Abstract
We aimed to assess the antitumor activity of Orobanche crenata methanolic extract and evaluate its cytotoxic effect on different cancer cell lines to develop an effective natural anticancer drug. Components of O. crenata methanolic extract were analyzed using gas chromatography–mass spectrometry. The extract’s antioxidant activity was assessed by 2,2-diphenyl-1-picrylhydrazyl and ferric reducing antioxidant power procedures and cytotoxicity of the extract was assessed by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and lactate dehydrogenase assays. Caspase-3 activity was also estimated. O. crenata methanolic extract shows powerful antioxidant activity. The extract inhibited the propagation of human hepatocellular carcinoma (HepG2), human prostate cancer (PC3), human breast adenocarcinoma (MCF-7), and human colon carcinoma (HCT-116) in a dose-dependent manner. O. crenata–treated cells displayed obvious morphological structures distinctive of apoptosis. MTT assay exposed that the extract presented prevention of cell persistence in a dose-dependent means and revealed extremely cytotoxic activity against HepG2, PC3, MCF-7, and HCT-116 with 50% inhibitory concentration values 30.3, 111, 89.6, and 28.6 µg/mL, respectively, after 24 h of incubation. In addition, treatment of HCT-116 with various concentrations of the extract caused the release of lactate dehydrogenase and induction of caspase-3 activity in a dose-dependent way. In conclusion, our findings suggested that the O. crenata extract possesses potent antioxidant, cytotoxic activity, and anticancer properties which are possibly due to the principal bioactive phytochemical composites existing in this plant. These results can be used to develop new drugs for cancer treatment.
Introduction
The idea of screening therapeutic plants is to search for bright lethal anticancer agents to human cancers. Despite the amazing developments in modern treatment, cancer remains a global health problem, thus attempting the exploration for new substitute approach. The nature as a vast treasured supplier of possible origin for chemotherapeutic agents has been reviewed. 1
The plant kingdom is interesting since its derived compounds control oxidative stress and hence affect cancer development. Pharmacological activities in plants are powerfully interrelated with natural antioxidants. 2 Antioxidants showed a capability to diminish opposing results of reactive species and handling of many diseases and severe disorders including cancer.3,4 Plant-derived compounds such as phenolic compounds, flavonoids, and others have antitumor properties and may diminish disagreeable side effects.5,6 In addition, plants contain various molecular bioactive compounds, and consequently plant kingdom is a probable foundation of antineoplastic and cytotoxic agents.7,8
Several studies have shown successful results for the screening of natural antioxidants from many plant materials. However, even with the huge amount of investigations about the screening of bioactive composites from plants in diverse portions of the world, up to now little is identified about the probable active composites from the parasitic plants.
Orobanche crenata (Broomrape) is a holo-parasite harmful to leguminous plants. Orobanchaceae is a well-recognized family in conventional remedy for its antispasmodic, antihyperglycemic, hypotensive, analgesic, and antimicrobial effects. 9 California Broomrape or cancer root is used superficially to treat skin infections produced by Streptococcus, as a poultice or wash for wounds, ulcers, and herpes lesions. Ancient Chinese herbalists have also used Broomrape varieties for various ailments. Broomrape was a food supplement for the ancient wandering nomads living in northwestern China. 10
In Egypt, Orobanche represents a major constraint in Middle and Upper Egypt. 11 Orobanche is an unconventional edible plant (like asparagus) that belongs to the local flora in Egypt and is readily consumed by many people. Thus, it appears to be relatively safe to ingest. The results of the acute lethal toxicity test carried out on rats showed that Orobanche extract is safe up to 5 g/kg with no mortalities. 10
Therefore, this study was commenced with the objective to assess the antitumor activity of O. crenata methanolic extract and evaluate its cytotoxic effect on different cancer cell lines in agreement with the apparent variations of cell morphology upon exposure to the extract which possibly subsidize to the progress of a hopeful anticancer agent.
Materials and methods
Reagents
Dimethyl sulfoxide (DMSO) and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Sigma (St. Louis, MO, USA). Fetal bovine serum, Dulbecco’s Modified Eagle Medium (DMEM), RPMI-1640 medium,
Collection and identification of plant materials
Orobanche crenata plant was obtained from agricultural field crop, Delta, Egypt in wintertime 2017. Authentication of the plant was conducted at Botany Department, Faculty of Science, Ain Shams University. Voucher specimens are deposited in department herbarium. Plant name has been checked with http://www.theplantlist.org/.
Preparation of plant extract
The dried residue of the plant (10 g) was extracted in 250 mL of methanol and stirred on a magnetic stirrer for 12 h along with the described method by Kalia et al. 12 The plant/methanol mixture was then allowed to rest overnight. The extract was then filtered by Whatman filter paper and concentrated with rotary evaporator at a constant temperature of 40°C. The dried extract was collected and stored in the refrigerator for further tests.
