Abstract
Fracture-related infection (FRI) is a complication that impacts care costs, quality of life, and patient function. Great strides have been made in the last decade to obtain a common language for definition and diagnosis with the contribution of the Fracture-Related Infection Consensus. Although FRI treatment requires the participation of clinical specialists in infectious diseases for the management of antibiotics, it is necessary to understand that this complication is an eminently surgical pathology. The orthopedic surgeon must play a leadership role in the prevention and treatment of this complex disease. In this review, the most relevant aspects of prevention are updated, and a strategy for a sequential and comprehensive approach to the patient with this complication is presented.
Keywords
Introduction
Fracture-related infection (FRI) is a complication that impacts care costs, quality of life, and patient function. According to Iliaens et al., 1 direct costs are eight times higher than a patient without infection, and physical function is significantly diminished. Multiple factors are associated with its development, 2 most of which are not modifiable as they are present in the patient (e.g. comorbidities, age, nutritional factors) or are related to the energy of the initial trauma (e.g. open fractures, bone loss, or severe soft tissue injuries). 3 On the other hand, there are also modifiable factors related to medical and surgical interventions, which are essentially helpful for prevention. The surgeon plays a fundamental role here, not only with the proper compliance of the strategies supported by the evidence in the current guidelines4–6 but also with the execution of a rational surgical technique, which leads to minor damage to the soft tissues and bone, preserving vascularization and providing the mechanical stability necessary for the consolidation phenomenon to occur efficiently. Although FRI treatment requires the participation of clinical specialists in infectious diseases for the management of antibiotics, it is necessary to understand that this complication is an eminently surgical pathology. It is necessary to start with a timely diagnosis and arrive at the correct surgical strategy for each case, but as far as possible to build a standard of management to compare the results between centers dedicated to the treatment of this pathology.
In recent years it has been possible to advance in consolidating scientific evidence using expert consensus. 7 In the absence of high-quality evidence, these consensuses are presented as a guide to homogenize the language surrounding the pathology 8 and define guidelines for diagnosis 9 and treatment.10,11
The purpose of this review is to present updated information on surgical prevention and treatment strategies in the management of FRI, highlighting the fundamental role of the surgeon in this process, and finally to mention a strategy that allows the surgeon to establish a sequential, logical, verifiable, and comparable flow for dealing with this type of patient.
Risk factors of fracture-related infection and tools to predict the risk
Proportion of fracture-related infections according to anatomical area.

Risk factors for fracture related infections in two predictive models.

Prevention
FRI prevention measures can be divided into three stages: preoperative, operative, and postoperative, according to National Institute for Health and Care Excellence (NICE), 4 Centers for Disease Control (CDC), 5 and World Health Organization (WHO) 6 guidelines.
Preoperative measurements
The measures described in this phase are several: patient bathing, decolonization of Staphylococcus aureus, hand hygiene, preparation of the surgical site, and antibiotic prophylaxis.
Asymptomatic carriers of Staphylococcus aureus in trauma are present between 5.2% and 17%15,16; for this reason, detection by screening tests and decolonization through 2% nasal mupirocin is a cost-effective intervention.17–19 Constant hand washing with antimicrobial soap and water or alcohol-based antiseptics by health care workers is a simple intervention that needs to be repeated each time a patient is examined, at shift changes, when returning from breaks, or after using the restroom.
