lower risk for recurrent infection for CoNS is likely explained by the high occurrence of polymicrobial (n = 8, 18.2%) and enterococcal (n = 6, 13.6%) PJIs in this cohort, which are less common but notorious for high treatment failure rates.39,40 More importantly, overall outcomes for all patients were dismal with a recurrent or persistent reinfection rate of 52.3%, and the virulence of offending pathogens seen in this cohort emphasize the importance of an intact soft tissue envelope in preventing infections. Regarding host status, patients with a pre-operative diagnosis of rheumatoid arthritis also demonstrated higher risk for infection, commensurate with prior reports of rheumatoid arthritis as an independent risk factor for worse outcomes after PJI. 41
Flap success is also dependent on the timing of flap placement with improved rates of limb salvage and component retention in patients with prophylactic as opposed to reactive flap placement.23,36,37,42 However, flap placement timing in the setting of a 2-stage revision for PJI has not been sufficiently studied. In a sub-analysis, we found no significant differences in infection clearance rates between patients with flaps placed at prosthesis explantation (n = 10/25, 40%) or reimplantation (n = 3/6, 50%) in the 2-stage revision (p = .676). Similarly, a study by Warren et al. showed that 37.5% (n = 3/8) of patients receiving flap coverage at explantation and 40% (n = 4/10) at reimplantation retained their TKA. 32 While no differences were noted for 2-stage timing, our study revealed patients receiving flaps concurrently with DAIR had relatively improved outcomes in regards to amputation compared to the 2-stage group. DAIR patients had higher rates of minor flap-related complications such as repeat STSG (Table 2). All cases of flap failure and most cases of wound dehiscence and additional flap placement occurred in the 2-stage group. Although all flaps were “reactive” in this cohort, the DAIR group had fewer prior septic revisions, representing flap placement at an earlier stage in their overall treatment course for PJI. Our study supports the importance of early recognition of soft tissue insufficiency and appropriate planning for wound management when primary closure is not attainable as compounding wound defects can lead to functional limb loss or amputation. 15
Flap complications were not uncommon in this cohort with a 1- and 5-years complication-free flap survivorship of 83.9% and 77.9%, respectively. Assessing the vascular viability of flaps and not over-tensioning the flap is crucial to avoiding flap failure and necrosis.15,28 Additionally, infections can precipitate sinus tract formation, wound dehiscence, and flap failure requiring additional flap reconstruction. In this study, there were 5 patients with wound dehiscence and 4 patients requiring additional flaps, three of which received pedicled fasciocutaneous flaps and one required a rotational rectus femoris flap. In a post-hoc analysis, there was no difference in failure rates for medial (n = 3/39) and lateral gastrocnemius (n = 1/4) flaps (p = .333). Patients requiring repeat STSG after failed graft take were common reasons for readmission within the 90-day period and were most common in the DAIR group. Flaps are not without complications and these additional risks should be discussed with the patients when discussing treatment options for limb salvage.
There are several limitations to this study with the major limitation being our study’s small sample size. Exclusion of patients to diminish confounders through a more homogenous cohort reduced the power of our analysis. Next, functional outcome scores for patients were sparse and unattainable for this cohort. Therefore, we are unable to comment on the quality of life for patients with varying final joint outcomes. Additionally, this was a single-institution study and the regional frequencies and virulence of pathogens detected in this study may not reflect those of other institutions. Furthermore, treatment decisions by a variety of infectious disease providers and surgeons over the 13-years study time period may affect outcomes. Nevertheless, the authors believe the present study provides relevant and informative findings on rotational muscle flaps after prosthetic knee infection.
Conclusion
Rotational muscle flaps for soft tissue coverage after prosthetic knee infections have high rates of reinfection, amputation, and mortality. This study is the first to report associations between offending pathogen and risk for recurrent or persistent infection following PJI treatment and flap placement. Recognition of soft tissue insufficiency and flap reconstruction early in a patient’s surgical course for prosthetic knee infection may provide better outcomes for limb salvage. Despite the ability to provide soft tissue coverage with rotational muscle flaps, patients should be advised on the high rates of treatment failure and functional limb loss.
Supplemental Material
Supplemental Material - Risk factors for failure after rotational muscle flap coverage for prosthetic knee infections
Supplemental Material for Risk factors for failure after rotational muscle flap coverage for prosthetic knee infections by Billy I Kim, Colleen M Wixted, Andrew M Schwartz, William A Jiranek, Sean P Ryan and Thorsten M Seyler in Journal of Orthopaedic Surgery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