Gas chromatography–mass spectrometry analysis
Gas chromatography–mass spectrometry (GC-MS) analysis was used for identification of the bioactive compounds in O. crenata methanol extract. The study was accomplished with a GC (Agilent technologies 7890A) connected with a mass-selective detector (MSD, Agilent 7000) armed with a polar Agilent HP-5ms (5%-phenyl methyl polysiloxane) capillary column (30 m × 0.25 mm i.d. and 0.25-μm film thickness). Helium as a carrier gas with the linear velocity of 1 mL/min was used.
The determination of all the identified compounds was made using a percent relative peak area. A provisional identification of the components was made in function with the relative retention time and the mass spectra with those of the National Institute of Standard and Technology, NIST Willy library data of the GC-MS system.
Measurement of antioxidant activity
Determination of the total antioxidant capacity and total phenolic contents
Total antioxidant capacity of the O. crenata extracts was evaluated by the phospho-molybdenum method 13 using ascorbic acid as a reference antioxidant compound. The test sample’s optical density was measured at 695 nm. The antioxidant capacity was expressed for the samples as ascorbic acid equivalents (AE). The total phenolics were measured according to Folin–Ciocalteu’s method. 14
Determination of radical scavenging activity
Radical scavenging activity of O. crenata methanolic extract against stable 2, 2-diphenyl-1-picrylhydrazyl (DPPH) was determined spectrophotometrically according to Brand-Williams et al. 15 The use of O. crenata extract as antioxidants was measured by its scavenging activity. The capacity of the antioxidant to scavenge the free radical is estimated as a percentage of discoloration of the DPPH in solution in methanol. The percentage of antioxidant activity was determined according to the subsequent calculation:
% Antioxidant activity = (OD control − OD sample/OD control) × 100. The results are the mean of three separate quantities ± standard deviation
IC50 or 50% inhibitory concentration is the concentration of the extract sample needed to reduce 50% of the DPPH free radical.
Determination of antioxidant reducing power capacity
The reducing power of the extracts was determined by ferric reducing antioxidant power (FRAP) assay according to the way of Oyaizu. 16 The existence of reductants in the solution causes the reduction of the ferric to the ferrous form. Increase in absorbance of sample with concentrations designates high reducing power of the samples.
In vitro anticancer activity
Culturing of cell lines
Different cell lines such as human hepatocellular carcinoma (HepG2), human prostate cancer (PC3), human breast adenocarcinoma (MCF-7), and human colon carcinoma (HCT-116) were provided from American Type Culture Collection (ATCC, Rockville, MD, USA) VACSERA Tissue Culture Unit. HepG2, PC3, and MCF-7 cells were cultured in RPMI-1640 medium accompanied with 10% inactivated fetal calf serum and 50 µg/mL gentamycin. HCT-116 cells were cultured in DMEM supplemented with 10% fetal bovine serum, 1% penicillin–streptomycin, and 1%
For anticancer assays, cancer cell lines were suspended in medium at concentration 5 × 104 cell/well in 96-well tissue culture plates, and then incubated for 24 h. Afterward, various concentrations of O. crenata methanolic extract (3.9–500 µg/mL) were prepared using DMSO then were added into 96-well plates (three replicates), untreated cells were used as negative control, 0.5% DMSO were run for each 96-well plate as an untreated control. After incubating for 24 h, viable cells number was identified by the MTT test and lactate dehydrogenase (LDH) assay.
Assessment and comparison of in vitro cytotoxic activity of O. crenata methanolic extract was accomplished using MTT assay 17 on diverse cancer cell lines.
MTT test is planned to be used for the quantification of both cell propagation and survival by determining the activity of a mitochondrial enzyme succinate dehydrogenase. The assay assumes that the MTT yellow water-soluble tetrazolium salt reduction to the blue/magenta water-insoluble MTT formazan occurs by the mitochondrial succinate dehydrogenase of living cells. In this way, formazan crystals’ concentration can be measured using a spectrophotometer and it is in parallel association with the number of metabolically active cells. 18
In brief, the media was detached from the plate and substituted with 100 µL of new culture RPMI 1640 medium, at that moment 10 µL of the 12-mM MTT stock solution (5 mg of MTT in 1 mL of phosphate buffer saline (PBS)) were added to each well counting the untreated negative controls. Then, the 96-well plates were incubated at 37°C and 5% CO2 for 4 h. After that 85 µL aliquot of media were discarded from the wells, and 50 µL of DMSO were added to each well, mixed, and incubated for 10 min at 37°C. The absorbance was detected at 590 nm with the microplate reader (SunRise; TECAN, Inc, Morrisville, NC, USA) to identify the number of survival cells and the percentage of viability was considered from (1 − (Abs.t/Abs.c)) × 100%, where Abs.t is the absorbance of treated wells with the extract and Abs.c is the absorbance of untreated wells.
The relation among viable cells and extract concentration is designed to get the viability curve of all cancer cell line after treatment with O. crenata extract concentrations. The concentration required to cause toxic effects in 50% of intact cells (IC50) was valued from graphic plots of the dose–response curve for each concentration using Graphpad Prism software (San Diego, CA, USA).