Surgical site preparation includes hair removal, preoperative bathing, and skin antisepsis. Preoperative hair removal is not absolutely necessary and should not be performed with razors but by using a clipper. In patients with fractures or trauma, preoperative bathing is difficult given the nature of the patient’s injuries; however, it is a recommended practice for most orthopedic surgeries; this can be performed with soap and water or with chlorhexidine and can also be performed with commercial towels impregnated with chlorhexidine. Skin antisepsis should ideally be performed with alcohol-based antiseptic solutions with chlorhexidine or with alcohol-based antiseptic solutions with iodine. 6
Finally, a single-dose prophylactic antibiotic administered 30–60 min prior to the incision is a strongly recommended practice. The use of antibiotics that cover the skin bacteria, in general Gram-positive, is preferred, so first or second-generation cephalosporins are recommended. The dose of the prophylactic antibiotic should be reinforced in case of surgeries lasting more than 4 h and significant intraoperative bleeding. 20 In closed fractures, the preoperative dose with or without reinforcement in the transoperative period is sufficient, and additional doses are not recommended in the postoperative period. In open fractures, the duration of antibiotic therapy should not exceed 72 hours.21–23
Intraoperative measurements
This stage includes the surgical environment, surgical technique, patient metabolic measures, and wound management.
The surgical environment is constituted by the surgical personnel’s clothing, with anti-fluid surgical gowns and gowns, surgical masks, goggles, and gloves. Handwashing prior to surgery, with initial washing with soap and water with chlorhexidine for 3–5 min at the beginning of the day and applying alcohol-based antiseptic solutions with chlorhexidine. In orthopedic trauma surgery, the use of double gloves is recommended due to the perforation that can occur in up to 21% of the cases,24, 25 for which a permanent revision should be performed and changed every 90 min. The use of laminar flow is a useful but not strictly necessary measure to maintain the sterility of the surgical field. Similarly, reducing the number of personnel in the operating room to the strictly necessary number and reducing their circulation during the procedure reduces the risk of infection. Adhesive drapes are another frequently used measure, but the evidence does not demonstrate superiority over not using them. 6
Careful surgical technique is crucial in infection prevention, proper handling of soft tissues and instruments to incise the skin, separate soft tissues and reduce fractures, saline and intermediate pressure lavage of open fractures, debridement of devitalized tissues in open and closed fractures, and irrigation at the end of surgery decrease the risk of infection.
Metabolic measures are also necessary; It is recommended to maintain the patient’s core temperature above 36°C to avoid hypothermia and maintain adequate oxygenation and optimal glycemic levels. 6
It is recommended to close the wound in planes with atraumatic surgical technique and without tension, using adequate sutures and with separate stitches, and then cover with sterile dressings that can remain for 8–10 days until the sutures are removed.
Postoperative measurements
In the postoperative period, personnel hygiene and wound care measures should be considered. Explaining the wound care to the patient very well, keeping the sterile dressing clean, avoiding the use of prophylactic antibiotics after wound closure, and performing adequate rehabilitation is critical in the post-surgical management of patients.
Surgical treatment
FRI in an eminently surgically managed condition; empirical antibiotic therapy without surgical debridement and microbiological identification show variable results 26 with the risk of chronicity, antibiotic toxicity, and increased bacterial resistance.
SOSTAC® Strategy for planning management of fracture related infections.