Morphological detection
Cells of each cell line were planted in six-well plates and incubated with 0, 31.25, 125, and 500 μg/mL of O. crenata methanolic extract for 24 h. Cells’ morphological variations were visualized with light microscope (Olympus, Center Valley, PA, USA).
Cytotoxicity LDH assay in HCT-116 cell line
LDH assay is a test used to identify the cytotoxicity of an anticancer agent on cancer cells. LDH is a cytosolic enzyme, which is released into the nearby culture medium upon cell injury or lysis during apoptosis and necrosis. Therefore, LDH activity in any cell culture medium could be used as an indicator of cell membrane integrity and development, and thus measure cytotoxicity.19,20 The integrity of membrane was judged by assessing the quantity of LDH leakage existing in the culture media. LDH assay was carried out using a BioVision LDH-cytotoxicity colorimetric assay kit according to the manufacturer’s instructions. Briefly, cells were planted and treated as described in MTT assay. After 24-h incubation (5% CO2, 90% relative humidity, 37°C), the culture medium was aspirated and centrifuged at 4000 r/min for 5 min. Then, 100 μL of the supernatant was moved to a new 96-well plate that previously contained 100 μL of the reaction mixture and incubated for 30 min in dark room temperature. HCl of 1 N was used to stop the reaction and the optical density was measured at a wavelength of 450 nm in a microplate reader (Sunrise™; TECAN, Männedorf, Switzerland). Experiments were achieved in triplicate and used for calculations. The positive control cells used to show total LDH leakage were treated with 1% Triton X-100 in culture media. LDH levels in the media were quantified and compared to control values consistent with the kit instructions.
Quantification of caspase-3 activity in HCT-116 cell line
HCT-116 (5 × 104 cell) were grown in each well of six-well plate in 2 mL of complete culture medium after treatment with the extract (50, 25, and 10 μg/mL) according to the predetermined IC50 of extract for 24 hours, drifted and adhesive cells were washed in cold PBS, then cells were diffused in PBS and subjected to one freeze/thaw cycle at −80°C. Lysed cells were centrifuged at 2000 r/min at 4°C for 10 min to eliminate cell remains, after which caspase-3 activity assay was performed in the supernatant fluids and compared to cell treated with extract-free media (control group) for 24 h.
The activity of caspase-3 assay was carried out according to caspase-3 activity assay ELISA Kit (Quantikine®; R&D Systems, Inc, Minneapolis, MN, USA). The absorbance of each well was identified within 30 min, using a SunRise (Tecan, USA) microplate reader. The caspase-3 concentrations were determined from a concentration-known standard curve. Protein amounts were measured according to the assay of Bradford 21 and all the results were expressed per mg protein content.
Results
GC-MS analysis results
The GC-MS analysis of O. crenata methanolic extract revealed the existence of several bioactive compounds (Figure 1), the plant extract’s compounds along with their retention time (RT), molecular formula, and peak area are given in Table 1. The major identified bioactive constituents in the O. crenata (i.e. carvacrol, glycerol, erythritol, mannitol, actinobolin, tyrosol, pyrogallol, acetamide, etc.) are characterized by antimicrobial, antifungal, anticancer, antioxidant, and anti-inflammatory properties.

GC-MS chromatogram represents the separated bioactive constitutes of O. crenata methanol extract
Bioactive compounds identified in methanol extract of O. crenata.

Antioxidants’ activity
Orobanche crenata methanolic extract exhibited good antioxidant activity relative to that of ascorbic acid as a standard antioxidant (866.63 ± 10.21 mg AE/g extract). Also, the amount of total phenolic content was measured and expressed in mg Gallic acid equivalent per gram (mg GAE/g). The quantitative study of phytoconstituents of O. crenata methanolic extract revealed the presence of total phenols of 762.5 ± 11.5 mg GAE/g (Table 2).
Antioxidant properties and phenolic yield of O. crenata methanolic extract.

AE: acid equivalent; GAE: gallic acid equivalent; SD: standard deviation.
Results are expressed as mean ± SD.
The antioxidant activities of O. crenata extract with various concentrations were explored using DPPH free radical scavenging activity (IC50 = 408 µg/mL). The presence of antioxidants in the methanol extract was exposed to FRAP at different concentrations (Table 3).
Antioxidant activity of O. crenata methanolic extract using DPPH and FRAP.
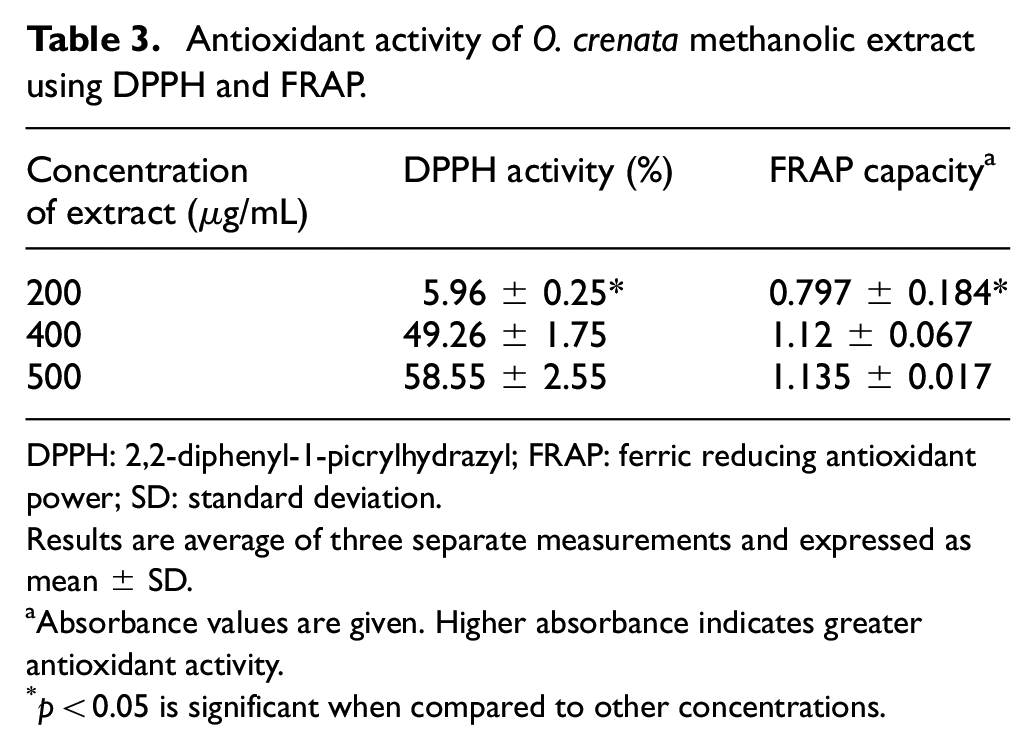
DPPH: 2,2-diphenyl-1-picrylhydrazyl; FRAP: ferric reducing antioxidant power; SD: standard deviation.
Results are average of three separate measurements and expressed as mean ± SD.
Absorbance values are given. Higher absorbance indicates greater antioxidant activity.
p < 0.05 is significant when compared to other concentrations.
Anticancer activity
In the current study, the cytotoxicity of methanolic extract of O. crenata was determined using MTT assay in HepG2, PC3, MCF-7, and HCT-116 cell lines exposed to 3.9, 7.9, 15.6, 31.25, 62.5, 125, 250, and 500 μg/mL of the extracts at incubation period of 24 h.
Cell viability analyses designated that O. crenata extract caused development inhibition of HepG2, PC3, MCF-7, and HCT-116 cell lines in dose-dependent manners (Figure 2(a)–(d)). O. crenata was found to have a great cytotoxic effect on HepG2, PC3, MCF-7, and HCT-116 cells with a IC50 value of 30.3, 111, 89.6, and 28.6 µg/mL, respectively.
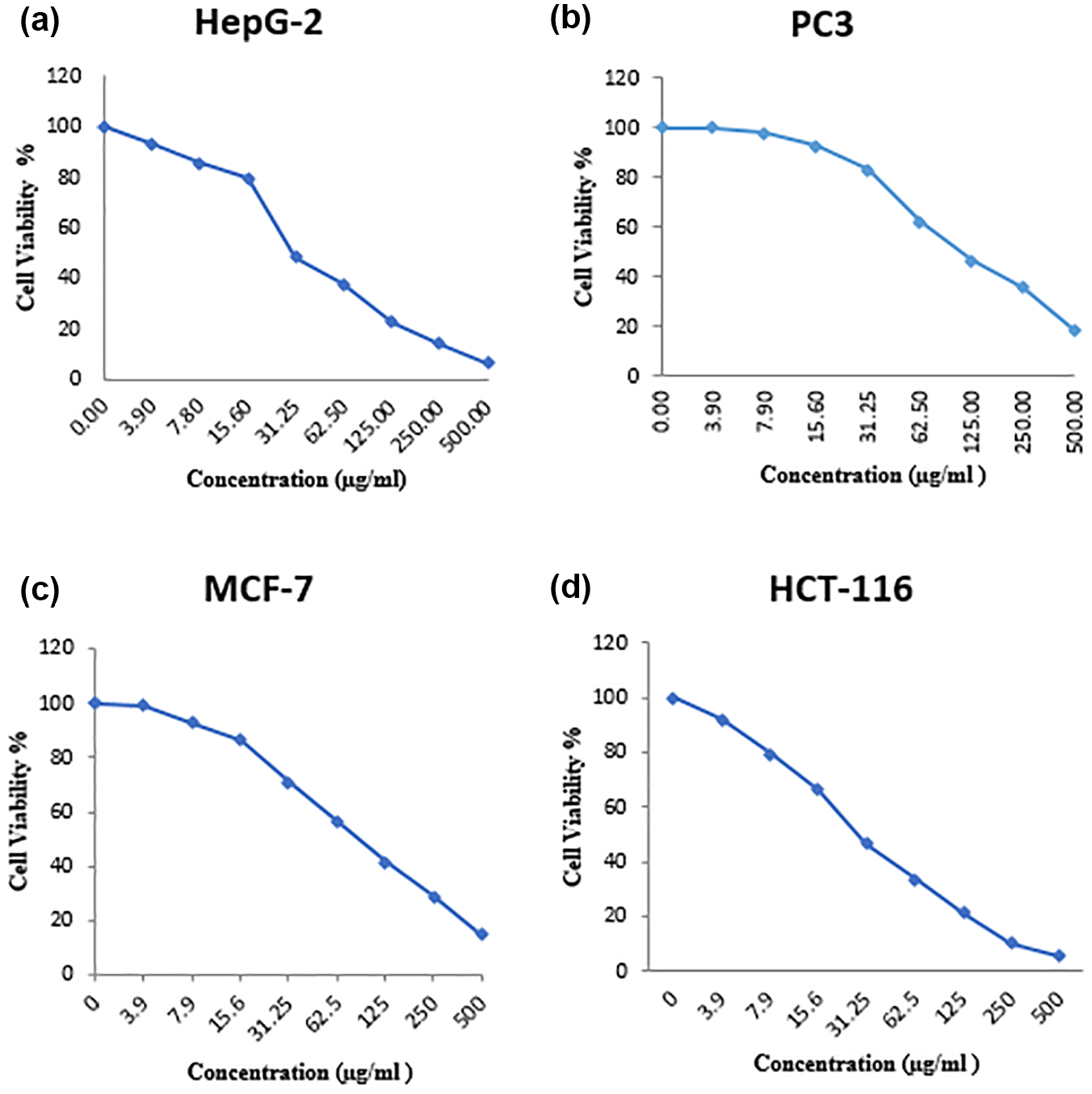
Toxicity effects of the O. crenata methanolic extract against cancer cell line: (a) HepG-2 (IC50 = 30.3 µg/mL), (b) PC3 (IC50 = 111 µg/mL), (c) MCF-7 (IC50 = 89.6 µg/mL), and (d) HCT-116 (IC50 = 28. 6 µg/mL) after 24 h of incubation.