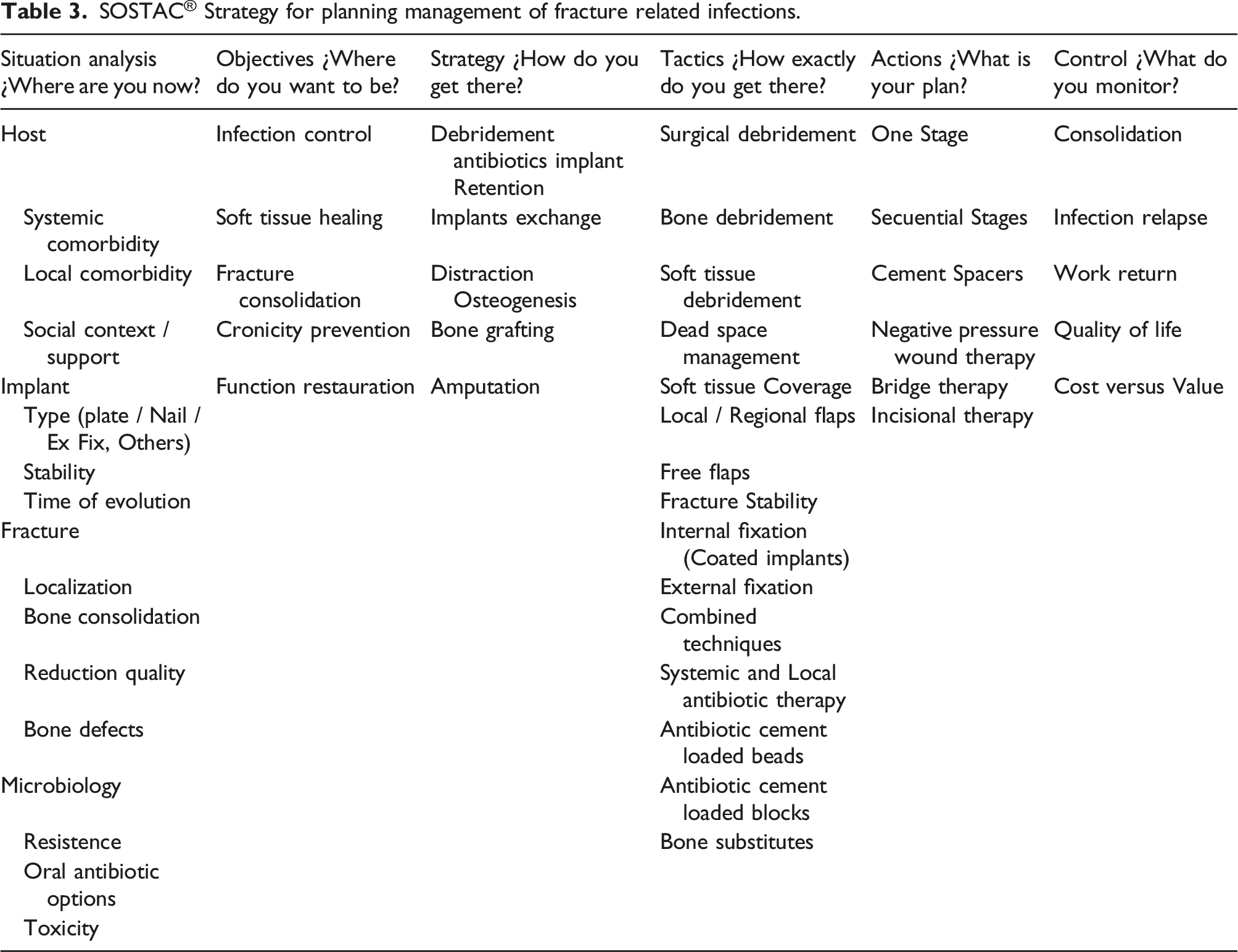
Situation analysis
The first step will be to determine the current status of the four main components.
First, the host’s status in terms of systemic comorbidity (e.g. diabetes, immune status, nutritional status, smoking, Etc.), local comorbidity (peripheral vascular or neurological disease), previous functional status, social and economic support for treatment and recovery, this last point will help to define if it is a good candidate for major reconstructive procedures that could take months and years since in some patients it is possibly more efficient to opt for an early amputation.
Second, it is necessary to evaluate the type of implant, its stability, and the time elapsed from application to the time of infection. According to the Willenegger and Roth 28 classification, early (<3 weeks) and some delayed (3–10 weeks) infections could be treated with implant retention. In the presence of intramedullary implants, it is not possible to perform an efficient debridement of the medullary canal without removing the implant; so, in delayed or late infections, removal, cleaning of the medullary canal, and new intramedullary fixation or change of stabilization method should be considered, since the biofilm is mature and the risk of not achieving control of the infection and relapses in the medium and long term will be greater.