An initial approach to cell death by O. crenata was shown by morphological comparison with untreated controls showing that after 24 h of treating the four cell lines with the different concentrations, the number of cells diminished, and the remaining cells reduced in size with obvious cell shrinkage, destruction of the monolayer, and chromatin clumping. Untreated cells with no change in morphology were shown in the negative control group (Figure 3).
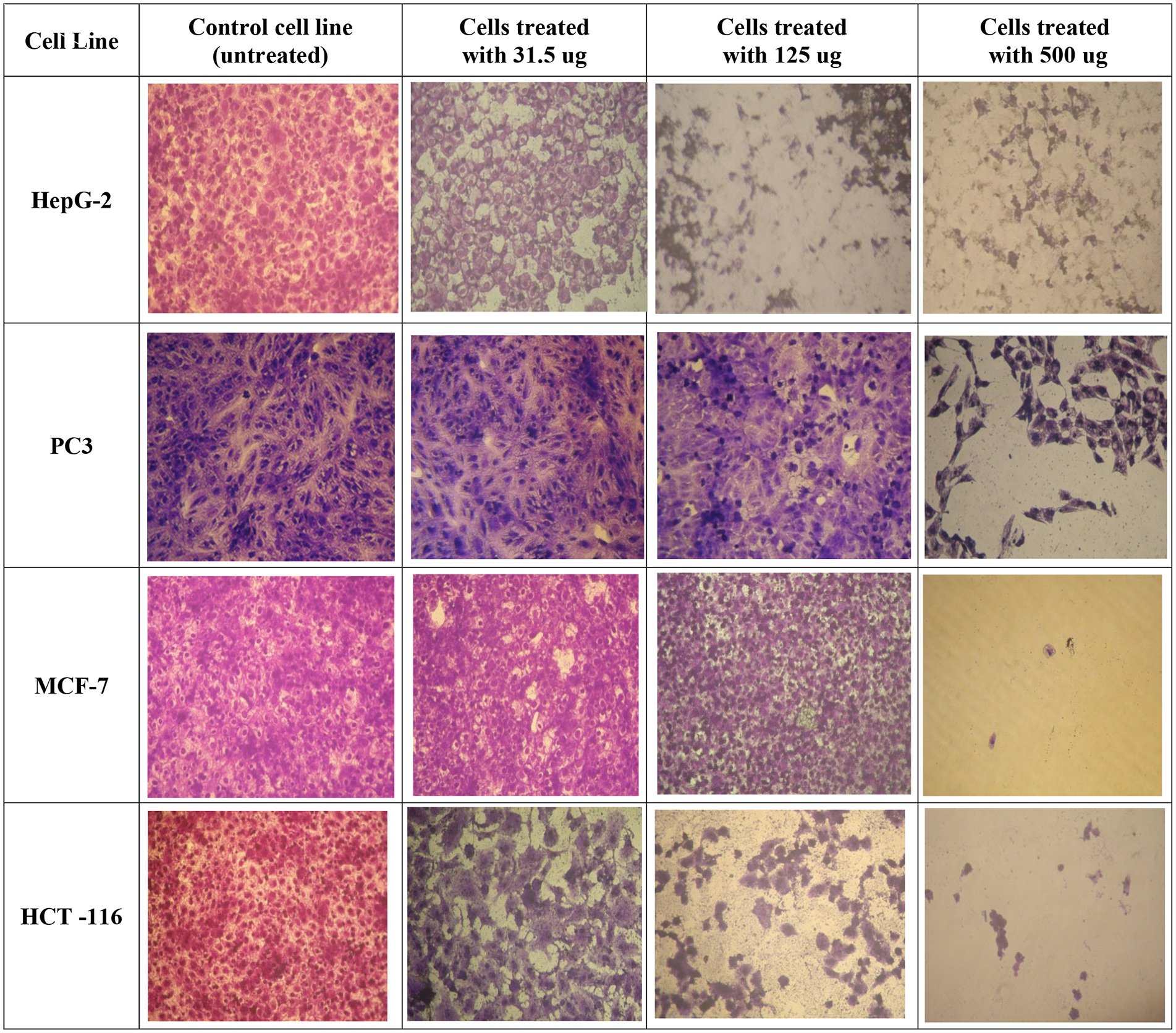
Microscopic photographs of the morphological changes induced by the methanolic extract of various concentrations of O. crenata on different cell lines after 24-h treatment and comparison with untreated controls. The medium was removed, and the cells were rinsed and visualized under light microscope.
Although the initial viability (toxicity) was studied in four cell lines (HepG2, PC3, MCF-7, and HCT-116), the more detailed MTT, LDH, and caspase-3 assessment was performed in HCT-116 only because this is just a preliminary study for the anticancer effect of O. crenata.