Third, the state of the fracture should be evaluated in terms of its location, quality of the reduction, state of consolidation, and presence of bone defects. Infection control in fractures with a favorable soft tissue environment (upper limb, pelvis, or hip) is usually faster and easier. In contrast, infection is more difficult to control in sites with less soft tissue support, such as the tibia or calcaneus, and requires more elaborate reconstructive efforts. In poor reductions and infection, the opportunity should be taken to improve joint alignment and congruence to obtain better functional results. On the other hand, the presence of bony defects will broadly define the need for advanced reconstructive procedures such as distraction osteogenesis or the Masquelet technique. 29 Finally, in the presence of a consolidated fracture and infection, implant removal should always be considered to reduce the possibility of future relapses. 30
Last but not least, the microbiological diagnosis will determine the antibiotic therapy that is complementary to surgical management. It is necessary to obtain an adequate number of samples for cultures, ideally five deep tissue samples, 31 never take samples from fistulous tracts, and in some occasions, sonication and pathological anatomy could be used as complementary tools. The ideal scenario for culture sampling is when the patient has not received antibiotic therapy for at least 2 weeks before, surgical debridement is performed prior to sampling, and deep tissues such as muscle, bone, or tissues adjacent to the implant are sampled. With the microbiological identification of multiresistant bacteria, it is suggested to avoid internal fixation due to the risk of infectious relapse. In FRI, the prolonged use of antibiotics is required, up to 12 weeks, with increased risk of toxicity; therefore, the orthopedic surgeon should be aware of drug interactions, alterations of renal and hepatic function, and the presence of other cutaneous, hematological or gastrointestinal adverse reactions.
Objectives
Once the main components of the patient’s current condition have been identified, it is necessary to be clear about treatment goals. The FRI consensus has defined five goals. 7
Although consolidation can be achieved even in the presence of infection, the first objective should be to control the infection to have a healthy local and systemic environment to avoid toxicity and induction of resistance due to inadequate or prolonged use of antibiotics; the following two objectives are complementary, achieving soft tissue healing and bone consolidation. By meeting these first three objectives, it will be possible to meet the last two, which refer to the prevention of chronicity and the functional restoration of the patient as close as possible to the previous one. It is essential to recognize that infection related to fractures is also a bone infection and that given the mechanisms of resistance to treatment by bacteria, 32 a risk of relapse of 10–20% can be expected according to the follow-up time, and therefore it is a disease that is controllable but not curable.
Strategy
To meet the five objectives mentioned above, a broad spectrum of surgical techniques can be grouped into five strategies: 1. DAIR (Debridement, antibiotics, and implant retention) 2. Changing implants 3. Distraction osteogenesis 4. Bone grafting with or without spacers 5. Amputation
Using the Willenegger and Roth classification as a guide, it can be said that for early (<3 weeks) and some delayed (3–10 weeks) infections, performing surgical debridement, achieving adequate microbiological identification, and providing prolonged antibiotic therapy (up to 12 weeks) with implant retention can offer up to 90% success defined as consolidation. Morgenstern et al.
33
consider that the time of evolution of the infection is only one more component to make decisions regarding implant retention. In addition to the time, the implant’s stability, the location of the fracture, and finally, the time of evolution of the infection should be considered for implant removal or retention (Figure 1). Flow chart to define implant retention or removal. This applies to fractures that have not healed. Any consolidated fracture with infection and internal fixation requires removal of the implant. Three basic questions: 1. Is the implant stable ? 2. Where is the fracture ? 3. How long does the initial fixation surgery take?