The cell-membrane damage of HCT-116 cells after the treatment with O. crenata extract was determined by the leakage of LDH into the incubation medium. The LDH release curve for HCT-116 cell line treated with diverse concentrations of the O. crenata proposed that the extract cytotoxic effect was concentration-dependent (Figure 4). Cell viability was detected using LDH assay with IC50 = 46.3 ± 5.71 µg/mL.
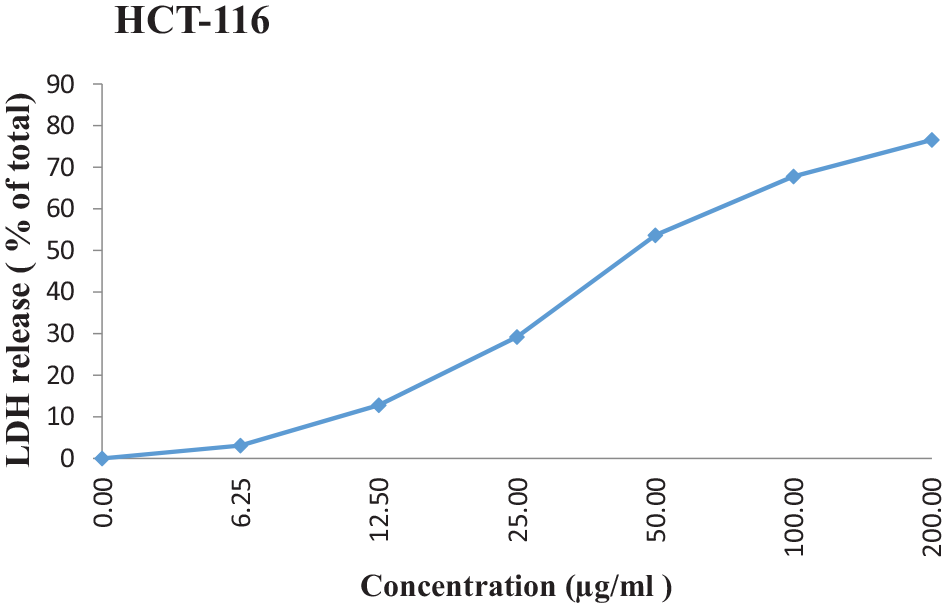
The effect of increasing concentration of O. crenata extract on LDH release in HCT-116 cell line. Each point represents the mean of the data from three independent experiments; IC50 = 46.3 ± 5.71 µg/mL.
The O. crenata treatments triggered a higher activity of LDH than the control non-treated cells (Figure 5). There was a significant increase in the LDH with increasing concentrations of O. crenata (p < 0.01) (Figure 5). Higher concentrations of the extract produced greater LDH release on cell line.

LDH activity with different concentration treatment by O. crenata extract. Values are expressed as mean ± SD of three independent assays.
Four concentrations of the extract had been submitted for human caspase-3 determination by ELISA in HCT-116 treated and untreated cells. From the ELISA result, O. crenata extract with a concentration of 50 μg/mL shows higher caspase-3 concentration (1.13 ng/mg protein) than O. crenata extract with concentration of 25 and 10 μg/mL (Figure 6).