The orthopedic surgeon needs to recognize and appropriately manage FRI in the acute phase, which will undoubtedly improve the chance of success. 34 In unconsolidated fractures without bone defects greater than three cm and delayed or late infections (>10 weeks), it will be necessary to consider changing the implant and even the fixation technique. For more extensive bone defects, it will be necessary to consider techniques such as distraction osteogenesis with external fixation (Ilizarov type circular systems, monoplanar or combined techniques with internal and external fixation) or the use of bone grafts preceded or not by cement spacers with or without antibiotic (Masquelet technique). Finally, in the presence of recalcitrant infection, large bone defects, or hosts with high comorbidity burden or poor social support, it is possible that amputation may offer similar functional outcomes and should be an option to consider in the acute phase and more so in the late phases of reconstruction when multiple infectious relapses have occurred. Despite the severity of FRI, the proportion of amputation remains around 10%. 35
Tactics
The cornerstone of FRI treatment is rational and timely surgical debridement. One of the main doubts at the debridement is defining the extent of bone and soft tissue resection. Although the use of images such as MRI or nuclear medicine images such as PET has been mentioned to define the extent of resection, 36 these images can overestimate the extent of bone involvement and lead to unnecessary resections. Recently, a promising technique has been described to identify bone viability intraoperatively 37 ; however, this is not yet widespread, nor has it been validated in studies of higher methodological quality. Therefore, the most practical and reproducible criterion will be the presence of punctuate bleeding in the cortical bone (Paprika’s sign), and the debridement of the soft tissues will be guided by the bleeding capacity of the subcutaneous cellular tissue and muscle.
Debridement should focus on removing all infected and necrotic tissue that may favor chronicity; so, a frequent consequence of debridement is the presence of soft tissue and bone defects, so surgeons faced with the management of this pathology should have training in soft tissue reconstruction or within their teams have surgeons with training in plastic surgery and microsurgery. The choice of soft tissue reconstruction method depends on each case and the availability of resources in each hospital. In early or delayed infections, regional muscle or fasciocutaneous flaps are an excellent option for managing soft tissue defects. However, in late cases, with a history of multiple interventions, increased fibrosis, and poorer quality of the surrounding soft tissues, free flaps are likely to offer more significant advantages in terms of cosmetic results or secondary infections. 38 The results in the literature are variable, and there is no clear recommendation as to whether one type of flap is superior to another; however, it is necessary to promote the concept of orthoplastic management of these patients, which has been shown in different series to reduce complications and improve outcomes. 39
Bone stability is essential for infection control, soft tissue healing, and bone healing. The method of fixation (internal vs external) will depend on each case. However, in our experience, we favor the use of internal fixation in case of the absence of bone defects or defects smaller than three cm and that the infection is caused by bacteria that can be treated with oral antibiotics with biofilm activity, ideally including rifampicin (for Gram-positive) or a quinolone (for Gram-negative), for at least 12 weeks. 11 If the above two criteria are not met, treatment with external fixation techniques is recommended.
The use of local antibiotics is essential for the management of FRI; it is a fundamental part of the management of dead space secondary to infection or surgical debridement, allows high local concentrations of the specific antibiotic, and also compartmentalizes bone defects, facilitating secondary reconstruction either with distraction osteogenesis or bone grafting. 10 The local antibiotic release is more efficient with cement beads than blocks, but the latter is more beneficial for compartmentalization of the defect and helps as structural support. In any case, we recommend using local antibiotics directed by microbiological isolation. In most cases, the handmade mixture is made with a maximum of 10% of the weight of the cement, using, among others, doses of vancomycin 3 g or meropenem 3 g.
The specialists in infectious diseases will guide the systemic antibiotic. However, the orthopedic surgeon must be aware of the possible adverse reactions and promote the early change to oral antibiotic therapy, supported by the OVIVA study 40 and the combination of antibiofilm antibiotics, such as rifampicin or quinolones, once local control of the infection is achieved.
Actions
There are multiple surgical techniques for the reconstructive management of patients with FRI (Figure 2). For patients with early infections (<3 weeks), DAIR may be the best treatment option. For patients with delayed and late infections, techniques may be performed at one or more stages depending on local control of infection, microbiological identification, and associated bone or soft tissue defects. Case 1 (Top). Male, 26 years old, FRI delayed (5 weeks), MRSA infection (a); Management in two stages. Implant removal, sequestrectomy and cement spacer with antibiotic (b); Bone transport with circular fixator (c); 3-year follow-up without infection relapse (d). Case 2. (Middle) Female, 35 years old, 3B open fracture, partial bone defect, reconstruction with Masquelet is planned (a–b); late infection (8 weeks after graft) (c); Wide resection and bone transport are performed with a monoplanar fixator (d); removal of the fixator and refracture in the docking site (e), internal fixation with a conventional compression nail; final result 2 years of follow-up without infectious relapse (f). Case 3. (Bottom). Male 32 years old; Late infection (2 years in treatment without improvement), non-union with partial bone defect (a); partial secuestrectomy (b), definitive management with vascularized fibula flap (c); 28 months of follow-up without infectious relapse (d).
According to the meta-analysis of Bezstarosti et al., 41 the results obtained with these techniques are not substantially different, and between 85 and 90% of success defined as consolidation is achieved; however, a higher proportion of relapse is described with the Masquelet technique, and this should require greater vigilance of the initial surgical technique, specifically of the debridement.
Dead space management is fundamental in the early phases of treatment and is achieved with antibiotic cement beads, cement blocks, or bone substitutes such as calcium sulfate or ceramics. 10 However, the evidence for the latter is limited, and their high cost means that their availability in low or middle-income countries is restricted.
The use of negative pressure therapy in the presence of FRI is one of the most controversial topics at present. 42 It is a valuable tool to keep the wound isolated from the hospital environment, reduce the number of dressings and maintain a cleaner environment for the patient; it also helps to decrease the dead space and the size of the soft-tissue defect, which may eventually reduce the complexity of the coverage. 43 In addition, it offers the potential for wound edge contraction, and the low oxygen tension benefits angiogenesis. However, we believe this technique should be used as bridge therapy for the shortest time possible until definitive coverage of soft tissue defects with regional or free flaps is achieved. In patients who have undergone multiple previous interventions and have wounds at high risk of suffering or dehiscence, the use of incisional negative pressure therapy has shown promising results.
In general, the evidence in the literature is heterogeneous and does not allow making absolute recommendations in favor of one or the other intervention; however, surgical treatment of FRI should be guided by common sense, efficient surgical debridement, and the use of the resources available in each particular center adjusted to local realities and the needs of each particular patient.
Control
The measurement of outcomes in the management of FRI is a fundamental part of the interdisciplinary management process. The goals of treatment have been clearly defined by consensus, but to our knowledge, there is no homogeneity in the clinically meaningful outcomes to be considered in this scenario. Most of the literature focuses on defining consolidation and infectious relapse; according to Bezstarosti’s meta-analysis, 35 consolidation does not seem to be a significant problem; while infectious relapse has been poorly defined and most studies are not homogeneous in terms of follow-up time, which is recommended to be at least 1 year. 44 The clinically meaningful outcomes should be patient-centered, and there are already efforts to create scales with methodologically sound validation processes that in the future will allow us to compare results in a more objective way.45, 46 In the meantime, we must recognize that the results are variable, depend on multiple variables, and the impact on the function and quality of life is very high and, unfortunately, these results are barely regular.47,48
Conclusions
The surgeon must be responsible for prevention, leadership, education, training, and teamwork. Each surgeon must prevent the complications inherent to the procedures he/she performs, including postoperative infection. As has been mentioned, the surgeon plays a critical role in prevention, from in informing the patient about his risks and how to prevent them, as well as in the education of the medical and paramedical staff, in simple topics such as hand washing, behavior in operating rooms, courses related to infection related to fractures, transmitting the teaching of professionalism and behavior to the trainees.
In addition, the construction of work teams including specialists in infectious diseases, internal medicine, plastic surgery, nursing, anesthesiologists, pharmacy technicians, clinical laboratory, and microbiology department. It is the surgeon who must take the lead in the care of this complex surgical situation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