Caspase-3 concentration with different concentration treatment by O. crenata extract. Values are expressed as mean ± SD of three independent assays.
Discussion
In spite of the outshine by current synthetic chemistry as a technique of drug detection and invention, the influence of new products from probable bioactive plants or their extracts for disease handling and inhibition is still enormous. 22 Besides, plant-derived medications had led to utmost spread within the vicinity of antitumor upon where drugs were described to improvise the chemotherapy of some cancers. 23 Plants comprise nearly limitless capacity to create phytoconstituents that attract scientists in the search for innovative and original chemotherapeutics. 24 The exploration for new anticancer compounds in plant remedies and traditional foods is a truthful and auspicious strategy for its inhibition. 25
Plants used in folk and traditional medicines have been acknowledged as primes for healing drug progress in modern medicine. O. crenata was used by some North American tribes for externally treating ulcers and cancerous growths. 10 Moreover, O. coerulescens of the same family has a synergistic effect with 5-fluorouracil in renal cancer treatment, which hinders tumor metastasis. According to that study, O. coerulescens disables nuclear factor-κB which decreases the expression of intercellular adhesion molecule-1 (ICAM-1) so that apoptosis is achieved by its synergistic effect. It appears to likewise prevent metastasis through this mechanism. 26 Hence in the present study, cytotoxicity and anticancer activity of O. crenata extract were evaluated in vitro on different cancer cell lines.
GC-MS analysis of O. crenata methanolic extract revealed the existence of many bioactive compounds. From GC-MS spectra results, some compounds have been formerly stated to possess anticancer activity, including pyrogallol moiety which was found to exhibit cytotoxic effect against cancer cells; 27 carvacrol which owns an extensive range of biological activities useful for clinical applications as antimicrobial, antioxidant, and anticancer activities; 28 actinobolin which is found to prevent development of both Gram-positive and Gram-negative bacteria and have weak antineoplastic activity; 29 and acetamide derivatives have also been stated to cause anticancer activity. 30 In addition, other components in O. crenata methanolic extract have been shown to display antioxidant activity such as the non-toxic tyrosol, 31 erythritol, 32 and mannitol, 33 while others show antimicrobial and anti-inflammatory activities such as carvacrol, 34 glycerol, 35 and acetamide. 36 Plants of Orobanchaceae are famous for the accumulation of large amount of flavonoids, iridoides, phenyl-propanoides, and their glycosides that could explain the various reported biological effects such as antibacterial, hypoglycemic, hypotensive, anti-inflammatory, anti-obesity, hepatoprotective, cytotoxic, sedative, and neuroleptic in addition to the stimulation of immune response. 37 Studies have detected the presence of many bioactive compounds in the methanolic extracts of O. crenata counting polyphenols, tannins, alkaloids, saponins, and flavonoids which display numerous biological activities.9,38
The antioxidant activity of phenolics plays vital role in the neutralization of free radicals. 39 The anticarcinogenic and antimutagenic activity of polyphenols is due to their antioxidant activity, which deactivates direct carcinogens/mutagens and prevents the stimulation of indirect carcinogens/mutagens extracellularly. Phytochemicals, such as phenolic compounds, improve the level of cellular antioxidative system and activate the cytochrome P-450 resulting in cleansing the activity of carcinogens intracellularly. 40 Our results showed that O. crenata plant extract investigated in this study is rich with phenolic compounds. Also, the plant extract exhibited good antioxidant activity relative to that of ascorbic acid as a standard antioxidant. Previous studies have stated that plants exerting a high quantity of phenolic content displayed a respectable antioxidant activity. 41
Curative plants comprise very robust secondary metabolites bearing antioxidant, radical scavenging agents could help in inhibition of cancers and autoimmune disorders. Reducing activities are serious due to the fatal properties of free radical ions existing in biological systems through reactive oxygen species (ROS) which ultimately effects cancers. 42 Free radical scavenging activity is a mechanism for anti-oxidation. 43
To measure the antioxidant properties of plant sample, two different procedures at least must be used since antioxidants existing in natural products can neutralize radicals through two main mechanisms: single electron transfer (SET) and hydrogen atom transfer (HAT). 44 DPPH assay is an example of the HAT reaction while FRAP assay is a SET reaction. Antioxidant activity of O. crenata methanolic extracts was evaluated by the DPPH method and FRAP assay. DPPH radical scavenging activity displayed high scavenging activity consistent with concentrations increase of plant extract also, ferric reducing power capacity exhibited dose-dependent increase with plant concentration.
Reducing activity is a strong indicator of the antioxidant activity and the reducing characteristics are commonly linked with the occurrence of reductants, which apply antioxidant action through breaking the free radical chain by providing a hydrogen atom or stopping peroxide formation. 45 The obvious reducing activity of the methanolic extract of Orobanche seems to be attributable to the occurrence of phenolic compounds that may act as reductants by providing the electrons and reacting with free radicals to change them to more steady products and terminate radical chain reaction. 46
Consequently, the synergistic effects of phenolic, ascorbic acid, and antioxidant capacity may be responsible for the apoptotic effects detected in this study. Taken together, our findings confirmed that O. crenata methanol extract may confer a defensive role against different cancers which was more likely due to the bioactive compounds contained in the extract. Some of the compounds that exist in the extract have promising anticancer activity, and these properties could lead to the anticancer abilities detected in the extract. Hence, in this study O. crenata methanolic extract was assessed as a novel anticancer agent.
Cytotoxic effects of O. crenata extracts were appraised in cells from the human HepG2, PC3, MCF-7, and HCT-116 using the MTT assay and the LDH leakage assay. These two tests are widely used in in vitro toxicology studies. They are designed for finding cytotoxic effects on cell survival succeeding exposure to test materials. 47
O. crenata could treat many cancers such as hepatic, prostate, breast, and colon cancers and it shows more effective cytotoxicity on colon cancer followed by hepatic then breast and prostate cancers (as indicated by IC50 values).
Cancer is characterized by an uncontrolled increase in cell propagation and/or a decrease in cell apoptosis. Apoptosis induction and growth inhibition of cancer cells were supposed to be approaches for the cancer treatment. 48 O. crenata significantly repressed the growth of colon cancer cells, which was established by the diminished cell thickness. Moreover, caspases which represent family of cysteine proteases, play role in the control of apoptosis. Caspase-3, a key executioner of apoptosis, is thought to be a biomarker for cell apoptosis. 49
In the current study, HCT-116 cell line was treated with the plant extract then caspase-3 enzymatic activity within the cells was quantified. Since caspase-3 activity increases while cells die and cell numbers drop, it was vital to normalize caspase-3 activity to protein content, to obtain more accurate results. Measuring caspase-3 activity in cells is considered a straight way for the determination of apoptosis. A significant dose-dependent increase of the caspase-3 activity was observed in HCT-116 cell line–treated cells with increasing concentrations of methanolic extract of O. crenata.
Low caspase-3 activity level was noticed in the untreated cells which was apparently owing to the insignificant slight number of apoptotic cells present in the growing cell population. Among the execution caspases, caspase-3 was found to play an important role in intrinsic and extrinsic pathways of apoptosis through cleaving several critical cellular proteins. 50 This cleavage enables disassembly of cell, which is apparent from the morphological variations, such as cell shrinkage, membrane blebbing, chromatin condensation, and DNA fragmentation. 51 These morphological variations were also detected from the O. crenata–treated cells and powerfully proposed the caspase-mediated apoptosis in HCT-116 cells.
Finally, previous researches on mode of action of O. crenata as a holo-parasitic plant noxious to legumes, stated that throughout the initial steps of diffusion, the parasitic plant liberates enzymes that permit diffusion of disturbing cells between host cells 52 and an adherent substance that enables inner attaching of the parasite to host cell walls. 53 It is presumed that the parasite (i.e. O. crenata) can defeat some host barriers by discharging its secretion at the point of resistance, as suggested before by Joel et al. 53 The secretion released when resistance is met, either at initial diffusion phases or later when some vascular connections have already been known, plays role as an inner attaching device and as a fulcrum for extra physical efforts to break into “stubborn” host tissues. The secretion contains peroxidases 54 and pectin methyl esterases 52 which may aid in altering the structure of host cell walls, thus may make them fragile and more vulnerable to attack. We assumed that similar mechanism could be followed by O. crenata when attacking cancer cells in vitro.
Conclusion
In the light of these findings, it is apparent that O. crenata could be considered as a vital source of antioxidants. It is observed that methanol extract of O. crenata had significant anticancer activity in different cancer cell lines using in vitro models. These findings highlight the abilities of O. crenata extract in treatment of many cancer diseases such as hepatic, prostate, breast, and colon cancers. Existence of polyphenols in O. crenata may consequently be responsible for its total antioxidant potential. Thus, it could assist as an ideal candidate for bio-pharmaceutical therapeutic products and nutraceutical products. Futures perspectives of operating an in vivo study and exploring O. crenata as an alternative food preservative agent to replace the synthetic food preservatives could be a promising step.
The concept of using the drug combination of O. crenata–derived molecules (or their natural cocktails present in the extract) to target a wider range of signaling pathways in cancer cells seems to be substantially beneficial compared to using single anticancer O. crenata–derived molecule. The future preclinical and clinical research focused on O. crenata–derived compounds or extracts influencing the carcinogenesis should be directed toward the several important issues as clarifying the molecular targets and signaling pathways involved in anticancer activity, determining of an effective (and non-toxic) doses in humans, assessing a combined effect of several O. crenata–derived components or extracts targeting several relevant genetic, epigenetic, and immunomodulatory pathways.
Footnotes
Acknowledgements
We would like to express our gratitude to our colleagues in Botanical Department, Faculty of Science Ain Shams University for their help in identification of plant.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study does not involve any primary biological material from humans or animals. Ethical approval not applicable.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
